Abstract
More than 20% of bacterial proteins are noncytoplasmic, and most of these pass through the SecYEG channel en route to the periplasm, cell membrane, or surrounding environment. The Sec pathway, encompassing SecYEG and several associated proteins (SecA, SecB, YidC, SecDFYajC), is of interest as a potential drug target because it is distinct from targets of current drugs, is essential for bacterial growth, and exhibits dissimilarities in eukaryotes and bacteria that increase the likelihood of selectively inhibiting the microbial pathway. As a step toward validating the pathway as a drug target, we have adapted a mechanism-based whole-cell assay in a manner suitable for high-throughput screening (HTS). The assay uses an engineered strain of Escherichia coli that accumulates beta-galactosidase (β-gal) in its cytoplasm if translocation through SecYEG is blocked. The assay should facilitate rapid identification of compounds that specifically block the Sec pathway because widely, toxic compounds and nonspecific protein synthesis inhibitors prevent β-gal production and thus do not register as hits. Testing of current antibiotics confirmed that they do not generally act through the Sec pathway. A mini-screen of 800 compounds indicated the assay’s readiness for larger screening projects.
The bacterial Sec pathway is a conserved mechanism of protein export whose details have been revealed through extensive genetic and biochemical studies. 1 Preproteins destined to pass into or through the inner membrane are fed into a translocase channel consisting of three subunits (SecYEG; Fig. 1 ). Preproteins are guided to the channel either by the signal recognition particle (SRP) and FtsY or by the SecB and SecA proteins. Chemical energy for SecB/SecA-mediated translocation comes from the hydrolysis of adenosine triphosphate (ATP) by SecA, a molecular motor that is loosely associated with the SecYEG channel. A newly synthesized preprotein with a signal sequence specifying export binds first to the SecB chaperone protein; SecA then recognizes the SecB-preprotein complex, releases SecB, and threads the preprotein into SecYEG via multiple rounds of ATP hydrolysis. In most cases, preproteins are then processed by a signal peptidase and fold into their mature structure on the trans side of the membrane. (Most inner-membrane proteins are handled by SRP, although some appear also to require SecA. 1 )
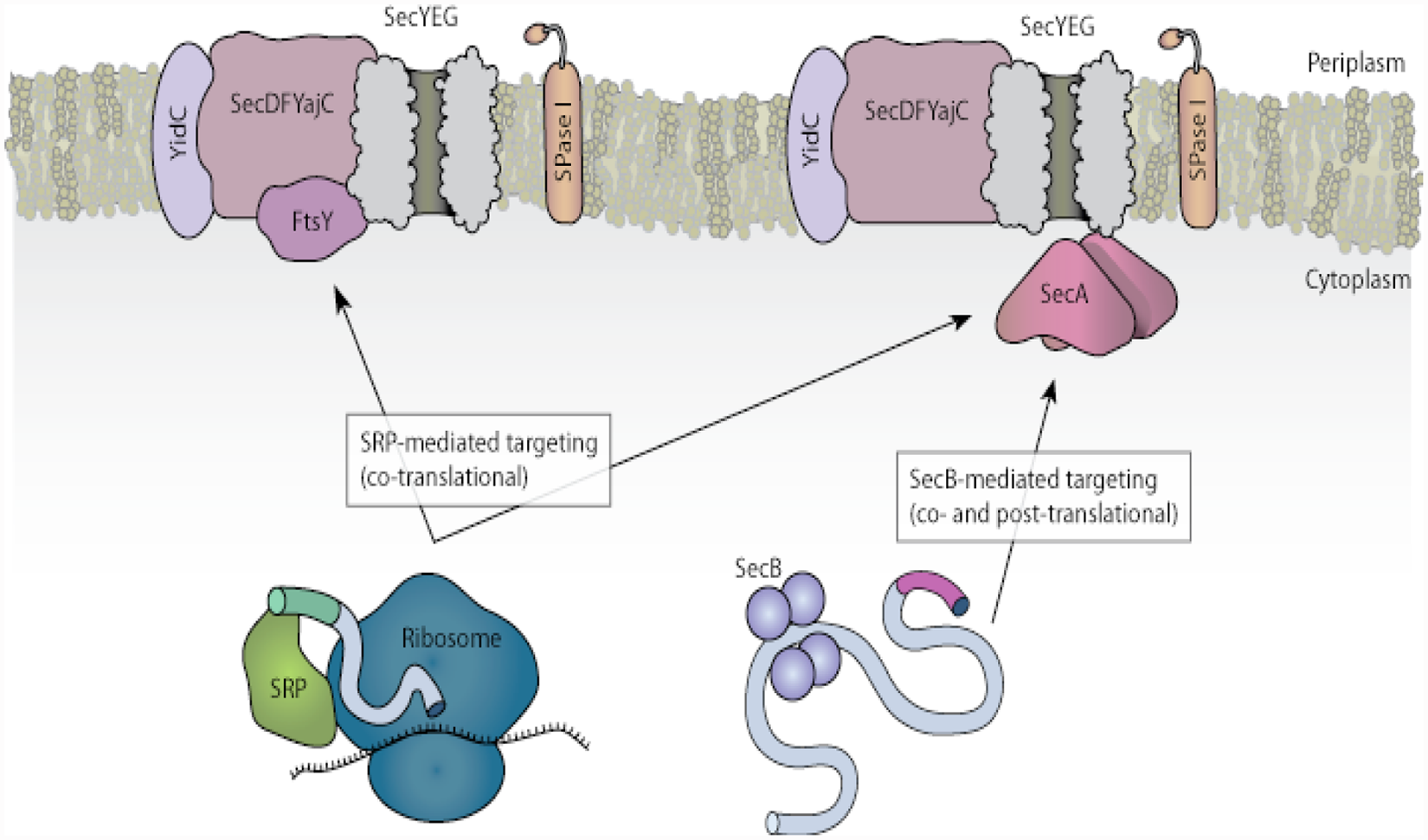
The Sec and signal recognition particle (SRP) pathways of protein translocation. Figure adapted from Papanikou et al. 1 by Stephen Nakazawa Hewitt. SPase, signal peptidase.
A recent review article 2 makes the case for the Sec pathway as a drug target in Mycobacterium tuberculosis, and similar justifications apply for gram-negative bacteria. 3 In brief, the Sec pathway is distinct from the targets of current drugs, is essential for bacterial growth and/or virulence, and exhibits dissimilarities in eukaryotes and bacteria that increase the likelihood of selectively inhibiting the microbial proteins. These dissimilarities include the localization of the eukaryotic channel (called Sec61) to the endoplasmic reticulum rather than the inner membrane and the mechanism of translocation through the channel, described as “ratcheting” in eukaryotes and “pushing” in bacteria. 4 Although the druggability of the pathway is uncertain, SecA’s crystal structure has been interpreted as containing druggable binding pockets. 3
The literature on experimental Sec-targeted screens5–10 indicates that there is considerable interest in this pathway from the perspective of bacterial drug development, but previous screens have not generally led to “developable” hits. For example, compounds identified as hits by Alksne et al. 5 had pleiotropic effects such as damage to the inner membrane, whereas a SecA inhibitor isolated from a natural product screen had very weak antibacterial activity. 7 A structure-based virtual screen for SecA inhibitors likewise did not identify superlative hits; potencies of the two best compounds, initially reported as Kis of 18 µM and 40 µM, 11 were later revised to IC50s of ~100 µM. 12 Furthermore, a medicinal chemistry effort to find SecA inhibitors through chemical synthesis rather than screening did not yield viable leads either, the most potent IC50s reported being 135 to 200 µM. 13
In an attempt to overcome such problems, we have adapted a mechanism-based whole-cell assay suitable for high-throughput screening (HTS) using an Escherichia coli strain that produces beta-galactosidase (β-gal) protein with a LamB signal sequence. The signal sequence causes SecB/SecA-mediated translocation of β-gal out of the cytoplasm, where the oxidizing environment renders it inactive. 14 Therefore β-gal activity in this strain remains low unless the Sec export pathway is disrupted. This approach offers several potential advantages. First, it assesses the function of the Sec pathway as a whole, so inhibitors of any important component of the pathway may be found. (This is in contrast to screens using a single protein such as SecA, which may or may not be druggable.) Second, it employs intact E. coli cells rather than an isolated protein in solution, so non-drug-like molecules unable to enter cells will not register as hits. Third, compounds that kill E. coli cells via mechanisms unrelated to protein export will not increase β-gal activity and will not register as hits. Finally, the readout of translocation is direct: If β-gal does not get exported, its activity increases. (This is in contrast to the more circuitous approaches of Alksne et al., 5 who assumed that any block in secretion would derepress SecA expression and thus increase the activity of a SecA-LacZ fusion protein, and Moir et al., 6 whose transgenic strain of Pseudomonas aeruginosa increases transcription of the luxCDABE operon and thus luminescence in response to SecA depletion.)
Here we report development of this β-gal-based assay of Sec pathway inhibition and assess its readiness for HTS.
Materials and Methods
The E. coli strains used here, MC4100 (lamB-lacZ) Hyb 42-1 and MC4100 (lamBΔ60-lacZ) Hyb 42-1, are gifts from the lab of Dr. Thomas Silhavy at Princeton University, which created them. We refer to these strains as EC626 and EC627, respectively. 15 In strain EC626, β-gal is exported to the periplasm via the Sec pathway, but in strain EC627, β-gal lacks an export motif and therefore stays in the cytoplasm. GC5, an E. coli strain that does not express β-gal, was obtained from GeneChoice (Frederick, MD).
Chemical compounds were generally purchased from Sigma-Aldrich (St. Louis, MO). The 96-well plates of drugs at clinically relevant concentrations were obtained from Trek Diagnostic Systems through the University of Washington Medical Center’s clinical microbiology lab. The gram-positive plate included the following drugs (with final concentrations in µg/mL): ampicillin (0.125–4), cefazolin (4–8), cefoxitin (3), ceftriaxone (0.5–16), clindamycin (0.125–1), daptomycin (0.25–2), doxycycline (0.5–4), erythromycin (0.125–2), gentamicin (2–4), imipenem (0.5–4), levofloxacin (0.25–2), linezolid (0.25–2), moxifloxacin (0.125–1), nitrofurantoin (16–32), oxacillin (0.125–1), penicillin (0.015–4), quinupristin/dalfopristin (0.125–1), rifampin (0.5–1), streptomycin (500), tetracycline (1–4), tigecycline (0.06–0.5), trimethoprim/sulfamethoxazole (0.5/9.5–2/38), and vancomycin (0.25–8). The gram-negative plate included the following drugs: amikacin (8–16), ampicillin (4–8), ampicillin/sulbactam (4/2–8/4), aztreonam (0.5–4), cefazolin (1–8), cefepime (0.125–8), cefotaxime (0.125–1), cefotetan (8–16), ceftazidime (0.25–8), ceftriaxone (0.125–1), ciprofloxacin (0.03–1), doxycycline (0.5–4), ertapenem (0.06–0.5), gentamicin (2–4), imipenem (0.25–4), levofloxacin (0.25–2), meropenem (0.5–4), minocycline (0.5–4), moxifloxacin (0.125–1), nitrofurantoin (16–32), piperacillin/tazobactam (4/2–32/2), ticarcillin/clavulinic acid (8/1–32/1), tigecycline (0.125–1), tobramycin (1–4), and trimethoprim/sulfamethoxazole (1/19.5–2/38). The 96-well plates of compounds from the MicroSource Discovery Systems “Spectrum Collection” were provided by Zhongsheng Zhang and Erkang Fan of the University of Washington Department of Biochemistry. The plate reader used was an ELx800 instrument made by BioTek Instruments (Winooski, VT).
Our previous assay for inhibition of the Sec pathway 15 was suboptimal for screening in that it used three microtiter plates: one for inducing β-gal expression, a second for permeabilizing the cells, and a third for assaying β-gal activity. Following the precedent of Alksne et al., 5 we now perform these steps in a single plate as follows. Strain EC626 is grown overnight at 37 °C with vigorous aeration in LB media with 0.2% glucose, which suppresses lamB/lacZ expression. The stationary-phase cell culture is twice centrifuged and resuspended in a volume of phosphate-buffered saline equal to the culture volume. For a 96-well-plate assay, 25 to 50 µL of the cell suspension is added to each well along with an equal volume of LB media containing the compound to be tested, for a total volume of 50 to 100 µL per well. The plate is incubated at 37 °C for 60 min. Then, 12.5 to 25 µL of “ZOB” buffer, 5 which contains the detergents hexadecyltrimethylammonium bromide (CTAB) and sodium deoxycholate to permeabilize cells and the β-gal substrate ortho-nitrophenyl-beta-galactoside (ONPG) to assess β-gal activity, is added. After 40 min at room temperature, 12.5 to 25 µL of 8 M urea with 1 M sodium carbonate is added to enhance color development, and absorbance is read at 405 nm to gauge the production of ortho-nitrophenol. Increases in A405 should reflect increased cytoplasmic β-gal activity presumably due to inhibition of β-gal export.
Note that compounds with a yellow color could increase A405 independently of an effect on β-gal. To determine whether these compounds merely increase baseline absorbance, they can be tested with 37 °C incubation periods of both 0 min and 60 min. Alternatively, these compounds could simply be removed from consideration before or after screening.
Results and Discussion
A405 Reflects Activity of Nonexported β-gal
Previous studies of EC626 and EC62714,16 have shown that maltose induces β-gal production in both strains and that this induction jams the SecYEG channel in EC626, causing cytoplasmic β-gal accumulation. We confirmed this behavior in our assay ( Fig. 2 ). As expected, β-gal activity in EC627 exceeds that of EC626 in the absence of maltose because EC626 exports β-gal to the periplasm, where it is inactive. However, 0.01% maltose increases β-gal levels beyond what can be exported in EC626, leading to a buildup of cytoplasmic β-gal. GC5 cells, which do not make β-gal, show only a baseline level of A405 whether maltose is present or not, providing further evidence that increases in A405 specifically reflect β-gal activity. In addition, the effect on β-gal is specific to maltose, as dextrose, fructose, and galactose do not increase A405 (data not shown).
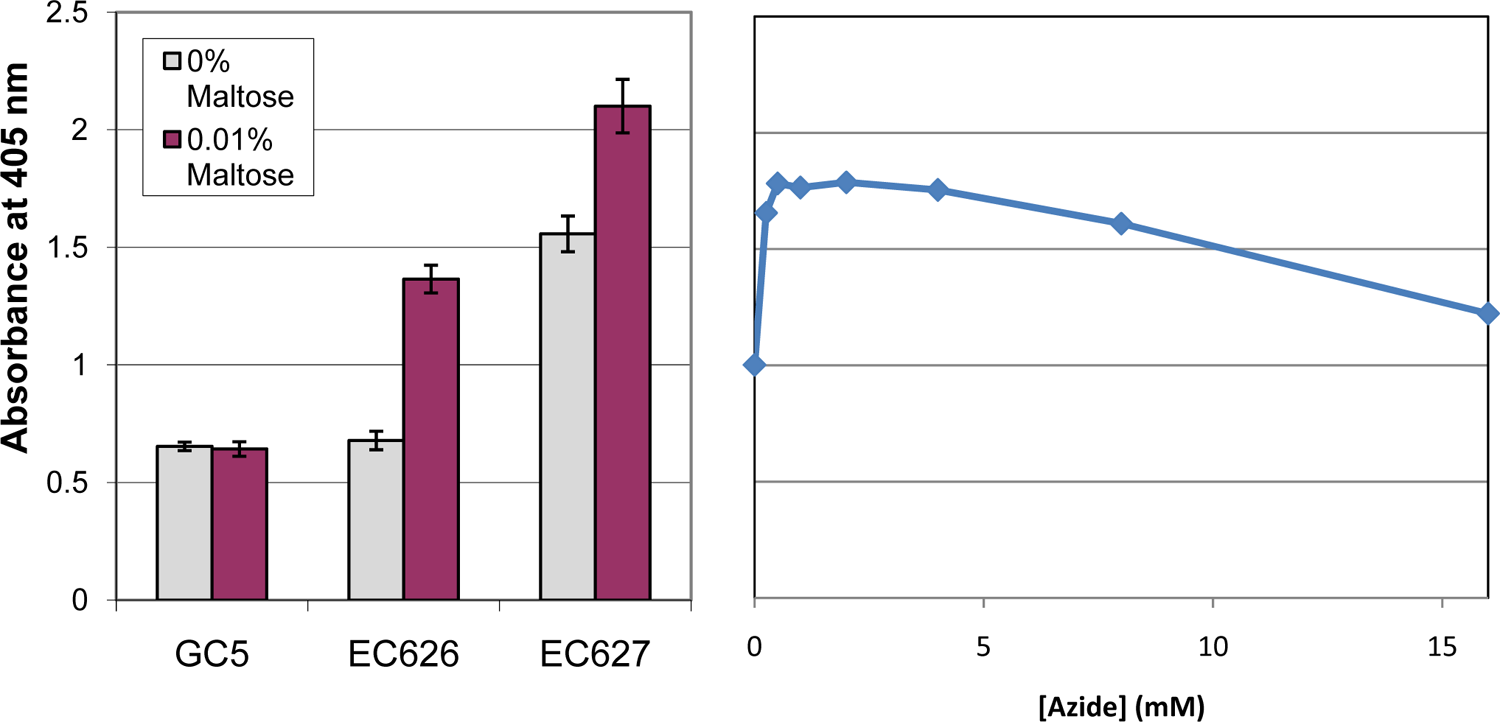
Escherichia coli strains respond as expected to maltose and azide in an assay of Sec pathway inhibition. Absorbance at 405 nm increases when β-galactosidase converts ortho-nitrophenyl-beta-galactoside (ONPG) to ortho-nitrophenol. Left: A representative experiment with E. coli strains GC5 (does not produce β-gal), EC626 (exports β-gal), and EC627 (does not export β-gal). Error bars here and elsewhere indicate standard deviations. Right: A405 increases in EC626 cells in the presence of sodium azide, consistent with azide’s inhibition of SecA and thus the Sec pathway.
The known SecA inhibitor azide was studied in a separate type of control experiment. As expected, azide concentrations ≥0.25 mM led to increased β-gal activity in EC626 ( Fig. 2 ), consistent with blockage of β-gal export due to inhibition of SecA. However, because results obtained with azide were less consistent than those obtained with maltose (possibly due to azide’s effects on other targets such as other ATPases and the electron transport chain), we adopted maltose as our primary control compound.
The maltose-induced accumulation of cytoplasmic β-gal in EC626 was studied further in dose-response experiments. Although previous studies had included β-mercaptoethanol in assay buffers, presumably to maintain cytoplasmic β-gal in a reduced and active state, 14 it did not appear necessary to preserve β-gal function under our assay conditions; EC626’s response to maltose was similar with and without β-mercaptoethanol ( Fig. 3 ). Thus, in performing this assay, the unpleasant odor of β-mercaptoethanol may be avoided.
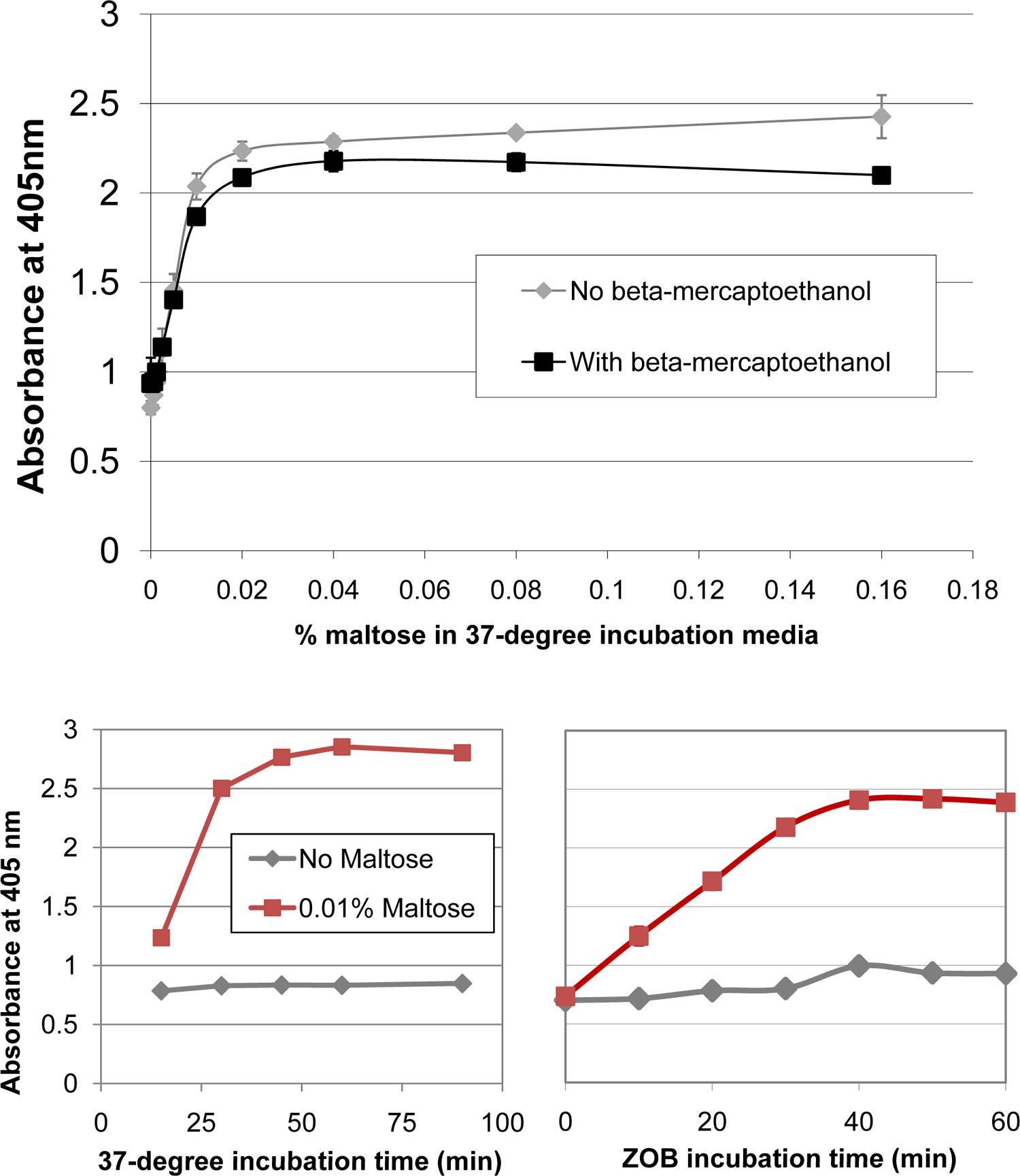
Optimization of assay conditions. Top: Accumulation of cytoplasmic LacZ in EC626 cells in response to various levels of maltose. Under our assay conditions, β-mercaptoethanol appears unnecessary to maintain β-gal function. Bottom: Effect of 37 °C incubation time (left) and ZOB incubation time (right) on assay readout. Although A405 depends on the length of these incubation periods, a good separation between the maltose and no-maltose conditions is evident over a range of times.
Sensitivity of Results to Variations in Assay Protocol
Assays suitable for HTS should be tolerant of small variations in parameters such as incubation time, which may vary somewhat when many plates are handled simultaneously. Therefore, we explored the assay’s sensitivity to assay conditions by systematically varying several of its parameters. Figure 3 shows that, although our default incubation times of 60 min at 37 °C and 40 min following addition of ZOB buffer are fine, there are many sets of times for which Sec-blocked (maltose-induced) cells can easily be distinguished from unblocked cells. The incubation time following addition of sodium carbonate and urea did not affect A405 over a range of 0 to 18 h (data not shown). We also asked whether our results are affected by the duration of cells’ time in stationary phase (following overnight growth) prior to the assay. To explore this issue, we let cells grow for a few hours before making 10-fold serial dilutions and letting growth continue overnight, thus creating cultures that would reach stationary phase at different times before the following day’s assay. Our data (not shown) show that Sec-blocked cells are easily distinguished from unblocked cells over a wide range of dilutions, suggesting that the results are relatively independent of the preassay time spent in stationary phase. Likewise, our results are not strongly dependent on the glucose concentration in the overnight growth medium; 0.05%, 0.1%, and 0.2% all work well (data not shown).
Although most of our assays have been done in 96-well plates, good results are also obtained with 384-well plates (data not shown).
Effect of Current Antibacterial Drugs on the Sec Pathway
The idea that the Sec pathway could be a novel drug target assumes that it is not a primary target of current drugs. To test this assumption, we performed our assay with plates of predispensed drugs: one for gram-positive bacteria and another for gram-negative bacteria (see Materials and Methods). Each plate was assayed three times; no drug consistently elevated A405 above baseline at any of the concentrations tested ( Fig. 4 and data not shown). To confirm that these negative results do not reflect problems delivering the drugs to the cells, we also tested these same plates with strain EC627, which constitutively makes cytosolic β-gal. We hypothesized that inhibitors of DNA gyrase and ribosomes, respectively involved in transcription and translation, would suppress β-gal production and thus reduce absorbance at 405 nm, whereas other drugs would have no effect. This hypothesis was shown to be partially correct in three replicates of each plate. All DNA gyrase inhibitors tested—moxifloxacin (0.12–1 µg/mL), ciprofloxacin (0.03–1 µg/mL), and levofloxacin (0.25–4 µg/mL)—did indeed suppress β-gal activity in EC627 to some extent (as indicated by A405’s more than three standard deviations below the baseline of drug-free cells), but results for drugs with alternative mechanisms of action were not uniform. β-Gal suppression did occur consistently with nitrofurantoin (16–32 µg/mL) and with the ribosome inhibitors minocycline and doxycycline (0.5–4 µg/mL). These data, although somewhat surprising in the details, suggest overall that known effects of the plated drugs can be seen and, therefore, that their apparent avoidance of the Sec pathway is genuine rather than reflecting problems with degradation or sensitivity.
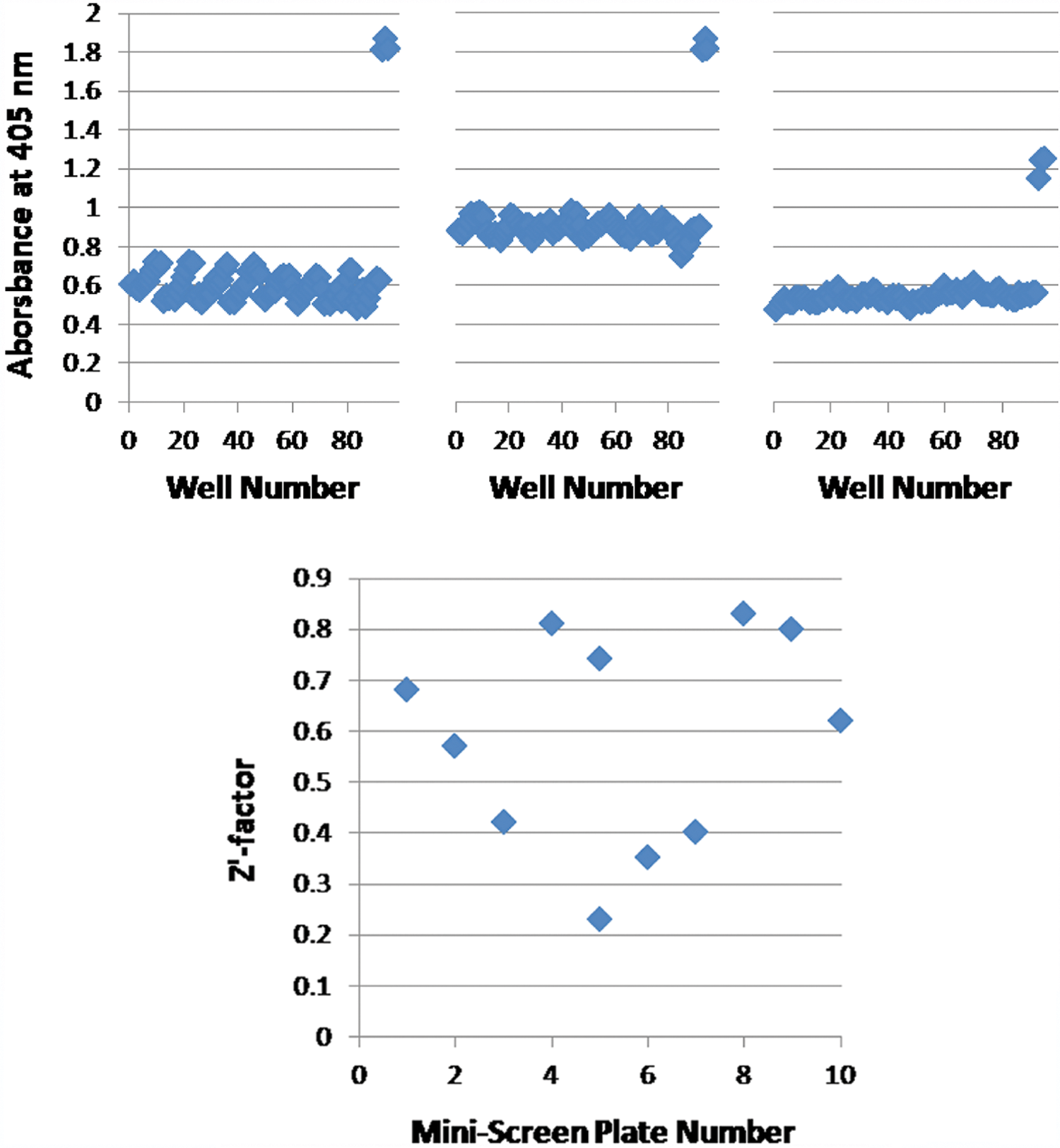
Screening of bioactive compounds with a Sec pathway assay. Top: Response of EC626 cells to drugs relevant to gram-negative bacteria in three replicate experiments run on separate days. The high-absorbance wells are controls with 0.01% maltose. Bottom: Z′ factors for a 10-plate mini-screen of 800 compounds from the MicroSource Discovery Systems “Spectrum Collection.” Plate 5 was run twice because poor results were obtained the first time.
Mini-Screen of 800 Compounds
As a further test of our assay’s readiness for HTS, we performed a small-scale screen of 800 small molecules taken from a commercial collection of structurally diverse, biologically active compounds. These compounds were tested at a final concentration of 100 µM in 96-well plates; 0.01% maltose and maltose-free media were used as controls in each plate. Although no reproducible hits were obtained, comparison of the maltose-exposed and maltose-free cells yielded an average Z′ factor of 0.60 ( Fig. 4 ). These Z′ factors seem good, especially considering that all materials were pipetted by hand, whereas HTS facilities generally use robotics to reduce variations in pipetting.
Our Sec Pathway Assay: Features and Follow-Up
The screening approach presented here falls into the general category of mechanism-based whole-cell screens, which “combine a targeted approach with a biologically integrated context.” 17 The focus on a particular pathway as a target (the Sec pathway, in our case) ensures that initial hits can be scrutinized carefully with respect to mechanism of action and can be developed relatively quickly through medicinal chemistry and structural biology. The assay’s cellular context ensures that compounds identified as hits can disrupt the target in vivo (avoiding compounds that are unsuitable as leads because they cannot permeate cells, for example) and permits searches for inhibitors of an entire pathway (rather than targeting a single protein whose druggability may be uncertain). Previous applications of this hybrid strategy to bacterial screening have resulted in the discovery of inhibitors of the cytokinesis protein FtsZ and the fatty acid biosynthesis enzymes FabF/B, for example. 17
Our screening strategy, like that of Moir et al., 6 is unusual in that hit compounds generate a positive signal (a yellow color corresponding to an increased A405) rather than extinguishing a signal (as in most bacterial growth assays). This is both advantageous and disadvantageous in certain respects. As discussed above, compounds that kill cells through mechanisms unrelated to the Sec pathway will not increase A405 and thus will not distract from the goal of finding specific modulators of the Sec pathway. On the other hand, if a compound does act through the Sec pathway but is screened at a concentration that kills cells within the 1-h incubation period, new β-gal will not be produced, and the Sec-inhibiting activity of the compound will not be detected. Because a bona fide Sec pathway inhibitor may not register as a hit if its concentration is either too low or too high, the choice of compound concentration at which to screen may be especially important for this assay. Indeed, if resources permit, screening at two or three different concentrations might be advisable.
If Sec pathway inhibitors are indeed discovered via HTS, follow-up work will likely test whether these inhibitors are effective in killing a variety of pathogenic bacteria beyond E. coli. The conservation of the Sec pathway among all known bacteria means that broad-spectrum activity is plausible; however, interspecies differences in compounds’ penetrance and degradation, along with possible differences in the redundancy of export machinery, may result in differences in activity. It is now recognized that narrow-spectrum antibiotics, although not as commercially attractive to pharmaceutical companies, may still have significant clinical value and should also be developed if possible. 17 Follow-up work on any HTS hits will also attempt to identify the specific component of the Sec pathway that is inhibited. This target identification may require a combination of genetic and biochemical approaches, as in previous work. 17
In conclusion, this article presents a novel strategy to test for inhibitors of the Sec pathway of protein translocation, along with data showing the feasibility of applying this strategy to HTS. We have developed an assay in which inhibition of protein translocation leads to an accumulation of β-gal in the cytoplasm of EC626, an E. coli strain engineered to export β-gal through the Sec pathway. Maltose may serve as a control “inhibitor” of the pathway through its induction of β-gal expression, which leads to jamming of the SecYEG channel.
Footnotes
Acknowledgements
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
