Abstract
The insulin-like growth factor I receptor (IGF1-R) system has long been implicated in cancer and is a promising target for tumor therapy. Besides in vitro screening assays, the discovery of specific inhibitors against IGF-1R requires relevant cellular models, ideally applicable to both in vitro and in vivo studies. With this aim in mind, the authors generated an inducible cell line using the tetracycline-responsive gene expression system to mimic the effects of therapeutic inhibition of the IGF-1R both in vitro and on established tumors in vivo. Inducible overexpression of IGF-1R in murine embryonic fibroblasts was achieved and resulted in the transformation of the cells as verified by their ability to grow in soft agar and in nude mice. Continuous repression of exogenous IGF-1R expression completely prevented outgrowth of the tumors. Furthermore, induced repression of IGF-1R expression in established tumors resulted in regression of the tumors. Interestingly, however, IGF-1R–independent relapse of tumor growth was observed upon prolonged IGF-1R repression. The IGF-1R cell line generated using this approach was successfully employed to test reference small-molecule inhibitors in vitro and an IGF-1R–specific inhibitory antibody, EM164, in vivo. Besides efficacy as a read-out, phospho-AKT could be identified as a pharmacodynamic biomarker, establishing this cell line as a valuable tool for the preclinical development of IGF-1R inhibitors.
Deregulation of the insulin-like growth factor I receptor (IGF-1R) has long been linked to tumorigenesis. Early experiments demonstrated that overexpression of IGF-1R in mouse embryo fibroblasts (MEFs) resulted in cell transformation. 1 Transformation occurred in a dose-dependent manner, as shown in cells deleted for their endogenous IGF-1R copy. 2 Activation of the IGF-1R mediates cell proliferation and survival but also transformation, angiogenesis, and metastasis.3,4 The receptor is activated by its ligands IGF-1 and -2, which result in receptor autophosphorylation and the stimulation of downstream signaling cascades including the IRS-1/PI-3K/PKB/S6K and Grb2/Sos/Ras/MAPK pathways. Interference with its activity by either gene disruption, 5 antisense oligonucleotides, 6 dominant negative mutants,7,8 neutralizing antibodies,9,10 or small-molecule inhibitors 11 was found to interfere with cell growth and proliferation. Thus, the IGF-1R is a promising drug target for the development of novel anticancer drugs. 12
The discovery of new IGF-1R–targeting compounds requires suitable model systems, both for initial screens and the further characterization of potential lead compounds. In vitro enzyme assays, which are fast, reliable, and comparably easy to set up, may serve for the initial hit generation. However, to characterize compound selectivity and potency and to allow for the measurement of efficacy and identification of potential biomarkers, cellular and in vivo models are required that are much more complex to establish. Cellular models for transforming receptor tyrosine kinases (RTKs) may readily be generated via stable transfection of the corresponding cDNA into untransformed cell lines (e.g., mouse fibroblasts) or using tumor cell lines that endogenously express the gene. However, in either case, it is difficult to prove that the given RTK is indeed the single transforming principle. Furthermore, when used for animal tumor models, it is usually impossible to distinguish between a requirement of the RTK for original cellular transformation or tumor maintenance. Stable transformation of a given gene may alter the general gene expression pattern of the cell leading to the expression of other oncogenes. The situation is even more complex in cells that express the gene of interest endogenously.
We therefore chose to establish an inducible cell system based on the tet-repressor that allows for reversible expression of a given gene. 13 This approach has been successfully used for the ErbB2 receptor.14,15 Thus, here we describe the generation of an inducible cellular model system for the IGF-1R receptor using an MEF cell line harboring a plasmid encoding the tet-repressor. The resulting cell line was tested for IGF-1 responsiveness in vitro and for transformation via anchorage-independent growth in soft agar and tumor growth in vivo. Finally, the established cell line was used to characterize the effect of known and putative IGF-1R inhibitors in vitro as well as in vivo and the identification of phospho-AKT as a potential downstream biomarker.
Materials and Methods
Cell Lines and Antibodies
The cell line expressing the anti-myc–specific monoclonal antibody 9E10 was purchased from Developmental Studies Hybridoma Bank (Iowa City, IA). The anti-IGF-1R β (C-20; sc-713), PY99 (sc-7020B), and anti-AKT1 (C-20) antibodies were obtained from Santa Cruz Biotechnology (Santa Cruz, CA). The antiphospho-AKT antibody (9275) was from Cell Signaling Technologies (New England Biolabs, Hitchin, Herts, UK). MEFTOFF (Clontech, Heidelberg, Germany) were kept in DMEM (61965-026; Life Technologies, Darmstadt, Germany) supplemented with 10% FCS/penicillin/streptomycin at 37 °C/10% CO2.
Generation of an MEF Cell Line Inducibly Expressing the IGF-1R
The cDNA encoding wild-type human IGF-1R, supplemented with a C-terminal 2myc6His tag, was inserted into pTRE (Clontech), and that construct was co-transfected with the plasmid pSV Hygro into the tet-off MEF (MEFTOFF) cell line. Successfully transfected cells were selected using 200 µg/mL hygromycin.
FACS/Western/Phospho-ELISA Analysis
For the fluorescence-activated cell sorting (FACS)/Western/phospho-enzyme-linked immunoadsorbent assay (ELISA) measurement, cells kept in the presence or absence of doxycycline for >48 h were plated at 750 000 cells/well into six 10-cm dishes each, keeping the respective doxycycline status. Twenty-four hours after plating, five plates were washed twice with phosphate-buffered saline (PBS), and the medium was replaced with DMEM containing 0.1% FCS only. Another 24 h later, 0, 5, 50, 100, and 200 ng IGF-1 (I3769, Sigma, Hamburg, Germany) was added, and the cells were incubated for an additional 24 h. On the following day, the medium supernatant was removed, the plates were washed with PBS, and the cells trypsinized and combined with the medium supernatant. The cells were collected by centrifugation and resuspended in 500 mL PBS. Two hundred fifty microliters of the cell suspension was fixed with methanol, rehydrated in PBS, stained with 25 µg/mL propidium iodide in PBS containing 100 µg/mL RNAse H, and analysed in a FACSCalibur (Becton Dickenson, Heidelberg, Germany). The remainder of the cells was pelleted and lysed in buffer A (20 mM TRIS pH 8/135 mM NaCl/1% NP-40/10% glycerol/1.5 mM sodium vanadate) and 1 tablet complete protease inhibitors (Roche, Mannheim, Germany). The protein content was determined using the Bradford method, 16 and the lysates were used for ELISA and Western blot analysis. For the ELISA, 100 µg protein was applied into 96-well plates precoated with anti-myc (9E10) antibody. PY-99 antibody was used to detect phosphorylated IGF-1R. For the Western blots, 30 µg of protein was blotted onto nitrocellulose membranes and probed with anti-myc (9E10), anti–IGF-1R, anti-AKT1, and a phospho-AKT antibody (all at 1:200).
Soft Agar and Immunofluorescence Assays
A soft agar assay as well as immunofluorescence detection of IGF-1R were performed using cells that had been grown for >48 h in the presence or absence of doxycycline. For soft agar assays, the cells were embedded in 0.4% agarose containing DMEM, FCS, keeping the respective doxycycline status, and plated onto a layer prepared of 0.8% agarose. For immunofluorescence analysis, the cells were plated onto cover slips, fixed in 4% paraformaldehyde, permeabilized, and the IGF-1R was detected using the 9E10 antibody followed by an FITC-labeled secondary anti-mouse antibody. DAPI was used as a nuclear counterstain.
Cellular Phosphorylation Assays
Cells kept for >48 h in the presence or absence of doxycycline were seeded in 48-well plates. After serum starvation for 18 h, the serially diluted compounds (in DMSO, final concentration of DMSO: 1%) were added for 90 min followed by IGF-1 for 10 min. The cells were then lysed and the ELISA was performed as described above.
Animal Experiments
All animal experiments were performed according to German Animal License Regulations (Tierschutzgesetz) identical to UKCCCR Guidelines for the welfare of animals in experimental neoplasia. NMRI nude mice were obtained from Harlan Winkelmann GmbH, Germany.
The injection of 4 × 107 cells in 0.2 mL aliquots into the subcutaneous space of the left flank was performed using a 29G needle syringe. Volumes of primary tumors were taken macroscopically by calipering and multiplying the distances of all three dimensions. Therapy or doxycycline treatment (2 mg/mL in drinking water containing 1% sucrose) started after the primary tumor had reached a volume of about 200 mm3, or as indicated in the figure legend. During the course of the study, animal weights were measured three times per week. Animal behavior was monitored daily. The criteria to evaluate the antitumor activity of the IGF-1R antibody were tumor regressions:
Complete regression (CR) = regression below the limit of palpation (<63 mm)
Partial regression (PR) = regression greater than 50% reduction in tumor mass (CR was included in the PR)
For BrdU labeling of sections, the animals were injected intraperitoneally with 100 to 200 µL of a 10 mg/ml solution of BrdU in sterile 1X DPBS (BD Pharmingen, Heidelberg, Germany) 24 h before necropsy.
IGF-1R Antibody EM164
The murine anti–IGF-1R monoclonal antibody, EM164,17,18 was prepared by Immunogen Inc. (Waltham, MA) and provided by Sanofi (94400 Vitry-sur-Seine, France). EM164 was formulated in PBS without Ca2+ and Mg2+ and administered intraperitoneally twice a week.
IGF-1R Protein Expression in MEF/IGF-1R Tumor Samples
Tumors were cut into small pieces and then grinded mechanically by bead beating in a Qiagen Mill Mixer MM301 in ice-cold lysis buffer (1% Triton X-100, 100 mM NaCl, 10 mM Tris-HCl [pH 7.5], 1 mM EDTA [pH 8], 1 mM EGTA [pH 8], 1 mM NaF, 20 mM Na4P2O7, 1 mM Na3VO4, 10% glycerol, and one tablet protease inhibitors; Roche Diagnostics, Mannheim, Germany). After a 2-h incubation at 4 °C, the tumor lysates were centrifuged at 16 000 g for 15 min at 4 °C, and the supernatants were transferred to fresh tubes on ice. The total protein concentration was determined using a Bradford assay 16 according to the manufacturer’s instructions (BIO-RAD protein assay). The lysates were stored at −80 °C until analysis. IGF-1R expression and AKT phosphorylation in tumor lysates were determined using a commercially available ELISA assay (DY391; R&D Systems, Lille CEDEX, France) according to the manufacturer’s instructions.
Immunohistochemistry
Seven-micrometer cryosections were prepared from snap-frozen tumors and fixed using 4% paraformaldehyde. Endogenous peroxidases were blocked using 3% H2O2 in methanol. Exogenous and total IGF-1R were detected using a biotinylated 9E10 antibody followed by a streptavidin-horseradish peroxidase (PO) conjugate or a rabbit anti–IGF-1R β antibody followed by a secondary PO-labeled anti-rabbit antibody, respectively. TUNEL and BrdU stainings were done according to manufacturers’ instructions (Roche and BD Pharmingen, respectively).
Results
Generation of a Tet-Repressable IGF-1R MEF Cell Line
The insulin-like growth factor receptor has been a promising cancer drug target for more than a decade. However, although small-molecule inhibitors as well as antibodies against IGF-1R are in clinical development, to date no therapy against IGF-1R has been approved.
The discovery of novel molecules requires relevant testing systems. So to generate a tool for the cellular and in vivo testing of IGF-1R antagonists, we planned to generate a mouse fibroblast cell line with an inducible IGF-1R allele. An MEF cell line expressing the Tet-Off transactivator protein was transfected with a vector carrying the full-length human IGF-1R cDNA under the control of a tet-sensitive minimal CMV promoter. 13 After selection and clonal expansion, cell lines were tested for tet-repressable expression of the IGF-1R target.
Cell clone 28 showed robust expression of the receptor in the absence and complete suppression in the presence of the repressor doxycycline. Expression of the IGF-1R transgene is detectable 24 h after removal of the repressor (
IGF-1R activation induces autophosphorylation of tyrosine residues on the C-terminal cytoplasmic tail of the kinase, which triggers the binding of signaling molecules that mediate, among others, proliferation and protection from apoptosis.19,20
In the presence of 10% FCS, the growth behavior of the cells was independent of the presence of the IGF-1R. However, upon serum starvation and subsequent supplementation with increasing amounts of IGF-1, a stimulation of cellular proliferation and a concomitant reduction of apoptosis were observed, dependent on IGF-1R expression (
The parental MEF cell line is nontumorigenic in nude mice, but the IGF-1R has been reported to transform mouse fibroblasts. As a test for anchorage-independent growth, a hallmark of cellular transformation, we first checked whether the expression of the IGF-1R enables the cell clone 28 to grow in soft agar. Because colonies were detectable, albeit few (data not shown), the cells were next implanted into nude mice and their growth monitored over time. Growth was very slow in the beginning, but exponential growth was observed after day 40 (
However, despite these promising results, the slow growth of the cells in the mouse model and the resulting rather large tumor size differences precluded the use of this cell line in a robust animal model.
Generation of Subclones 28.1 and 28.3 with Improved Growth Characteristics
Picking cell clones that grow in soft agar is one way to isolate cells with better growth characteristics in a setting selective for transformed cells. Subclones from clone 28 were expanded after isolation from soft agar, and two clones, 28.1 and 28.3, were characterized as described in the previous section.
Western blot analysis demonstrated that both subclones retained their doxycycline responsiveness, with similar kinetics as clone 28 ( Fig. 1 ). In the presence of 10% FCS, IGF-1R expression did not affect proliferation characteristics of the cell lines, although a slight decrease of apoptotic cells was apparent ( Fig. 2A , B ). However, serum starvation induced massive apoptosis and some reduction of the number of proliferating cells. Addition of IGF-1 to the cells kept in the absence of doxycycline fully reversed this phenotype. Higher IGF-1 concentrations (100 and 200 ng/mL) stimulated proliferation above even the levels found in the control grown in 10% FCS, whereas apoptosis levels dropped below ( Fig. 2A , B ). No (28.1) or a much less pronounced (28.3) IGF-1–induced effect was observed if the cells had been kept in the presence of doxycycline ( Fig. 2A , B ). Levels of IGF-1 that induced cellular proliferation and rescued cells from apoptosis also led to high levels of receptor tyrosine kinase autophosphorylation ( Fig. 2C ).
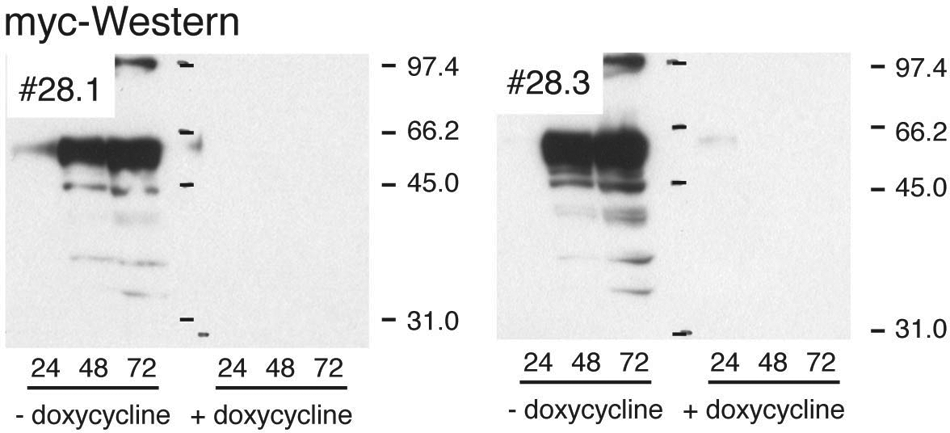
Insulin-like growth factor I receptor (IGF1-R) Western blot analysis of two sublines, 28.1/3, generated from clone 28. (
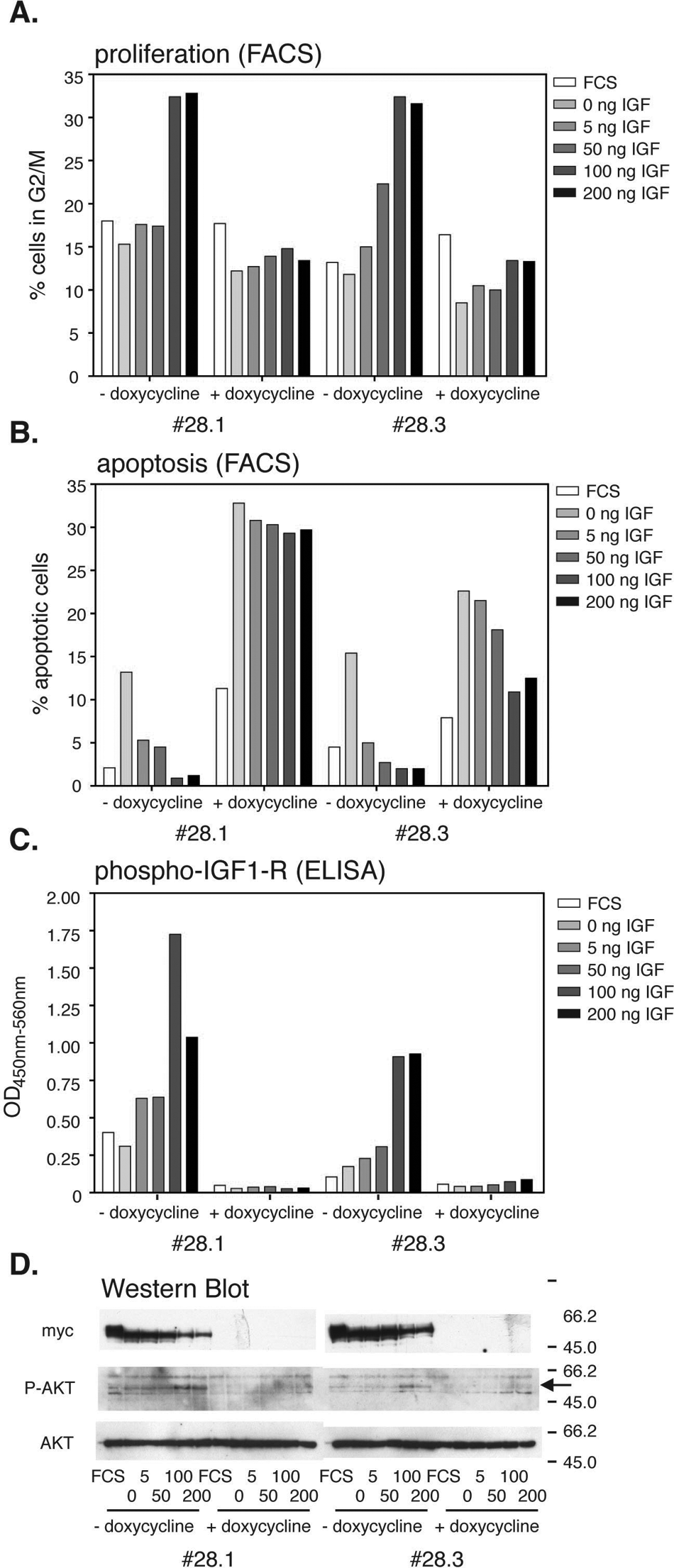
Fluorescence-activated cell sorting (FACS), enzyme-linked immunoadsorbent assay (ELISA), and Western blot analysis of insulin-like growth factor I receptor (IGF-1R). Cells from the IGF-1R expressing lines 28.1 and 28.3 were grown in the presence or absence of doxycycline and serum starved for 24 h before increasing amounts of IGF-1 were added to the medium. Forty-eight hours later, cells were either fixed and stained for cell cycle FACS analysis or lysed for Western blot analysis. (
Western blot analysis confirmed the absence of the IGF-1R in the doxycycline-treated cells ( Fig. 2D ). Phosphorylation of AKT-1, as a downstream target of the IGF-1R, and a potential pharmacodynamic marker for IGF1-R inhibition, could be detected in cells treated with the two highest IGF-1 concentrations. Total AKT levels served as a loading control ( Fig. 2D , bottom panel).
Immunofluorescence analysis was used to verify the expression of IGF-1R on a single cell level. Most cells expressed the transgene at easily detectable levels, with the highest levels apparently in cells piling on top of each other. Addition of doxycycline completely abolished the signal ( Fig. 3A ). Finally, the cells were checked for their ability to grow anchorage independently. In contrast to the parental clone 28, both subclones formed large colonies in soft agar ( Fig. 3B ).
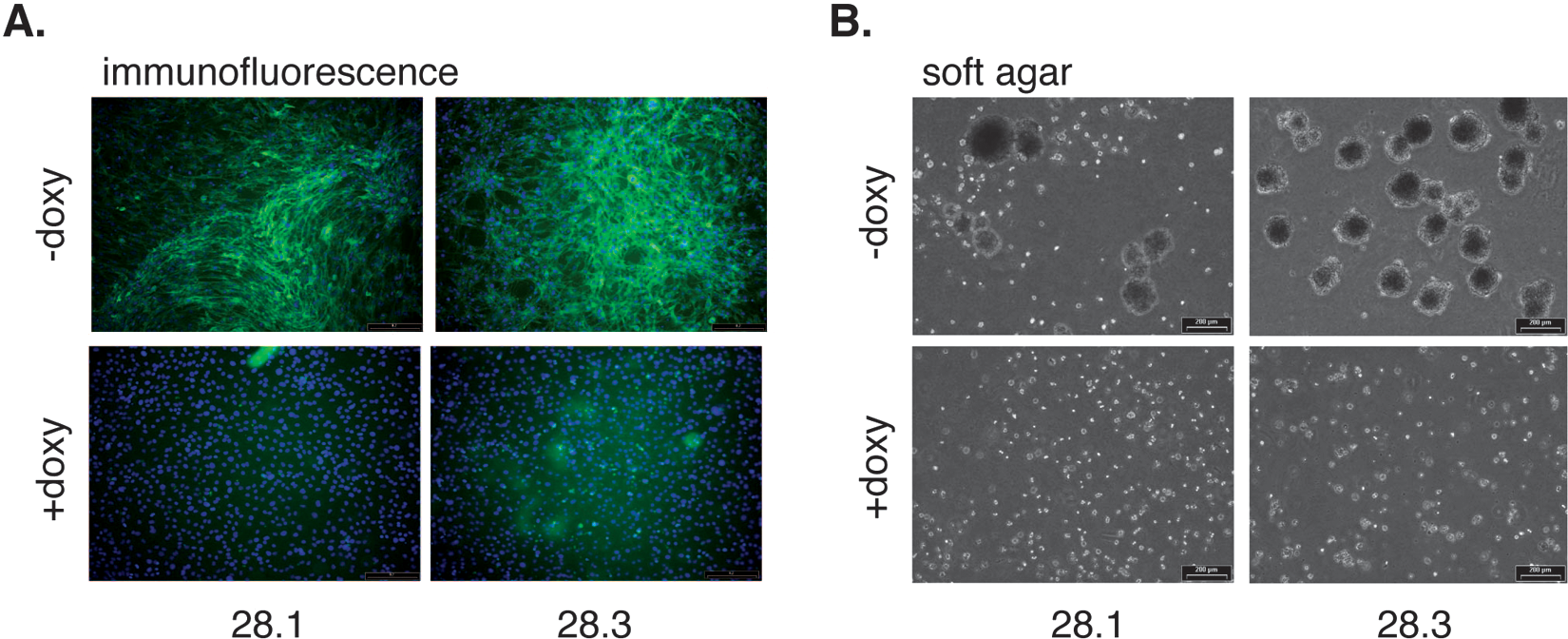
Immunofluorescence and soft agar assays. (
Next, growth of subclones 28.1 and 28.3 was tested in nude mice.
Reversible Growth In Vivo of Subclones 28.1 and 28.3
Cells from both subclones were implanted into the left flanks of a total of nine mice each, along with a negative control using the original, nontransformed MEF cell line. Three mice bearing 28.1 or 28.3 cells, respectively, received doxycycline directly after implantation, as a control for the IGF-1R dependency of the tumor growth. Of the other six animals, three animals received doxycycline as an anti–IGF-1R treatment after about 21 d, when the tumors reached an average size of about 200 to 300 mm3 (compared with parental clone 28: 40 d;
Tumors from untreated mice reached a size of about 2000 mm3 after 45 d (3/3; Fig. 4A , left panel, black line). Doxycycline treatment of tumors at a size of 200 to 300 mm3 induced regression, and tumors were undetectable after 12 d ( Fig. 4A , dark gray line). However, when doxycycline was removed, tumors relapsed and exponential growth was observed after another 3 to 4 wk (3/3, both cell lines; Fig. 4A , dark gray line). If, after 5 wk, doxycycline was removed from animals that had been treated from day 0, two of three animals bearing the 28.1 cell line developed a tumor (28.3: 0/3; Fig. 4A , left panel, light gray line). Last but not least, tumors that had been treated with doxycycline at a late stage (>1000 mm3) also regressed ( Fig. 4A , left panel, light gray line; right panel, black line). But, after about 8 d, relapse was observed even in the presence of doxycycline ( Fig. 4A , right panel, black line).
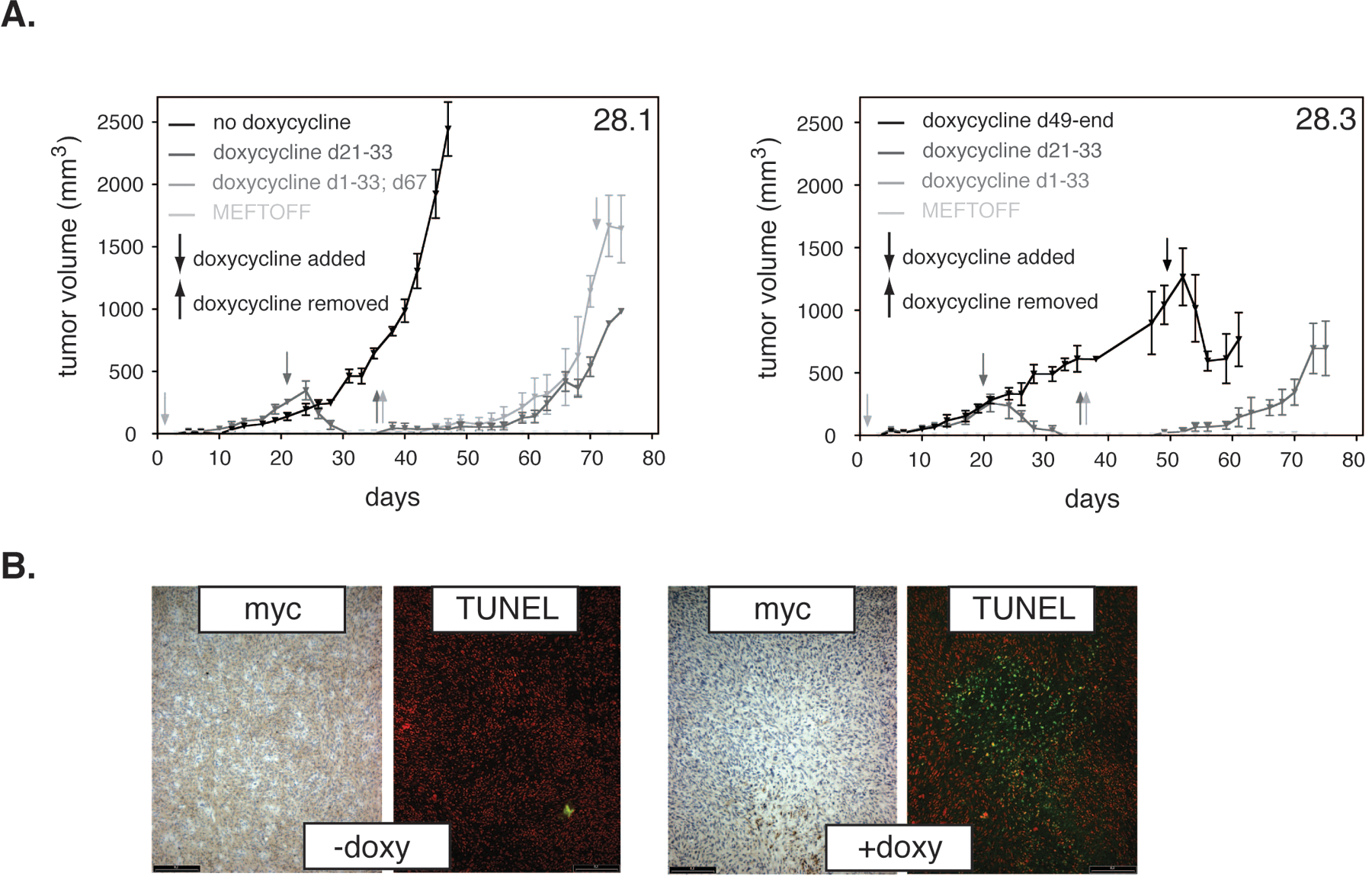
In vivo growth of the sublines 28.1/28.3 and growth inhibition by doxycycline. (
To confirm that doxycycline treatment shut down IGF-1R expression in vivo, tissue sections were prepared from tumors of the 28.1-untreated control group ( Fig. 2A , left panel, black line) and the late-stage treated group ( Fig. 4A , left panel, light gray line). Besides the exogenous IGF-1R receptor, which was identified by its myc-tag, a potential induction of apoptosis as a consequence of IGF-1R repression was analyzed via TUNEL staining.
Doxycycline treatment completely abolished expression of IGF-1R in vivo. Whereas a robust, relatively homogenous staining was obtained from sections of untreated tumors, doxycycline treatment reduced the signal from the myc-tagged transgene to undetectable levels ( Fig. 4B ). Furthermore, TUNEL-positive areas that were absent from sections of untreated tumors were readily detected in those of their treated counterparts ( Fig. 4B ).
The relapse of tumor growth observed in the presence of doxycycline was puzzling ( Fig. 4A , right panel, black line). Potential explanations for this unexpected finding are, among others, that (1) it was an isolated event, specific for clone 28.3, (2) the cells had acquired resistance against doxycycline, or (3) the cells had overcome their dependence on the IGF-1R for survival. To distinguish between these possibilities, cells of the clone 28.1 were implanted into nude mice and left either untreated or treated with doxycycline. Although some tumors of the latter group were harvested during regression, others were kept under treatment to check whether a relapse of tumor growth could be observed. Sections of the tumors were then stained either for myc, to determine the level of exogenously expressed protein, or IGF-1R, to test whether endogenously expressed IGF-1R might complement for the loss of the exogenously expressed protein. Furthermore, the proliferation status of the tumors was determined via BrdU incorporation.
Three exemplary tumor growth curves are shown in Figure 5 : one control tumor, which grew to a size of approximately 2500 mm3 in approximately 50 d ( Fig. 5 , black line), and two tumors that had been treated with doxycycline: one isolated while in regression ( Fig. 5 , light gray line) and the other treated until growth resumption ( Fig. 5 , dark gray line). Regrowth of two other tumors that received continued doxycycline treatment was also observed, indicating that the relapse was a reproducible event (data not shown). Immunohistochemical staining of tumor sections from the respective samples taken after doxycycline treatment demonstrated a complete elimination of the myc-signal ( Fig. 5 , left immunohistochemistry [IHC] panels). This result was in line with a net reduction of total IGF-1R, as demonstrated using an antibody recognizing both human and mouse IGF-1R protein ( Fig. 5 , middle IHC panels). No induction of endogenous IGF-1R expression was observed from tumors that regrew in the presence of doxycycline ( Fig. 5 , compare the middle IHC panels), indicating that the tumors activated an alternative survival pathway.
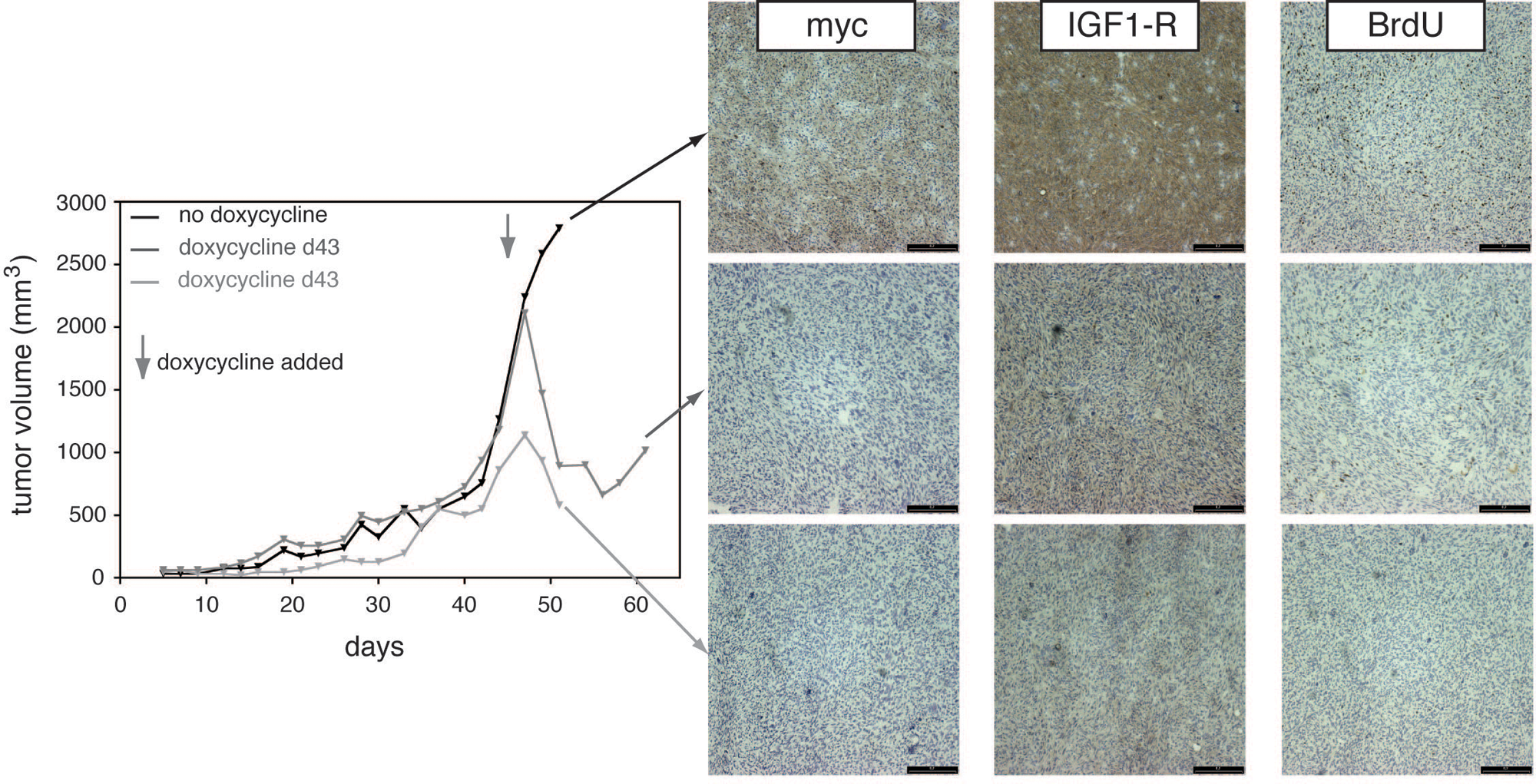
In vivo growth of the sublines 28.1/28.3 and growth inhibition by doxycycline. Repeat of Figure 4 , but only one animal per group is shown (total n = 3 per group). Animals were sacrificed at given time points during tumor growth, regression, or relapse, and the tumors were analyzed with immunohistochemistry. 9E10 (exogenous), as well as an insulin-like growth factor I receptor (IGF-1R) antibody (total IGF-1R) and BrdU stains are shown. A quantification of the results of the BrdU staining is shown in Table 1 .
Effect of Insulin-like Growth Factor I Receptor Repression on Tumor Cell Proliferation In Vivo

Per mm2.
With respect to control.
The rate of proliferation as determined via BrdU incorporation, as expected, mirrored the position of the tumor in the growth curve. The noninhibited exponentially growing tumor had the highest percentage of BrdU-positive cells, followed by the relapsing tumor. The tumor in remission was essentially depleted of S-phase cells ( Fig. 5 , right IHC panels; Table 1 ).
Thus, the repression of the IGF-1R as the original transforming principle led to a significant, but only transient, tumor regression in this cell system. The reason for the relapse was not a leakiness of the tet-system but most likely a switch to another survival pathway. Nevertheless, the original clear dependence of the cell lines on the IGF-1R signaling for survival and proliferation under starvation conditions in vitro and the rapid induction of tumor remission (even if transitory) make this a valuable cellular test system for IGF-1R inhibitors in vitro and in vivo.
Characterization of Agents Directed against IGF-1R In Vitro and In Vivo
Two kinase inhibitors were tested on the cell line in vitro: NVP-AEW541 and staurosporine. NVP-AEW541 is a small molecule that inhibits Ins-R and IGF-1R in in vitro kinase assays with similar IC50 values (0.15 µM) but distinguishes between the two highly similar receptor tyrosine kinases in cellular assays (IC50s of 2.3 and 0.086 µM, respectively 11 ). Staurosporine is a rather general kinase inhibitor, with IC50s in the nanomolar range for many kinases. 21 The 28.1 cells were serum starved followed by a preincubation with the inhibitor and a subsequent stimulation by IGF-1.
The IC50 for NVP-AEW541, 54 nM, corresponded very well with the literature value. Staurosporine was 10 times less effective with an IC50 of 650 nM ( Fig. 6 ).
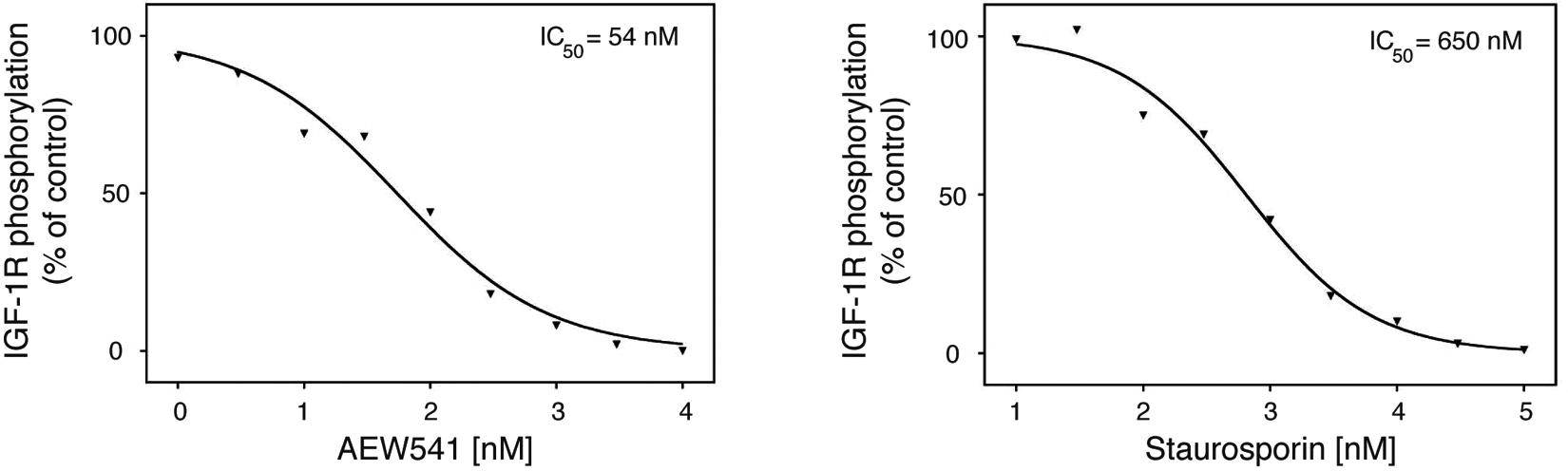
Cellular assays using 28.1. (
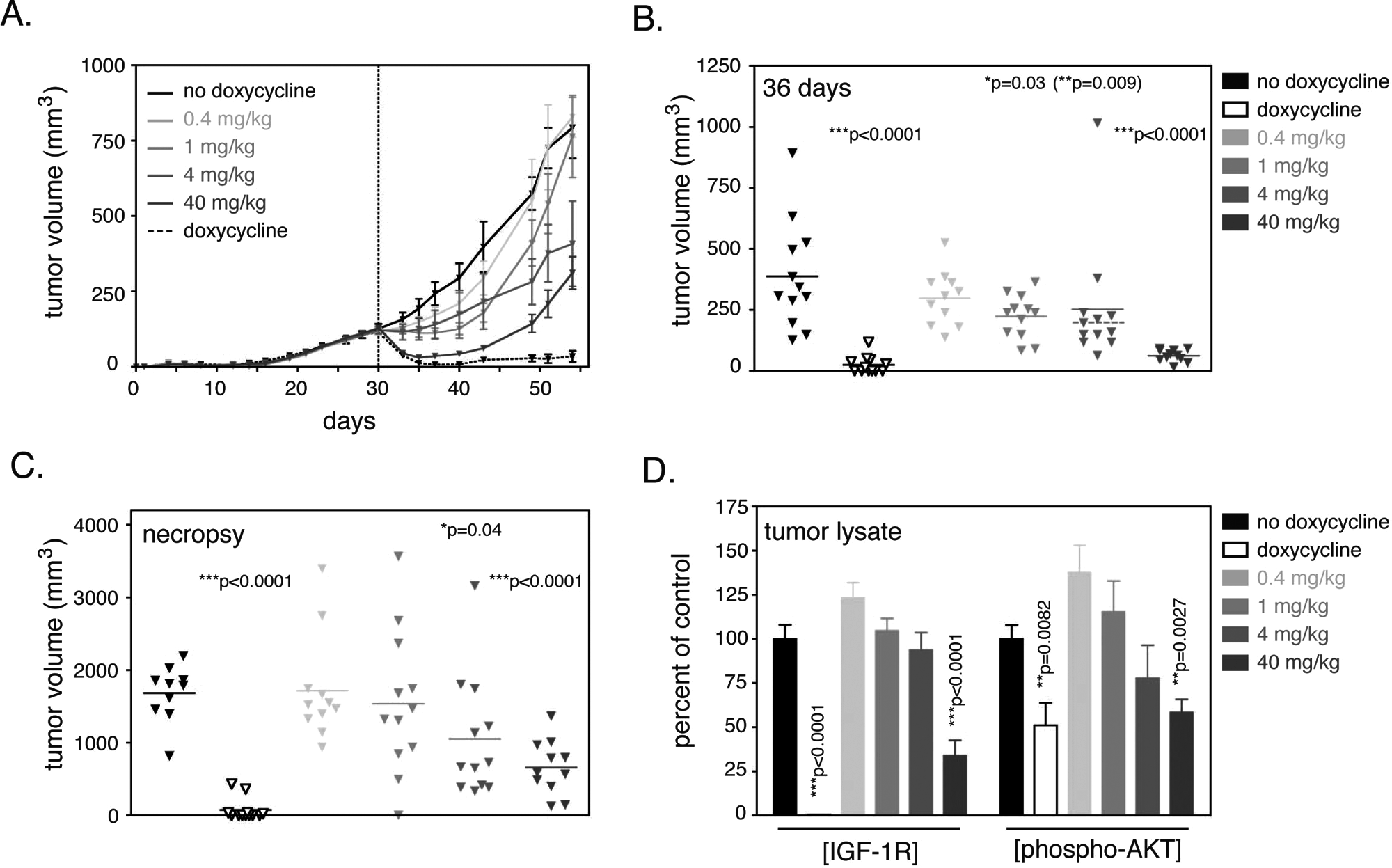
A murine anti–IGF-1R monoclonal antibody, EM164, served to test the model system in vivo. EM164 is a potent antagonist of human IGF-1R that blocks binding of IGF-1 to the receptor, inhibits receptor activation, promotes degradation of IGF-1R, and exhibits activity against tumor cell lines in vitro and in vivo.17,18 Mice engrafted subcutaneously with #28.1 cells were randomized into six groups at day 30 when the tumor volumes reached sizes of ≥200 mm3. Group 1 received vehicle, group 2 doxycycline (2 mg/mL) in their drinking water as a positive control (days 30–54), and groups 3 to 6 increasing doses of EM164 (0.4, 1, 4, and 40 mg/kg/injection) administered intraperitoneally twice weekly for 3 wk (six injections; days 30–48).
Tumor growth curves demonstrated a nice dose-dependent effect of EM164, although doxycycline was the most effective treatment ( Fig. 7A ). Five days after onset of treatment, doxycycline induced 100% complete tumor regressions, confirming that tumor growth was IGF-1R dependent ( Fig. 5B ; ***p < 0.0001). At the same time, antitumor activity of EM164 was observed at 40 mg/kg (11/11 PR, 6/11 CR; ***p < 0.0001), 4 mg/kg (1/12 PR, 1/12 CR; not significant; if the outlier, which exceeded 450 mm3 at the time of randomization, is omitted, **p = 0.009), and 1 mg/kg (3/12 PR, 2/12 CR; *p = 0.03), whereas the lower dose was inactive ( Fig. 7B , right upper panel). When compared at the time of necropsy (day 54 [some animals: day 51, see figure legend]), doxycycline (***p < 0.001), as well as the two higher doses of EM164 (*p = 0.04 for the 4 mg/kg group and ***p < 0.001 for the 40 mg/kg group, respectively), led to significantly reduced tumor sizes when compared with the untreated control ( Fig. 7C ). Taken together, these findings established EM164 as a dose-dependent inhibitor of IGF-1R function that suppresses tumor growth in vivo.
In vivo inhibition of 28.1 tumor growth by the anti–insulin-like growth factor I receptor (IGF-1R) antibody EM164. (
As a biomarker for the effect of EM164, IGF-1R down-regulation and a potential reduction of phospho-AKT levels as an important downstream target of IGF-1R were measured in tumor lysates. In untreated tumors, human IGF1R was expressed at very high levels (1094 ± 87.3 ng/mg of total protein), also when compared to tumor cell lines (e.g., 6.45 ng/mg of total protein in pancreas BXPC-3 or 6.02 ng/mg in multiple myeloma L363). Doxycycline treatment essentially abolished IGF-1R expression (4.4 ± 1.01 ng/mg of total protein; –99.6%), but EM164, albeit only at the highest dose of 40 mg/kg, also significantly reduced the levels of IGF-1R (–64%; Fig. 7D ). Lower doses showed increased rather than decreased IGF-1R levels, possibly indicating a compensatory effect by the tumor. Phosphorylation of AKT was also strongly reduced by doxycycline and the highest EM164 dose, indicating that the IGF-1R signaling cascade was effectively inhibited ( Fig. 7D ).
Discussion
The present study characterizes MEF cell lines, which are reversibly transformed due to a regulatable overexpression of the receptor tyrosine kinase IGF-1R. The original clone, 28, showed good overexpression of the transgene and clear IGF-1 dependency when grown in the absence of serum. However, when embedded into soft agar, or implanted into nude mice, growth was very slow, indicating that the transforming potential of the IGF-1R had not yet been fully exploited. Sublines generated from clone 28 via soft agar selection yielded cell lines that still depended on IGF-1R expression but grew much faster both in soft agar and in the mouse model.
The molecular differences between clone 28 and its sublines 28.1 and 28.3 have not yet been fully elucidated. No significant difference was found between the parental and the sublines with respect to IGF-1R expression levels (compare
The relapse of tumor growth in the mouse in the presence of doxycycline was an unexpected finding ( Fig. 4 ). However, this was not due to leakiness of the tet-system, because exogenous IGF-1R levels—as detected via 9E10 IHC—remained low. Also, total IGF-1R levels remained much lower than in the induced state, indicating that elevated expression of endogenous IGF-1R did not compensate for the loss of human IGF-1R expression ( Fig. 5 ). Similar observations were also made in a comparable model using ErbB2 as a doxycycline-controlled transforming receptor tyrosine kinase. 15 The expression and/or activation of other receptors might provide a potential explanation for this phenomenon. EM164 treatment of hepatocellular carcinoma was shown to lead to EGFR/HER3 activation, and a synergy was found between the antibody and the EGFR inhibitor gefitinib. 23 Similar observations were reported for a small-molecule IGF-1R inhibitor, BMS-536924. 24 Interestingly, the reverse also seems to work, that is, IGF-1R appears to mediate resistance to the HER-2 antibody trastuzumab. 25 However, at least in colon cancer, EGFR treatment resistance does not appear to sensitize tumors to IGF-1R treatment. 26 Thus, a similar scenario may be responsible for the doxycycline resistance/IGF-1R independence observed in our MEF cell lines.
The window of IGF-1R dependency is, however, large enough, and the cell line described here has been successfully employed in a preclinical discovery campaign to establish the IGF-1R–mediated antitumor activity of EM164, a murine IGF-1R–specific antibody.17,18 Treatment with EM164 resulted in a dose-dependent tumor regression, demonstrating the principle that EM164 can block IGF-1R function and suppress in vivo the growth of IGF-1R–dependent tumors. IGF-1R protein levels as well as AKT phosphorylation as pharmacodynamic biomarkers were strongly reduced upon EM164 treatment, establishing that the antibody reached its target in the tumor cells and blocked IGF-1R signaling by inducing its degradation ( Fig. 7A , B ).
Moreover, a proof-of-concept study demonstrated that this cell line is a valuable tool to discover blood-based biomarker via secretome proteomics. Vascular endothelial growth factor and osteopontin were differentially detected by LC-MS/MS in vitro, and osteopontin could indeed be detected via ELISA in the plasma of mice bearing IGF-1R–expressing tumors but not their doxycycline-treated counterparts. The increase of osteopontin levels paralleled tumor growth, qualifying the molecule as a potential plasma biomarker for IGF-1R–dependent tumors. 27
In conclusion, the cell lines described here represent important tools for IGF-1R–related research and drug discovery.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
