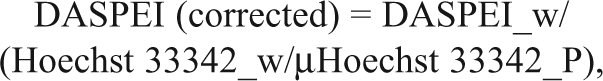Abstract
Mitochondrial dysfunction is a hallmark of several diseases and may also result from drugs with unwanted side effects on mitochondrial biochemistry. The mitochondrial membrane potential is a good indicator of mitochondrial function. Here, the authors have developed a no-wash mitochondrial membrane potential assay using 2-(4-(dimethylamino)styryl)-N-ethylpyridinium iodide (DASPEI), a rarely used mitochondrial potentiometric probe, in a 96-well format using a fluorescent plate reader. The assay was validated using 2 protonophores (CCCP, DNP), which are known uncouplers, and the neuroleptic thioridazine, which is a suspected mitochondrial toxicant. CCCP and DNP have short-term depolarizing effects, and thioridazine has long-term hyperpolarizing effects on the mitochondrial membrane potential of Chinese hamster ovary (CHO) cells. The assay also detected changes of the mitochondrial membrane potential in CHO cells exposed to cobalt (mimicking hypoxia) and in PC12 cells exposed to amyloid β, demonstrating that the assay can be used in cellular models of hypoxia and Alzheimer’s disease. The assay needs no washing steps, has a Z′ value >0.5, can be used on standard fluorometers, has good post liquid-handling stability, and thus is suitable for large-scale screening efforts. In summary, the DASPEI assay is simple and rapid and may be of use in toxicological testing, drug target discovery, and mechanistic models of diseases involving mitochondrial dysfunction.
Introduction
D
Materials and Methods
Cell lines and cultures
CHO cells were maintained in HAM F12 medium supplemented with 10% fetal bovine serum, 200 mM L-glutamine, 200 U/mL penicillin, and 5 µg/mL streptomycin. Undifferentiated PC12 cells (American Type Culture Collection, LGC Standards AB, Boras, Sweden, CRL-1721) were maintained in Dulbecco’s modified Eagle’s medium (DMEM) 1965 medium supplemented with 10% fetal bovine serum, 5% horse serum, 200 mM L-glutamine, 200 U/mL penicillin, and 5 µg/mL streptomycin. Zero glucose medium consisted of DMEM minus glucose (11966-025; Invitrogen, Carlsbad, CA) supplemented with 10% fetal bovine serum, 10 mM galactose, 5 mM HEPES sodium salt, 1 mM sodium pyruvate, 200 mM L-glutamine, 200 U/mL penicillin, and 5 µg/mL streptomycin. PC12 cells were differentiated by adding 100 ng/mL nerve growth factor to the growth medium over a period of 3 days before use. All cells were cultured on Nunclon™ Δ surfaces in 35-mm or 100-mm Petri dishes (172958, 153066; Nunc, Roskilde, Denmark) or in 96-well plates (3628; Corning Costar, Copenhagen, Denmark) in a standard CO2 incubator at 37°C and 5% CO2.
Chemicals
The following items were produced and purchased locally from the Panum Institute Substrate Department: HAM F12, DMEM 1965 medium, penicillin, streptomycin, L-glutamine, and trypsin. Fetal bovine serum (10106-169) and horse serum (16050-122) were from Invitrogen. A 2% CoCl2 solution was from Bie & Berntsen (Copenhagen, Denmark). DASPEI (stock: 40 mM in DMSO), TMRM (stock: 10 mM in DMSO), Hoechst 33342 (stock: 10 mM in water), N-methyl-D-glucamine (NMDG), HEPES sodium salt, HEPES acid, nerve growth factor–2.5S (NGF), amyloid β–protein fragment 25-35 (Aβ [25-35]), Trypan blue, and thioridazine (stock: 20 mM in DMSO) were purchased from Sigma-Aldrich (St. Louis, MO). The fluorescence quencher F-QUENCH (stock: 25 mM in water) was obtained from electropharma (Copenhagen, Denmark). F-QUENCH has an absorbance spectrum that covers the 500- to 700-nm range and a maximal absorbance wavelength (λmax) in water of 589 nm (supplier information). Control experiments showed that F-QUENCH quenched fluorescence from 100 µM TMRM with an EC50 of 35 µM (Hill: −2.2) and 100 µM DASPEI with an EC50 of 34 µM (Hill: −1.0). Aggregates of F-QUENCH were noted when combined with TMRM but not combined with DASPEI.
Cell handling, loading buffer, and fluorescence measurements
The DASPEI assay was run on cells either grown to confluence in 96-well plates or in suspension that were allowed to settle at the bottom of 96-well plates. In the suspension version, 2 confluent 100-mm Petri dishes were trypsinized, and the cells were suspended in a total of 16 mL NMDG-buffer solution. One 96-well plate was prepared with 100 µL loading buffer and appropriate test compounds in each well, and 50 µL cell suspension was injected into each well. The loading buffer was an NMDG-buffer solution that was composed of 169 mM N-methyl-D-glucamine, 20 mM HEPES acid, pH adjusted to 7.4 by HCl (from 5M stock), with additional 100 µM DASPEI and 25 µM Hoechst 33342. The probe-loading time depended on setup but was usually 60 min in an incubator at 37°C and 5% CO2.
Fluorescence from DASPEI, TMRM, and Hoechst 33342–loaded cells was measured using a FLUOstar OPTIMA plate reader (BMG Labtech, Offenburg, Germany). The reader was maintained at 25°C, and the following filters were used: 485 ex/555 em for DASPEI, 544 ex/590 em for TMRM, and 320 ex/460 em for Hoechst 33342. The 96-well plates were read from the bottom with 10 measurements per reading and orbital averaging in a 3-mm radius. The fluorescence quencher F-QUENCH was added following DASPEI, Hoechst 33342, and test compound coloading; plates were kept at room temperature and were read at variable time intervals following F-QUENCH addition. The same plate reading method was used with adherent cells grown at the bottom of a well or trypsinized cells in suspension that had settled at the bottom of a well during the loading period. Hoechst labeling shows linearity with cell number in microplates, 16 and thus when using trypsinized cells in suspension, DASPEI fluorescence was partially corrected for differences in cell number between individual wells using the following formula:
where DASPEI_w is the DASPEI fluorescence measured from the well, Hoechst 33342_w is the Hoechst 33342 fluorescence from the same well, and µHoechst 33342_P is the mean of fluorescence across the whole plate or the desired area to correct. During assay optimization, vehicle/CCCP ratios were determined to identify the largest signal-to-noise ratios under different conditions.
CoCl2-induced apoptosis in CHO cells
CHO cells were cultured to confluence in 96-well plates. The normal growth medium was replaced with 100 µL medium containing 0, 0.05, 0.5, or 1.0 mM CoCl2 and incubated for 2 to 24 h at 37°C and 5% CO2. The CoCl2-containing medium from the wells was replaced with 150 µL loading buffer, followed by 1-h incubation at 37°C, addition of F-QUENCH, and 30-min incubation at room temperature prior to fluorescence measurements. Cell viability was also assessed by counting viable and dead cells in 0.4% Trypan blue using an inverted microscope with oblique light.
Amyloid β–induced cell death in PC12 cells
PC12 cells were cultured to confluence in 96-well plates. The normal growth medium was replaced with 100 µL media containing 0, 10, and 1000 nM amyloid β (fragment 25-35) in an oligomeric form (incubated for 24 h at 37°C before use) and incubated for 6 to 48 h. At specific time points, the media were removed from the wells and replaced with 150 µL loading buffer and incubated for 1 h, followed by addition of F-QUENCH and 30-min incubation at room temperature prior to fluorescence measurements.
Photomicrographs and data analysis
CHO cells that had grown overnight on polylysine-coated glass cover slips or trypsinized CHO cells that had settled on the glass cover slips were loaded with 50 µm DASPEI for 1 h at 37°C. Photomicrographs were taken on a fluorescence microscope using ex: 470 ± 20 nm, em: 515LP filters.
Data are expressed as either the mean ± SD or SEM, and Student t test was used for statistical comparisons (p < 0.05 considered significant). Curve fittings and plots were performed using Prism v. 5.0 (GraphPad Software, Inc., San Diego, CA).
Results
Fluorescence from DASPEI-loaded cells was measured, without the need for washing steps, by coapplying DASPEI and test compounds in 96-well plates, quenching fluorescence in the liquid phase using F-QUENCH, and reading plates from the bottom. DASPEI is a positively charged styryl dye that labels mitochondria, and we reasoned that a maximally hyperpolarized plasma membrane potential would facilitate loading of DASPEI into cells for subsequent accumulation in mitochondria. Thus, the loading buffer (NMDG-buffer) contains zero sodium and potassium, which theoretically should hyperpolarize the resting membrane potential of CHO cells by reducing inward leak sodium current and increasing outward leak potassium currents. Indeed, CHO cells showed an increased fluorescence over time, which was markedly larger in NMDG-buffer compared to normal culture medium (
2-(4-(dimethylamino)styryl)-N-ethylpyridinium iodide (DASPEI) labeling of Chinese hamster ovary (CHO) cells in suspension or adhered. (
Fluorescence microscopy of adherent CHO cells, grown on polylysine-coated glass cover slips, loaded with 50 µM DASPEI for 1 h at 37°C showed a fluorescent DASPEI label throughout the cell, with distinct accumulation in tubular mitochondria-like structures surrounding the nucleus (
Once inside the cell, DASPEI accumulates in the mitochondria dependent on the mitochondrial membrane potential.
13
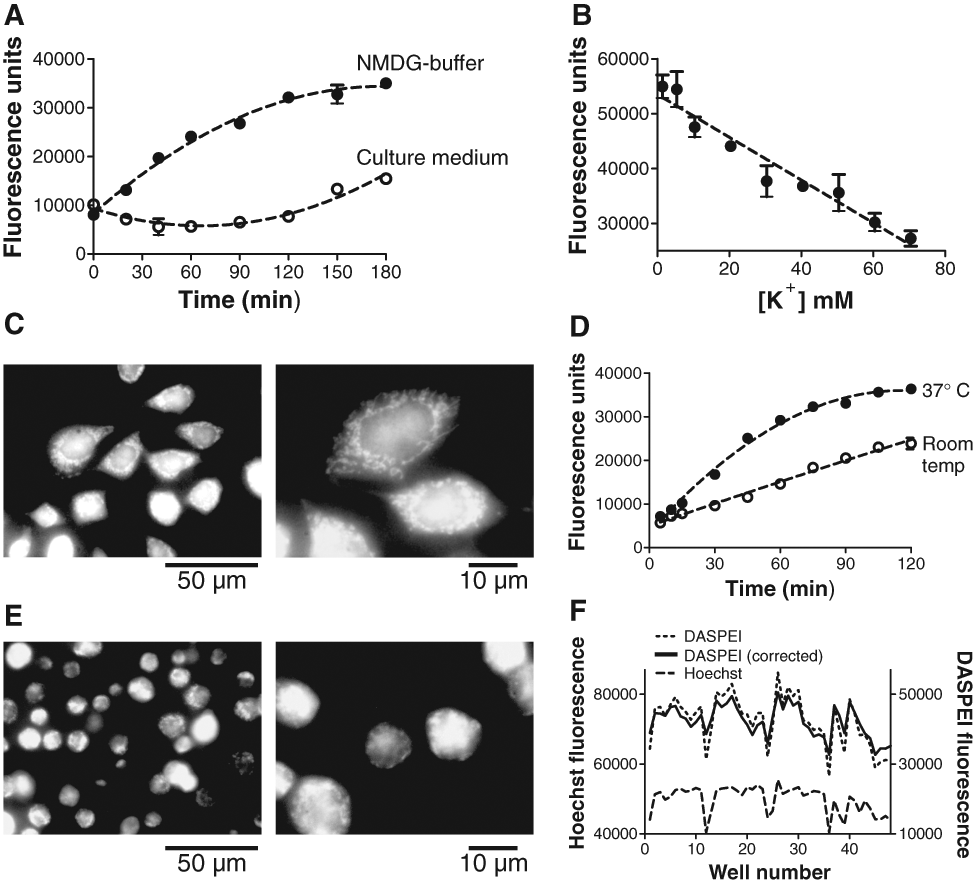
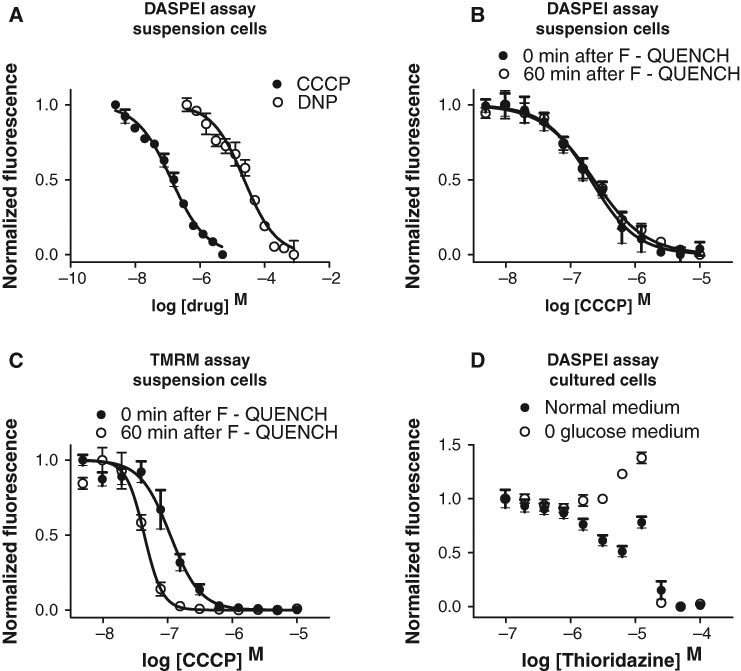
We used the potent protonophore CCCP, which depolarizes mitochondria, to validate that DASPEI loading is dependent on mitochondrial membrane potential. When 10 µM CCCP was present during DASPEI loading, the fluorescence, read immediately after a 60-min loading time, was reduced compared to vehicle (DMSO) at a DASPEI-loading concentration of ~10 µM and above (
Fluorescence from vehicle and CCCP-treated Chinese hamster ovary (CHO) cells at increasing 2-(4-(dimethylamino)styryl)-N-ethylpyridinium iodide (DASPEI) and tetramethylrhodamine methyl ester (TMRM) concentrations and at increasing time lags to reading. (
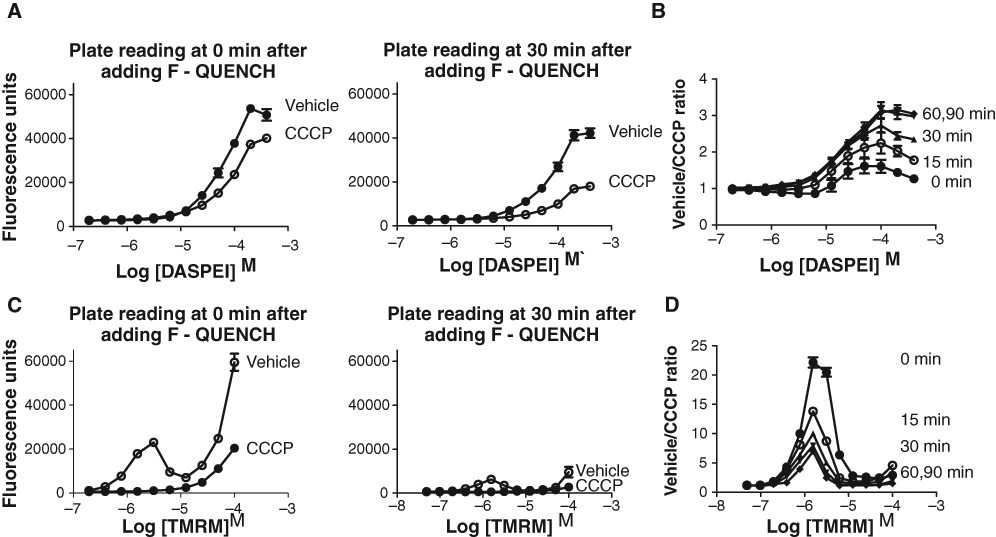
Using this optimized assay procedure, dose-response relationships for 2 reference compounds (CCCP, DNP), which both are protonophores with a short-term effect on collapsing the mitochondrial membrane potential, gave the following EC50 values: CCCP = 132 nM (Hill slope: −0.8), and DNP = 23 µM (Hill slope: −0.9;
Dose-response relationships for 3 test compounds using 2-(4-(dimethylamino)styryl)-N-ethylpyridinium iodide (DASPEI) and tetramethylrhodamine methyl ester (TMRM) assays. (
DMSO tolerance in the DASPEI assay was determined by incubating CHO cells with increasing concentrations of DMSO (0%-30% v/v) with vehicle (DMSO) or 10 µM CCCP present (
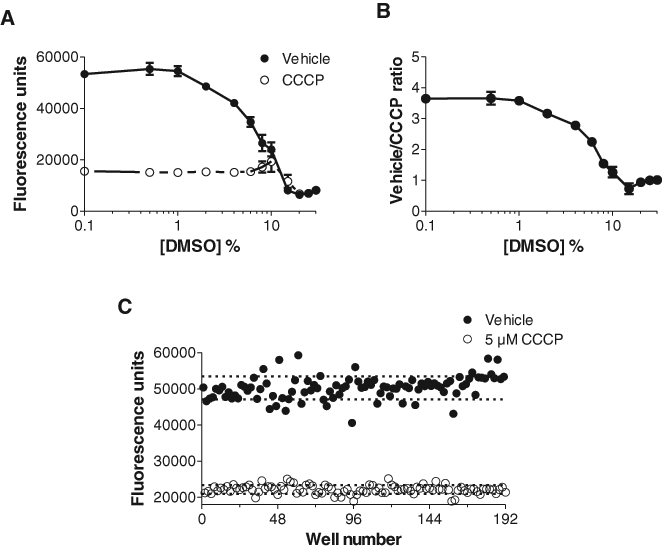
DMSO tolerance and Z′ determination. (
To further validate the DASPEI assay and its potential use in biochemistry and disease-related studies, we used ionomycin and cobalt applications in CHO cells and amyloid β applications in PC12 cells. Ionomycin, which allows the free passage of divalent cations across the plasma membrane and membrane of intracellular organelles, was used to raise the intracellular level of calcium. Mitochondrial calcium overload is known to be accompanied by depolarization of the mitochondrial membrane in other cell types.
19
A dose-dependent decrease in DASPEI fluorescence, indicating depolarization of the mitochondrial membrane, was observed after a 3-h application of ionomycin (EC50: 728 nM, Hill slope: −2.6) in CHO cells with 1 mM extracellular CaCl2 (
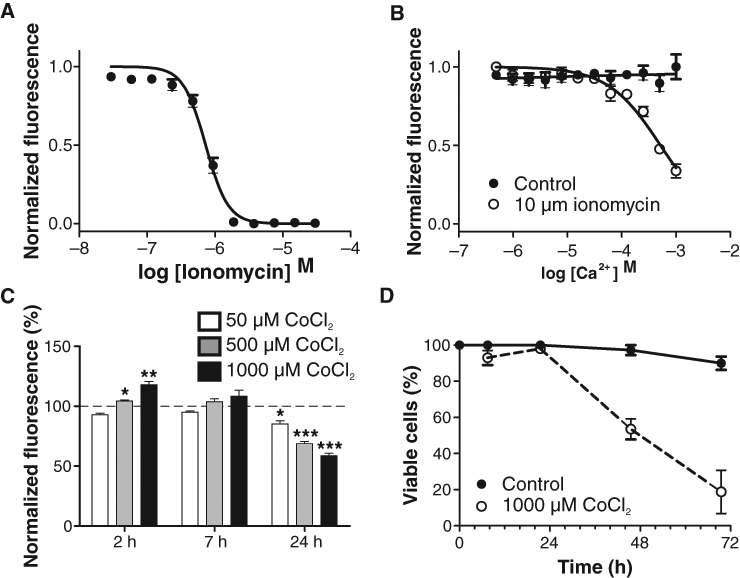
Effect of ionomycin and cobalt on the mitochondrial membrane potential of Chinese hamster ovary (CHO) cells. (
NGF induces a neuronal phenotype of PC12 cells, and differentiated PC12 cells are often used in cellular studies of neuronal disease. Undifferentiated trypsinized PC12 cells show the same DASPEI-loading pattern as trypsinized CHO cells (data not shown). However, PC12 differentiation by NGF is a process that takes several days, and the resulting neurite-bearing PC12 cells are not easily trypsinized. We therefore tested if DASPEI fluorescence could be measured on cultured undifferentiated PC12 and NGF-differentiated PC12 cells in 96-well plates. CCCP and DNP dose-dependently reduced DASPEI fluorescence in undifferentiated PC12 cells (CCCP-EC50: 874 nM, Hill slope −1.2; DNP-EC50: 81 µM, Hill slope −1.7) and NGF-differentiated PC12 cells (CCCP-EC50: 549 nM, Hill slope −0.9; DNP-EC50: 46 µM, Hill slope −0.6;
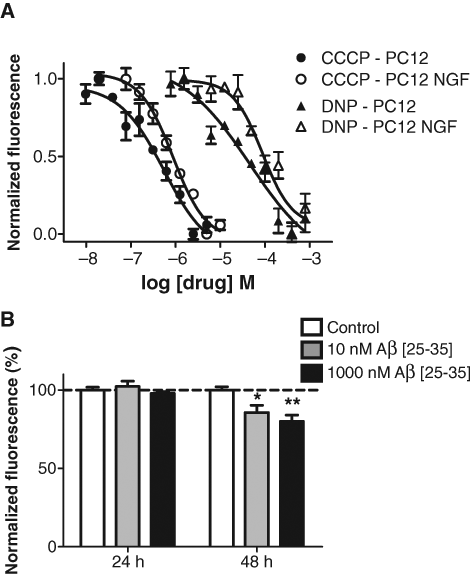
Dose-response relationships for 3 test compounds in PC12 cells. (
Amyloid β, the main constituent of amyloid plaques in the brains of Alzheimer’s disease patients, depolarizes the mitochondrial membrane and leads to cell death in PC12 and NGF-differentiated PC12 cells.
21
Differentiated PC12 cells cultured in a 96-well plate and exposed to 10 and 1000 nM Aβ [25-35] showed a dose-dependent decrease in DASPEI fluorescence at 48 h compared to control, indicating a mitochondrial membrane depolarization (10 nM Aβ, p ≤ 0.05; 1000 nM Aβ, p < 0.01;
Discussion
The simple and rapid method presented here measures mitochondrial membrane potential in intact cells in suspension or growing adherently in microplates and may be of use in toxicological testing and drug target discovery identifying compounds that change mitochondrial membrane potential. The DASPEI assay needs no washing steps, has a Z′ value >0.5, can be used on standard fluorometers, has good post liquid-handling stability, and thus is suitable for large-scale screening efforts.
Fluorescent lifetime measurements from DASPEI-labeled mitochondria in XTH2 cells suggest that DASPEI is partially inserted into the outer leaflet of the inner mitochondrial membrane. 22 A model was proposed where DASPEI preferentially locates its positively charged methyl pyridinium ring in the aqueous interface facing the outer mitochondrial membrane and its hydrophobic moiety along the hydrocarbon chain. When the mitochondrial membrane depolarizes the charge of the positively charged methyl pyridinium, moiety increases, leading to a more polar structure of DASPEI at the inner membrane, and the molecules are able to diffuse freely to other compartments of the cell. The fluorescent intensity of DASPEI is dependent on the local molecular environment, with reduced quantum efficiency in polar environments. 14 If this model is correct, the reduced DASPEI fluorescence in the assay described here under conditions where, for example, CCCP is present can be explained by a reduced accumulation of DASPEI in the outer leaflet of the inner mitochondrial membrane during loading. DASPEI molecules that have entered the cell due to the plasma membrane potential will preferentially be located in the polar environment of the cytosol and consequently display a lower quantum yield.
The optimal DASPEI concentration for measurement of the mitochondrial membrane potential was found to be 100 µM. This is a high concentration compared to other mitochondrial membrane potential probes used in assays, such as JC-1 and TMRM, which use 10 µM and 50 nM, respectively, 9,23 and may in itself influence mitochondrial biochemistry. 14 However, DASPEI does not have the self-quenching limitations of TMRM 24 or the aggregation properties of JC-1, 25 which allows higher concentrations and gives a greater fluorescence yield and thus a clearer signal in microplates. The high loading concentration of DASPEI also means high concentrations of DMSO, which severely affects the performance of the assay if the total DMSO concentration is above 8%. Two mitochondrial uncouplers (CCCP and DNP) were used to validate the DASPEI assay and gave EC50 values comparable to those found in the literature. 8 The post liquid-handling stability of the EC50 values was good, staying constant for at least 60 min. However, when TMRM was used together with F-QUENCH, a very steep Hill slope (−2 to −3) of the dose-response curve for CCCP was observed, together with a systematically decreasing EC50 with increased time lag to reading. This phenomenon was not observed when washing was used instead of quenching with F-QUENCH. We suspect that the self-quenching properties of TMRM may be cooperative at accumulating TMRM concentrations in the mitochondrial matrix and that this effect may continue after liquid-handling-producing unstable EC50 determinations. In addition, time-dependent aggregation (see Materials and Methods) of F-QUENCH induced by the presence of TMRM may also contribute. However, using F-QUENCH with DASPEI, blocking fluorescence from the microwell liquid phase, was useful because the assay could be run without any washing steps. In experiments not shown here, F-QUENCH did not reduce DASPEI loading but significantly reduced Hoechst 33342 loading into the cell. This suggests that the quencher may in some way interact with Hoechst 33342, as it did with TMRM. Therefore, a homogeneous assay with the probes and quencher mixed and a “just add cells” setup is not possible if Hoechst 33342 is used.
One important problem with the current assay is the dependency of the plasma membrane potential. CCCP depolarizes the plasma membrane potential at a high concentration (>2.5 µM), and micromolar concentrations of ionomycin have a similar effect. 26 Thus, the effect of CCCP and ionomycin in this study may partially be explained by a reduced plasma membrane potential, which on its own will decrease DASPEI loading, combined with the effect on the mitochondrial membrane potential. Thus, direct action of test compounds on plasma membrane potential should always be kept in mind when interpreting effects on DASPEI fluorescence from intact cells in this assay. In this respect, the assay may be inferior to assays based on TMRM, which can be set up to be independent of plasma membrane potential. 8 However, the plasma membrane potential dependency may be an advantage under conditions where toxic compounds with effects on both plasma and mitochondrial membrane potential are to be tested because the combined effect will give a large signal-to-noise ratio. The optimized assay procedure, maximizing the plasma membrane potential, may also increase the accumulation of many cationic lipophilic drugs.
The DASPEI assay also detected an effect on the mitochondrial membrane potential after long-term exposure to a suspected mitochondrial toxicant. Thus, acute cellular toxic effects of high concentrations of thioridazine were observed, followed by a relative hyperpolarization of the mitochondrial membrane potential after 2 to 3 days’ exposure at lower thioridazine concentrations, particularly when the cells were forced to use oxidative phosphorylation by replacing glucose with galactose in the medium. Several compounds are known to induce hyperpolarization of the mitochondrial membrane potential (e.g., hydrogen peroxide). 27 Thioridazine binds to the inner mitochondrial membrane 28 and inhibits NOX4 activity. 29 However, the mechanism by which thioridazine induces long-term hyperpolarization of the mitochondrial membrane potential and the associated effects on long-term cellular functions remain to be determined.
The DASPEI assay may also be used to measure mitochondrial membrane potential in cellular models of neurodegenerative diseases. CoCl2 dose-dependently induces apoptosis in CHO cells, 20 possibly by inhibiting degradation of the regulating α subunit of the hypoxia-inducible factor–1 (HIF-1) transcription factor. Here we observed that the mitochondria of CHO cells exposed to CoCl2 initially hyperpolarized (6 h) followed by depolarization, with a decrease in cell viability (24 h and on). This is consistent with findings that HIF-1 activation by cobalt hyperpolarizes and fragments mitochondria in smooth muscle cells. 30 CoCl2 induces apoptosis in PC12 cells in a similar fashion through ROS production. 31 Thus, the DASPEI assay may be used in CHO and PC12 cells in conjunction with CoCl2 exposure to model hypoxia-linked neurological disorders. Oxidative stress induced by amyloid β has been implicated in the neurodegeneration observed in Alzheimer’s disease. Yet, Alzheimer’s is a multifactorial disease, and using amyloid β in a simple PC12 cell setup will by no means encompass the complex Alzheimer’s pathology. However, a cellular model using PC12 cells and amyloid β, as presented here, can be used in screening for compounds that protect against amyloid β–mediated mitochondrial depolarization.
In conclusion, the combination of using a fluorescence quencher (F-QUENCH), a loading buffer that maximally hyperpolarizes the plasma membrane potential, optimized assay parameters for DASPEI concentration, loading temperature, loading time, and postquencher reading time defines a no-wash DASPEI assay for mitochondrial membrane potential that may be of use in toxicological testing, drug target discovery, and mechanistic models of diseases involving mitochondrial dysfunction.
Footnotes
Acknowledgements
We thank Lis Hansen for technical assistance.
This work was supported by The Lundbeck Foundation, Aase og Ejnar Danielsens Fondation, Agnes and Poul Friis Foundation, Sofus Carl Emil Friis and Wife Olga Doris Friis Foundation, 17-12-1981 Foundation, and the Owensenske Foundation.