Abstract
Many G-protein-coupled receptors (GPCRs) have been shown to form heteromeric complexes primarily by biochemical methods, including competitive radioligand binding assays or measurements of changes in second-messenger concentration in lysed cells. These results are often cell line specific, and the expression of other cell surface proteins makes it difficult to detect potential functional consequences of GPCR interaction. Here, 2-electrode voltage clamping in Xenopus oocytes was used as a bioassay to explore heterodimerization of bradykinin type 2 receptor (Bk2R) and beta 2 adrenergic receptor (β2AR), using chloride channels as outputs for receptor activation. The data show for the first time that these 2 receptors heterodimerize with functional consequences. Stimulation with bradykinin induced activation of Gαq- and transactivation of Gαs-coupled pathways in oocytes expressing Bk2R and β2AR. To corroborate these data, potential receptor interaction was examined in PC12 cells, a cell line that endogenously expresses both receptors, and confirmed that stimulation with bradykinin transactivates β2AR. In both oocytes and PC12 cells, transactivation was ablated by Bk2R or β2AR inverse agonists, suggesting that transactivation occurred directly through both receptors. This is the first evidence of Bk2R/β2AR physical interaction, forming a functional heterodimer. The oocyte system may prove highly useful for exploration of GPCR heterodimerization and the functional consequences thereof.
Keywords
Introduction
G-
Despite increasing evidence of functional dimers, few reports have demonstrated physiologically relevant consequences of heterodimerization; even fewer of these in vivo dimer pairs originate from GPCRs that activate different classes of G-proteins. One such example is the Gαs-coupled beta adrenergic receptors that have been shown to heterodimerize with the Gαi-coupled opioid receptors (OPRs) and modify opiate tolerance. 8 Interactions between ATR1 and beta adrenergic receptors led to ATR1 function being inhibited by beta-blockers. 9
Attempts to discover new GPCR heterodimer pairs have been exhaustive and often plagued with skepticism because of the biochemical methods used to identify them. Given the lack of specificity for many antibodies, as well as the potential for protein aggregation, co-immunoprecipitation studies require additional experiments to confirm any putative physical association between GPCRs. Newer technologies such as bioluminescence resonance energy transfer (BRET) or Förster resonance energy transfer (FRET) rely on heterologous expression of proteins with fluorescent tags placed in close enough proximity to one another to overlap emission and excitation spectra. These methods are often difficult and time-consuming and must be done in conjunction with competitive radioligand binding studies or other complex pharmacology experiments to prove the connection between physical association and functional interaction. However, neither of these methods provides a real-time, functional assay of coexpressed GPCRs that can provide insights into both physical interaction and functional consequences thereof, and neither of these methods is applicable to the study of endogenously expressed receptors.
Injection of cRNA into Xenopus laevis oocytes and subsequent experimentation in these cells using electrophysiological methods is most often used for studying ion channels, as oocytes express relatively few membrane proteins. However, oocytes are often a useful model system for the functional study of heterologously expressed receptors because oocytes express the same intracellular components of signal transduction pathways as do other cell types. 10 Because many GPCRs activate signaling pathways that impinge on ion channels, this assay provides an elegant means for studying 1 or 2 proteins of interest using channel activity as an output of receptor activation in a controlled environment. Two-electrode voltage clamp (TEVC) electrophysiology allows one to measure changes in current from receptor-activated channels in response to a manipulation; the magnitude of current change is proportional to the change in activity of the channels and hence of the receptors. Furthermore, the use of electrophysiological methods enables the detection of a signal of interest above all of the stochastic events occurring in a single cell because the ion channel effectors amplify signal magnitude. The approach described here uses TEVC in Xenopus oocytes as a means of discovering and characterizing new GPCR heterodimer pairs.
Both Bk2R and β2AR play a number of regulatory roles, including in cardiovascular and smooth muscle function. Given the colocalization of Bk2R and β2AR in several tissues, their dysfunction in normal and disease states, and their propensity to heterodimerize with other receptors of their same classes, we hypothesized that Bk2R and β2AR undergo physical interaction that modulates individual receptor-mediated signaling events. To examine the interaction of these receptors, we performed TEVC studies in Xenopus oocytes expressing these receptors along with the cystic fibrosis transmembrane conductance regulator (CFTR) as an output to monitor Gαs pathway activation. To confirm these findings in oocytes, we performed similar experiments in pheochromocytoma (PC12) cells, a rat adrenal medulla tumor cell line that natively expresses both Bk2R and β2AR. We show here that stimulation of Bk2R leads to a novel signaling cascade via direct transactivation of β2AR in both oocytes and PC12 cells. Co-immunoprecipitation (co-IP) studies in oocytes transiently expressing these receptors, in PC12 cells, and in mouse ventricular cardiac myocytes show here for the first time that Bk2R and β2AR physically associate in their native environment, as well as in a heterologous expression system, thereby correlating physical interaction with function interaction. Using TEVC as a screening method for the functional interaction of GPCRs can prove to be a highly useful and accurate method for discovering new GPCR heterodimers, studying the interaction between GPCRs, and identifying downstream effectors of GPCR heterodimer signaling.
Materials and Methods
Oocyte isolation
Stage V to VI defolliculated X. laevis oocytes were prepared as previously described. 11 Oocytes were injected with 5 to 25 ng of cRNAs from in vitro transcription of constructs encoding Bk2R (with or without a hemagglutinin [HA] tag; UMR cDNA resource center), β2AR (a generous gift from Dr. Brian Kobilka, Stanford University), serotonin type 2C receptor (5HT2C-R) (a generous gift from Dr. H. Criss Hartzell, Emory University), and/or CFTR 12 36 to 72 h prior to experimentation. Methods for animal handling are in accordance with the National Institutes of Health (NIH) guidelines, and the protocol was approved by the Animal Use and Care Committees of the Georgia Institute of Technology and Emory University.
Electrophysiology
TEVC recordings were performed using a GeneClamp 500B amplifier (Axon Instruments, Sunnyvale, CA). Bath recording solutions contained (in mM) 96 NaCl, 2 KCl, 1 MgCl2, and 5 HEPES (pH 7.5). TEVC electrodes were filled with 3M KCl and had resistances of 0.2 to 2 MΩ. Oocyte recordings were performed using a holding potential of Vm = −60 mV, and data were acquired at a 500-µs per point sampling rate. Dose-response curves were recorded using a step protocol of Vm = −60 to +40 mV, acquired at a sampling rate of 2500 µs per point. Points show the peak current in response to each concentration of terbutaline, normalized to the maximum current at the highest concentration in each oocyte.
Cell culture
PC12 cells (kindly donated by Dr. Victor Faundez, Emory University) were maintained in Dulbecco’s modified Eagle’s medium (DMEM; Mediatech, Manassas, VA) with 10% horse serum (Rockland, Gilbertsville, PA) and 5% fetal bovine serum (Mediatech) with 1 mg/mL penicillin and streptomycin (Sigma-Aldrich, St. Louis, MO). Chinese hamster ovary (CHO) cells were maintained in DMEM with 10% fetal bovine serum with 1 mg/mL penicillin and streptomycin. Freshly dissociated murine ventricular myocytes were kindly provided by Dr. Yanggan Wang (Emory University).
Co-immunoprecipitation
PC12 cells were lysed in 20 mM Tris-HCl, 137 mM NaCl, 2.7 mM KCl, 1 mM MgCl2, 1 mM CaCl2, 1% Nonidet P-40, 10% glycerol, 1 mg/mL bovine serum albumin (BSA), 0.2 mM phenylmethanesulphonylfluoride (PMSF), and 0.5 mM NaVO4 (pH 6.8). Fifteen to 20 oocytes tested for expression prior to co-immunoprecipitation were lysed in 20 mM Tris-HCl, 5 mM MgCl2, 5 mM Na2HPO4, 1 mM EDTA, 80 mM sucrose, and 1 mM PMSF (pH 7.4). Cardiac myocytes and CHO cells were lysed in 1% NP40, 1% sodium deoxycholate, 0.1% sodium dodecyl sulfate (SDS), 150 mM NaCl, 0.01 M NaPO4, 2 mM EDTA, 50 mM NaF, 0.02% sodium azide, and 0.1 mM PMSF (pH 7.2). Protease inhibitor cocktail (Sigma-Aldrich) was added to all lysis buffers on the day of the experiment. Lysates were tumbled in an end-over-end agitator for 1 h and spun down for 30 min at 4°C at 16,000 g. Precleared cell lysates were tumbled in an end-over-end agitator at 4°C with either anti-β2AR antibody (Santa Cruz Biotechnology # sc-569, Santa Cruz, CA) or anti-Bk2R antibody (Santa Cruz Biotechnology, # sc-15050) for 1 h prior to conjugation to Protein A agarose beads (Roche, Indianapolis, IN). If appropriate, precleared lysates were instead tumbled with anti-HA antibody-conjugated agarose beads (Roche). A recent publication by Pradidarcheep and coworkers 13 questions the specificity of one anti-β2AR antibody produced by Santa Cruz. We used a different commercially available anti-β2AR antibody here. Cell lysate and antibody-conjugated beads were tumbled overnight at 4°C prior to elution and Western blotting. Blots were then incubated with anti-Bk2R (EMD Biosciences, San Diego, CA), anti-5HT2C-R (Santa Cruz), or anti-β2AR antibodies for 1 h at room temperature at a 1:1000 dilution. After thorough washes, blots were incubated with the appropriate AlexaFluor 680-conjugated secondary antibodies (Invitrogen, Carlsbad, CA) at a 1:5000 dilution, and membranes were visualized using a LI-COR Odyssey Scanner (LI-COR Biosciences, Lincoln, NE).
Enzyme immunoassay
cAMP levels were measured using a cAMP enzyme immunoassay (EIA) kit per manufacturer’s instructions (GE Amersham, Piscataway, NJ). Briefly, approximately 30,000 PC12 cells were seeded in either triplicate or quadruplicate for each condition in a 96-well plate 24 h prior to ligand treatment and lysis. Cells were treated with 10 to 50 µM 3-isobutyl-1-methylxanthine (IBMX) to inhibit the breakdown of cAMP, plus 10 µM GF 109203X and/or 10 µM propranolol, if appropriate, for 10 min prior to addition of either 5 µM isoproterenol (ISO) or 1 µM bradykinin (BK) for 10 min prior to cell lysis. After lysis, cells were diluted 1:1 with media prior to the start of determination of cAMP.
Statistics
Results are expressed as a mean ± SEM for n observations. Comparisons are by Student’s paired t-test to determine statistical significance and are deemed significant when p ≤ 0.05. Confirmation of statistical significance was performed using a nested analysis of variance (ANOVA). Statistical analysis was performed using SigmaPlot (Jandel Scientific, San Rafael, CA) and JMP (Cary, NC).
Results
Receptor stimulation induces Bk2R/β2AR cross-talk
We first expressed Bk2R and β2AR individually in oocytes to gain insight into the electrophysiological waveforms of the ion channels activated by these receptors. β2AR can respond multiple times to ligand and repeatedly stimulate the protein kinase A (PKA)–activated CFTR chloride channel, used here to monitor Gαs-mediated signaling ( Fig. 1A ). Activation of CFTR is characterized by a slow increase in current, followed by a prolonged return to baseline. In contrast, Bk2R signals through the Gαq-coupled pathway to activate endogenously expressed calcium-activated chloride (Cl(Ca)) channels via intracellular calcium release ( Fig. 1B ). Activation of Cl(Ca) channels is characterized by a sharp, transient spike reflective of chloride exit from the cell, followed by a rapid return to baseline. In contrast to β2AR, Bk2R undergoes persistent desensitization ( Fig. 1B ) for up to 24 h via a protein kinase C (PKC)–independent mechanism (data not shown). This desensitization is not apparently at the level of the Cl(Ca) channels; 5HT2C-R can respond numerous times to ligand, activating Cl(Ca) channels repeatedly ( Fig. 2A ).
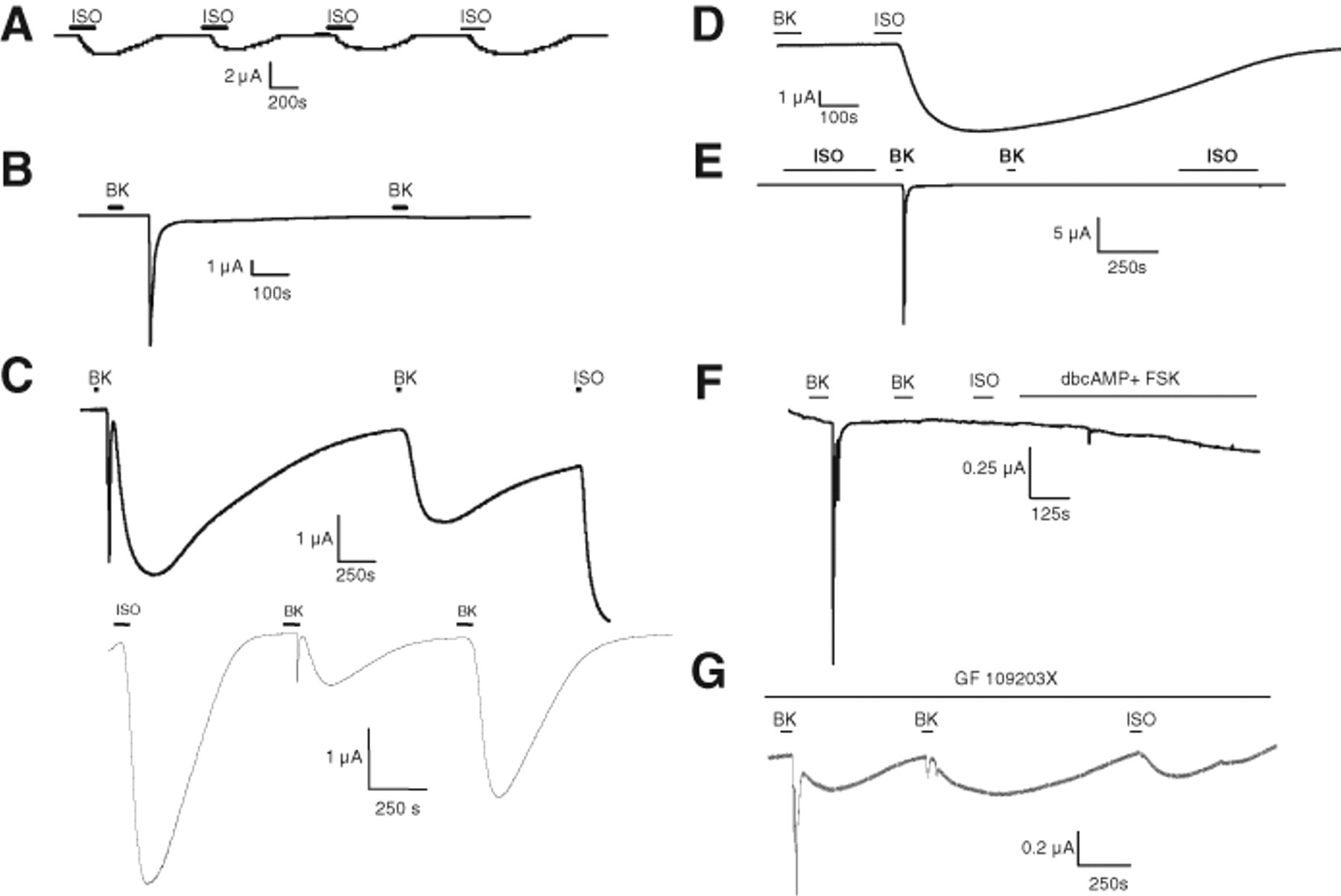
Bk2R and β2AR exhibit very different physiological profiles when expressed singly; upon coexpression, BK stimulation provokes novel signaling events. For each experiment, n > 4. (
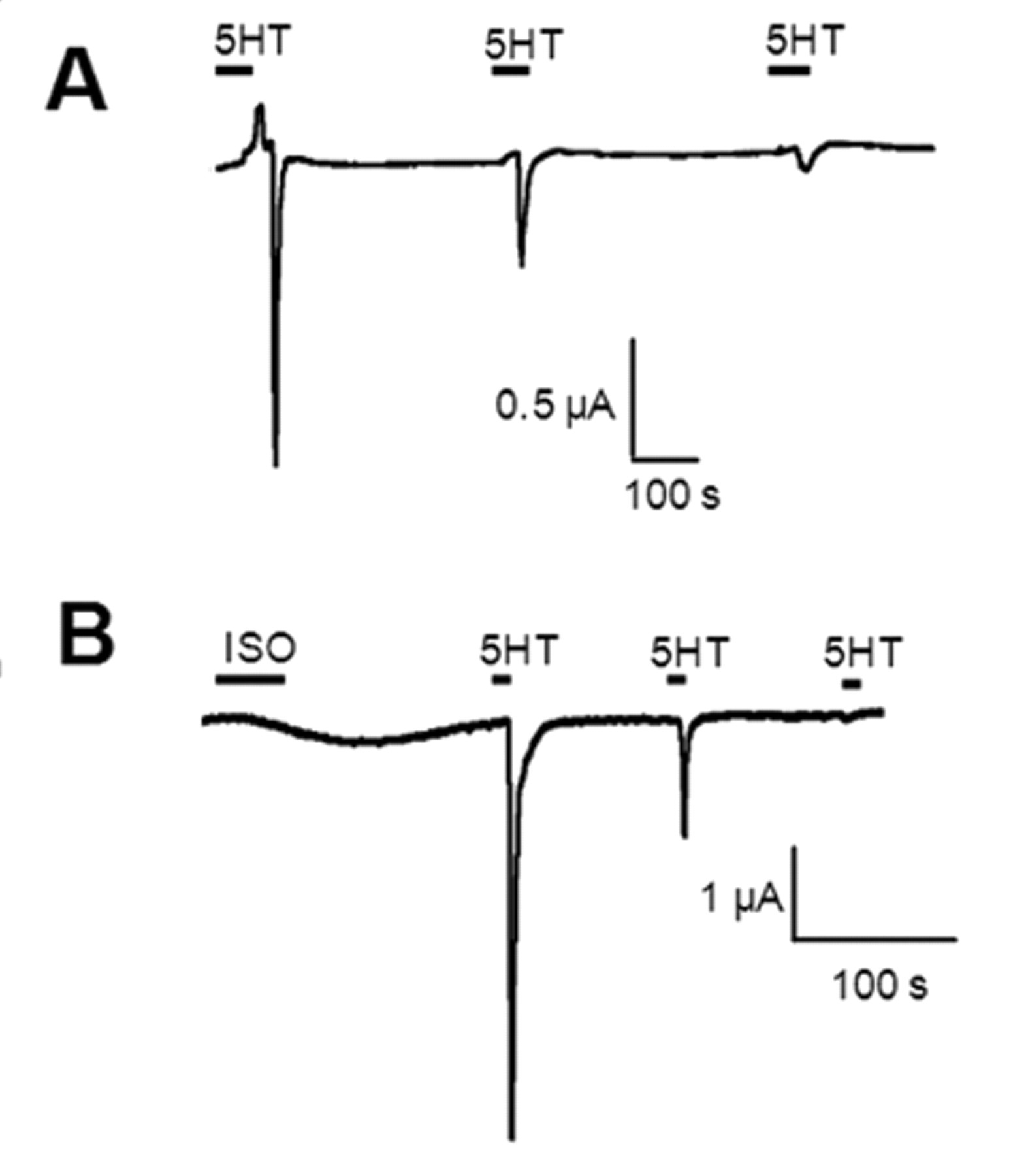
β2AR and 5HT2C-R do not functionally interact. (
In oocytes coexpressing Bk2R plus β2AR and CFTR, activation of Bk2R led to the appearance of novel BK-induced signaling, with Bk2R activating the Gαq pathway as expected but ostensibly also activating the Gαs pathway, potentially via stimulation of β2AR ( Fig. 1C ). The first stimulation with BK activated both the Gαq and the Gαs signaling pathways, as both the transient spike reflective of Cl(Ca) channel activation and the slow activation and deactivation characteristic of CFTR were observed. When these cells were stimulated with BK a second time, the Gαq-mediated activation of Cl(Ca) channels exhibited desensitization as expected from Figure 1B , but Gαs-mediated signaling persisted, as evidenced by activation of CFTR. Finally, stimulation with ISO also activated CFTR, confirming β2AR expression. Transactivation was independent of ligand order; stimulation with ISO first did not prevent cross-talk ( Fig. 1C , bottom). Transactivation such as that shown in Figure 1C has been observed in >50 cells expressing both receptors. One potential interpretation of these results is that stimulation of Bk2R in a Bk2R/β2AR heterodimer leads to the activation of β2AR directly. Other experiments, described below, were performed to test this interpretation.
Because the electrophysiological profile of 5HT2C-R was dissimilar to that of Bk2R despite both receptors coupling to Gαq, we asked if serotonin (5HT) was capable of transactivating β2AR ( Fig. 2B ). In oocytes expressing 5HT2C-R, β2AR, and CFTR, stimulation with 5HT did not activate β2AR or, apparently, any component of the Gαs pathway. In these cells, activation with ISO confirmed expression of β2AR and CFTR. These data suggest that the ligand-induced transactivation of β2AR by a Gαq-coupled receptor is specific to Bk2R.
Characterization of mechanism underlying Bk2R/β2AR cross-talk
We next sought to identify the role that β2AR plays in BK-induced transactivation of the Gαs signaling pathway by performing a number of controls to confirm ligand specificity and eliminate other potential mechanisms. First, to confirm that β2AR could not be activated directly by BK, oocytes expressing β2AR and CFTR alone were exposed to BK; these cells did not exhibit a response to BK but did respond to ISO ( Fig. 1D ). Next, cells expressing β2AR and Bk2R were tested to confirm that the Gαs-like response induced by BK stimulation was via CFTR, a PKA-activated chloride channel. Without expression of CFTR, exposure to BK activated only the transient current attributed to Cl(Ca) channels. No transactivation could be seen in these experiments because there are no endogenously expressed ion channels that are activated by components of the Gαs pathway in oocytes ( Fig. 1E ). Furthermore, the ability of one of these receptors to transactivate the other is unidirectional; stimulation with ISO did not activate Bk2R and therefore Cl(Ca) channels. We then addressed the possibility that BK-induced transactivation is caused by second messengers of the Gαq pathway activating the Gαs pathway indirectly. Specifically, a few studies have shown cross-talk between PKC and PKA pathways 14 ; thus, if Bk2R-mediated activation of Gαq pathway second messengers increased the intracellular levels of Gαs pathway second messengers, this could lead to activation of CFTR without requiring β2AR. However, in cells only expressing Bk2R and CFTR without β2AR, BK-induced activation of CFTR did not occur ( Fig. 1F ), strongly suggesting that BK-induced signaling via both Gαq and Gαs pathways requires the presence of β2AR. In these experiments, expression of CFTR was confirmed by stimulation of PKA-mediated activation with 5 µM dibutyryl-cAMP and 1 µM forskolin. In addition, these results show that transactivation seen previously ( Fig. 1C ) was not due to direct interaction of Bk2R and CFTR. To further discount the possibility of PKC-mediated increases in PKA activity, we preincubated cells expressing β2AR, Bk2R, and CFTR with 100 nM bisindolylmaleimide (GF 109203X, IC50 = 8-20 nM), a PKC inhibitor, for 10 min prior to and during exposure to BK. GF 109203X had no effect on transactivation, as both episodes of BK-induced activation of the Gαs pathway persisted ( Fig. 1G ). Taken together, these experiments strongly suggest that β2AR is required for Bk2R-mediated transactivation of the Gαs pathway and that transactivation does not occur due to cross-talk between signaling pathways downstream of the receptors.
Because β2AR can signal multiple times, we asked if co-expression of Bk2R alters β2AR pharmacology by injecting cRNAs encoding β2AR and CFTR and generating dose-response curves using terbutaline, a β2AR-selective agonist. Dose-response curves generated from oocytes expressing β2AR and CFTR alone provided a calculated EC50 of 10−7.40 M ( Fig. 3 ). Other studies have reported the EC50 of terbutaline to be as varied as 13.8 nM to 2.14 µM; our calculated EC50 is on the lower end of that spectrum. 15,16 Interestingly, coexpression of Bk2R along with β2AR and CFTR shifted the dose-response curve for β2AR rightward ( Fig. 3 ), increasing the EC50 to 10−6.93 M, suggesting that Bk2R dampens the ability of β2AR to signal through its own pathway. Because terbutaline is most likely activating both β2AR proteins that are in heterodimers as well as β2AR proteins that are in homodimers or monomers, we must consider the likelihood that the above-referenced results underestimate the effect of heterodimerization upon receptor function.
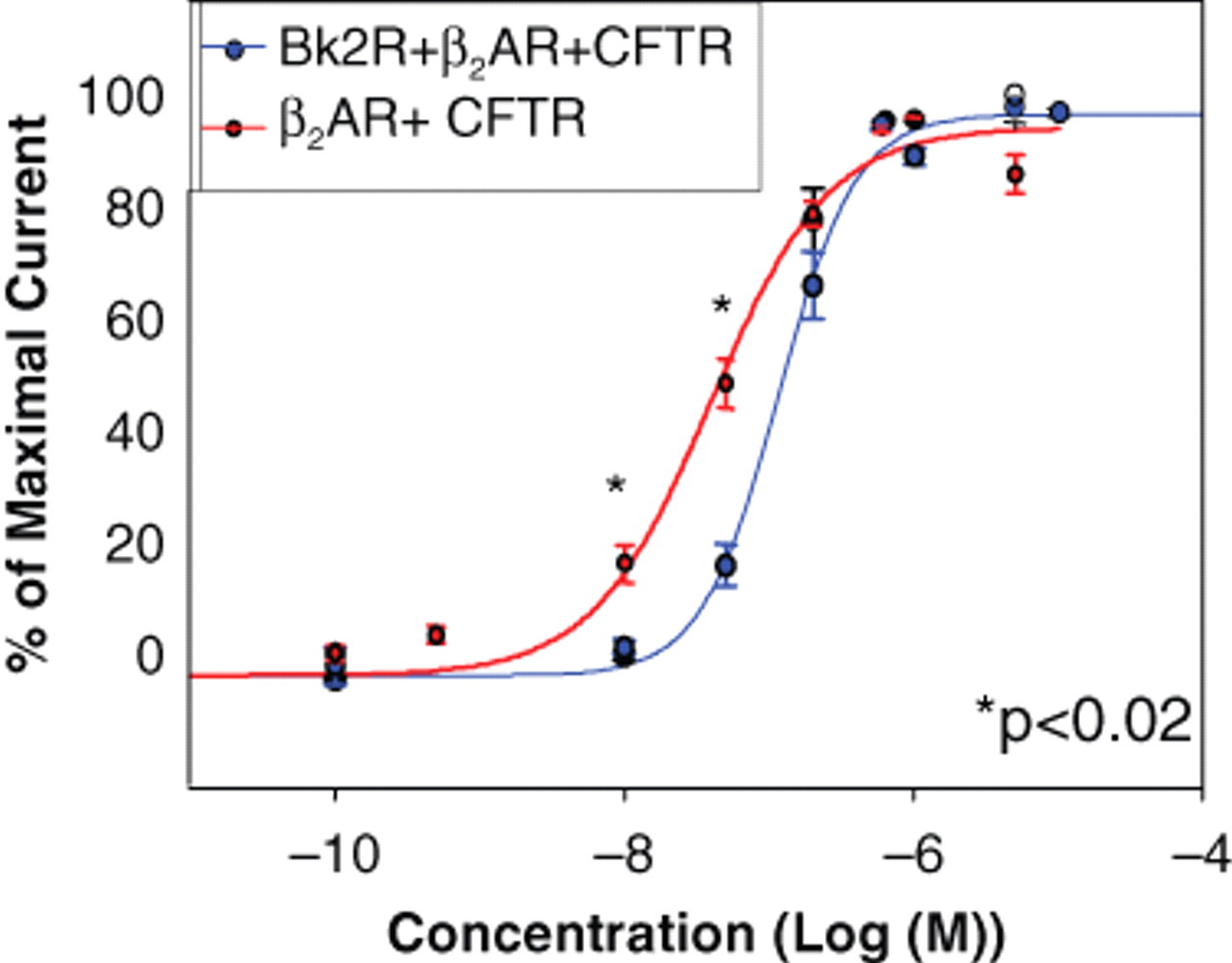
Dose-response curve for terbutaline in cells expressing either β2AR and cystic fibrosis transmembrane conductance regulator (CFTR) or Bk2R, β2AR, and CFTR. Maximal response to each concentration of terbutaline was measured at Vm = +40 mV.
Transactivation requires Bk2R and β2AR to undergo conformational changes
To further examine the mechanism of Bk2R transactivation of β2AR, we used the inverse agonists HOE-140 and propranolol to determine whether the active conformations of Bk2R and β2AR, respectively, are required for BK-induced transactivation of β2AR. Oocytes expressing Bk2R, β2AR, and CFTR were incubated with 5 µM propranolol for 10 min ( Fig. 4A , C ), then stimulated with BK or ISO in the continuing presence of propranolol. As shown in Figure 4A (left panel), cells incubated with propranolol after first BK stimulation were unable to activate β2AR or any component of the Gαs pathway via either ISO or BK. In the experiment shown in Figure 4A , cells were stimulated with BK and then ISO to confirm expression of all 3 cRNAs prior to repeated exposures to ligands in the presence of propranolol. If cells were instead incubated with propranolol prior to the first activation of ligand, Bk2R activation only produced the Gαq response of similar magnitude to the Gαq response in the absence of propranolol but did not activate the Gαs signaling response ( Fig. 4A , right panel); furthermore, a second exposure to BK in the presence of propranolol did not produce any response. These data indicate that the BK-induced activation of Gαs signaling via Bk2R/β2AR transactivation required β2AR to attain its activated conformation. As a control to confirm propranolol-induced block of β2AR, cells were exposed to ISO in the presence of propranolol, which caused no increase in CFTR-mediated current ( Fig. 4A ). Inhibition of Bk2R/β2AR transactivation by propranolol was significant, reducing the magnitude of the first BK-induced Gαs-mediated currents 5-fold and of the second BK-induced Gαs-mediated currents almost 7-fold ( Fig. 4C ). Similarly, incubation with HOE-140 also prevented transactivation ( Fig. 4B ). In the presence of HOE-140, oocytes expressing Bk2R, β2AR, and CFTR did not respond to BK, but ISO was still able to activate β2AR ( Fig. 4B , right panel). After HOE-140 was washed out, stimulation with BK activated both Gαq- and Gαs-coupled pathways, as expected. Figure 4C summarizes these data, showing that HOE-140 reduced the magnitude of the first BK-induced Gαs response 5-fold and of the second BK-induced Gαs response 3-fold.
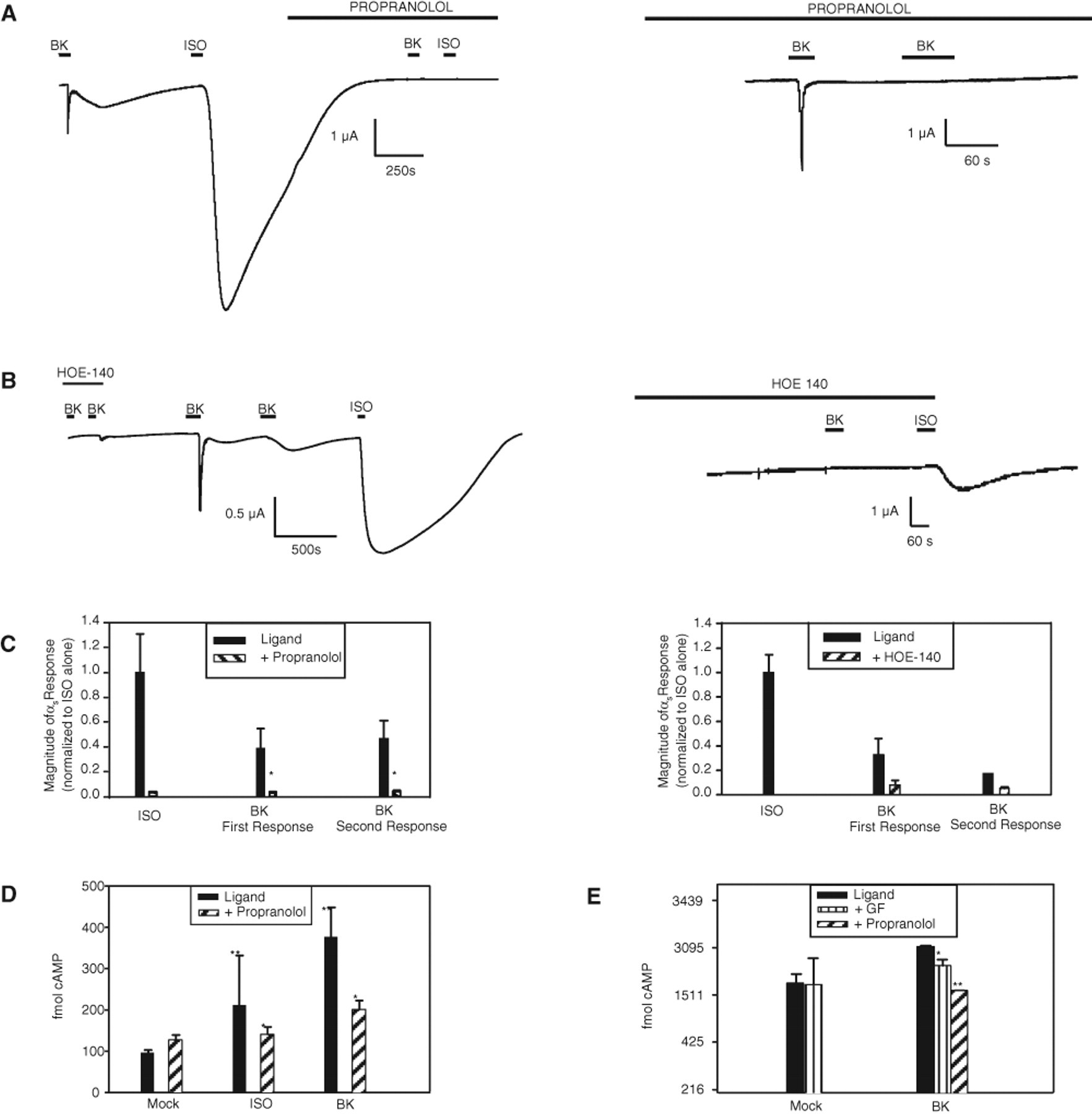
Transactivation requires conformational changes in both Bk2R and β2AR. (
Because these data strongly suggested that stimulation with BK led to transactivation of β2AR, we next asked if this functional interaction seen in oocytes also occurred when receptors are at endogenous expression levels by using EIA in PC12 cells to monitor changes in cAMP levels. PC12 cells were pretreated with 10 µM IBMX to prevent cAMP breakdown for 10 min prior to addition of either 5 µM ISO or 1 µM BK ( Fig. 4D ). Treatment with both BK and ISO elevated cAMP levels above basal levels, as expected. However, pretreatment with propranolol before addition of either ISO or BK significantly dampened the response to both ligands (p < 0.05), suggesting that β2AR mediates BK-induced increases in cAMP levels in PC12 cells.
Graness and others 17 also have shown that BK can mediate increases in cAMP in PC12 cells, but they concluded that this was via PKC-mediated activation of the Gαs pathway. Therefore, we also examined the role that PKC may play in BK-mediated increases in cAMP concentration by comparing the inhibitory effects of GF 109203X and propranolol on BK-mediated increases in cAMP. Graness and coworkers preincubated PC12 cells with 100 µM IBMX and 1.25 µM GF 109203X; for these experiments, we preincubated PC12 cells with 50 µM IBMX and 4.1 µM GF 109203X for 10 min prior to the addition of ligand to compare with inhibition of BK-mediated increases in cAMP by 5 µM propranolol ( Fig. 4E ). Similar to previous reports, GF 109203X did decrease the amount of cAMP accumulated in response to BK-induced stimulation. However, preincubation with propranolol caused a more dramatic decrease in cAMP in response to BK compared to GF 109203X (p < 0.05), suggesting not only that there is more than one mechanism responsible for BK-mediated increases in cAMP in PC12 cells but also that Bk2R transactivation of β2AR is the major route for the BK-induced increase in the abundance of this Gαs pathway second messenger. These results combined illustrate that the data collected from expression in oocytes can be confirmed in an endogenous expression system, supporting the validity of TEVC as an appropriate screening tool for GPCR heterodimerization.
Bk2R and β2AR physically associate
Bk2R, a Gαq-coupled receptor, and β2AR, a Gαs-coupled receptor, have been shown to interact with other receptors within their same classes, 4,18 but it is not known if these 2 receptors form heterodimers with each other. Given our data suggesting that Bk2R and β2AR are functionally interacting, we next asked if these 2 receptors are physically coupled by attempting to co-IP these proteins from cells expressing them heterologously or at native levels.
We first tested the specificity of our antibodies by performing some control experiments. Because CHO cells express a small amount of β2AR and no Bk2R, we performed co-IP experiments in these cells to test for nonspecific binding of either the β2AR antibody conjugated beads or the anti-Bk2R antibody 19 (data not shown). In lysed CHO cells that were immunoprecipitated with an anti-β2AR antibody and immunoblotted with an anti-Bk2R antibody, no bands were detected. A faint band was detected in CHO cell lysate that was both immunoprecipitated and immunoblotted with an anti-β2AR antibody, confirming its endogenous expression in CHO cells. We next confirmed that there was not any cross-reactivity between the anti-β2AR and anti-Bk2R antibodies by performing the co-IP protocol in the absence of any cell lysate (data not shown). No bands were detected on the nitrocellulose membrane, confirming that there is no cross-reactivity between anti-Bk2R and anti-β2AR antibodies.
We examined the extent of interaction of Bk2R and β2AR using co-IP in cells transiently expressing both receptor subtypes. Xenopus oocytes injected with cRNAs encoding HA-tagged Bk2R and β2AR and shown to be expressing these proteins by electrophysiological experiments were lysed under mildly reducing conditions, and receptor complexes subsequently were immunoprecipitated with anti-β2AR antibodies. Immunoblot analysis with anti-Bk2R antibody revealed the presence of a ~95- to 100-kDa β2AR/Bk2R immunoreactive band ( Fig. 5A , left panel). These experiments were also repeated in uninjected oocytes, and no visible bands were detected ( Fig. 5A , middle panel). We also performed the inverse experiment, wherein we immunoprecipitated using an anti-HA antibody and immunoblotted using an anti-β2AR antibody; the heterodimers band was faint but present ( Fig. 5A , right).
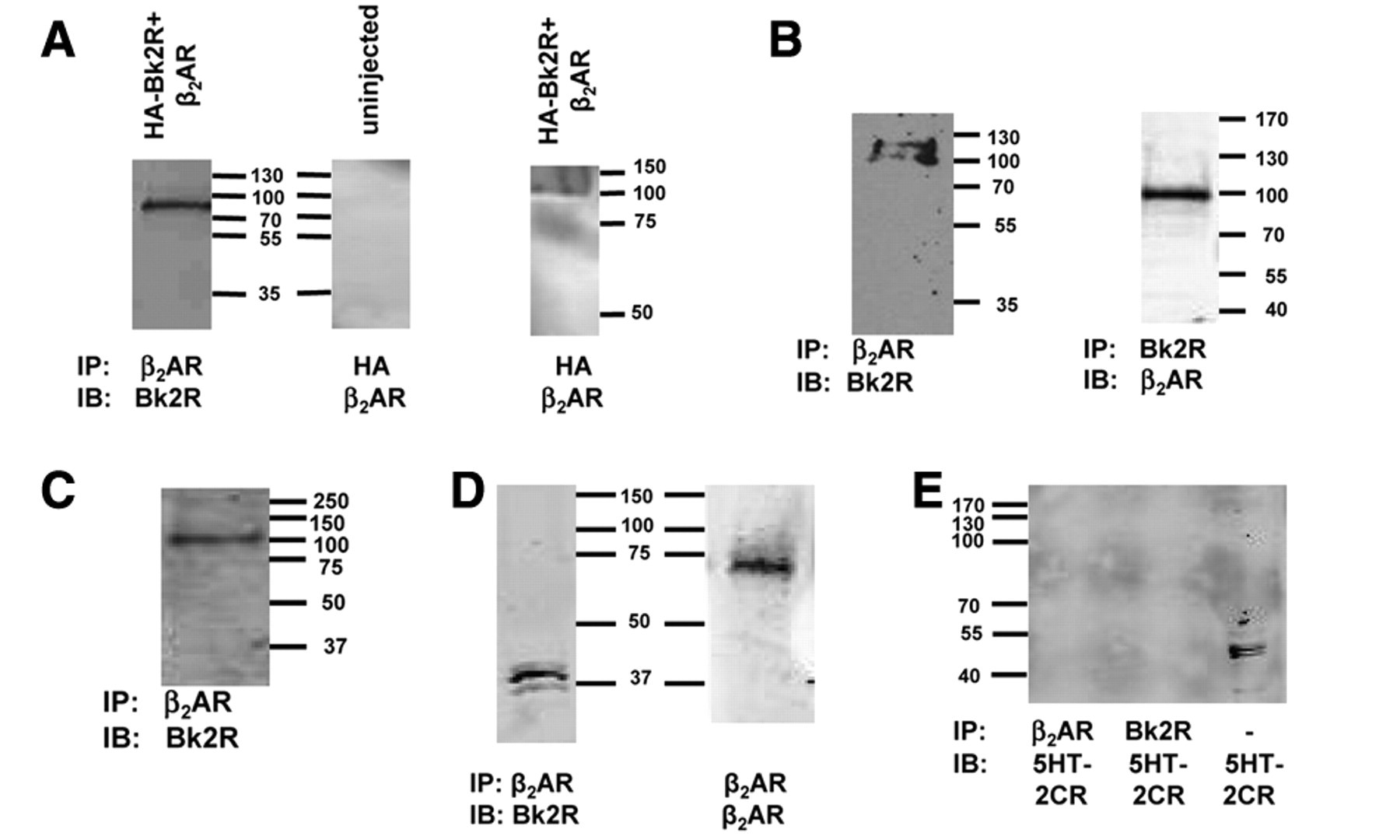
Bk2R and β2AR physically associate. Receptor interactions were assessed by co-immunoprecipitation (co-IP); for each of these experiments, Bk2R monomers were predicted to be ~40 kDa, β2AR monomers were predicted to be ~63 kDa, and Bk2R/β2AR heterodimers were predicted to be ~95 kDa. (
Because our data suggest that there is a correlation between functional interaction and physical association, we next asked if Bk2R/β2AR heterodimers were observed in cells endogenously expressing both receptors. Following immunoprecipitation of endogenous β2AR in rat PC12 cells, immunoblotting with anti-Bk2R antibody revealed the presence of bands representing the heterodimer at ~100 kDa ( Fig. 5B , left panel). We also performed the inverse experiments, immunoprecipitating using an anti-Bk2R antibody and immunoblotting with an anti-β2AR antibody. Again, the ~100-kDa band representing the Bk2R/β2AR heterodimer was apparent ( Fig. 5B , right panel), supporting the conclusion that these 2 receptors heterodimerize.
Because both Bk2R and β2AR are highly expressed in cardiovascular tissues, we next examined their physical interaction in murine ventricular cardiac myocytes. Co-IP experiments in cardiac myocytes suggested that Bk2R and β2AR also heterodimerize in these cells ( Fig. 5C ).
In the experiments described thus far, Bk2R and β2AR heterodimers were detected under mildly reducing conditions, suggesting a strong interaction between these 2 receptors. To determine the strength of the Bk2R/β2AR interaction, we performed co-IP in PC12 cells and added a 3-fold higher concentration of reducing agent during elution of the co-IP receptor complex. As shown in Figure 5D , the heterodimer could be disassembled under strongly reducing conditions, but both Bk2R and β2AR were still detected.
To further validate the correlations between physical association and functional interaction, we next asked if 5HT2C-R physically associated with β2AR despite there being no functional interplay between these 2 receptors. From PC12 cells, we immunoprecipitated using either anti-β2AR or anti-Bk2R antibodies and immunoblotted against endogenously expressed 5HT2C-R ( Fig. 5E ). Neither Bk2R or β2AR co-immunoprecipitated with 5HT2C-R, but the presence of 5HT2C-R in these cells was confirmed in total lysate. Taken together, our data suggest a strong corollary between functional interaction and physical association for Bk2R and β2AR when expressed heterologously or endogenously.
Discussion
An increasing number of GPCRs have been shown to oligomerize both in vitro and in vivo. A small percentage of these GPCR dimer pairs arise from different signaling classes, and an even smaller set has shown evidence of functional consequences arising from their interaction. 6,9,20,21 Here, we show the first example of physical association of the Gαq-coupled Bk2R and the Gαs-coupled β2AR in both endogenous and transient expression systems. Furthermore, this is the first example of Bk2R-mediated transactivation of the Gαs signaling pathway via its direct association with β2AR in both endogenous and heterologous expression systems. Coexpression of Bk2R along with β2AR changed the ability of β2AR to respond to its selective ligand, suggesting that Bk2R acts as a negative allosteric modulator of β2AR.
Use of electrophysiological methods in oocytes to examine receptor interactions has a number of advantages over biochemical methods in cultured cells. Many studies that investigate GPCR dimerization used transfection of cDNAs of interest into a cultured mammalian cell line, followed by application of biochemical techniques to examine fluctuations in second-messenger concentration or changes in ligand binding. Although somewhat effective, these methods can be both time-consuming and inaccurate. Because oocytes express few membrane proteins, it is possible to identify the effects of GPCR coexpression and dimerization more readily than in other systems because the output (ion channel activity) is correlated only to the activation of a given receptor. Thus, in contrast to biochemical methods that measure GPCR dimerization by global changes in second-messenger abundance (which could potentially be influenced by other proteins expressed in these same cells), ion channel activity is only modulated by fluxes in second-messenger abundance large enough to be from activation of the receptor of interest. Using the oocyte method also provides a faster way of identifying potential interactions between GPCRs; channel activity indicative of receptor activation is noticed typically within seconds of ligand addition. Furthermore, the oocyte system does not require cell lysis to obtain results, which can be highly useful for comparing responses to increasing concentrations of ligand or comparing the magnitude of response to different ligands within a single cell. Given the number of advantages of the oocyte system, high-throughput formats of Xenopus oocyte electrophysiology are currently available. Molecular Devices (Sunnyvale, CA) manufactures the OpusXpress 6000A, an automated parallel 2-electrode voltage clamp electrophysiology system that can test 8 cells simultaneously while controlling chamber volume. Thus, it would be possible to identify other GPCR heterodimers using the oocyte system in either single or high-throughput formats.
The data presented here obtained from oocytes are congruous to the data from PC12 cells. This suggests that the oocyte system may provide an accurate recapitulation of an endogenous expression system. There have been instances in which published data characterizing GPCR heterodimerization in a cultured cell line transiently expressing GPCRs of interest were not successfully reproduced by other labs, 22 likely due to differences in abundance of heterologously expressed receptors. Even though the oocyte system also requires exogenous addition of cRNAs encoding GPCRs, only a small amount (nanograms) of cRNA is required to get detectable expression. This is because channel activation amplifies the GPCR’s response to ligand. Thus, our data show that the oocyte system is a reliable and accurate method in which to study the functional consequences of GPCR dimerization.
The present study suggests that the mechanism underlying transactivation involves direct receptor-receptor interaction. Our data showing that inverse agonists against either Bk2R or β2AR inhibit transactivation, as well as activation of cognate signaling pathways, indicate that both receptors must be able to adopt an active conformation in order for stimulation of Bk2R to activate β2AR. Hence, one possible interpretation is that ligand binding to Bk2R induces a conformational change in this receptor, leading to docking and activation of Gαq at this receptor’s cytoplasmic domain, and the physical association of the 2 receptors allows the change in Bk2R conformation to be transduced to the β2AR, inducing a similar conformational change in that receptor that then leads to docking and activation of Gαs at its cytoplasmic domain. The first exposure to ligand leads to desensitization of Bk2R with respect to activating its own cognate signaling pathway, most probably due to phosphorylation of the Gαq-binding site. However, the receptor remains at the plasma membrane and can still undergo ligand-induced conformational change upon second exposure to BK, such that the desensitized Bk2R is still capable of transactivating β2AR. Stimulation with BK facilitates Bk2R-mediated signaling through the other member of the dimer and allows subsequent signaling events to occur after the native signaling pathway of Bk2R has been desensitized, thereby providing an additional mechanism of activating Gαs. Further studies are required to identify the ligand-binding properties of the Bk2R/β2AR heterodimer, as compared to individual receptors, and to determine whether receptor stimulation affects the oligomerization state of the Bk2R/β2AR heterodimer.
Here, we provide evidence of heterodimerization of Bk2R and β2AR, including BK-induced transactivation of β2AR and its signaling pathway, and evidence that Bk2R also acts as a negative allosteric modulator of β2AR; these interactions between these receptors may underlie the side effects seen with ligands thought to be selective for one receptor or the other. Bk2R and β2AR are coexpressed in a number of locations in the body, including cardiac and vascular tissues. Both receptors play regulatory roles in cardiac function, and both receptors are less effective in states of cardiac disease. Others already have shown that beta-blockers also reduce ATR1 signaling ability. 9 Alternatively, the ability of Bk2R to also modulate β2AR may prove beneficial by providing an alternative route to the stimulation of Gαs-mediated signaling. Using the Xenopus oocyte system to gain an understanding of the functional consequences of GPCR heterodimerization may provide insight into the concurrent regulation of signaling pathways in any given system but may also present a method of screening novel targets for existing and emerging therapeutics. Given the clinical importance of pharmaceutics that affect Bk2R and β2AR, our results may provide new approaches for future drug development or refinement of existing drugs.
Footnotes
Acknowledgements
We thank Dr. R. A. Hall for helpful discussions and insights on this project and Dr. J. M. Hansen for his technical expertise. This project was supported in part by a seed grant from the Emory-Egleston Children’s Research Center.
