Abstract
FluxOR™ technology is a cell-based assay used for high-throughput screening measurements of potassium channel activity. Using thallium influx as a surrogate indicator of potassium ion channel activity, the FluxOR™ Potassium Ion Channel Assay is based on the activation of a novel fluorescent dye. This indicator reports channel activity with a large fluorogenic response and is proportional to the number of open potassium channels on the cell, making it extremely useful for studying K+ channel targets. In contrast to BTC-AM ester, FluxOR™ dye is roughly 10-fold more thallium sensitive, requiring much lower thallium for a larger signal window. This also means that the assay is carried out in a physiological, normal-chloride saline. In this article, the authors describe how they used BacMam gene delivery to express Kv7.2 and 7.3 (KCNQ), Kir2.1, or Kv11.1 (hERG) potassium ion channels in U2-OS cells. Using these cells, they ran the FluxOR™ assay to identify and characterize channel-specific inhibitory compounds discovered within the library (Tocriscreen™ Mini 1200 and Sigma Sodium/Potassium Modulators Ligand set). The FluxOR™ assay was able to identify several known specific inhibitors of Kv7.2/7.3 or hERG, highlighting its potential to identify novel and more efficacious small-molecule modulators.
Introduction
I
Kv7.2/7.3 (KCNQ2 and KCNQ3) coassemble to underlie an “M current” in the CNS, 3 and mutations cause benign familial neonatal convulsions (BFNC), dominantly inherited epilepsy, and myokymia. 4-6 hERG (Kv11.1, KCNH2) is a widely screened channel that encodes a voltage-gated potassium channel known to repolarize cardiac muscle following an action potential. Rigorous hERG screening and safety testing is required of all new drugs in preclinical development. 7 Kir2.1 is a strong inward rectifier expressed predominantly in heart, brain, and skeletal muscle. Kir2.1 mutations underlie the dysmorphic features, periodic paralysis, and cardiac arrhythmias of Anderson’s syndrome. 8
Using thallium influx as a surrogate indicator of potassium ion channel activity, we describe here our efforts with the FluxOR™ Potassium Ion Channel Assay. The FluxOR™ assay is based on the activation of a novel fluorescent dye with a high affinity for thallium. This indicator reports channel activity with a large fluorogenic response and is proportional to the number of open potassium channels on the cell, making it extremely useful for studying K+ channel targets.
In contrast to BTC-AM ester, 9,10 FluxOR™ dye is roughly 10-fold more thallium sensitive, requiring much lower thallium concentrations for a larger signal window. Importantly, this means that the assay can be carried out in physiological saline with normal chloride concentrations. This was an important limitation of BTC, for which chloride-free conditions were necessitated by the solubility coefficient of thallium chloride, which is about 4 mM at physiological pH.
In the present study, we used BacMam gene delivery to express these 3 different potassium ion channel targets in U-2 OS cells. We then ran the FluxOR™ assay on the cells to identify and characterize channel-specific inhibitory compounds discovered within the library (Tocriscreen™ Mini 1200 and Sigma Sodium/Potassium Modulators Ligand set).
Methods and Materials
Cell culture
The human U-2 OS cell line was obtained from the American Type Culture Collection (ATCC number HTB-96; ATCC, Manassas, VA) and maintained and cultivated twice weekly in McCoy’s 5A Medium Modified Growth Serum (GIBCO 12330; GIBCO, Carlsbad, CA) with fetal bovine serum (Cosmic Calf FBS; Hyclone, Logan, UT) at a final concentration of 10%. Cells were cultured in 5% CO2 at 37 °C, and subcultures were made with divalent-free phosphate-buffered saline (PBS) rinse followed by dissociation with TrypLE™ Express (Invitrogen, Carlsbad, CA). A Countess™ automated cell counter (Invitrogen) was used to quantitate the cell numbers before plating into Greiner poly-D-lysine-coated 96- or 384-well microplates.
BacMam K channel transduction using BacMam
For multiplicity of infection (MOI) studies: U-2 OS cells were harvested for resuspension in divalent-free PBS at 1 × 106 cells/mL. Aliquots were prepared in 15-mL tubes with 1 mL of cells (106) and viral stock needed to achieve the indicated MOI. This suspension was incubated at room temperature for 60 min before adding complete medium and plating.
For screening: U-2 OS cells were trypsinized and transduced in fresh-10 cm dishes as follows: 2.5 × 106 cells in 9.5 mL McCoy’s Media (Invitrogen) + 10% fetal calf serum (FCS; Hyclone) were used per 10-cm dish. BacMam ion channel targets were added at 10% v/v (1 mL of BacMam stock at approximately 2 × 108 pfu/mL) when cells were diluted for plating. For the Kv7.2/7.3 dual transduction, 8 mL media was used, and the BacMam constructs for each channel were added at a 1:1 ratio (1 mL each for a total of 2 mL). Cells were incubated at 37 °C with 5% CO2 for 5 to 6 h. Following incubation, cells were trypsinized and plated in 384-well plate(s) at 5000 cells/well in 30 µL media.
Compound preparation and handling
The Tocriscreen™ Mini collection of 1120 compounds (Tocris Bioscience, Bristol, UK) and Sigma Sodium/Potassium Modulators Ligand set collection of 69 compounds (Sigma-Aldrich, St. Louis, MO) were aliquoted into four 384-well plates at a 5-mM concentration in 100% DMSO. These master plates were used to make a working stock plate of compounds at a 100-µM concentration in Hank’s Buffered Saline Solution (Invitrogen) and 10% DMSO (Sigma-Aldrich).
The compounds were added to the screening plates as a 10× solution yielding a 10-µM final concentration. The final DMSO concentration was 1%.
Potassium channel assay
The FluxOR™ potassium channel assay was performed as outlined in the product information sheet and performed on the Hamamatsu FDSS 6000 (Hamamatsu Photonics, K.K., Hamamatsu, Japan). Briefly, FluxOR™ loading buffer was made from Hank’s Balanced Saline Solution (HBSS) buffered with 20 mM HEPES and pH adjusted with NaOH to 7.4. Powerload™ concentrate and water-soluble probenecid were used as directed by the kit to enhance the dye solubility and retention, respectively. Media were removed from the cell plates manually, and 20 µL of loading buffer containing the FluxOR™ dye mix was applied to each well with a Multidropper Combi (Thermo Fisher, Waltham, MA). Once inside the cell, the nonfluorescent AM ester form of the FluxOR™ dye was cleaved by endogenous esterases into a thallium-sensitive indicator. The dye was loaded for 60 min at room temperature and then removed manually. The cell plates were subsequently washed once with dye-free assay buffer, before adding a final volume of 20 µL assay buffer containing water-soluble probenecid. Cell plates received 2 µL per well of the screening compounds, delivered with a Hamamatsu FDSS 6000 (Hamamatsu Photonics, K.K.), and were then incubated at room temperature (23-25 °C) for 30 min to allow equilibration of the test compounds in the cultures. Prior to injection, stimulation buffer was prepared from the 5× chloride-free buffer, thallium, and potassium sulfate reagents provided in the kit to contain 10 mM free thallium (5 mM Tl2SO4) and 50 mM free potassium (25 mM K2SO4). These concentrations resulted in final added concentrations of 2 mM free Tl+ and 10 mM free K+ after 1:5 dilution upon injection of the stimulus buffer into cells that had been loaded with FluxOR™ dye.
Data analysis
Data for the FluxOR™ screen were analyzed using the CeuticalSoft™ program (CeuticalSoft, Hudson, NY). The max-min fluorescence value for each well was determined from a 25-s wide window spanning 1 s preinjection and 24 s postinjection. Hits were chosen as greater then 50% inhibition when compared to DMSO controls. Autofluorescence was determined by measuring the change in fluorescence when compounds were added to the plate. If the baseline fluorescence increased by more then 50 RFUs upon compound addition, the compound was determined to be autofluorescent and not included in further analysis. Eighteen compounds were identified as autofluorescent and disregarded, and 10 µM cisapride functioned as a positive control for hERG channels. DMSO at the same concentration used for compound addition served as the negative control for all channels assayed. Values were graphed using GraphPad® Prism (GraphPad Software, San Diego, CA).
Dose-response analysis and hit confirmation
Selected compounds determined as “hits” from the screen were picked from the library comprising the Tocriscreen™ Mini 1120 (Tocris Bioscience) plus Sigma Sodium/Potassium Modulators Ligand set (Sigma-Aldrich) and diluted over an 8-point dose-response curve. Each compound was measured in 4 replicates. Automatic fitting routines in GraphPad® Prism (GraphPad Software) were subsequently used to define the dose-response activity and associated apparent IC50 values.
Results and Discussion
Cells were transduced with the appropriate potassium channel target as outlined in Methods and Materials and plated into 384-well microplates for use the following day. Cells were loaded with FluxOR™ at room temperature, treated with compound, and assayed. hERG, Kv7.2/7.3, and Kir2.1 were assayed by adding stimulus buffer with thallium and 10 mM additional KCl. To determine the transduction conditions that resulted in the largest assay window, we tested different amounts of virus per cell and cell number per well. U-2 OS cells were transduced with BacMam Kir2.1 (KCNJ2) at approximately 200 MOI and incubated overnight. The cells were harvested and plated at densities of 200, 500, 1000, 2000, 5000, and 10,000 cells/well into a Greiner poly-D-lysine-coated 384-well microplate. Cells were allowed to adhere for 2 h before FluxOR™ dye loading. The FluxOR™ potassium channel assay was run on the FDSS 6000 as described in Methods and Materials. Responses were recorded as normalized departure from baseline (ΔF/F) at 60 s after the stimulus, obtaining values of 0.22 ± 0.04, 0.30 ± 0.04, 0.55 ± 0.06, 1.06 ± 0.06, 1.05 ± 0.05, and 0.91 ± 0.05 in correspondence with cell density (
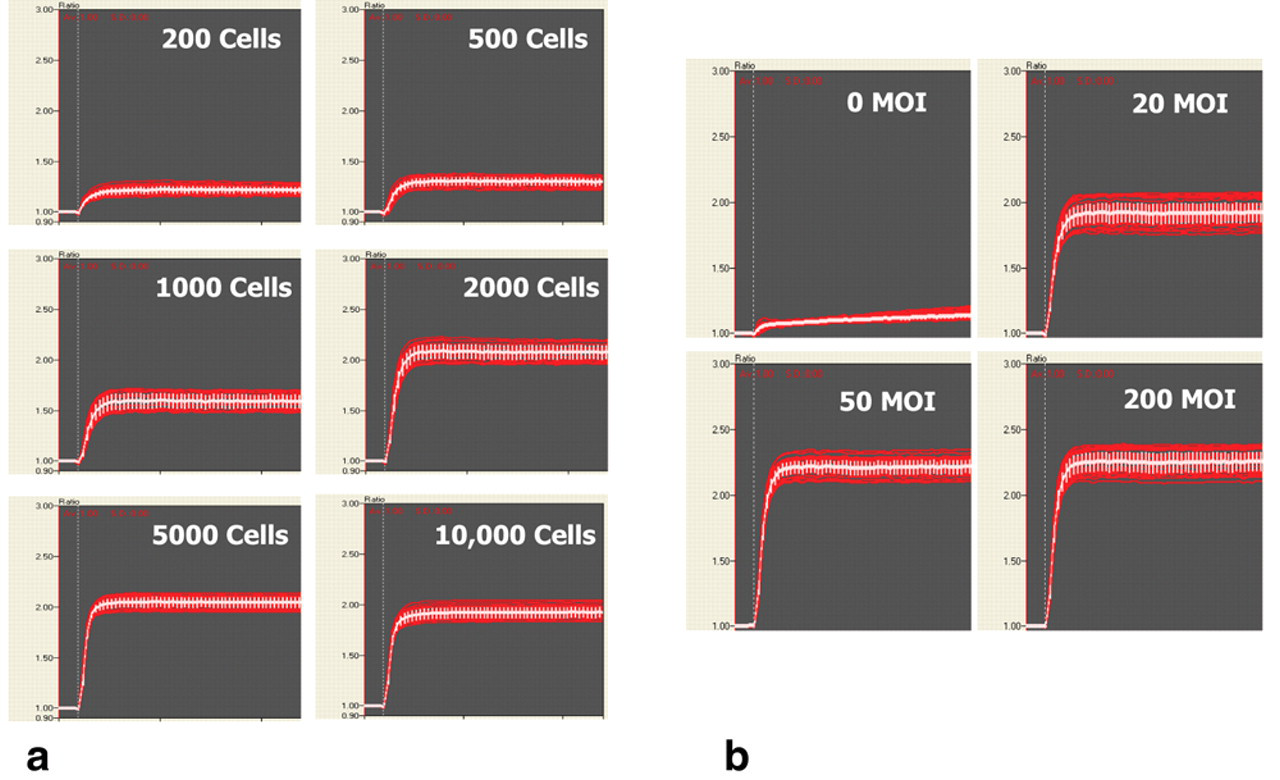
(
Cells were also treated to differing numbers of viral particles per cell. U-2 OS cells were transduced with BacMam Kir2.1 stock solutions at approximately 0, 20, 50, and 200 MOI and plated at 5000 cells/well into a 384-well plate and incubated overnight. The following day, cells were assayed for a potassium channel–mediated thallium response using FluxOR™. Responses were (ΔF/F) 0.11 ± 0.02, 0.90 ± 0.05, 1.20 ± 0.05, and 1.27 ± 0.06. We found that at 5000 cells per well, ~200 viral particles per cell resulted in the largest assay window (
To demonstrate that the FluxOR™ assay can be an effective high-throughput screening (HTS) reagent, we performed a screen of a library consisting of the compounds in the Tocriscreen™ Mini 1200 and Sigma Sodium/Potassium Modulators Ligand collection. We used BacMam transduced cells expressing functional hERG, Kv7.2/7.3, and Kir2.1 channels as described in Methods and Materials. Screening results are shown in
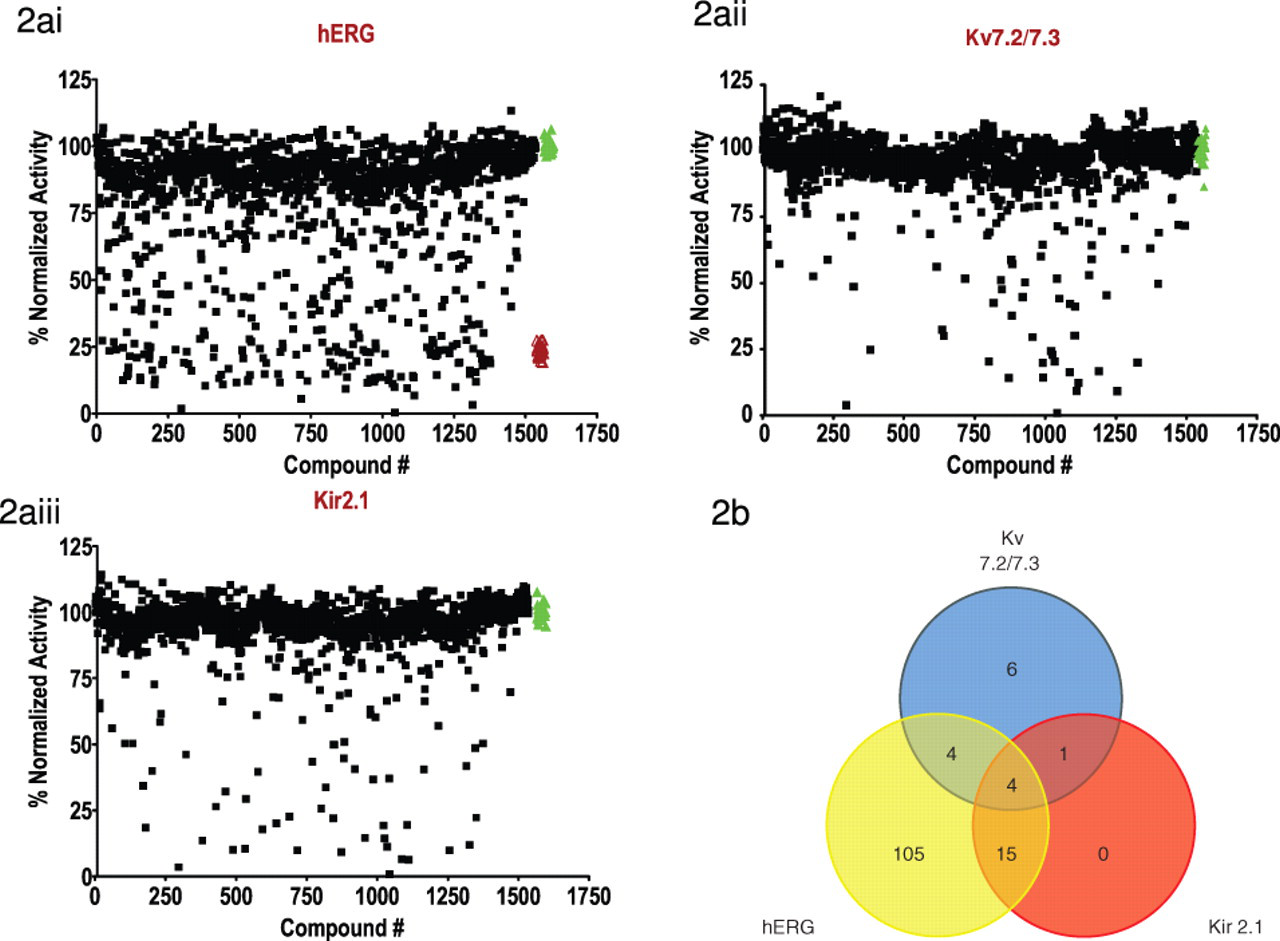
Results from the library screen of U-2 OS cells expressing Kv7.2/7.3, Kir2.1, or hERG using FluxOR™. (
To validate the feasibility of the FluxOR™ assay as a screening tool for potassium channels, we went on to test the pharmacology of known potassium channel modulators identified as hits in
Apparent IC50s across hERG and Kv7.2/7.3

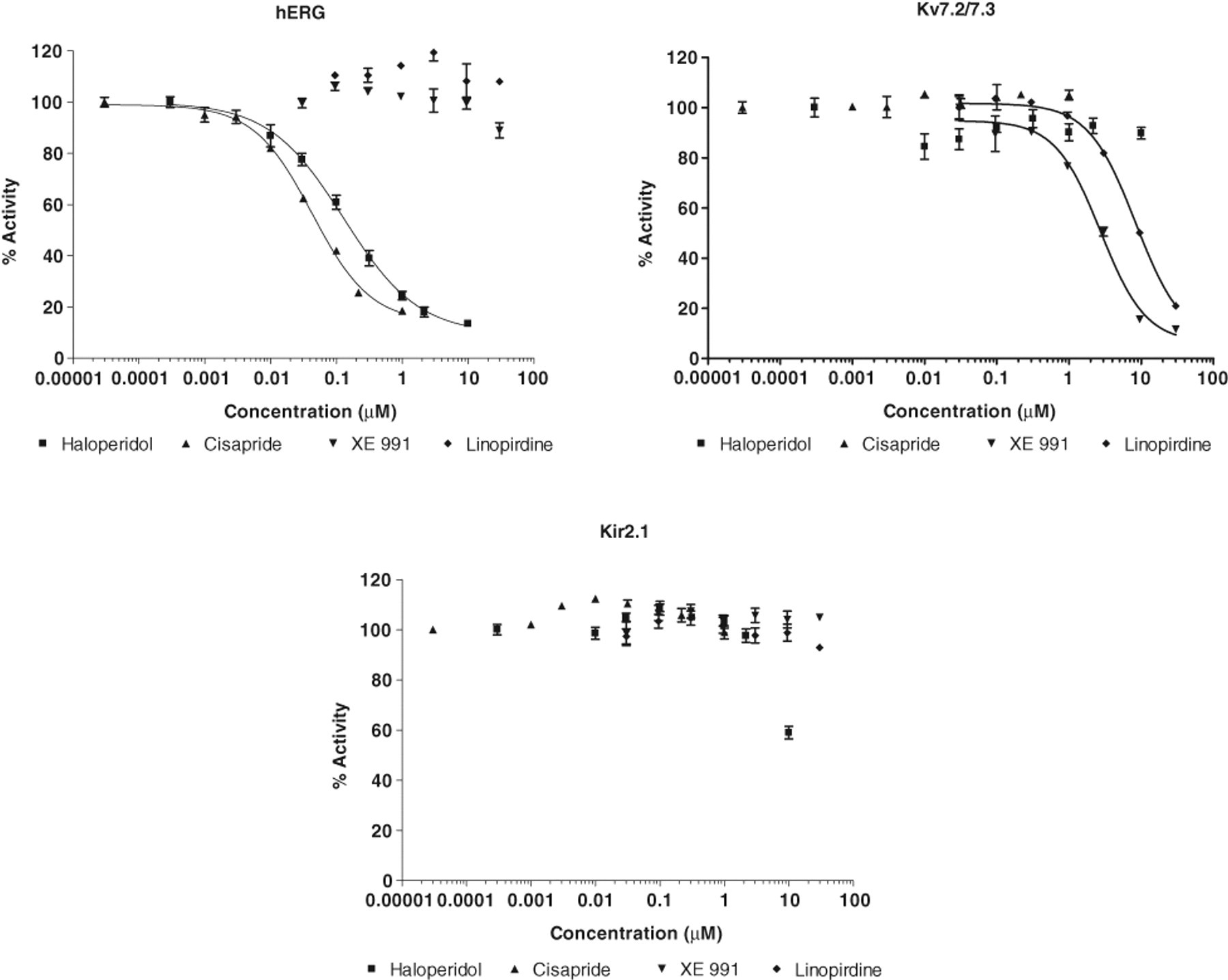
Dose response for inhibition of the FluxOR™ signal by haloperidol, cisapride, XE991, and linopiridine at hERG, Kv7.2/7.3, or Kir2.1 channels. Channel activity with no inhibitor present is defined as 100%. For each sample, the max-min fluorescence value was calculated and then normalized, with the largest change being set to 100%. Each point is the average of 4 replicates, and error bar represents the standard error of the mean.
Summary
HTS is a highly evolved and increasingly automated environment with large unmet needs in the field of ion channels. Potassium channels in particular are an important class of membrane proteins implicated in a variety of cardiac and CNS diseases; they are expressed in virtually every mammalian cell and regulate electrical excitability and synaptic communication. Potassium channels represent a class of drug target that has thus far proven intractable to HTS interrogation.
The purpose of this study was to show that the signal obtained with the FluxOR™ potassium channel assay can be easily incorporated into a high-throughput workflow while showing pharmacological fidelity and a predictive capacity. A screen of 3 different potassium channels showed that FluxOR™ is able to identify several known specific inhibitors of Kv7.2/7.3 or hERG channels, highlighting its potential to identify novel and more efficacious small-molecule modulators. When a subset of the hits was further studied, the IC50 values we obtained for these compounds, as measured in the fluorescent FluxOR™ assay, were in agreement with those reported in electrophysiology recordings, confirming the functionality of this assay platform.
