Abstract
CD36, a member of the class B scavenger receptor, is a high-affinity receptor for oxidatively modified low-density lipoprotein (oxLDL). Extensive evidence points to a significant role of CD36 in atherosclerosis and suggests that CD36 could be a potential target for treatment of atherosclerosis. Here, the extracellular domain of human CD36 (Gly30-Asn439) was expressed in Escherichia coli as His6-tagged soluble CD36 (sCD36), which could bind oxLDL specifically and effectively inhibit the uptake of oxLDL by murine macrophage RAW 264.7 cells. An enzyme-linked immunosorbent assay (ELISA)–like high-throughput screening (HTS) assay was developed for the discovery of CD36 antagonists, based on the competition of sCD36 binding to immobilized oxLDL and detection with a monoclonal antibody against His-tag. This assay was suitable for HTS in a 96-well format and was robust and reliable according to the evaluation parameter Z′ value of 0.82. The developed HTS assay was applied to both pure chemical compounds and microbial secondary metabolite crude extracts to identify CD36 antagonists. Three active compounds—sodium danshensu (DSS), rosmarinic acid (RA), and salvianolic acid B (SAB)—were shown to be antagonistic to sCD36-oxLDL binding and further validated by their inhibition of oxLDL uptake in RAW 264.7 cells. These results suggest that the ELISA-like assay represents a promising screening for identifying bioactive molecules targeting atherosclerosis at the level of CD36-ligand binding.
Introduction
A
Human CD36 is an 88-kDa heavily N-linked glycosylated single-chain transmembrane protein found on the surface of many cell types. 2,3 It consists of 472 amino acids comprising a large extracellular domain and, in all likelihood, 2 transmembrane α-helices, one each at the amino and carboxy terminal. 8,9 CD36 binds many ligands in addition to modified low-density lipoprotein (LDL), including thrombospondin, long-chain fatty acids, the growth hormone-releasing peptides (GHRPs) such as hexarelin and EP 80317, apoptotic cells, Plasmodium falciparum–infected erythrocytes, advanced glycated products (AGE), and native LDL, high-density lipoprotein (HDL), and very low-density lipoprotein (VLDL; see Febbraio and Silverstein 2 and references therein). The multiligand binding activity of CD36 is mediated by the extracellular domain, within which several distinct functional domains were defined by competition studies with synthetic or recombinant CD36 peptides. 10-13 The extracellular domain of CD36, expressed as a fusion protein with the Fc domain of IgG1, was reported to be folded correctly and to bind oxLDL efficiently. 14 Therefore, a recombinant soluble form of CD36 may be useful in the identification of small-molecule antagonists targeting atherogenesis at the level of CD36-oxLDL binding.
In this article, the extracellular domain of human CD36 (sCD36), expressed as a His-tagged protein, was demonstrated to bind oxLDL specifically. With hexarelin serving as a positive control, this functional sCD36 was adopted to establish an enzyme-linked immunosorbent assay (ELISA)–like high-throughput screening (HTS) assay for CD36 antagonists in an in vitro ligand-binding system. After optimization of the parameters, this cell-free HTS assay presented an excellent Z′ value, showing that it is robust and reliable. Then the assay was applied to screen pure chemical compounds and a microbial secondary metabolite crude extract library. Three chemical compounds were shown as small-molecule antagonists of CD36, with IC50 lower than hexarelin in the developed assay. Further studies showed that these compounds effectively inhibited cellular uptake of oxLDL and subsequent lipid accumulation in RAW 264.7 cells. Thus, the reported ELISA-like assay based on the recombinant sCD36 represents a promising method for the discovery of novel CD36 antagonists.
Materials and Methods
Materials
Escherichia coli DH5α was used as the host for cloning experiments. PCR fragment cloning vector pGEM-T was from Promega (Madison, WI). E. coli BL21(DE3) and the expression vector pET-30a(+) were purchased from Invitrogen (Carlsbad, CA). Endonucleases (BamHI, EcoRI, and NotI) and Pfu DNA polymerase were obtained from TransGen Biotech (Beijing, China). Ampicillin, X-gal, and isopropyl-β-D-thiogalactoside (IPTG) were from Sigma (St. Louis, MO).
Mouse monoclonal antibody against His-tag, rabbit anti- human CD36 polyclonal antibody H-300, and horseradish peroxidase (HRP)–conjugated goat antimouse and antirabbit IgG antibodies were obtained from Santa Cruz Biotechnology (Santa Cruz, CA). The rabbit polyclonal antibody against apolipoprotein B (apoB) was from Calbiochem-Behring (San Diego, CA). oxLDL and acetylated LDL (acLDL) were purchased from the Institute of Basic Medical Sciences (Chinese Academy of Medical Sciences, Beijing, China). 1,1′-Dioctadecyl-3,3,3,′3′-tetramethylindocarbocyanine perchlorate-labeled acLDL (DiI-acLDL) was purchased from Biomedical Technologies (Stoughton, MA). Polypropylene microplates (96-well) and 24-well plates were purchased from Corning (Acton, MA). Cell culture media and fetal bovine serum (FBS) were from Hyclone (Logan, UT).
Cell culture
RAW 264.7 murine macrophage cells were cultured in Dulbecco’s modified Eagle’s medium (DMEM, high glucose) containing 10% FBS (v/v). Human leukemic monocyte lymphoma U937 cells were grown in suspension in RPMI1640 medium containing 10% FBS (v/v). Both cells were grown at 37°C in a humidified 5% CO2 incubator. For differentiation, U937 cells were cultured in 24-well plates at a density of 50,000 cells each well and stimulated with 0.1 µg/mL PMA (Sigma) for 2 days.
Cloning and expression of human recombinant soluble CD36 in E. coli
Total RNA was extracted from PMA-induced U937 cells using the QuickPrep Total RNA Extraction Kit (GE Healthcare, Piscataway, NJ). The reverse transcription was performed by SuperScript III First-Strand (Invitrogen) with the oligo dT primer. The human soluble CD36 (sCD36) cDNA encoding the extracellular domain of CD36 (Gly30-Asn439) was subsequently amplified by PCR, with primers sCD36 forward 5′-AAGAATTCGGAGACCTGCTTATCCAGAAG-3′ and sCD36 reverse 5′-AGCGGCCGCTTAGTTTATTTTTCCAGTTACTTGAC-3′ (the italicized sequences were the recognition sites of the restriction enzymes EocRI and NotI, respectively). The PCR products were purified with gel cleanup kit (TransGen Biotech), cloned into the pGEM-T vector, and confirmed by sequencing. Then the designated sCD36 cDNA fragment was inserted between the EcoRI and NotI sites of the vector pET-30a(+), resulting in pET-sCD36.
E. coli BL21(DE3) containing plasmid pET-sCD36 was grown overnight in Luria-Bertani (LB) media at 37°C until the OD600 was between 0.4 and 0.6. At this time, 0.1 mM IPTG was added into the media and incubated at 37°C for 4 h. After induction, the bacteria were harvested and lysed by sonication in the TGT buffer (50 mM Tris-HCl [pH 8.0], 100 mM NaCl, 20% glycerol, 1% Triton X-100). Then the inclusion bodies were collected by centrifugation (10,000 rpm for 10 min) and resuspended completely in the wash buffer (50 mM Tris-HCl [pH 8.0], 100 mM NaCl, 20% glycerol, 1% Triton X-100, 2 M urea). The wash step was repeated 2 more times, and the inclusion body was dissolved in lysis buffer (50 mM Tris-HCl [pH 8.0], 100 mM NaCl, 20% glycerol, 8 M urea, 100 mM β-mercaptoethanol). Before being used in the HTS assay, the protein was refolded by 50-fold dilution with phosphate-buffered saline (PBS) buffer (137 mM NaCl, 2.7 mM KCl, 4.3 mM Na2HPO4, 1.4 mM KH2PO4 [pH 7.3]). The bacteria containing pET-30a(+) (vector control) were grown and lysed as described above. The protein concentration of refolded sCD36 and the whole-cell extract of the vector control strain were adjusted to 2 mg/mL, using BCA Protein Assay Kit (Pierce Biotechnology, Rockford, IL).
Western blot assay
Protein samples were subjected to 12% sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS-PAGE) and electroblotted onto a 0.45-µm polyvinylidene difluoride (PVDF) membrane (Millipore, Bedford, MA) by a Trans-Blot SD Semi-Dry Electrophoretic Transfer Cell (Bio-Rad, Hercules, CA). Ponceau S staining solution (0.2% [w/v] ponceau S [Sigma] in 5% [v/v] acetic acid) was used to detect proteins transferred onto the membrane and mark the molecular weight. The membrane was rinsed several times in distilled water and blocked in Tris-buffered saline with Tween (TBST) buffer (100 mM Tris-HCl [pH 7.5], 0.9% NaCl, 0.1% Tween-20 [Merck, Whitehouse Station, NJ]) containing 5% nonfat dry milk for 1 h at room temperature. Then, the membrane was incubated overnight at 4°C with the primary antibody against His-tag protein or human CD36 in TBST containing 5% nonfat dry milk. Subsequently, after being washed with TBST, the membrane was incubated with HRP-conjugated goat antimouse or antirabbit IgG antibody for 1 h at room temperature. After washing with TBST, detection was achieved using an enhanced chemiluminescence (ECL) detection system (Santa Cruz Biotechnology) and quantified using ChemiImager 5500 (Alpha Innotech, San Leandro, CA).
Ligand-binding blot assay
The process of the ligand-binding assay was similar to the Western blot assay described above except that the nondenatured PAGE was used. Simply, the protein samples were subjected to 12% nondenatured PAGE and then transferred to a 0.45-µm PVDF membrane. Then, the membrane was blocked for 1 h in TBST containing 5% nonfat dry milk and incubated with 20 µg/mL oxLDL for 4 to 6 h. Afterwards, it was incubated overnight with the rabbit polyclonal antibody against apoB in TBST with 5% nonfat dry milk, followed by being incubated with HRP-conjugated goat antirabbit IgG antibody for 1 h at room temperature. The detection was conducted using an ECL detection system.
ELISA-like HTS assay
Microplates (96-well) were coated with 100 µL 5 µg/mL oxLDL each well at 4°C for 8 to 12 h. After being washed with PBS buffer twice, the oxLDL-coated wells were blocked with PBS buffer containing 1% bovine serum albumin (BSA) for 1 to 2 h at 4°C. The wells were then washed with PBST (PBS buffer with 0.1% Tween-20) buffer twice, which was used in the following washing steps. After being incubated with 100 µL 30 µg/mL sCD36 or vector control protein at 4°C for 1 h, 1 µL various pure compounds (1 mg/mL) or microbial secondary metabolite crude extract samples (1:10 dilution) was added. Following a 2-h incubation, the plate was washed and then incubated with the mouse monoclonal antibody against His-tag protein at a final dilution of 1:1250 (4°C, 2 h). The wells were washed and incubated with HRP-conjugated goat antimouse IgG (1:5000 dilution, 4°C, 1 h). Subsequently, after incubation with 100 µL TMB/H2O2 (mixed immediately before incubation by 1:1 [v/v]) for 45 min at 37°C, 100 µL 1 M HCl was added to stop the reaction. The optical density (OD) value of each well was recorded using spectrophotometry with the microplate reader (Model 680 microplate reader, Bio-Rad) at 450 nm.
Preparation of the microbial secondary metabolite crude extract library
The microorganism strains for preparation of the microbial secondary metabolite crude extract library are from the Centre for Culture Collection of Pharmaceutical Microorganisms (Institute of Medicinal Biotechnology, Chinese Academy of Medical Sciences, Beijing, China). The crude extracts were prepared as previously described. 15
Oil red O staining assay
RAW 264.7 cells were seeded at 500,000 cells/well in a 96-well plate in DMEM. Then the cells were incubated with 80 µg/mL oxLDL in serum-free DMEM in the absence and presence of sCD36 or compounds for 24 h at 37°C. Uptake of oxLDL in RAW 264.7 cells was quantified by fixing the cells in 10% formaldehyde for 10 min, then washed twice with double-distilled water (dd H2O). The cells were treated with 60% isopropanol for 5 min and stained with 0.6% oil red O (Sigma) in 60% isopropanol for 1 h and then washed sequentially with 60% isopropanol and dd H2O. The stained cells were photographed using a light microscope (Olympus CKX41, Japan) with a digital camera. Quantification of lipid accumulation was achieved by extracting oil red O from stained cells with isopropanol and measuring the OD of the extracts at 490 nm.
Flow cytometry analysis for cellular uptake of DiI-labeled acLDL
RAW 264.7 cells were plated in 24-well culture dishes at 200,000 cells/well, followed by incubation with 2 µg/mL DiI-acLDL in the presence of vehicle (0.1% DMSO) or an indicated concentration of compound for 4 h at 37°C. Cells were washed with PBS and incubated with PBS containing 0.5% BSA and 2 mM EDTA for 1 h at 4°C and then detached from the plate by gentle pipetting. The cell suspension was centrifuged (800 rpm, 3 min, 4°C), the obtained pellet was resuspended in PBS buffer, and DiI fluorescence was analyzed using an Epics XL flow cytometer (Coulter Corporation, Miami, FL).
Surface plasmon resonance analysis
All experiments were performed in PBS buffer (pH 7.4) on a BIAcore T100 System (GE Healthcare). The recombinant human CD36 (R&D, Minneapolis, MN) was diluted to 50 µg/mL in 10 mM sodium acetate (pH 4.5) and immobilized on a CM5 sensor chip by amino coupling at a total signal of 16,400 response units (RU). A blank flow cell was used as a reference surface on the chip. Three different concentrations (1, 25, and 50 µM) of salvianolic acid B (SAB) were injected consecutively on the chip. The resulting binding and dissociation curves were analyzed with BiaEvaluation software (GE Healthcare). Between each run, the chip was regenerated with 10 mM NaOH.
Data analysis
To assess the robustness of the HTS assay, the Z′ value was calculated as described in Zhang et al. 16 All experiments except the primary HTS were repeated at least 3 times, and representative results are presented. Quantitative data are expressed as mean ± SEM. Statistical significance of the data was evaluated by Student t-test. Data from all ligand-binding and competition assays were analyzed by nonlinear regression analysis and curve fitting using GraphPad Prism, Version 4.03 (GraphPad Software, San Diego, CA).
Results
Expression of recombinant human soluble CD36 in E. coli
Recombinant human soluble CD36 (sCD36) composed of the entire extracellular domain (Gly30 to Asn439) was engineered with a His6-tag at its N terminus and expressed in E. coli BL21(DE3). Maximal protein expression was achieved 4 h postinduction with 0.1 mM IPTG, and the His-tagged sCD36 was mainly aggregated in inclusion bodies. After washing the inclusion bodies, the purity of sCD36 reached approximately 80%, as determined by SDS-PAGE ( Fig. 1A ). The expressed His-tagged sCD36 was validated by Western blot assay using an antibody against either human CD36 or His-tag ( Fig. 1A ). The protein has an apparent molecular weight of 53 kDa, which was consistent with the calculated molecular weight of the recombinant human sCD36.
The ligand binding ability of sCD36 was first examined by the ligand-binding blot assay. It was shown that sCD36 could bind oxLDL, which was detected by antibody against apoB, the main component of LDL ( Fig. 1A ). The vector control protein did not display the same specific binding band, although there were several nonspecific binding bands in the whole-cell extract ( Fig. 1A ). To further determine the ligand-binding properties of sCD36, a microplate-based ELISA-like assay was used. Binding of His-tagged sCD36 to potential ligands coated on the wells was detected using a primary antibody against His-tag, as described in Materials and Methods. As shown in Figure 1B , recombinant sCD36 bound to CD36 ligands oxLDL and acLDL but showed very minimal binding to BSA, indicating the specificity of sCD36 binding to oxLDL and acLDL. Furthermore, the biological activity of sCD36 was identified using uptake studies of oxLDL by macrophages in the presence of sCD36. oxLDL may lead to uncontrolled accumulation of lipids within monocytes/macrophages and foam cell formation, which is implicated in the progression of atherosclerosis. 3 As determined by staining neutral lipids with oil red O, incubation of RAW 264.7 cells with 80 µg/mL oxLDL resulted in increasing lipid droplets accumulation and accelerating foam cell formation. After treatment with 100 µg/mL sCD36, a marked decrease (~40%) in total cellular lipid and in the size of lipid droplets was observed, compared with treatment with vector control protein. The result indicated that sCD36 might act as a decoy for oxLDL and prevent the uptake of oxLDL by RAW 264.7 cells. These findings demonstrate that the recombinant human sCD36 retains its functional ability to bind oxLDL.
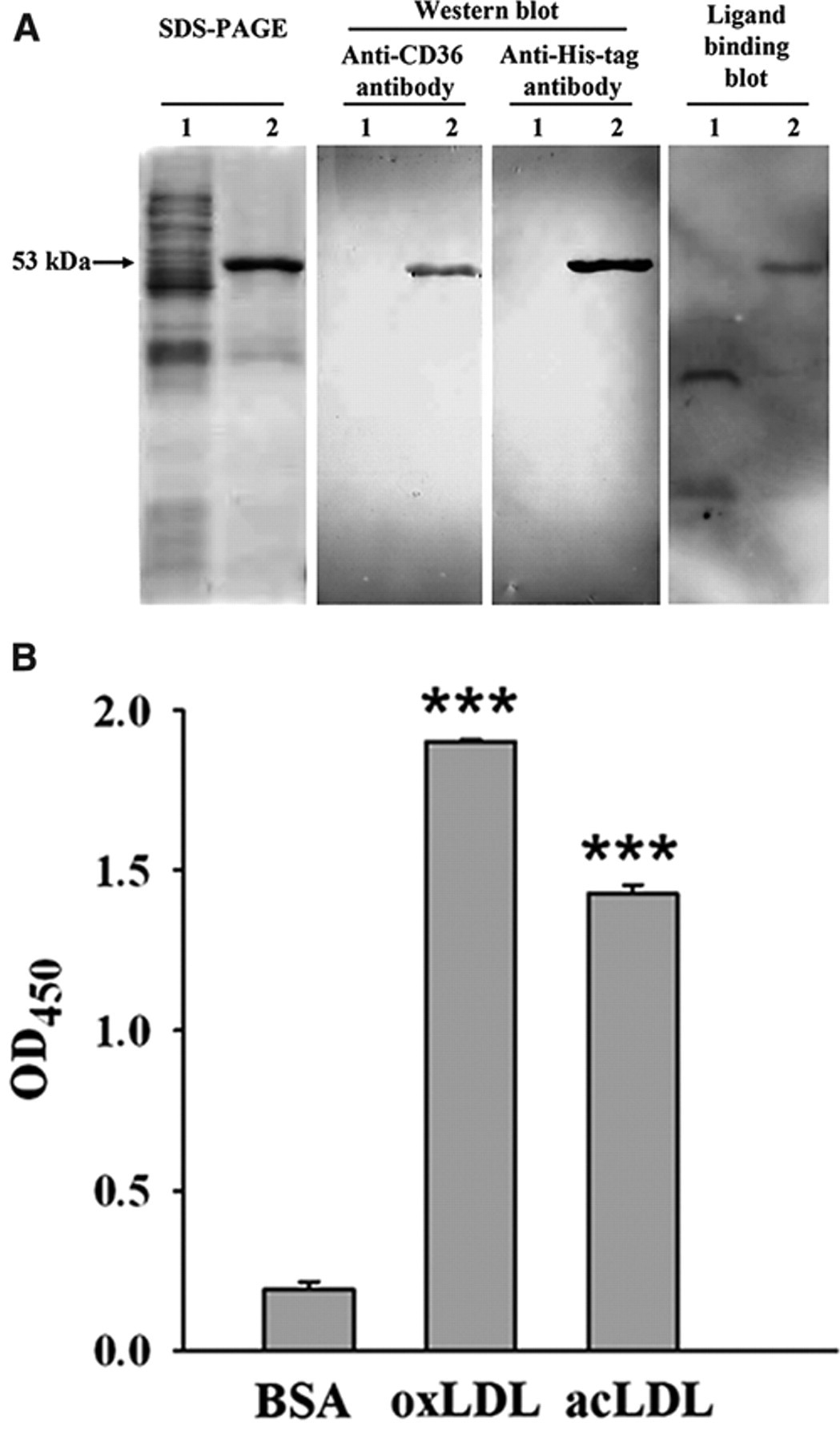
Expression and validation of recombinant human soluble CD36 (sCD36). (
Development of ELISA-like HTS assay for CD36 antagonists
The recombinant human sCD36, with specific oxLDL binding activity, was used to develop the ELISA-like assay suitable for HTS in a 96-well format. First, the immobilization of oxLDL was determined in a separate experiment detected by the anti-apoB antibody (inset of Fig. 2A ). It was shown that oxLDL bound on the well wall effectively within the concentration of 2 to 10 µg/mL. In addition, the plate coated with different concentrations of oxLDL was incubated with 30 µg/mL sCD36 and then detected by the anti-His-tag antibody. The result showed the binding of sCD36 to coated oxLDL in a similar dose-responsive manner ( Fig. 2A ), and 5 µg/mL oxLDL was used in the following assays. Second, the wash buffer was optimized in this assay. As shown in Figure 2B , the recombinant sCD36 was detected to bind oxLDL fixed on the plate but not when the vector control protein was incubated. The signal-to-background ratio (S/B) of the assay was 636.6 when using PBST as wash buffer, which was much higher than using PBS (S/B = 11.2). The signal-to-noise ratio using PBST (S/N = 25.3) was also better than PBS (S/N = 12.9), so PBST was used as wash buffer in the following assays. Then, series concentrations of sCD36 were applied to bind oxLDL coated on the plate and detected by antibody against His-tag protein or against human CD36, respectively. The results indicated that the binding of sCD36 to oxLDL was dose dependent ( Fig. 2C ). Compared with antibody against CD36, the anti-His-tag antibody was more reactive and less noisy. According to these results, 30 µg/mL sCD36 and anti-His-tag antibody were applied in the following assays. Then the optimization of the assay was focused on the titers of the anti-His-tag antibody ( Fig. 2D ) and the HRP-conjugated secondary antibody ( Fig. 2E ). The series of optimizations led to the following optimal screening conditions:
The concentration of oxLDL: 5 µg/mL ( Fig. 2A )
Wash buffer: PBST ( Fig. 2B )
The concentration of sCD36: 30 µg/mL ( Fig. 2C )
Primary antibody: anti-His-tag antibody ( Fig. 2C )
Primary antibody dilution: 1:1250 ( Fig. 2D )
Secondary antibody dilution: 1:5000 ( Fig. 2E )
With the optimized assay conditions, the specific binding of sCD36 to oxLDL was a saturable process that showed best fit to a 1-site binding model ( Fig. 2F ). The measured apparent K d value was 8.4 ± 0.2 µg/mL (n = 3). Several known CD36 ligands were used to confirm the feasibility of the sCD36-based HTS assay. acLDL could inhibit the binding of sCD36 to immobilized oxLDL in a dose-dependent manner. The measured IC50 value was 18.2 µg/mL ( Fig. 2G ). Hexarelin, a hexapeptide member of the GHRPs, was reported to bind CD36 and interfere with the CD36-mediated uptake of oxLDL by macrophages, featuring its cardioprotective effects. 10,17,18 The binding domain for hexarelin on CD36 has been demonstrated to overlap with that for oxLDL. 10 As shown in Figure 2H , hexarelin inhibited the sCD36 binding to oxLDL in a dose-dependent manner, with the measured IC50 value of 52.8 µM. In addition, several known antioxidants were evaluated using the developed assay. Probucol 19 and vitamins C and E 20 have been reported to inhibit foam cell formation by protection of LDL from oxidation. As predicted, these compounds showed no activity in the developed assay at the concentration range of 1 to 1000 µM ( Fig. 2H ). The HTS assay performance was quantitatively assessed by a Z′ factor using hexarelin as a positive control. The Z′ factor value of the developed ELISA-like assay for CD36 antagonists was 0.82 with 5 separate experiments ( Fig. 2I ).
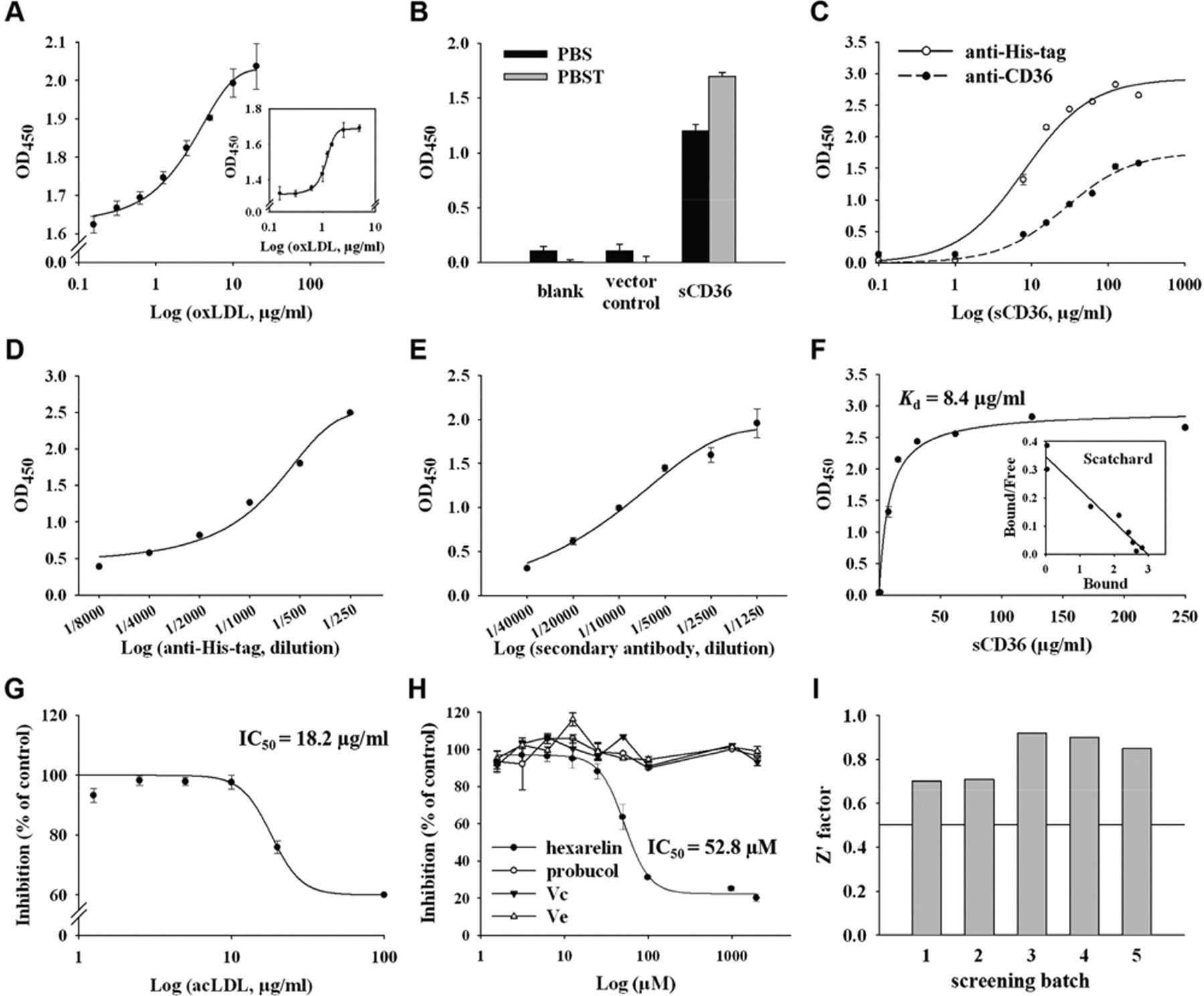
Development of the enzyme-linked immunosorbent assay (ELISA)–like high-throughput screening (HTS) assay. (
High-throughput screening
The developed HTS assay for CD36 antagonists was used in the following screening applications for both pure chemical compounds and microbial secondary metabolite crude extracts. The vehicle control groups containing 0.1% DMSO, which was used to dissolve the samples, did not exhibit any effect on the result of this HTS assay (data not shown). Samples with greater than 50% inhibition of sCD36-oxLDL binding were defined as hits in the primary screening and retest for confirmation. In total, 2640 compounds at 10 µg/mL were screened, and those chemicals efficiently preventing the bindingX between sCD36 and oxLDL in the primary screening were retested. The primary and confirmed positive rate for the chemicals reached 1.4% and 0.79%, respectively. The dose-response curves for these confirmed chemicals on inhibition of sCD36 binding to oxLDL were detected using the developed assay, and the IC50 was measured. About 0.5% of the compounds had an IC50 less than 50 µM, the level of IC50 value of hexarelin ( Fig. 2H ). After primary screening, the most effective compound, SAB, was chosen for further characterization, together with 2 structurally related positive compounds: rosmarinic acid (RA) and sodium danshensu (DSS). The structures of SAB, RA, and DSS and their consensus structure of catechol are shown in Figure 3A . The measured IC50 of the 3 compounds were 23.2, 25.1, and 37.8 µM, respectively ( Fig. 3B ).
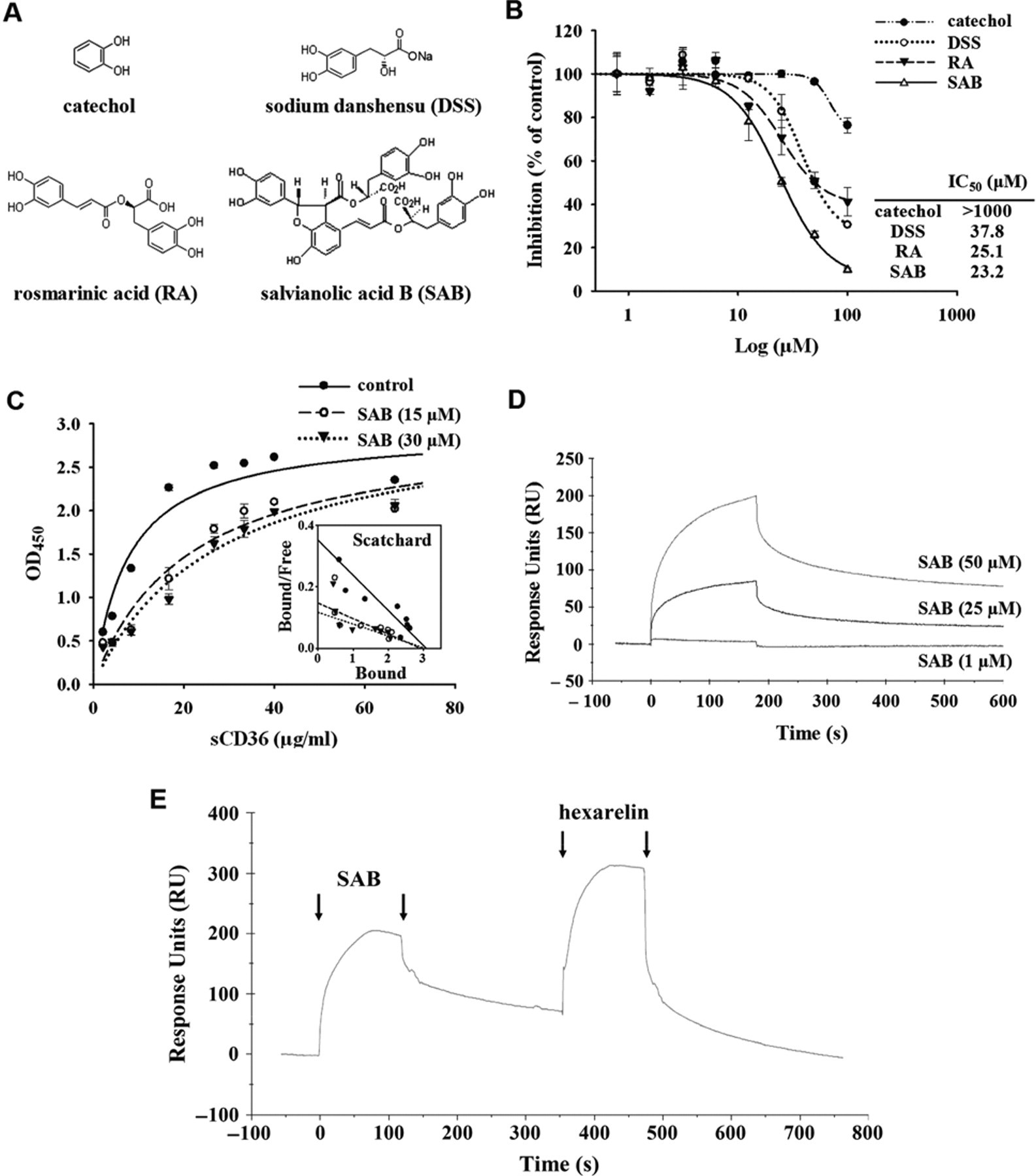
Positive compounds. (
To characterize the nature of the inhibition, the binding saturation experiments were performed in the presence of SAB. As shown in Figure 3C , the analysis of the binding data obtained in the presence of 15 or 30 µM SAB determined that the apparent K d was decreased from 8.7 to 19.8 µg/mL or 25.9 µg/mL, respectively, and the Bmax remained the same. Furthermore, the ability of SAB binding to CD36 was investigated by surface plasmon resonance (SPR) analysis, using recombinant human CD36 immobilized on a CM5 sensor chip. The binding assays demonstrate that SAB readily interacts with CD36 in a dose-dependent manner ( Fig. 3D ). After binding and dissociation of SAB (50 µM) to the immobilized CD36, 50 µM hexarelin was injected consecutively on the chip. The resulting binding and dissociation curves showed that hexarelin readily bound to the immobilized CD36 after the dissociation of SAB ( Fig. 3E ).
In addition to the pure compounds, 2000 samples from the microbial secondary metabolite crude extract library were screened using the developed assay. Of 4 primary hits (≥50% inhibition), 3 crude extracts of microbial secondary metabolites were confirmed to be validated hits by detecting their dose-response curves ( Fig. 4 ). The positive rate of this screening assay for the microbial samples reached about 0.15%. The positive microorganism strains (i.e., 08-863, 08-671, and 08-690) belong to actinomycetes according to primary identification mainly based on the morphological characteristics.
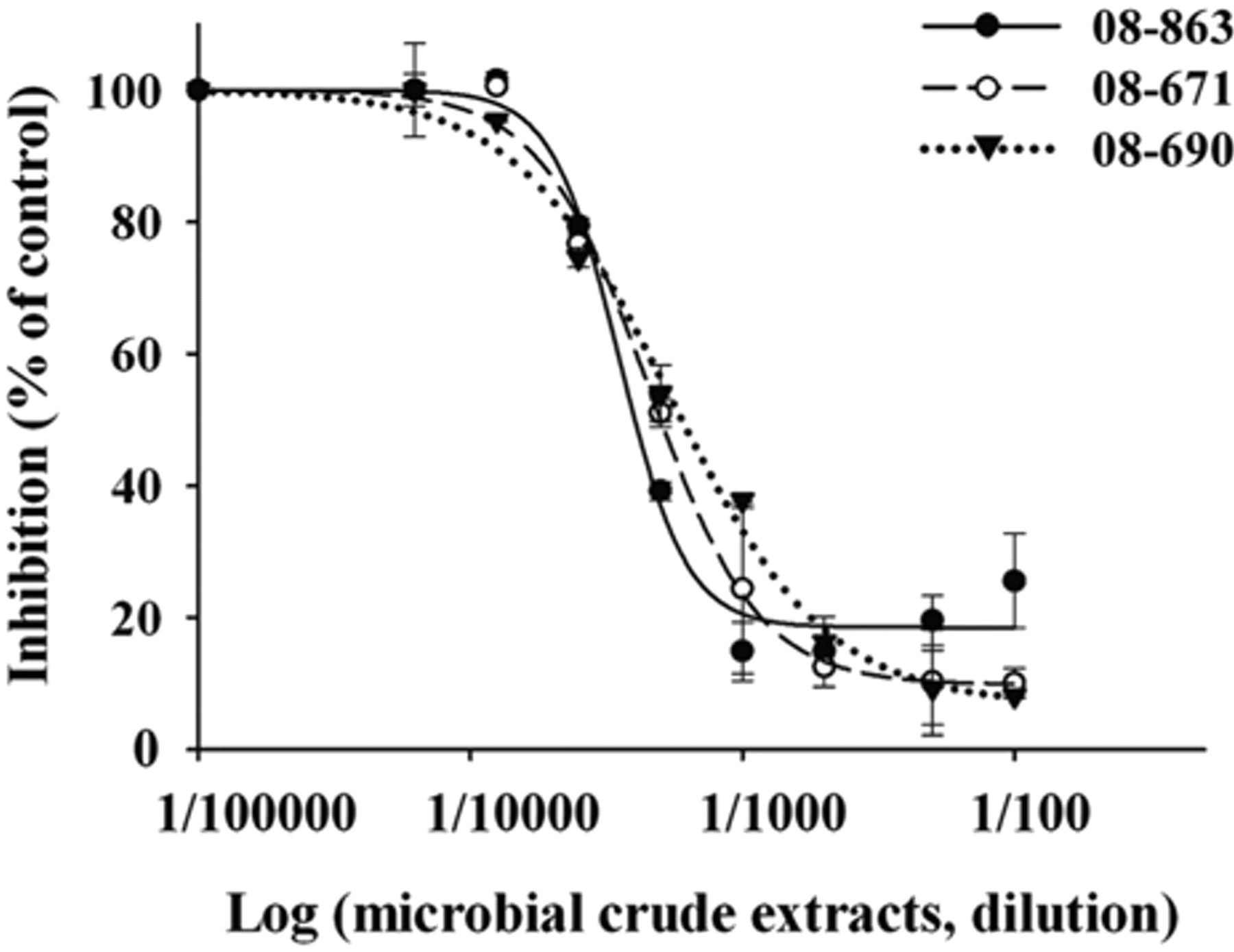
The effect of the positive microbial crude extracts on sCD36-oxLDL binding. The dose-response curves were detected using the developed assay as described in Figure 3B .
Characterization of positive compounds
To confirm the antagonistic activity of positive compounds identified in the HTS, RAW 264.7 cells were incubated with oxLDL in the absence or presence of the positive compounds. As described above, lipid accumulation and foam cell formation were readily apparent in cells treated with oxLDL alone, as identified by oil red O staining ( Fig. 5A , b ). The increase in the size and number of lipid vesicles upon oxLDL loading was markedly diminished by treating cells with the 3 positive compounds ( Fig. 5A , e - g ). Photometric measurement of lipid stained with oil red O showed that the 3 positive compounds reduced lipid loading in a significant and dose-dependent manner at 10 and 100 µM ( Fig. 5B ), suggesting that these compounds remarkably decreased the uptake of oxLDL into RAW 264.7 cells compared with the oxLDL-incubated control. Hexarelin was reported to prevent lipid accumulation from oxLDL uptake in macrophage cells 17 and was used as a positive control in this experiment ( Fig. 5A , h ), with a respective 37.6% and 68.0% inhibition at 10 and 100 µM ( Fig. 5B ). The most effective compound, SAB, was further investigated using flow cytometry assay to evaluate its effect on the uptake of fluorescence-labeled acLDL (DiI-acLDL) in RAW 264.7 cells. The result showed that 10 or 100 µM SAB reduced the uptake of DiI-acLDL into RAW 264.7 cells by 10.3% or 35.7%, respectively ( Fig. 5C ).
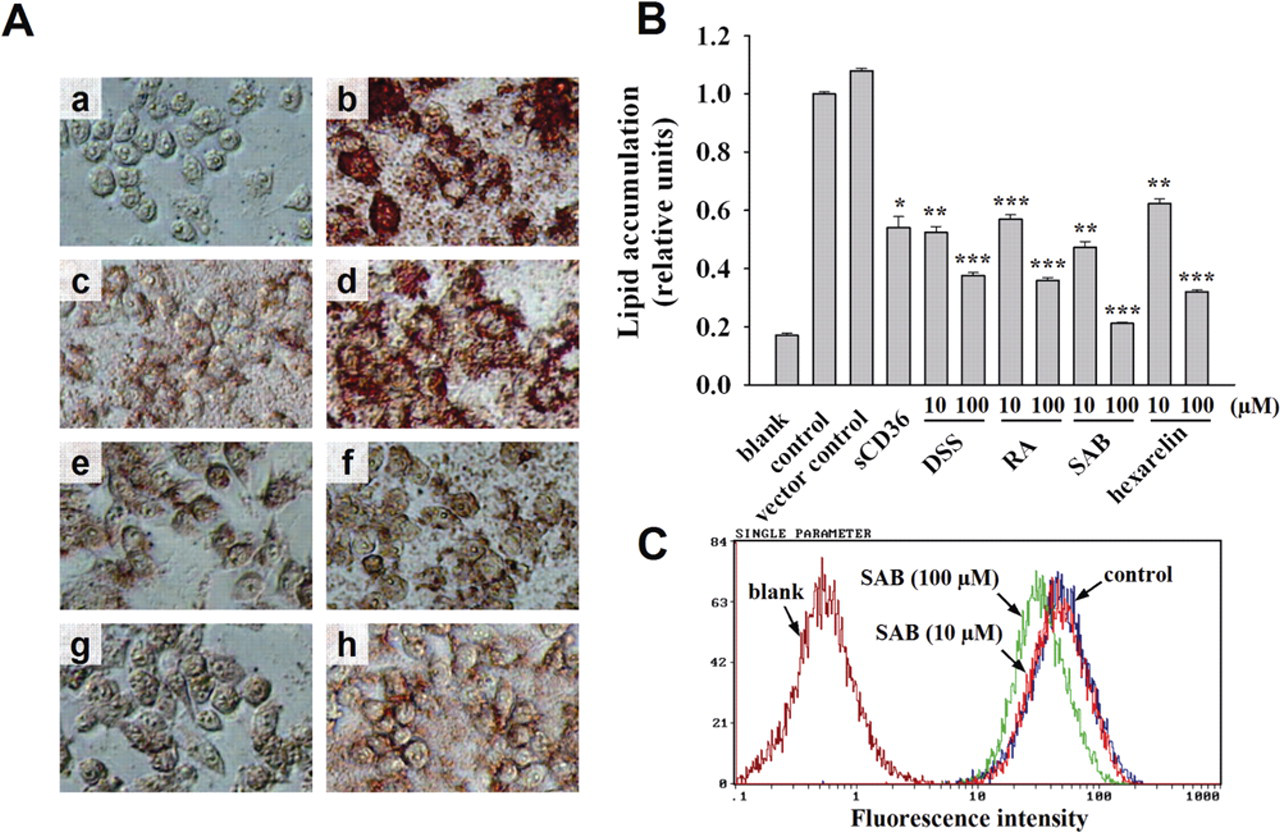
Characterization of recombinant sCD36 and positive compounds. (
Discussion
Since CD36 was discovered as a macrophage receptor for oxidized LDL, 21 the consensus of the literature has been that macrophage CD36 plays a major role in the binding and uptake of proatherogenic lipoproteins, which results in foam cell formation and the initiation of lesions in atherosclerosis. 2 CD36-apoE double-null animals developed significantly less atherosclerosis compared with apoE-null animals, 6,7 suggesting that elevated plasma cholesterol may not be as important as arterial wall macrophages for the initiation of atherogenesis. A recent report showed that the combined absence of CD36 and SR-A, another major macrophage receptor to bind oxLDL, provided no greater protection against atherosclerosis than the absence of CD36 alone, 22 which further confirmed the role of CD36. In agreement with this, EP 80317, a CD36 ligand of the GHRP family but devoid of any growth hormone–releasing activity, reduced atherosclerotic lesion development in apolipoprotein E (apoE) knockout mice. 23 Furthermore, the effects of EP 80317 were shown to be CD36 dependent. 23 Although there are many important targets taking part in atherosclerotic lesion development in vivo, these results suggest that CD36 plays a major role in the initiation of atherosclerotic process. Thus, CD36 antagonists might be useful in blocking oxLDL-CD36 binding and be developed to a novel class of antiatherosclerotic agents. However, no small-molecule, nonpeptide CD36 antagonist has been discovered that can block the binding of modified LDLs to CD36. In the present study, we report the development of an ELISA-like HTS assay for CD36 antagonists using a recombinant soluble form of human CD36.
In addition to the classical notion of receptors as transmembrane proteins, many receptors have endogenous soluble forms that are released into the extracellular space. A soluble form of CD36 recently was demonstrated to be present in plasma, and it was suggested as a novel marker for metabolic syndrome 24 and atherosclerosis, 25 although the mechanism of its generation is still unclear. Recombinant soluble receptors have been used to study structure/function analysis of receptors and, in some cases, therapeutically block the functions of its membrane counterpart (e.g., Cole and Rabasseda 26 ). A recombinant sCD36-Ig chimeric molecule has been shown to be able to bind oxLDL and to efficiently compete with the membrane-expressed CD36, 14 suggesting that CD36 without both the N- and C-transmembrane domain and terminal ends could fold in a conformation that does not affect its binding ability to oxLDL. In this study, the extracellular domain of CD36 was expressed as a His-tagged protein in E. coli. Although without eukaryotic posttranslational modifications such as glycosylation, the recombinant sCD36 showed specific binding to oxLDL both by a ligand-binding blot assay ( Fig. 1A ) and by a solid-phase-based ligand-binding assay ( Fig. 1B ). These results, together with the ability of sCD36 to inhibit the macrophage binding and uptake of oxLDL ( Fig. 5A , c and B ), suggest that sCD36 expressed in this study maintains its oxLDL binding properties and should be suitable for use in the HTS assay to identify novel CD36 antagonists at the level of ligand binding. Our results are consistent with the early studies of E. coli–expressed recombinant GST/CD36 fusion proteins, 12 as well as a recent report demonstrating that glycosylation on CD36 is not necessary for ligand binding but necessary for trafficking to the plasma membrane using different glycosylation site mutants. 27
Depending on the solid-phase-based ligand-binding assay, the in vitro ELISA-like HTS assay for the identification of bioactive small molecules was developed by optimizing the parameters, including immobilized concentration of oxLDL, concentration of sCD36 for binding, the type of detection antibody, and the dilution of the detection antibody and the secondary antibody. The polyhistidine tagged at the N terminus of sCD36 made it possible to use the anti-His-tag monoclonal antibody as the detection antibody ( Fig. 2C ), which is not only more effective but also less costly and more practicable compared with the CD36 polyclonal antibody. On the premise of these optimized conditions, we showed that the recombinant sCD36 was competent to bind oxLDL immobilized on the microplate wells, and the binding was specific and saturable in the absence of other associating cofactors ( Fig. 2F ), which was in agreement with the previous report that no cofactor is required for initial binding of modified LDL to recombinant CD36 expressed in insect cells. 28 Using the developed ELISA-like assay, the competition assay was conducted using acLDL and hexarelin, a known CD36 ligand of the GHRP family. 10 A dose-dependent response was observed for either acLDL ( Fig. 2G ) or hexarelin ( Fig. 2H ), which validated the feasibility of the sCD36-based HTS assay. On the other hand, several known antioxidants were used to evaluate the assay as negative controls ( Fig. 2H ). Probucol, a highly effective inhibitor of the oxidative modification of LDL, may inhibit atherogenesis by limiting foam cell formation and/or endothelial cell injury. 19 Vitamins C and E were also proved to protect LDL from oxidative damage. 20 The fact that different antioxidants (probucol, vitamins C and E) show no activity in the developed assay suggests that the antioxidant characteristics of the compounds will not interfere with the antagonistic effect detected by the assay. Furthermore, this assay resulted in high confidence in the ability of detecting compounds with CD36 antagonistic activity, as reflected by the excellent Z′ value (0.82).
The success of hit identification through HTS relies not only on the robustness of biological assays but also on the quality and diversity of screening libraries. Natural products from microorganisms and plants have been shown to be the most productive source of leads for new drugs, as they are more structurally varied and more drug-like than that of combinatorial chemical collections. The application of this developed assay in screening the pure chemical compounds mainly from natural products and the microbial secondary metabolite crude extracts resulted in some positive compounds and strains. Although the identification of active compounds from the positive microbial secondary metabolites is ongoing, 3 positive compounds with relative structures have been characterized in this study. Dose-dependent competition assays for DSS, RA, and SAB were performed, and their IC50 values were determined. The relative order of magnitude of the activity of these CD36 antagonists is SAB > RA > DSS > hexarelin, within which the IC50 of SAB and RA was a half lower than that of hexarelin. The chemical structure of DSS is 3-(3,4-dihydroxy-phenyl)-2-hydroxy-propionic acid, and RA and SAB could be roughly regarded as its dipolymer and tetrapolymer, respectively ( Fig. 3A ). Catechol, the phenolic part of DSS, lost almost all of the activities with its IC50 > 1000 µM. Thus, DSS may be considered a basic structure for the CD36 antagonistic activity. The binding saturation studies and Scatchard analysis ( Fig. 3C ) showed that SAB caused successive decreases in the slopes (indicative of an increased Kd value) with no decrease in the maximal binding, indicating that SAB is a reversible competitive inhibitor. 29 The direct interaction of SAB with recombinant human CD36 was confirmed by SPR analysis ( Fig. 3D ), suggesting that CD36 is the target of the compound. Furthermore, the readily binding of hexarelin to the immobilized CD36 was detected after the binding and dissociation of SAB ( Fig. 3E ), definitely demonstrating that the inhibition of SAB is reversible. The inhibition studies further demonstrate that the active small-molecule compounds found as CD36 antagonists are effective at blocking macrophage binding and uptake of modified LDL ( Fig. 5 ). These results indicate that the developed HTS assay should be suitable to search for novel CD36 antagonists to inhibit foam cell formation, a potential therapeutic approach in limiting the development of lipid-laden cells in the blood vessel wall during atherogenesis.
DSS, RA, and SAB are members of phenolic acid compounds, which are the main type of hydrophilic components from Danshen, the dried roots of Salvia miltiorrhiza. 30 Danshen is a commonly used traditional Chinese medicine for the treatment of atherosclerosis-related disorders such as coronary artery disease, hyperlipidemia, and cerebrovascular diseases (see Zhou et al. 30 and references therein). Various in vitro and in vivo studies have concentrated on pharmacological constituents of Danshen. The water-soluble compounds of Danshen were found to have antioxidative effect in vitro, 31 and this may contribute, at least in part, to the antiatherosclerotic effects of Danshen. SAB, a polyphenolic antioxidant, has been reported to effectively protect LDL from oxidative modification by scavenging free radicals in the bloodstream of cholesterol-fed rabbits. 32 However, several known antioxidants (probucol, vitamins C and E) showed no inhibitory activity in the developed assay ( Fig. 2H ), suggesting that the activity of these positive compounds may be ascribed to their antagonistic properties rather than their antioxidant effect. Recently, these compounds have been found to play roles in other aspects in the development of atherosclerosis. DSS was found to protect the endothelial cells against a homocysteine-induced adverse effect. 33 RA was reported to inhibit in vitro the angiogenic process in a concentration-dependent manner. 34 SAB was reported to inhibit tumor necrosis factor α (TNFα)–induced plasminogen activator inhibitor type 1 (PAI-1) expression, 35 display anti-inflammatory activities by decreasing COX-2 expression, 36 and show anti- migration properties by attenuating MMP-2 and MMP-9 expression. 37 However, to the best of our knowledge, this is the first time that DSS, RA, and SAB have been demonstrated to be antagonists for oxLDL binding with CD36. The results in this study may propose a novel antiatherosclerotic mechanism of the hydrosoluble salvianolic acids that might be helpful to further understand the beneficial effects of Danshen on the cardiovascular system.
In addition, CD36 has been identified as a multiligand molecule and demonstrated to be expressed in a diverse array of cells and tissues. 2,38,39 This multiple ligand-receptor interaction suggests possible roles for CD36 in a wide variety of physiological and pathological processes, including inflammation, lipid metabolism, phagocytosis, angiogenesis, migration of microglia, and atherosclerosis. 2,38,39 It will be of interest to study whether the developed assay could be adapted to other CD36 ligands, which could be useful in screening CD36 antagonists for different ligands in allusion to different diseases.
Footnotes
Acknowledgements
We thank Mr. Ming Liu for his helpful discussions. This work was funded by grants from the National Natural Science Foundation of China (grant no. 90813027) and the Key New Drug Creation and Manufacturing Program (grant nos. 2009ZX09302-004 and 2009ZX09301-003). Support is also acknowledged from the China Ministry of Education (NCET-06-0157).
