Abstract
Dopamine is a key monamine neurotransmitter, yet it can also exhibit toxicity to neuronal cells. There are suggestions that dopamine may be neurotoxic due to its propensity to induce the formation of reactive oxygen species, which may in turn adversely affect mitochondrial function and cell viability. In this study, the effects of dopamine or a dopamine reaction product on yeast growth and survival have been explored. Yeast is ideal for such a study because, unlike mammalian cells, yeast cells can be grown even when respiratory function is totally absent. Indeed, dopamine was found to be inhibitory to yeast growth in media where respiratory function was required and cytotoxic to yeast cells suspended in water. The inhibitory effects of dopamine were reduced greatly by the antioxidants ascorbate and glutathione, suggesting the involvement of reactive oxygen species in dopamine-mediated toxicity. It would appear that yeast may offer a convenient model to perform screens for further compounds that may provide protection against dopamine-mediated growth inhibition and toxicity.
Keywords
Introduction
D
It is possible that cytoplasmic or extracellular dopamine in or near these neurons may be associated with their loss because a number of studies show that dopamine can be toxic to cells (reviewed in Zigmond et al. 3 ). Dopamine is considered to be capable of generating reactive oxygen species (ROS), which can be lethal to cells. 4,5 This can occur spontaneously or in a reaction with monamine oxidase (MAO), which can generate additional toxic species, including oxygen radicals, semiquinone, and quinones. 4,5
Dopamine originates in the CNS by the hydroxylation of the amino acid L-tyrosine to L-dihydroxyphenylalanine (L-DOPA) via the enzyme tyrosine 3-monooxygenase. Subsequently, L-dihydroxyphenylalanine is decarboxylated to dopamine by the enzyme aromatic L-amino acid decarboxylase. 6 Dopamine is enzymatically degraded to 3,4-dihydroxyphenylacetic acid (DOPAC), which is O-methylated to 3-methoxy-4-hydroxyphenylacetic acid (homovanillic acid) 7 in an MAO-dependent pathway.
In the dopaminergic neurons, the synthesized dopamine is taken up into storage vesicles through a vesicular membrane transporter. When a dopaminergic terminal is depolarized, calcium ions enter the nerve terminal via ion channels and trigger vesicle fusion to the nerve membrane, leading to the release of dopamine into extracellular space. 8 The uptake of dopamine is mediated by a membrane transport protein called the dopamine transporter, which binds to synaptic dopamine and transports it into the pre- synaptic terminal. 9,10 The dopamine signal is transmitted by the dopamine receptors, and the release of dopamine is affected by which types of receptors the dopamine neurotransmitter binds to. 11
Dopamine has cytotoxic effects on mammalian neurons, 12-15 resulting in the generation of ROS and the reactive metabolite, dopamine quinone. ROS might occur via the normal metabolism of dopamine by MAO-producing hydrogen peroxide, superoxide radicals, and hydroxyl radicals. Coadministration of antioxidants can diminish dopamine toxicity and the extent of dopamine-induced protein modification. 16,17
In this study, dopamine was shown to exhibit inhibitory and cytotoxic effects on yeast. Yeast are simple eukaryotic cells that retain the basic cell biology processes of mammalian cells. This makes yeast useful for studying many human diseases, and yeast are particularly ideal for the study of disorders of human mitochondrial function. 18 Yeast also have the added advantage that growth on glucose can be maintained during the total inhibition of respiratory function. Yeast cells produce most of their adenosine triphosphate (ATP) by fermentation or by respiration, with both occurring on glucose medium. In contrast, respiration is the only way to produce ATP on ethanol- or glycerol-based media. It is expected that significant advantages can be made by discovering more about dopamine toxicity in a simple model system and that yeast can be used to assay inhibitors of dopamine-induced toxicity.
Materials and Methods
Chemicals
Dopamine was purchased from Fluka (Buchs, Germany). Glutathione, ascorbic acid, and DOPAC were purchased from Sigma Aldrich (St. Louis, MO).
Yeast strains and media
The yeast strains employed in these studies were Candida albicans ATCC 90028, Candida glabrata ATCC 90300, Candida tropicalis ATCC 750, Candida parapsilosis ATCC 22019, Pichia pastoris GS115 (his4), Kluveromyces lactis MW98-8c (MATα uraA1-1 lysA argA), and a collection of Saccharomyces cerevisiae strains: W303-1a (MATa ura3-52 leu2-3, 112 trp1-1 ade2-1 his3-11), FYH1 (MATa leu2 trp1 his3), DBY745 (MATα ade1 ura3 leu2 his3), YH1 (trp1 ura3 leu2), J69-1B (MATα ade1 his6), BY4741 (MATa his3Δ1 leu2Δ0 met15Δ0 ura3Δ0), J69-1B/BY4741 (MATα/MATα his3Δ1/HIS3 leu2Δ0/LEU2 met15Δ0/MET15 ura3Δ0/URA3 ade1/ADE1 his6/HIS6) from the cross of J69-1b, and BY4741 and BY4743 (MATa/MATα his3Δ1/his3Δ1 leu2Δ0/leu2Δ0 met15Δ0/MET15 LYS2/lys2Δ0 ura3Δ0/3Δ0) from the cross of BY4741 and BY4742. Strains were grown on YEPD (YEP, 2% dextrose), YEPE (YEP, 2% ethanol), and YEPG (YEP, 3% glycerol) media. YEP is 1% yeast extract plus 2% peptone. When required, media were solidified by the addition of 1.5% agar. Dopamine, glutathione, and ascorbic acid were freshly prepared as 1 M stocks and added to media at 55°C before pouring plates. To minimize the degradation of dopamine, plates were given minimal exposure to light.
Yeast viability measurements
For viability measurements, yeast cells in log phase growth were obtained from cultures grown in YEPD. Cells were suspended at a density of 104 cells/mL in sterile water with dopamine or 6-hydroxydopamine in 96-well microplates. After incubation at 30°C, aliquots of cell suspensions were plated onto solidified YEPD media. After incubation for 3 days at 30°C, the number of colony-forming units was determined. Tests were performed in triplicate.
Yeast growth measurements
For the analysis of yeast growth on solidified media, yeast freshly grown on YEPD media were suspended in water in serial 10-fold dilutions. This enabled aliquots of many strains at various cell concentrations to be dispensed onto different plates using a multipronged inoculator. This method was required because heavy inocula can carry over nutrients and metabolites in cells, causing spurious results.
Dopamine analysis by high-performance liquid chromatography
Chromatography of dopamine was performed using a HP 1090 HPLC with a Vydac C18 protein/peptide column (#218TP54). Absorbance was monitored at 214, 254, and 280 nm wavelengths. Solvent A was 0.1% trifluoroacetic acid (TFA) in water, and solvent B was 0.1% TFA in 70% acetonitrile run over a linear gradient: time 0 (5% B) to time 45 (100% B) at 1 mL/min at 40°C.
Results
Dopamine inhibits respiratory growth
The effects of dopamine on the growth of a variety of yeast species in standard yeast growth media are shown in Table 1 . Species included 8 S. cerevisiae laboratory strains, as well as 5 other strains that included Pichia and Kluyveromyces and 3 Candida species. On solidified YEPD media, dopamine, even at levels of 10 mM, exerted no significant growth inhibition ( Table 1 ). However, the growth of all strains on YEPE, which offered ethanol in the place of glucose as the carbon source, was strongly inhibited by dopamine ( Table 1 ). Of the non-Saccharomyces strains, K. lactis, C. albicans, and C. tropicalis displayed the least sensitivity to dopamine, whereas other strains were generally more sensitive.
Growth of Yeast Strains in the Presence of Dopamine
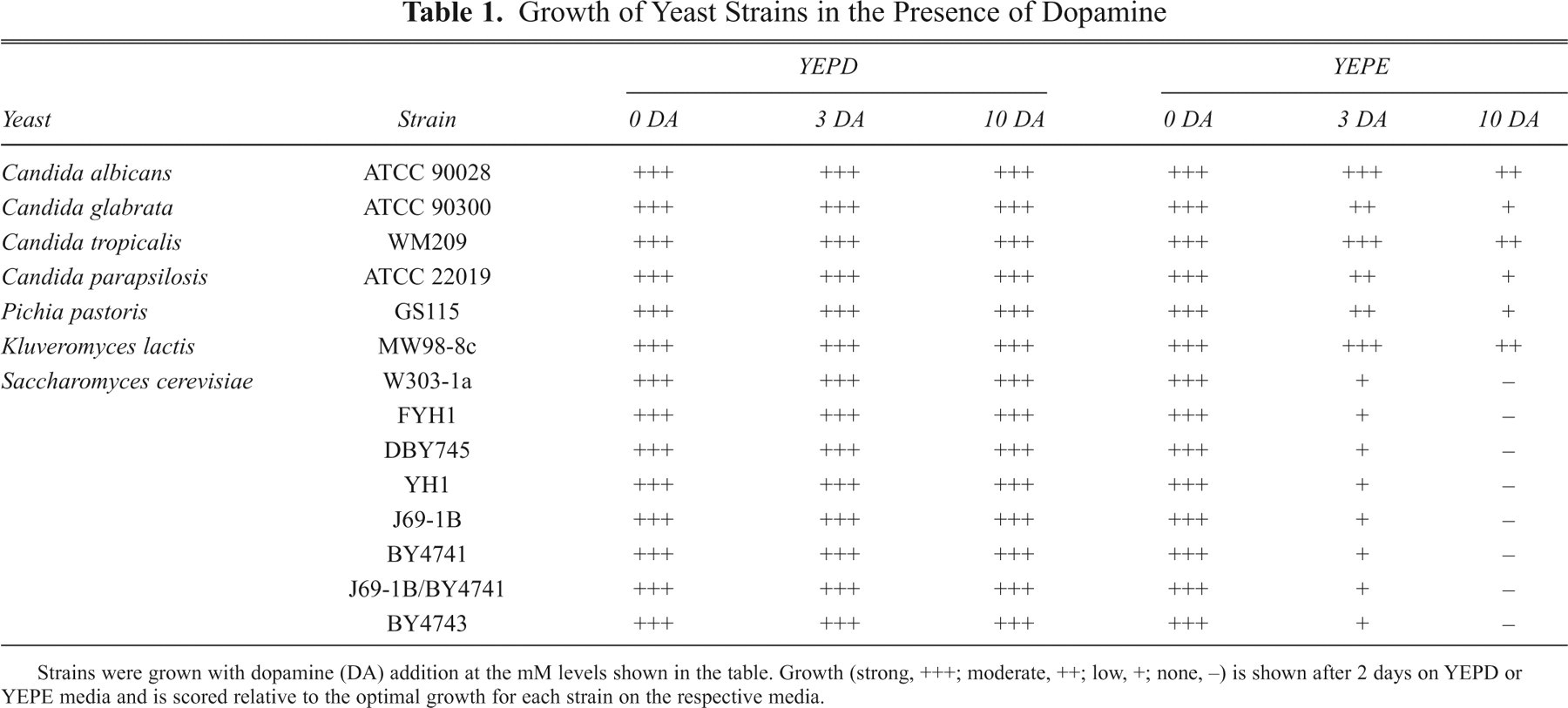
Strains were grown with dopamine (DA) addition at the mM levels shown in the table. Growth (strong, +++; moderate, ++; low, +; none, −) is shown after 2 days on YEPD or YEPE media and is scored relative to the optimal growth for each strain on the respective media.
An example of the dopamine-induced growth inhibition for 2 strains (C. glabrata and the S. cerevisiae strain W303-1a) is shown in Figure 1 . It is notable that in YEPE media, there is dark brown pigmentation that increases with the time of incubation. This pigmentation also develops when ethanol is replaced by glycerol as the carbon source, and growth inhibition occurs under these conditions as well ( Fig. 1 ). There is much less pigmentation when glucose is the sole carbon source (data not shown). This raises a possibility that a reaction product or products of dopamine might be responsible for the observed effects. Dopamine causes a cytostatic growth inhibition because after 3 days in the presence of YEPE media W303-1a cells remained viable.
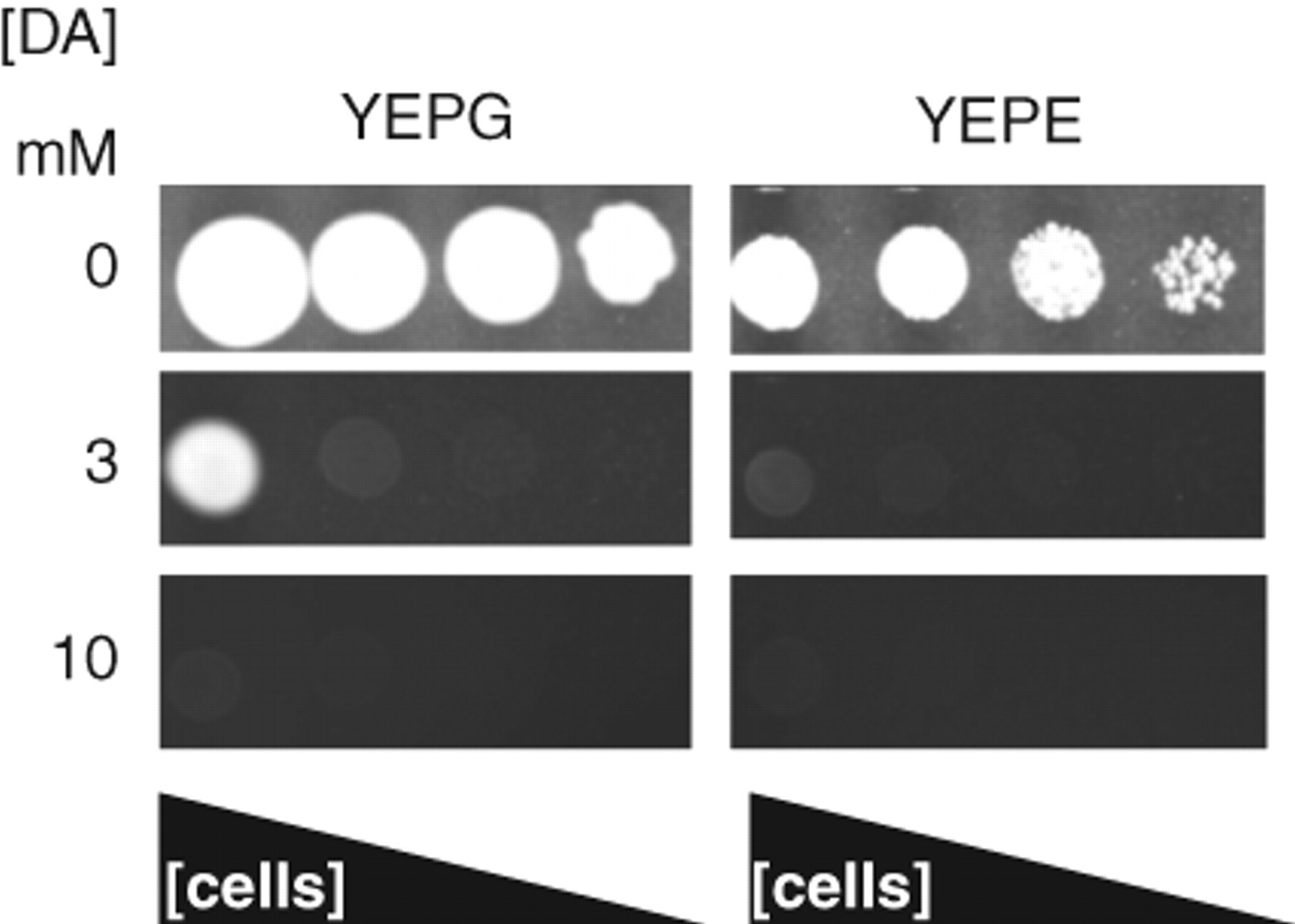
Growth of Candida glabrata ATCC 90300 on solidified YEPG and YEPE media with dopamine added at concentrations shown. Dilutions of cells (approx. 50, 500, 5000, and 50,000 cells) were added to each area of the plates as indicated. Plates were incubated for 2 days. The YEPE and YEPG media darkened in color after 2 days due to breakdown of the dopamine.
Sensitivity to dopamine was also observed in W303-1a cells suspended in water. If cells from log phase growth were suspended in water, their survival was diminished with the addition of dopamine ( Fig. 2 ). This cell killing was concentration and time dependent. In 2 h, 30 mM dopamine killed 30% of the population (data not shown), and after 16 h, 100% of the cells were killed ( Fig. 2 ). For a 16-h exposure to dopamine, the IC50 was 8.5 mM. The related compound, 6-hydroxydopamine, exhibited much greater toxicity, with 1 mM causing cell death of the entire population; the IC50 was 0.25 mM.
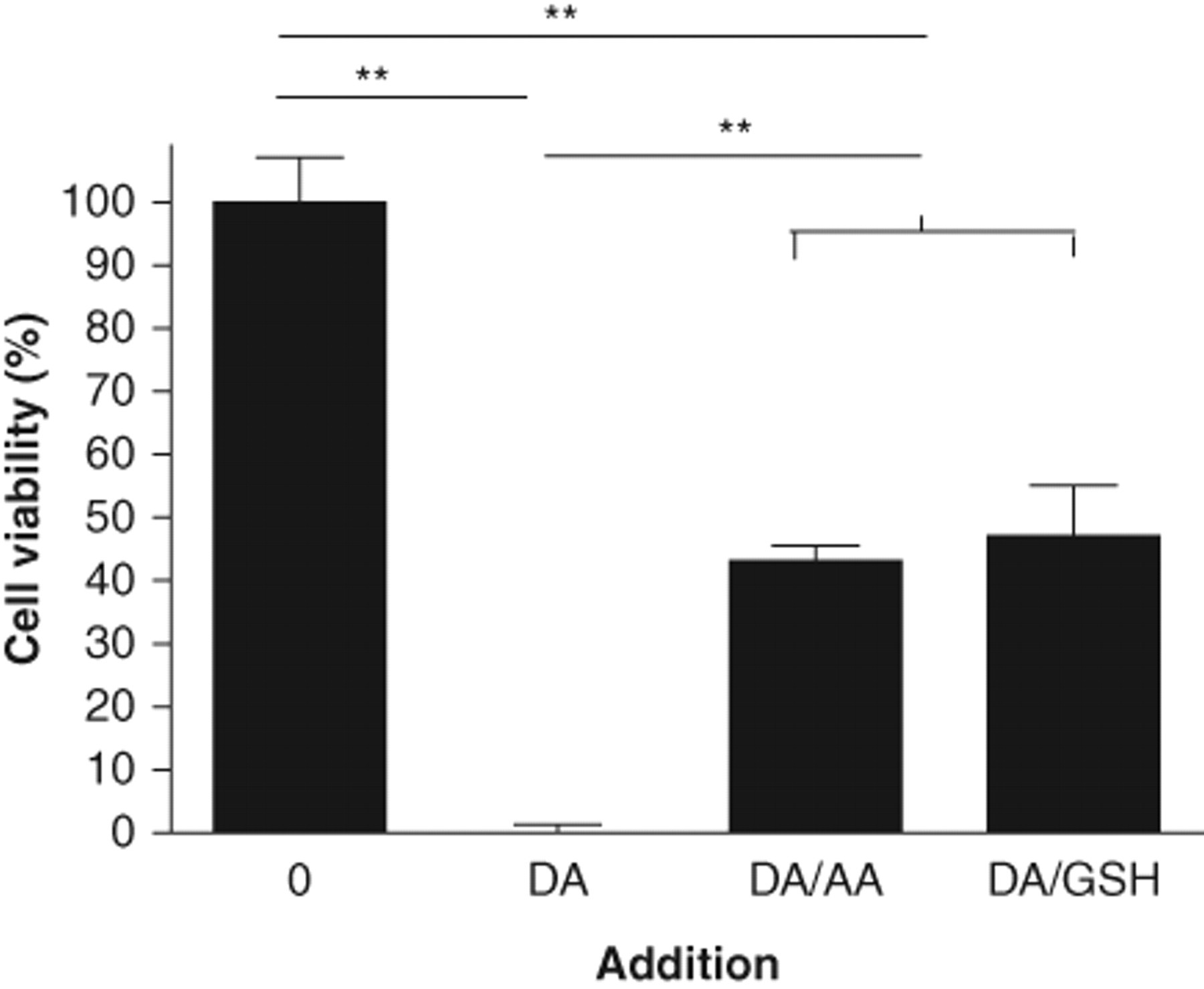
Inhibition of dopamine (DA)–mediated cell killing in the presence of ascorbate (AA) and glutathione (GSH). Saccharomyces cerevisiae strain W303-1a cells were suspended in water and treated with 10 mM dopamine, 10 mM ascorbic acid, and 10 mM reduced glutathione as indicated. After 16 h, cells were plated to determine viability. The mean and standard deviation are shown. Statistical analysis: **p < 0.01; n = 3.
Following treatment, the supernatant in which cells were treated was examined by high-performance liquid chromatography (HPLC). The profile exhibited a peak at 5 min ( Fig. 3 ), the same as a pure dopamine solution.
Analysis of the dopamine solution after exposure to cells. After 24-h exposure to yeast cells, 25 µL of the dopamine-containing culture supernatant was examined by high-performance liquid chromatography.
Ascorbate and glutathione counteract dopamine’s effects
Dopamine’s inhibition of yeast respiratory growth was capable of being alleviated somewhat by ascorbate or reduced glutathione. All strains that exhibited dopamine-induced growth inhibition were capable of having the inhibition rescued. Examples of this “rescue” for 2 yeast species, C. glabrata and S. cerevisiae, are shown in Figure 4 . It is also notable that the glutathione and ascorbate also reduce the production of the pigmented compound.
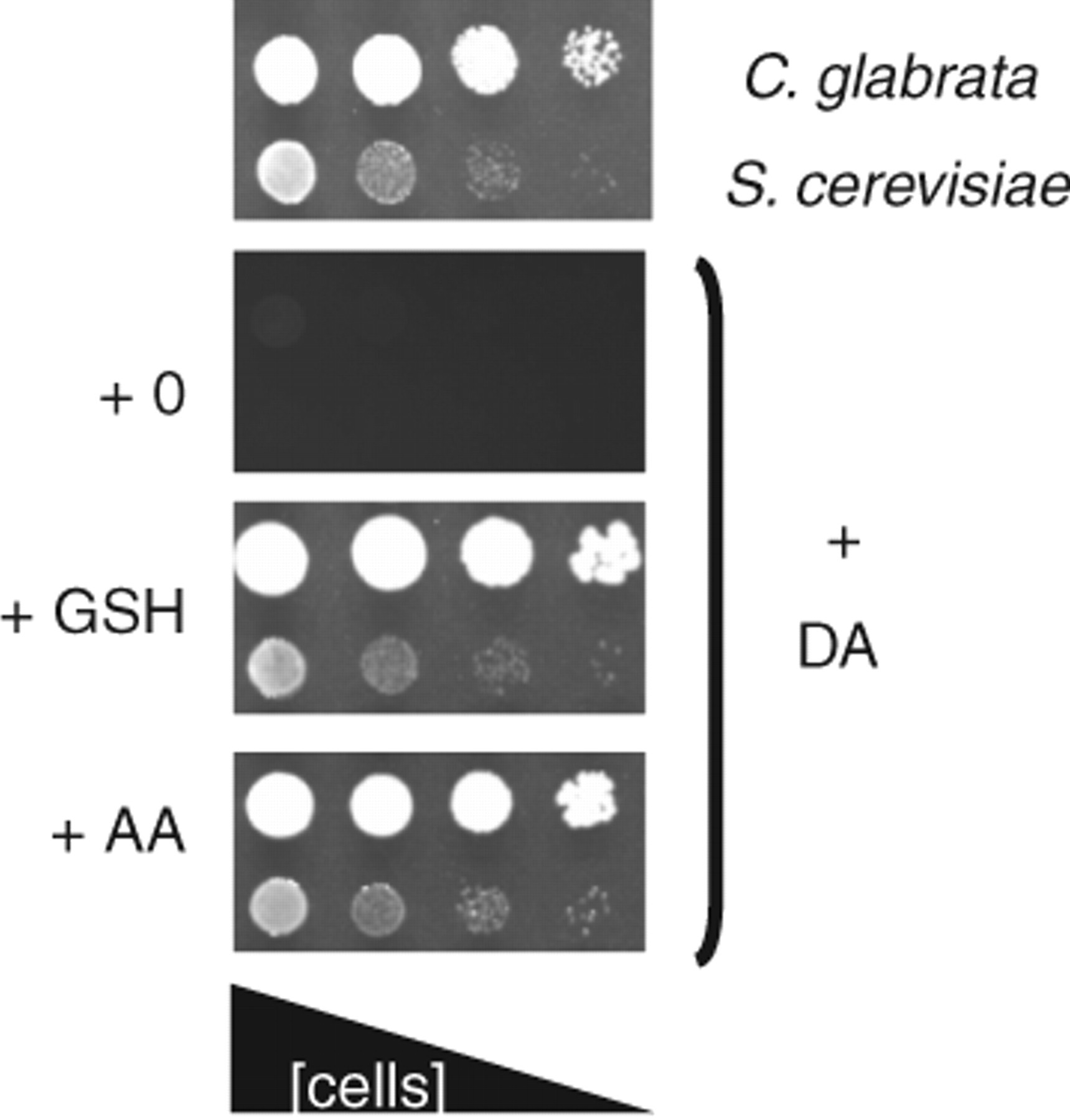
Growth of Candida glabrata and Saccharomyces cerevisiae W303-1a on solidified YEPE media in the presence of 10 mM glutathione, 10 mM ascorbic acid, and 10 mM dopamine as indicated. Other conditions are as described in Figure 1 .
Inhibition of dopamine-induced death was also investigated for S. cerevisiae W303-1a ( Fig. 2 ). In the experiment shown, 10 mM dopamine addition caused almost 100% cell death. With the further addition of 10 mM ascorbate or 10 mM glutathione, the cell killing was significantly inhibited: about half of the cells survived.
Discussion
In this study, dopamine was shown to exert concentration-dependent growth inhibition of yeast on YEP medium, containing ethanol or glycerol as the carbon source. The substitution of glucose as a carbon source allowed normal growth in the presence of dopamine. This is a classic growth phenotype that typifies inhibitors of yeast mitochondrial function. Such inhibitors include oligomycin, venturicidin, erythromycin, chloramphenicol, mucidin, and antimycin. The growth response demonstrates that dopamine’s effects were due to the inhibition of mitochondrial respiratory function.
Previous studies using neuronal tissue 19-22 have identified mitochondrial respiratory function as a target for dopamine. However, the use of neuronal cells requires sophisticated handling and assays. In some cases, isolated mitochondria from such tissue have been used 23 to examine other brain chemicals that interfere with mitochondrial function. For example, DOPAC has exhibited a significant effect, but in the current study, it had no effect. Possibly DOPAC was not taken up by the yeast cells.
Dopamine had a cytostatic effect in YEPE media and had the ability to kill yeast cells suspended in water. The killing could occur in the absence of growth, and the entire population was susceptible. 6-Hydroxydopamine also caused toxicity and with increased effectiveness. Both have been observed to kill cells in cultured cell lines, with 6-hydroxydopamine being more potent. 13,17 The levels of dopamine and 6-hydroxydopamine used in this study were very high compared to what has been shown to be toxic levels in mammalian cells that have a dopamine transporter, which would serve to recruit extracellular dopamine into the cell; yeast do not appear to have an equivalent transporter. In mammalian cells, levels as low as 10 µM dopamine have been shown to exert inhibitory effects to cells in culture. 24
Dopamine levels and the fate of the dopamine in the yeast cytoplasm during the experiments here have not been ascertained. Dopamine solutions in which cells were suspended appeared unchanged after overnight incubation, with yeast suggesting that all effects arose from intracellular dopamine or reaction product(s) derived from dopamine.
The inhibitory effect of dopamine was significantly reduced by equimolar amounts of reduced glutathione or ascorbic acid. The most likely explanation is that dopamine, or its reaction product(s), causes oxidative damage, and this is prevented by glutathione or ascorbic acid. Glutathione has previously been implicated in protection of cells from dopamine toxicity. PC12 cells, derived from a pheochromocytoma of the rat adrenal medulla, were protected from dopamine toxicity by glutathione, 25 whereas neuroblastoma cells depleted of glutathione by buthionine sulfoximine were shown to be more sensitive to dopamine. 26 Glutathione and ascorbate inhibited autoxidation, as well as toxicity of dopamine and L-DOPA, to Neuro-2A cells. 17 In the above cases, it is likely that dopamine’s effects are caused through its reaction products, whose production can be inhibited by ascorbate or glutathione. It could be worth addressing the fate of dopamine in yeast with radiolabeling experiments, but this is beyond the scope of the present study.
These effects of dopamine on yeast may be relevant to neurobiology, particularly Parkinson’s disease. When it comes to the examination of dopamine’s effects on mitochondrial function, yeast may provide an extremely useful experimental model because of the ability of yeast to grow with or without mitochondrial function. This growth characteristic is an easy attribute to work with and may be useful for further studies and to identify more antagonists of dopamine-induced effects.
Footnotes
Acknowledgements
We thank Kou Nga Io (Gloria Kou) for her excellent technical assistance and Drs. Tom Peat and Tim Adams for helpful discussions in the preparation of this manuscript.
