Abstract
Background:
Children with ADHD show attentional and cognitive control deficits compared to typically developing controls. However, it is unclear whether these deficits are function-specific to subcomponents of attention and cognitive control and/or disorder-specific, that is, unique to one clinical group.
Methods:
Here, we characterized subcomponents of attentional and cognitive control in children with ADHD (N = 37, 21.6% girls), ADHD and comorbid anxiety disorder (AnxD, N = 24, 50% girls), AnxD (N = 39, 30.8% girls), as well as typically developing controls (N = 36, 41.7% girls) aged 7 to 14 years, using behavioral measures from the Attention Network Test (ANT) and AX Continuous Performance Task (AX-CPT). Analysis of variance (ANOVA) and post hoc t-test were used to test for group differences. Cohen’s d was used as a measure of effect size for significant group differences.
Results:
Children with ADHD and children with ADHD and comorbid AnxD showed lower accuracy (Cohen’s d = −0.91/−1.08), higher reaction time variability (Cohen’s d = 0.81/1.13) in ANT-derived measures and lower accuracy (Cohen’s d = −0.89/−1.04) and A-cue bias (Cohen’s d = −0.92/−0.98) in AX-CPT -derived measures compared to controls. Children with ADHD and comorbid AnxD also exhibited higher reaction time (Cohen’s d = 0.83) in the ANT and lower d’-context (Cohens’ d = −0.97) in the AX-CPT compared to controls.
Conclusion:
The results indicate function-specific deficits in cognitive control, but not in attention, among children with ADHD and comorbid AnxD. We did not find strong evidence for disorder-specific deficits in attention or cognitive control.
Introduction
Attention deficit/hyperactivity disorder (ADHD) is a neurodevelopmental disorder with an estimated global prevalence of ~5% (Polanczyk et al., 2007), but the number of ADHD diagnosed children and adolescents has been rapidly increasing over the last decades (Xu et al., 2018). Thus, there is an urgent need to establish robust phenotypic profiles of ADHD to aid clinical assessments and treatments among children diagnosed with ADHD. Although impaired attentional and cognitive control functions have been long-standing central features of neurocognitive theories of ADHD (Barkley, 1997; Castellanos & Tannock, 2002), there is also substantial heterogeneity in attention and cognitive control functions among children with ADHD, potentially influenced by subgroups with comorbid psychopathological conditions. Indeed, individuals with ADHD show high rates of comorbidity with other childhood onset disorders (see Gnanavel et al., 2019 for a review), where ~25% to 50% also exhibit symptoms of anxiety disorders (Bowen et al., 2008; Overgaard et al., 2016; Quenneville et al., 2022; I. Souza et al., 2005). Anxiety disorders are characterized by altered attentional control (Bishop, 2007; Dudeney et al., 2015) and atypical performance in cognitive control tasks (Coussement et al., 2022; Eysenck et al., 2007; Majeed et al., 2023; Mogg et al., 2015; Pacheco-Unguetti et al., 2011). Cognitive interference theory and attentional control theory suggest that anxiety impairs cognitive performance due to external or internal threat stimuli (e.g., worrying thoughts), which may manifest itself in altered processing efficiency but not necessarily performance effectiveness (Eysenck & Calvo, 1992; Eysenck et al., 2007; Sarason, 1988). In ADHD, comorbid anxiety has been suggested to be a consequence of slow information processing and/or behavioral inhibition deficits that disrupts aspects of social functioning (Barkley, 1997; Schatz & Rostain, 2006). Interestingly, previous studies have also suggested that comorbid anxiety may improve ADHD-related inhibitory deficits (Manassis et al., 2000; Maric et al., 2018; Pliszka, 1992), modulate the relationship between ADHD severity and neural activity during a cognitive task (van der Meer et al., 2018), and can yield a distinct cognitive profile that deviate from either disorder (Manassis et al., 2000; Vloet et al., 2010). Indeed, it has been suggested that anxiety in ADHD may improve behavioral inhibition deficits seen in ADHD only (i.e., impulsivity and response inhibition), but may worsen working memory (Gnanavel et al., 2019). The impaired working memory may underlie selection efficiency (Vogel et al., 2005), indicating that attentional processes act, in part, as a gating mechanism for working memory. Moreover, attention and cognitive control are complex cognitive constructs posited to consist of several subcomponents (Braver, 2012; Petersen & Posner, 2012; Posner & Petersen, 1990; Posner & Rothbart, 2007), and it remains unclear which of these subcomponents are affected in children with ADHD and comorbid psychopathological conditions. Thus, studies that isolate subdomains of attentional and cognitive control functions, and takes comorbidity between ADHD and anxiety into account, are needed to gain insights into function-specific and disorder-specific profiles of ADHD.
Inattention is one of the core behavioral symptoms of ADHD and can include a broad specter of attentional deficits in everyday life. However, everyday situations may require recruitment of several subsystems of attention, including executive control (i.e., the ability to resolve conflict among responses), orienting (i.e., the ability to select relevant information from sensory input), alerting (i.e., the ability to achieve and maintain an alert state; Petersen & Posner, 2012; Posner & Petersen, 1990; Posner & Rothbart, 2007). Thus, methods that can better isolate subcomponents of attention are needed to gain insight into function-specific deficits in ADHD. The Attention Network Test (ANT) is a computer-based task that is designed to measure the efficacy of the executive control network, orienting network and alerting network (Fan et al., 2002). Previous studies have shown that children with ADHD exhibit worse performance in global indices derived from ANT data sets, including lower overall accuracy (Adólfsdóttir et al., 2008; Booth et al., 2007), slower reaction time (Booth et al., 2007; Casagrande et al., 2012; Suades-González et al., 2017), increased reaction time variability (Adólfsdóttir et al., 2008; Suades-González et al., 2017) and deficits in the alerting network (Bieleninik et al., 2023) compared to typically developing controls. Moreover, children with ADHD show better orienting scores compared to children with anxiety (Mogg et al., 2015), possibly pointing toward a unique profile of attentional deficits in ADHD with comorbid anxiety.
Beyond attention functions, cognitive control deficits have been proposed as a core cognitive phenotype of ADHD (de Zeeuw & Durston, 2017). According to the dual mechanisms of control (DMC) framework, cognitive control can be subdivided into proactive and reactive cognitive control (Braver, 2012). Proactive control is considered a top-down control mechanism and refers to the sustained maintenance of relevant information for goal-directed behavior or thoughts. In contrast, reactive control is a bottom-up process, where cognitive control mechanisms are transiently activated in response to a cognitive demanding task. The AX Continuous Performance Task (AX-CPT) has been widely used to examine cognitive control in mental and neurodevelopmental disorders (Chun et al., 2018; Hogeveen et al., 2018; Niendam et al., 2018), which provides behavioral measures of proactive and reactive cognitive control. Previous studies have found support for impaired reactive control, but not proactive control in children with ADHD or ADHD symptoms (Hulst et al., 2018; Pani et al., 2013; Suarez et al., 2021), whereas others have found deficits in both proactive and reactive cognitive control, which are also associated with behavioral problems in children with ADHD (Cai et al., 2023). However, it is still unclear whether the cognitive control deficits are due to reduced ability to recruit proactive or reactive cognitive control (de Zeeuw & Durston, 2017), emphasizing the utility of using cognitive tasks including measures of both reactive and proactive cognitive control.
To establish disorder-specific phenotypic profiles in children with ADHD and comorbid anxiety, there is a need for adequate comparison groups, including typically developing children and children with anxiety but without ADHD (Jarrett et al., 2016; Tannock, 2009). As such, comparison between ADHD with comorbid anxiety and anxiety can provide important insight into whether ADHD-related deficits are exacerbated by comorbid anxiety or if they are disorder-specific to ADHD. Thus, the current study aims to examine function-specific subcomponents of attention and cognitive control in children with ADHD, both with and without comorbid anxiety, anxiety, and typically developing controls. According to attentional control theory, anxiety impairs processing efficiency more than performance effectiveness, resulting in comparable accuracy at the expense of slower responses. Thus, comorbid anxiety in ADHD is hypothesized to improve accuracy, slow down responses, and improve inhibition.
Methods
Sample
A total of 136 children aged 7 to 14 years were included in the study. The clinical sample was recruited from outpatient child and adolescent psychiatric clinics and included children who met the DSM-IV criteria for ADHD and/or an anxiety disorder (AnxD), whereas the control sample was recruited from neighboring schools as described elsewhere (Skirbekk et al., 2011). The children’s parents were interviewed using the Kiddie Schedule for Affective Disorders and Schizophrenia (Kiddie-SADS) Present and Lifetime version, which is a semi-structured diagnostic interview designed to assess current and past episodes of psychopathology in children and adolescents according to DSM-IV criteria (Kaufman et al., 1997). Interviews were administrated by experienced clinicians. Thirty-nine randomly selected audiotaped interviews were used to assess interrater reliability, yielding high agreement for ADHD (Cohen’s kappa = .90) and any AnxD (Cohen’s kappa = .88). Children who met the diagnostic criteria for any AnxD or ADHD were invited to participate in the study. None of the participating children were receiving psychopharmacological treatment. The specific anxiety disorders represented in the ANX group were as follows: panic disorder (n = 2), separation anxiety (n = 15), specific phobia (n = 9), social phobia (n = 15), agoraphobia (n = 2), generalized anxiety disorder (n = 6), obsessive/compulsive disorder (n = 9). In the ADHD + AnxD group, the anxiety disorders included: separation anxiety (n = 12), specific phobia (n = 10), social phobia (n = 2), agoraphobia (n = 1), generalized anxiety disorder (n = 1), obsessive/compulsive disorder (n = 4). Note that some of the participating children meet the diagnostic criteria for more than one AnxD. The Wechsler Abbreviated Scale of Intelligence (WASI) was administered to estimate the intelligence quotient (IQ, Wechsler, 1999). The study was approved by the Regional Committee for Medical Research Ethics and the Norwegian Data Inspectorate. The parents, and the children aged 12 years and older, gave their written informed consent to participate in the study. Verbal informed consent was given from children younger than 12 years. Sample characteristics are presented in Table 1.
Sample Characteristics.
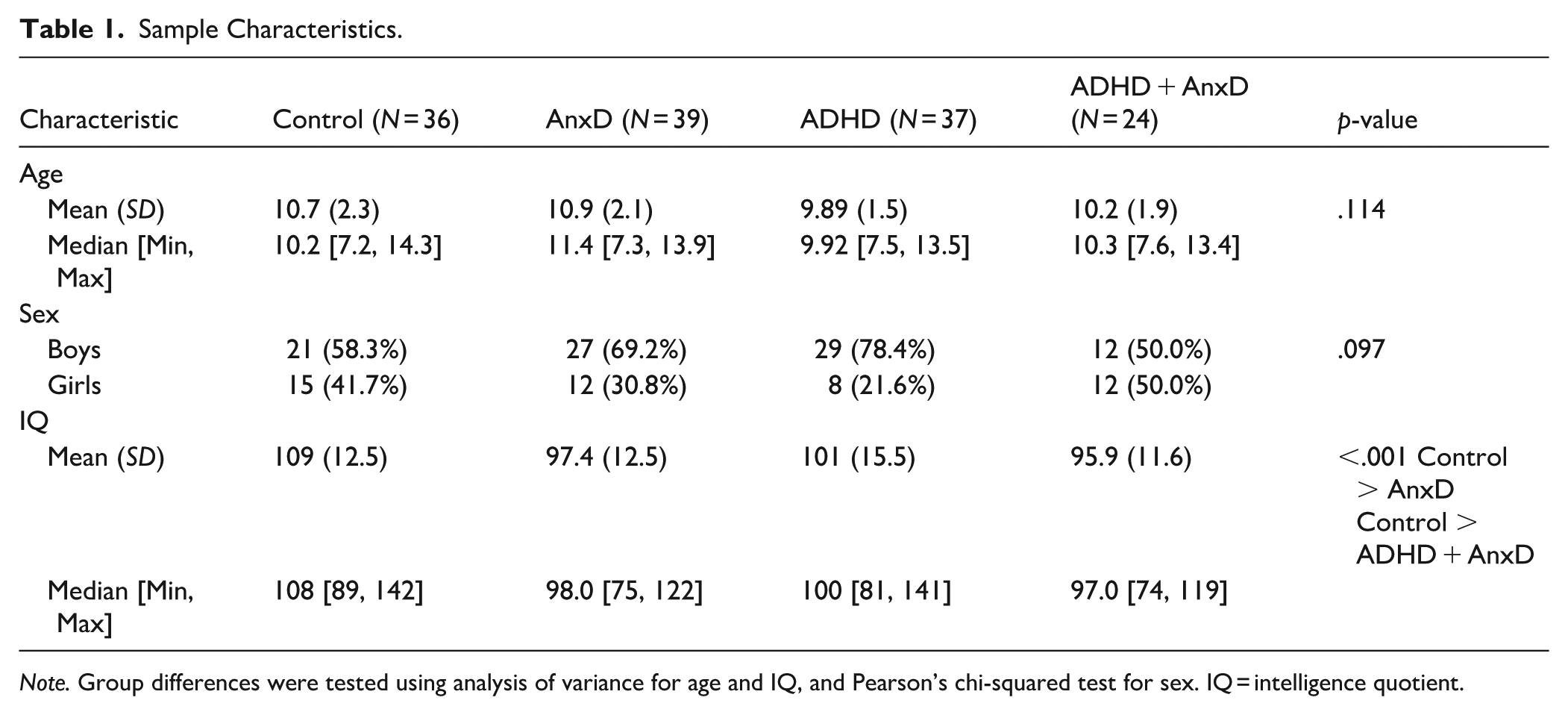
Note. Group differences were tested using analysis of variance for age and IQ, and Pearson’s chi-squared test for sex. IQ = intelligence quotient.
Derivatives of the Dependent Variables
Attention Network Test (ANT)
The child version of ANT (Rueda et al., 2004) is a computerized task designed to measure three types of attention networks, that is, alerting, orienting, and executive control, in children. The child version of ANT consists of a practice block of 24 trials and three experimental blocks with 48 trials each, where the participants respond to rightward or leftward target (i.e., line drawing of a yellow fish) by pressing a button corresponding to the direction of the target. A trial consists of one out of 12 conditions, with three target types and four cues, including a congruent (i.e., target flanked by yellow fish pointing in the same direction), incongruent (i.e., target flanked by yellow fish in the opposite direction), and neutral (i.e., no flankers) preceded by either a center cue, a double cue, a spatial cue, or no cue. Each trial was initiated with a fixation period for a random duration of 400 to 1,600 ms, followed by, if presented, the cue stimuli for 100 ms and then a fixation cross for 450 ms before the target stimuli. The target stimuli remained visible on the screen until response or for 1,700 ms. For the ANT, we calculated the overall accuracy, median reaction time (RT) and standard deviation (SD) of RT to get global measures. Furthermore, the combination of the flanker and cue conditions can be used to estimate measures of the executive control network, the orienting network and the alerting network using the median reaction time (RT) across trials. In line with previous research (Boen et al., 2021; Westlye et al., 2011), the ANT components were calculated based on median reaction times using a ratio procedure to isolate the attention scores from reaction time differences and calculated as follows: Executive control = (median RT incongruent trials − median RT congruent trials)/median RT congruent trials; Orienting = (median RT center cue − median RT spatial cue)/median RT spatial cue; Alerting = (median RT no cue − median RT double cue)/median RT double cue.
AX-Continuous Performance Task (AX-CPT)
The AX-CPT (Rosvold et al., 1956) is a widely utilized behavioral paradigm for assessing proactive and reactive cognitive control. This task involves the identification of specific cue-probe letter pairs, with the target pair consisting of the letter “A” followed by the letter “X.” During each trial, participants are required to determine whether the presented cue-probe pair constitutes a target (AX) or a non-target (e.g., AY, BX, and BY). In some trials, the cue (such as the letter “B”) predicts the correct response, allowing participants to prepare their response in advance. In other trials, specifically when an “A” cue is presented, the correct response can only be determined once the probe appears—an “X” following “A” signals a target, while a non-X letter signals a non-target. Due to the higher frequency of AX trials (70%) compared to other trial types (10% each), participants often develop an expectancy that an “A” cue will be followed by an “X,” leading to a tendency to commit AY errors (incorrectly anticipating a target response). Trials with such errors are considered to results from proactive control, while trials with BX errors are viewed as relying more on reactive control. In the task, capital letters, presented in red on a black background, were displayed sequentially on a computer screen. Each letter was shown for 300 ms, with a 3,000 ms stimulus onset asynchrony (SOA). The participants responded by depressing a non-target button on a response box for each non-target stimulus, and a target button for each target stimulus. Responses within 100 to 2,000 ms after stimulus onset were included in the analysis. The procedure included a practice block of 20 trials and four experimental blocks of 50 trials each. For the AX-CPT, we computed the overall accuracy and overall RT, and the following measures based on accuracy scores: d’-context (calculated by computing a d’-index from hits on AX trials and false alarms on BX trials as Z(H) − Z(F), with H representing hits on AX trials, F representing false alarms on BX trials, and Z representing the z-transform of a value), A-cue bias (calculated by computing a c criterion from hits on AX trials and false alarms on AY trials as 1/2(Z[H] + Z[F]), with H representing hits on AX trials and F representing false alarms on AY trials), and BX probe interference (BX − BY). Previous studies have interpreted higher d’-context and A-cue bias scores as indicative of a proactive cognitive control strategy (e.g., Filippi et al., 2022; Gonthier et al., 2016; Valadez et al., 2022) and higher BX probe interference as indicative of a reactive cognitive control strategy (e.g., Husa et al., 2022; Valadez et al., 2022).
Statistical Analysis
All statistical analyses were conducted in R v4.2.3. We regressed out the effect of age and sex on all the dependent variables, and the residuals where further z-scored across all groups. First, we correlated all the dependent variables across all groups for the ANT and the AX-CPT derived measures, respectively. The correlations were adjusted for multiple comparisons using Bonferroni correction for 15 correlations in the ANT derived measures and across 10 correlations for the AX-CPT derived measures. To test whether the significant correlations differed between groups, we compared the correlation coefficients using Fisher’s z-transformation implemented in the cocor package in R (Diedenhofen, 2022). We report significant difference between correlations after Bonferroni correction. Second, analysis of variance (ANOVA) was used to test for group differences among the four groups (i.e., ADHD, ADHD + AnxD, AnxD, and typically developing controls). Here, we used the eta2 as measure of effect size. Post hoc tests were computed for the significant ANOVA results. The post hoc tests were performed using t-test between all groups (i.e., six comparisons), where the p-values underwent correction for multiple comparison using Bonferroni correction. We report the Cohen’s d for the significant group differences. We also ran several sensitivity analyses on the significant findings, including adjustment for median RT on SD of RT and adjustment for IQ on all the significant group differences. Next, we investigated the associations between performance scores on the ANT and AX-CPT tasks and assessed whether these correlations differed between clinical groups and controls using Fisher’s z-transformation and Bonferroni correction for multiple comparisons. To examine age-related differences in ANT and AX-CPT scores across and between groups, we conducted separate linear regression analyses including age and age-by-group interaction terms. Sex was included as a covariate in all models, and p-values were Bonferroni-corrected for 11 and 33 comparisons, respectively.
Results
Group Differences in Measures of Attentional Functioning
The unadjusted values for executive control, orienting, alerting, accuracy, RT, and SD of RT derived from the ANT are presented in Table 2 and correlations between the age and sex adjusted values are presented in Figure 1. Here, the positive correlation between RT and SD of RT was found to be significant different between the AnxD and the ADHD group (z = 2.98, p = .005, Supplemental Table 1), indicating a stronger association for children with AnxD compared to ADHD only.
Overview of Mean Performances in the ANT Across Groups.
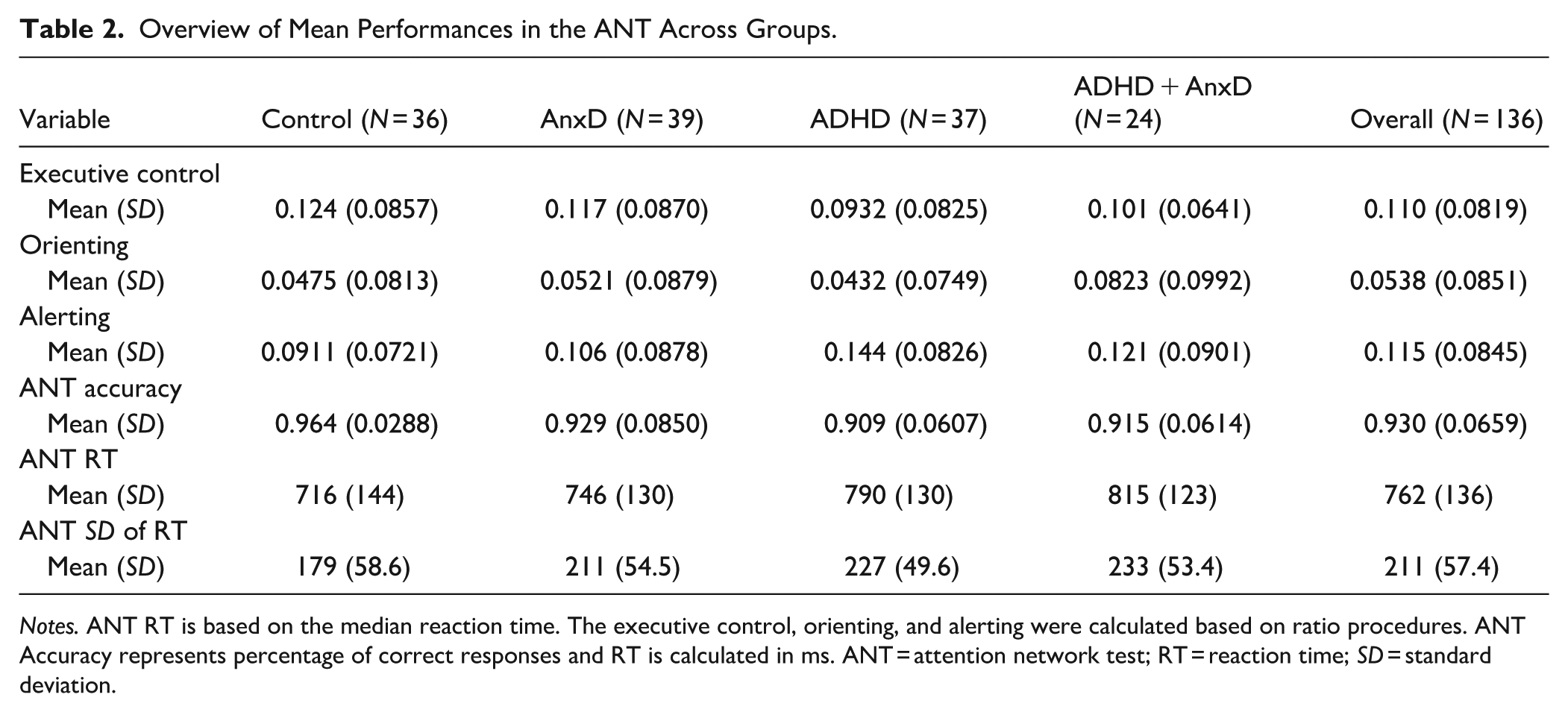
Notes. ANT RT is based on the median reaction time. The executive control, orienting, and alerting were calculated based on ratio procedures. ANT Accuracy represents percentage of correct responses and RT is calculated in ms. ANT = attention network test; RT = reaction time; SD = standard deviation.
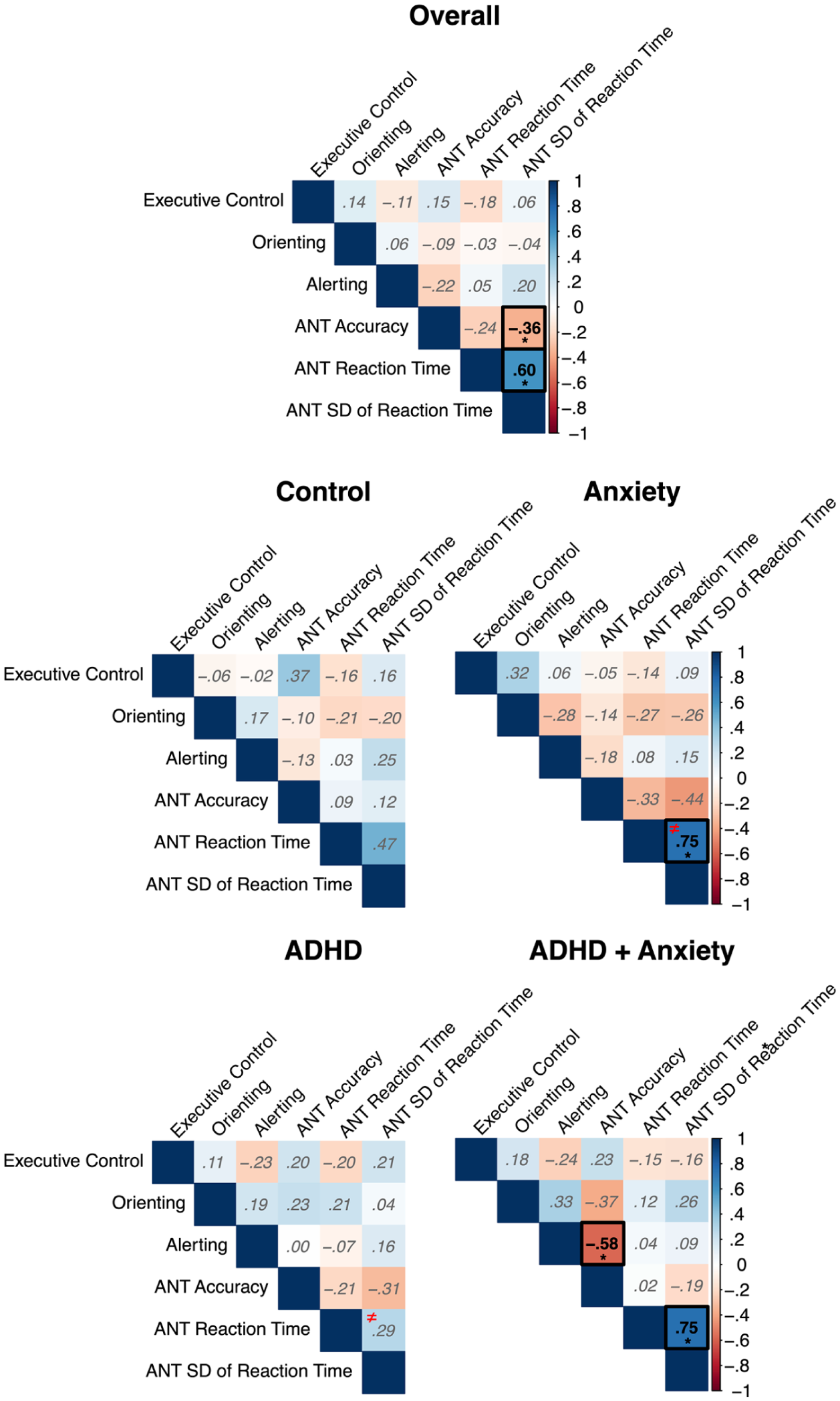
Pearson correlations between the derived measures from the Attention Network Test (ANT) for the overall sample and each of the clinical group.
The results showed a main effect of group for ANT accuracy (F (3, 132) = 4.96, p = 0.003; η2 = .10, 95% CI [0.02, 1.00]), where the ADHD (Cohen’s d = −0.91, 95% CI [−1.39, −0.42] ) ADHD with comorbid AnxD (Cohen’s d = −1.08, 95% CI [−1.62, −0.52]), and AnxD (Cohen’s d = −0.69, 95% CI [−1.15, −0.22] ) groups showed lower accuracy compared to typically developing controls (Figure 2(A)). The group differences in ANT accuracy remained significant after adjusting for IQ for ADHD (Cohen’s d = −0.74, 95% CI [−1.21, −0.26], p = .002), ADHD with comorbid AnxD (Cohen’s d = −0.74, 95% CI [−1.27, −0.21], p = .01) and AnxD (Cohen’s d = −0.48, 95% CI [−0.94, −0.02], p = .037) compared to typically developing controls. For median RT, we found a significant main effect of group (F(3, 132) = 2.94, p = .036; η2 = .06, 95% CI [0.00, 1.00]), where the post-hoc analysis showed a significant higher median RT in the ADHD with comorbid AnxD group compared to the control group (Cohen’s d = 0.83, 95% CI [0.29, 1.36], Figure 2(B)). However, after adjusting for IQ, the ADHD with comorbid AnxD group did not show higher median reaction time compared to typically developing controls (p = .056). Further, there was also a main effect of group on SD of RT (F(3, 132) = 6.36, p < .001; η2 = .13, 95% CI [0.04, 1.00]), where the ADHD (Cohen’s d = 0.81, 95% CI [0.33, 1.28] ), ADHD with comorbid AnxD (Cohen’s d = 1.13, 95% CI [0.57, 1.68]), and AnxD (Cohen’s d = 0.74, 95% CI [0.26, 1.20] ) groups showed higher SD of RT compared to typically developing controls (Figure 2(C)). The results remained significant after adjusting for IQ for the ADHD (Cohen’s d = 0.61, 95% CI [0.14, 1.08], p = .011), ADHD and comorbid AnxD (Cohen’s d = 0.78, 95% CI [0.25, 1.32 ], p = .006), and AnxD (Cohen’s d = 0.47, 95% CI [0.01, 0.93 ], p = .043.) groups compared to typically developing controls. Similarly, the results also survived adjustment for median RT for the ADHD (Cohen’s d = 0.56 95% CI [0.09, 1.03], p = .018), ADHD with comorbid AnxD (Cohen’s d = 0.74, 95% CI [0.21, 1.28], p = .004), and AnxD (Cohen’s d = 0.64, 95% CI [0.17, 1.10], p = .008) groups compared to typically developing controls. Finally, we did not find evidence for a significant main effect of groups for either executive control (F(3, 132) = 1.06, p = .368; η2 = .02, 95% CI [0.00, 1.00]), orienting (F(3, 132) = 1.19, p = .317; η2 = .03, 95% CI [0.00, 1.00]), or alerting (F(3, 132) = 2.36, p = 0.075; η2 = .05, 95% CI [0.00, 1.00]; Supplemental Figure 1).
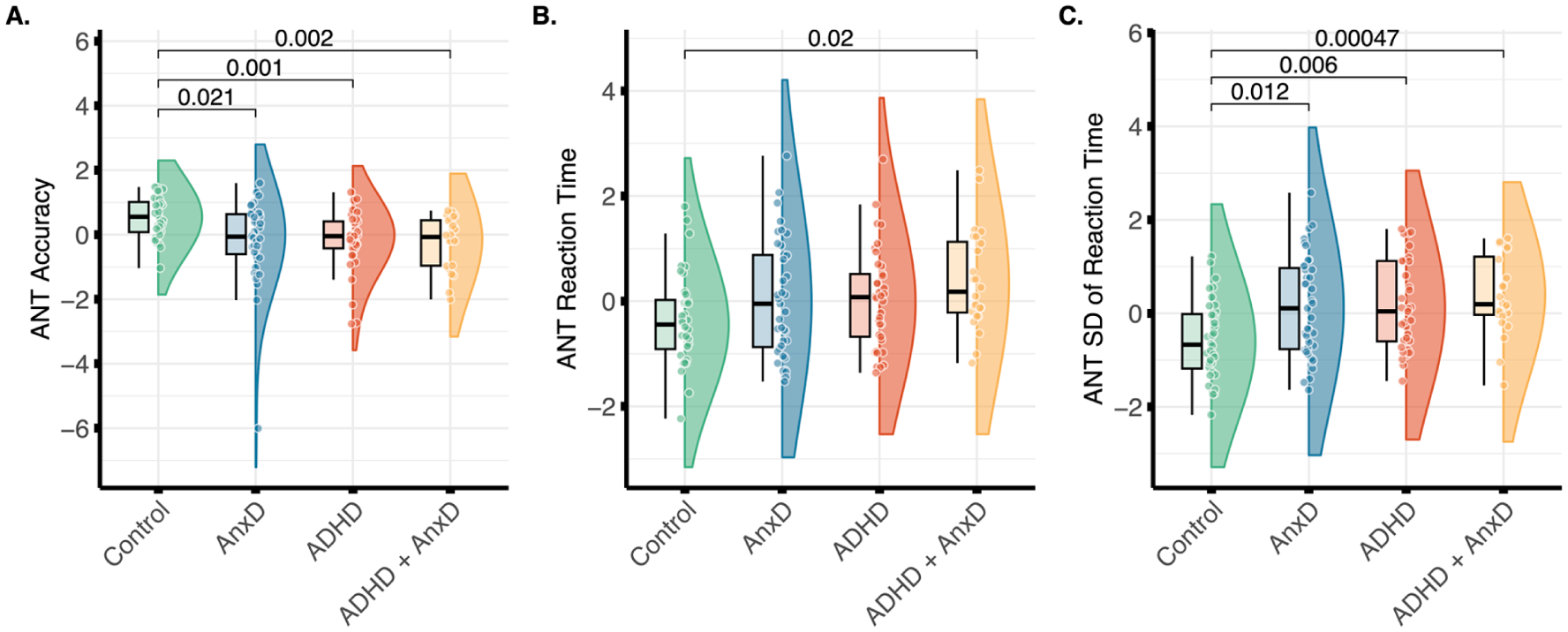
Group differences in A. overall accuracy, B. median reaction time, and C. standard deviation (SD) of the reaction time from the Attention Network Test (ANT). Significant group differences after Bonferroni correction are shown. The values were adjusted for age and sex, and z-transformed.
Group Differences in Measures of Cognitive Control Functions
An overview of the unadjusted values for the overall accuracy, overall reaction time, d’-context, A-cue bias, and BX probe interference is presented in Table 3, whereas the correlations between the age and sex adjusted values are presented in Figure 3. The positive correlation between accuracy and d’-context was found to be significant different between the control and the ADHD + AnxD group (z = −3.22, p = .001, Supplemental Table 2), indicating a stronger association for children with ADHD and comorbid AnxD compared to the control group.
Overview of Mean Performances in the – AX-CPT Across Groups.
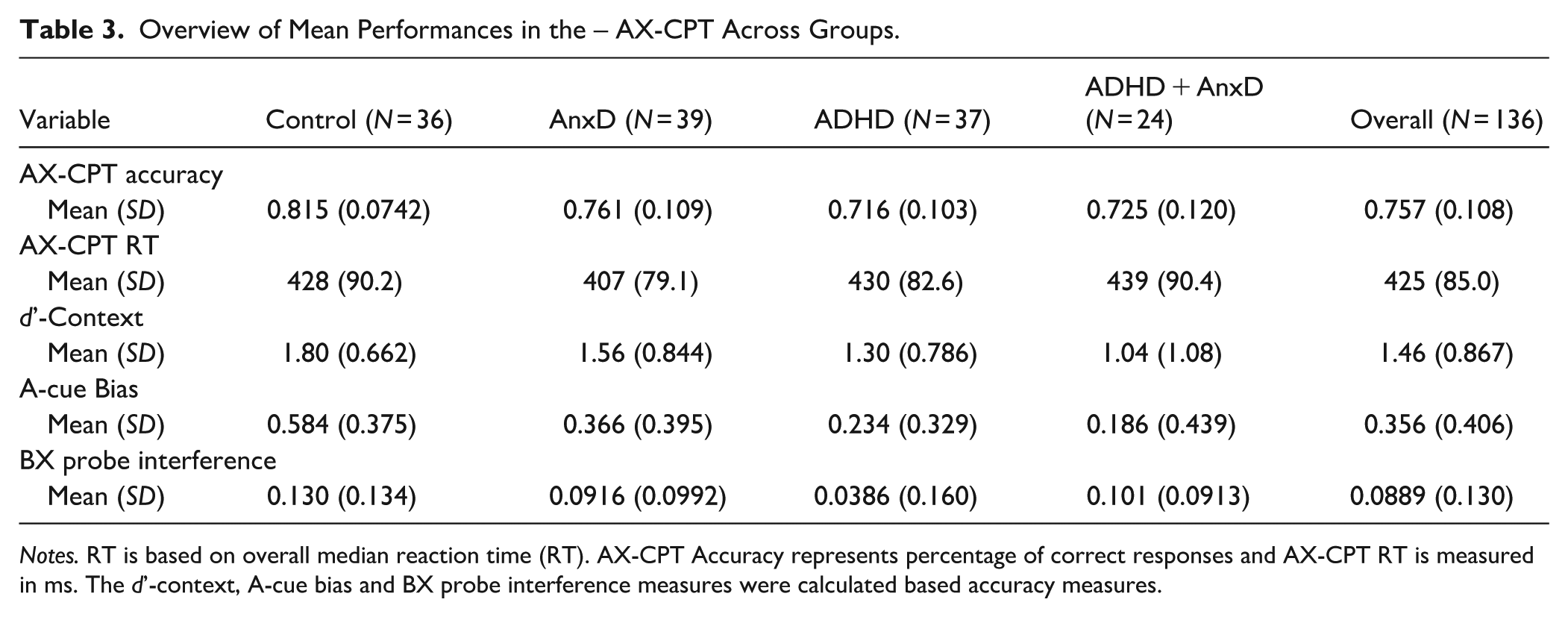
Notes. RT is based on overall median reaction time (RT). AX-CPT Accuracy represents percentage of correct responses and AX-CPT RT is measured in ms. The d’-context, A-cue bias and BX probe interference measures were calculated based accuracy measures.
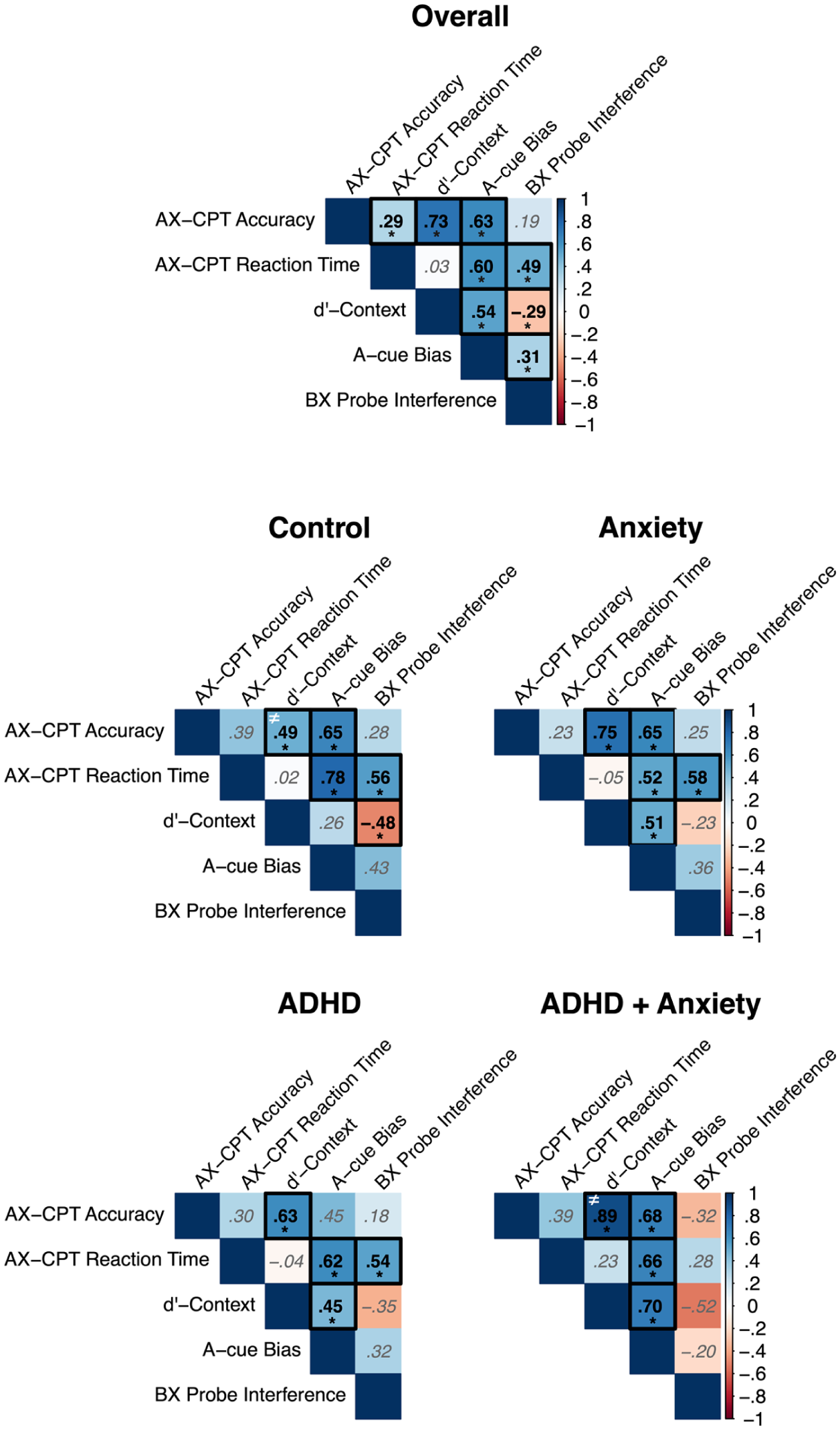
Pearson correlations between the derived measures from the AX-Continuous Performance Task (AX-CPT) for the overall sample and each of the clinical group.
For the AX-CPT performance, there was a statistically significant main effect of group on overall accuracy (F(3, 132) = 5.76, p < .001; η2 = .12, 95% CI [0.03, 1.00]), where the ADHD (Cohen’s d = −0.89, 95% CI [−1.37, −0.41]), ADHD with comorbid AnxD (Cohen’s d = −1.04 95% CI [−1.59, −0.49]), and AnxD (Cohen’s d = −0.77, 95% CI [−1.24, −0.30] ) groups showed lower overall accuracy compared to typically developing controls (Figure 4(A)).The results remained significant for the ADHD (Cohen’s d = −0.72, 95% CI [−1.19, −0.25], p = .003), ADHD and comorbid AnxD (Cohen’s d = −0.67, 95% CI [−1.20, −0.13 ], p = .026), and AnxD (Cohen’s d = −0.46, 95% CI [−0.92, 0.00], p = .048) after adjusting for IQ. There was no significant main effect of group on median RT (F(3, 132) = 0.27, p = 0.847; η2 = .01, 95% CI [0.00, 1.00]). For the d’-context measure, the main effect of group was found to be significant (F(3, 132) = 3.94, p = .010; η2 = .08, 95% CI [0.01, 1.00]), where the ADHD with comorbid AnxD group showed lower d’-context values compared to typically developing controls (Cohen’s d = −0.97, 95% CI [−1.52, −0.42], Figure 4(B)), indicating less reliance on contextual information. The group difference remained significant after adjustment for IQ (Cohen’s d = −0.65, 95% CI [−1.18, −0.12], p = .035). The results also showed a significant main effect of group on A-cue bias (F(3, 132) = 6.54, p < .001; η2 = 0.13, 95% CI [0.04, 1.00]), where the ADHD (Cohen’s d = −0.92, 95% CI [−1.40, −0.44] ) and ADHD with comorbid AnxD (Cohen’s d = −1.00, 95% CI [−1.54, −0.44]) showed lower values compared to typically developing controls (Figure 4(C)), indicating less A-cue bias. The results on A-cue bias remained significant after adjusting for IQ for both the ADHD (Cohen’s d = −0.87, 95% CI [−1.34, −0.38], p < .001) and the ADHD with comorbid AnxD (Cohen’s d = −0.90, 95% CI [−1.44, −0.36], p = .002). We also observed a main effect of group on BX interference (F(3, 132) = 3.01, p = .033; η2 = .06, 95% CI [0.00, 1.00]), however, none of the groups showed significant group differences after adjustment for multiple comparisons.
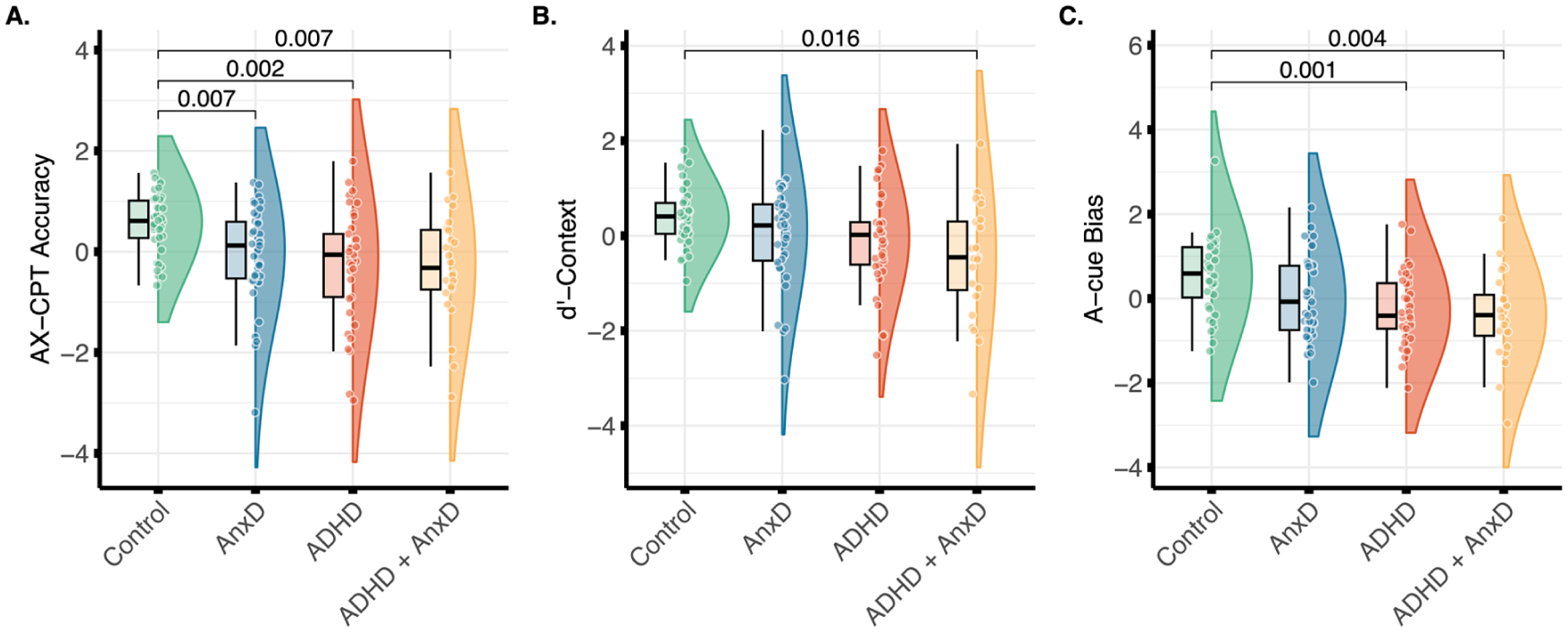
Group differences in A. overall accuracy, B. d’-context, and C. A-cue bias from the AX-Continuous Performance Task (AX-CPT). Significant group differences after Bonferroni correction are shown. The values were adjusted for age and sex, and z-transformed.
Age-Related Analyses and Cross-Task Correlations
Across all groups, there were significant age effects on accuracy (b = 0.02, p < .001), reaction time (b = −44.04, p < .001), and SD of RT (b = −14.26, p < .001) from the ANT-derived measures and on accuracy (b = 0.03, p < .001), reaction time (b = −14.97, p < .001), and d’-context (b = 0.17, p < .001) from the AX-CPT-derived measures, indicating that older participants tended to be more accurate, responded faster and less variable, and used a more proactive cognitive control processing style (Supplemental Table 3). We did not find evidence for significant age-related differences between the clinical groups and controls (Supplemental Table 4).
We further explored correlations across the ANT and AX-CPT performance. Overall, higher accuracy in the ANT was associated with higher accuracy and d’-context in the AX-CPT. In addition, there was a significant negative correlation between SD of RT during the ANT and accuracy and d’-context scores derived from the AX-CPT, indicating that children who exhibited less variable response times during the ANT tended to be more accurate and better at discriminating target and nontargets based on the cue (Figure 5). We also tested whether the correlations differed between groups. Here, we found that AnxD group exhibited a stronger association between accuracy derived from the ANT and accuracy derived from the AX-CPT compared to the ADHD group only (z = 3.28, p = .001), and a stronger association between the ANT-derived accuracy and AX-CPT-derived d’-context compared to the control (z = 3.56, p < .001) and ADHD only groups (z = 3.06, p = .002, Supplemental Table 5).
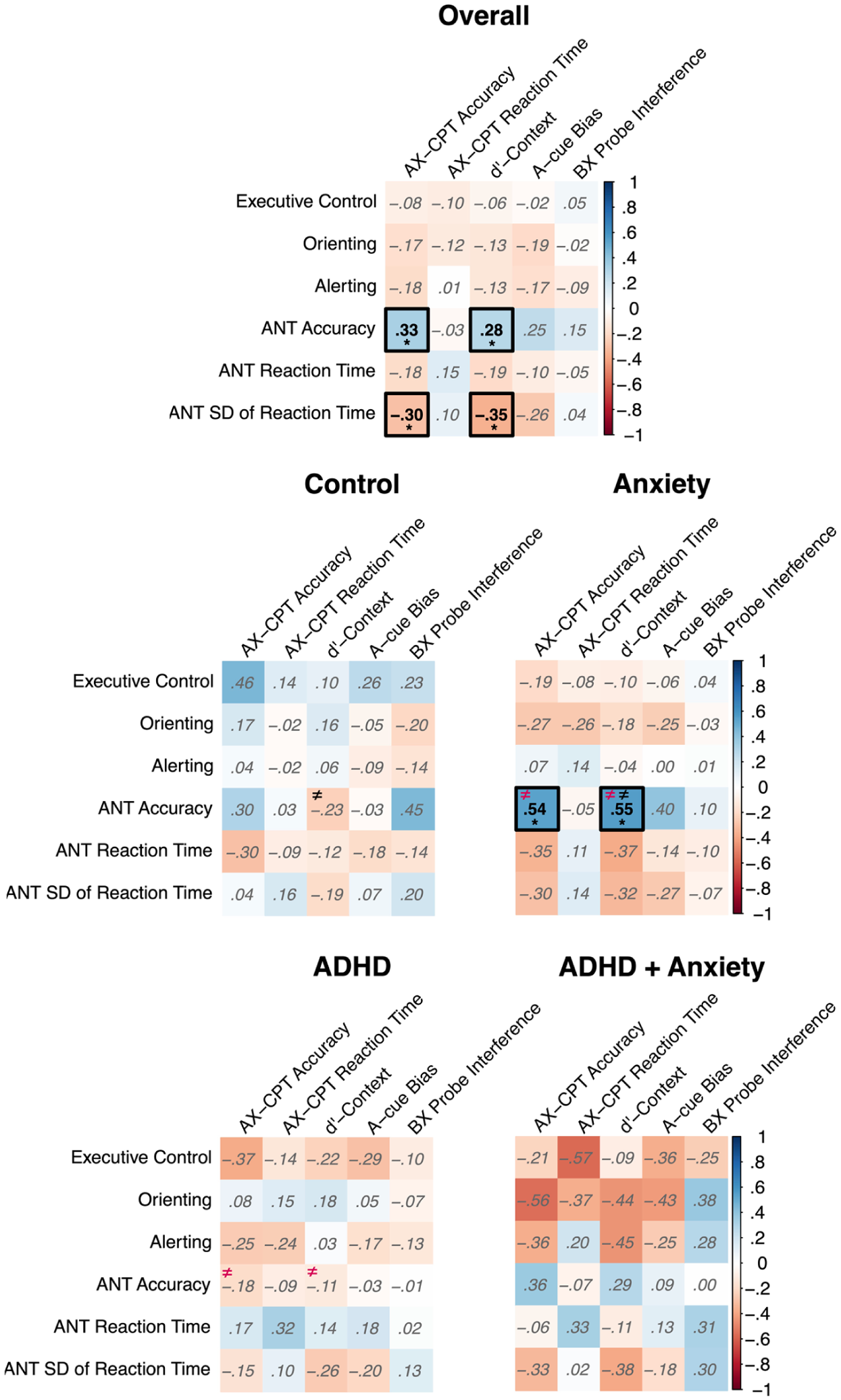
Pearson correlations across the Attention Network Test (ANT) and the AX-Continuous Performance Task (AX-CPT) derived measures for the overall sample and each of the clinical groups.
Discussion
The current study aimed to characterize attentional and cognitive control functions among children with ADHD and children with ADHD and comorbid AnxD. Specifically, we used the ANT and the AX-CPT to measure performance in attention and cognitive control, respectively. Overall, the children with ADHD and comorbid AnxD group showed evidence for worse performance in an attention-based task, however, we did not find support for function-specific deficits among the three subcomponents of attention (i.e., executive control, orienting, and alerting). In contrast, we did find evidence for worse proactive cognitive control among children with ADHD and comorbid AnxD compared to typically developing controls, supporting a function-specific deficit in cognitive control. Finally, we did not find support disorder-specific deficits in attentional and cognitive control among children with ADHD and comorbid AnxD beyond the ADHD-related deficits. Children with ADHD and comorbid AnxD showed an overall lower accuracy, slower RT, and more variable RT in the ANT compared to typically developing controls, extending previous findings of deficits in global indices among children with ADHD (Adólfsdóttir et al., 2008; Bieleninik et al., 2023; Booth et al., 2007; Suades-González et al., 2017). However, our findings diverge from the enhanced cognitive effort hypothesis, which would predict preserved or improved accuracy in the presence of comorbid anxiety. Instead, the results suggest that comorbid anxiety does not confer performance benefits in children with ADHD, but is associated with broader cognitive inefficiencies, including lower accuracy, greater response variability, and reduced proactive control. One plausible interpretation is that the co-occurrence of ADHD and anxiety in the present sample reflects greater overall neurodevelopmental burden rather than a distinct cognitive profile, consistent with evidence linking more severe symptom presentations to poorer cognitive performance in ADHD (Levy et al., 2018; Machida et al., 2019; van Lieshout et al., 2017). At the same time, it remains possible that the relationship between anxiety and cognitive performance in ADHD is not linear, such that higher levels of anxiety may overwhelm limited regulatory resources rather than enhance performance. However, without dimensional measures of anxiety severity, the present cross-sectional design does not allow these accounts to be disentangled. Thus, longitudinal data are needed to examine the interplay between cognitive deficits and comorbid anxiety in children with ADHD.
The lower accuracy and slower and more variable RT may indicate global deficits in cognitive tasks, altered speed-accuracy tradeoffs and/or processing speed in children with ADHD and comorbid AnxD. Indeed, previous studies have found evidence for deficits in the information processing speed, and not speed-accuracy tradeoffs (Karalunas et al., 2012), as well as slower internal clock and decision making speed in children with ADHD compared to controls (Shapiro & Huang-Pollock, 2019). This may also indicate a general processing speed deficit in children with ADHD and comorbid AnxD, Still, although slower RT may indicate worse processing speed, we urge caution when this is accompanied with higher RT variability, as this can simply be due to inattention to the task rather than reflecting slower processing speed per se (Kofler et al., 2013). Interestingly, variability in cognitive performance has been found to be associated with attention problems (Wiker et al., 2023) and to be a strong predictor of elevated ADHD symptoms in children (Arnett et al., 2022). Alternatively, slower RT may also be a consequence of general deficits in cognitive ability. Indeed, the difference in ANT RT was not significant after adjustment for IQ, indicating that the slower processing speed in ANT may, in part, be attributed to general cognitive deficits. Thus, if consistent across task, this may influence all ANT conditions equally, which could also explain the typical ratio scores for ANT subcomponents. Still, it should be noted that if RT and IQ measures are qualitatively distinct phenotypes in ADHD, the IQ-adjusted effects may also obscure the ADHD-related deficits in RT. Furthermore, we did find evidence for lower accuracy and more variable RT for the ADHD + AnxD group compared to controls in the ANT after adjusting for IQ. Thus, the results may indicate that altered accuracy and variable responses in attentional tasks are independent of global cognitive functioning deficits among children with ADHD and comorbid AnxD. Interestingly, we found a significant association between RT and RT variability in children with ADHD + AnxD and AnxD. The latter was also significantly different from the ADHD only group, possibly indicating similar cognitive strategies among the ADHD + AnxD and AnxD groups. Thus, to speculate, the lower accuracy and more variable RT in the absence of attention specific deficits and the correlation pattern in the ANT-derived test scores may point toward a differential but ineffective cognitive strategy in children with ADHD and comorbid AnxD compared to ADHD only. Deficits in cognitive control has been suggested to be a hallmark feature of ADHD, however, it has been unclear if the deficits are driven by proactive or reactive cognitive control (de Zeeuw & Durston, 2017). Previous studies have found evidence for reactive cognitive control deficits in ADHD using stop signal task and no-go task (Cai et al., 2023; Hulst et al., 2018; Pani et al., 2013), but the evidence for deficits in proactive cognitive control has been unclear (Cai et al., 2023; Fosco et al., 2019; Pani et al., 2013). In the current study, we found partial evidence for differences in proactive and reactive cognitive processing style, reflected by a significant main effect of group on BX interference. However, the follow-up analysis did not survive adjustment for multiple comparisons. Still, we do find lower scores in the A-cue bias and d’-context measures among children with ADHD and comorbid AnxD. Both the d’-context and A-cue bias measures are based on signal detection theory (Stanislaw & Todorov, 1999) that are used to measure the ability to discriminate between relevant and non-relevant information. The lower A-cue bias in children with ADHD indicates a stronger tendency to wait for the probe to appear instead of using the cue to prepare for the response, reflecting a reliance on reactive cognitive control. Moreover, the lower d’-context among children with ADHD and comorbid AnxD also suggest that this could be reflected by a reduced ability to maintain contextual information to guide behavior, potentially reflective of reduced recruitment of a proactive cognitive control mode in children with ADHD and comorbid AnxD. Thus, in relation to the DMC framework (Braver, 2012), we interpret the lower d’-context and A-cue bias as reflective of deficits in proactive cognitive control among children with ADHD, which may be worsened by comorbid AnxD,
Although ADHD and comorbid AnxD may worsen working memory, which is postulated to underlie attention control (A. S. Souza et al., 2018), we only found evidence for function-deficits in proactive cognitive control and not attention subcomponent. As suggested by others, anxiety may not impact attentional performance due to enhanced cognitive effort but at the expense of slower responses (Eysenck et al., 2007). Thus, comorbid anxiety in children with ADHD may exacerbate slow responses in unified manner across conditions, yielding typical performance in the attention subcomponent ratio scores. The lower accuracy and higher SD of RT could also reflect a tendency for post-error slowing to correct for failed trials, affecting the RT in subsequent trials regardless of the trial condition in the ANT. A deficit in proactive cognitive control, as indicated by AX-CPT performance, may indicate that an increase in cognitive effort may not be sufficient to improve overall accuracy. Thus, a lower overall accuracy could be accompanied by greater post-error slowing, yielding more variable RT in children with ADHD and comorbid AnxD. Indeed, previous results have shown that anxiety yield greater post-error slowing than controls (Rueppel et al., 2022) and altered error-related brain responses (Aarts & Pourtois, 2010; Meyer, 2017; Moser et al., 2013). To speculate, children with ADHD and comorbid AnxD may exhibit increased cognitive effort due to anxiety, but the benefits may be counteracted by the weaker preparatory bias (i.e., as indicated by lower A-cue bias) in ADHD. This is in line with a previous electrophysiological study that found altered preparatory processing in adults with ADHD during a cue task switching paradigm, indicating atypical information processing conveyed by cue information rather than general alertness (Sidlauskaite et al., 2020) There was also a positive correlation between accuracy and d’-context in the AX-CPT across all groups, indicating that the children who were better at maintaining and use context to guide responses made fewer mistakes. We also found that this association was stronger for the ADHD + AnxD group compared to controls, possibly reflecting the enhanced cognitive effort in children with ADHD with comorbid AnxD as predicted by attentional cognitive control theory. Indeed, higher accuracy in the ANT also predicted higher accuracy in the AX-CPT for children with AnxD that differed from ADHD only but not the other groups. Moreover, we did not find evidence for slower RT in the AnxD group but rather more variable RT. Thus, despite showing reduced overall accuracy, this may indicate that children with AnxD put more weight on responding correctly, possibly at the expense of being more variable in their RT, during cognitive tasks. To speculate, anxiety in children with ADHD may result in a greater emphasis on doing the task correctly due to increased cognitive effort, however, it does not improve the impaired task performance and RT in ADHD. Overall, the result indicates that comorbid AnxD in children with ADHD could exacerbate attentional and cognitive deficits as indicated by group differences across the ANT and AX-CPT. However, the correlational differences with accuracy measures suggest that ADHD and comorbid AnxD may lead to a different cognitive strategy compared to the control group, with a greater emphasis on accuracy across tasks. This is in line with previous results indicated altered brain-related activity during cognitive tasks in ADHD with anxiety, contributing to neurobiological heterogeneity in ADHD (van der Meer et al., 2018). As such, different neural mechanisms may be involved in ADHD with comorbid AnxD compared to ADHD only, which may be related to the differential response to psychopharmacological treatment in children with ADHD and with high levels of anxiety (Moshe et al., 2012). A meta-analysis also showed improvement in inhibitory control in children without a history of ADHD-medication compared to children that were taken off medication or with unknown medication status (Maric et al., 2018). Although it remains unclear whether the effect is due to symptom severity or medication effects, it may indicate that children with ADHD and comorbid AnxD require different treatments to enhance attention and cognitive functioning. Future research examining the interaction between psychopharmacological medication and anxiety symptoms on cognitive performance and cognitive strategies may yield important insight into targeted treatments in children with ADHD with comorbid AnxD.
The current study has strengths and limitations. By using a unique sample of children with (i) ADHD, (ii) AnxD, and (iii) ADHD and AnxD, we were able to examine the attentional and cognitive control profile of subgroups of children with ADHD using adequate comparison groups. It should be noted, however, that despite lack of group differences between the clinical groups, we cannot exclude the possibility of small differences, which would require larger sample size to reliably detect. However, to function as useful diagnostic markers, group differences should be of large effect sizes to be of clinical relevance. Still, our results show moderate to large differences in cognitive measures and reveal distinct associations between cognitive measures in children with ADHD and ADHD + AnxD. This underscores the importance of detailed cognitive assessments to delineate the cognitive profiles and strategies that may underlie everyday challenges, which could be used to inform individualized educational and clinical interventions. Moreover, due to limited sample size, we did not investigate sex differences in the current study. Although we did adjust for sex in our analyses, the different proportions of males and females within the groups may have influenced the results. Males are more likely to meet the diagnostic criteria for ADHD (Willcutt, 2012) and present hyperactivity and impulsivity symptoms compared to females (Li et al., 2019; Willcutt, 2012), whereas females with ADHD may exhibit elevated anxiety symptoms compared to males (Skogli et al., 2013). In addition, sex differences in cognitive profiles may also impact the results observed in the ADHD versus ADHD + AnxD groups. For instance, previous results have also demonstrated sex differences in cognitive control task among children with ADHD, where males exhibit lower accuracy and females exhibit slower responses compared to controls (Ojuri et al., 2024). As such, a higher proportion of males in the ADHD group may exaggerate accuracy and attenuate reaction time differences relative to the ADHD + AnxD group. Thus, more research is needed to examine sex differences in attention and cognitive control subcomponents. In addition, novel insight can be made by examining attentional and cognitive control profiles in larger sample sizes, as this allows for data-driven approaches to identify subgroups in children with ADHD and possibly disentangle clinical heterogeneity beyond AnxD diagnoses. It is also important to note that the current study utilizes cross-sectional data; hence, we urge caution when interpreting the results. Indeed, there are several plausible mechanisms that may have contributed to the observed differences in cognitive functioning, both between and within the clinical groups, which would require longitudinal data to examine. For instance, it is possible that cognitive control deficits increase the risk of developing ADHD or they can be an indirect consequence of other behavioral or medical issues associated with ADHD, such as sleep problems (Hansen et al., 2014; Hvolby, 2015). It is also worth noting that the AnxD group showed lower accuracy in the ANT and AX-CPT and higher RT variability in the ANT compared to controls, possibly reflecting neurodevelopmental issues among children with AnxD. Thus, to speculate, ADHD and comorbid AnxD may be a representation of a more severe neurodevelopmental disorder. Future longitudinal studies are needed to increase our understanding of the relationship between cognitive control deficits and the development of ADHD with and without AnxD, including deep phenotyping of neurodevelopmental problems.
Conclusion
Children with ADHD exhibit worse performance in global indices (i.e., accuracy and RT) in an attention-based task compared to typically developing controls, whereas we do not find evidence for deficits in subcomponents of attention (i.e., executive control, orienting, and alerting). In contrast, the children with ADHD and comorbid AnxD do show evidence for deficits in proactive cognitive control, indicating a function-specific deficit in cognitive control. The results do not indicate different attentional and cognitive control profiles between children with ADHD and children with ADHD and comorbid AnxD. However, the results may indicate that comorbid AnxD exacerbates ADHD-related deficits in cognitive control functions. More research is needed to establish disorder-specific attentional and cognitive control profiles in children diagnosed with ADHD, including the use of data-driven approaches in larger clinically heterogenous samples.
Supplemental Material
sj-docx-1-jad-10.1177_10870547261430076 – Supplemental material for Characterizing Attention and Cognitive Control Profiles of Children With ADHD and Anxiety
Supplemental material, sj-docx-1-jad-10.1177_10870547261430076 for Characterizing Attention and Cognitive Control Profiles of Children With ADHD and Anxiety by Rune Boen, Beate Oerbeck, Berit Hjelde Hansen, Benedicte Skirbekk, Hanne Kristensen, Christian K. Tamnes and Thomas Espeseth in Journal of Attention Disorders
Footnotes
Ethical Considerations
The study was approved by the Regional Committee for Medical Research Ethics and the Norwegian Data Inspectorate.
Consent to Participate
The parents, and the children aged 12 years and older, gave their written informed consent to participate in the study. Verbal informed consent was given from children younger than 12 years.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Research Council of Norway (#288083, #323951), Southern and Eastern Norway Regional Health Authority (#2021070, #2023012, #500189), and the Norwegian Centre of Expertise for Neurodevelopmental Disorders and Hypersomnias (to CKT).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
