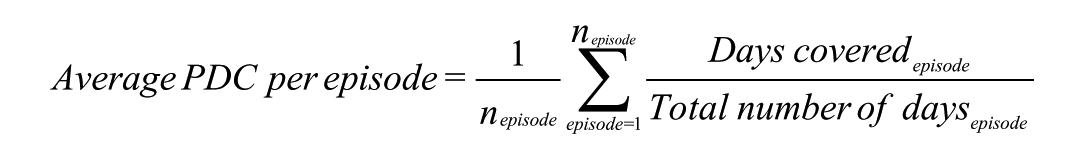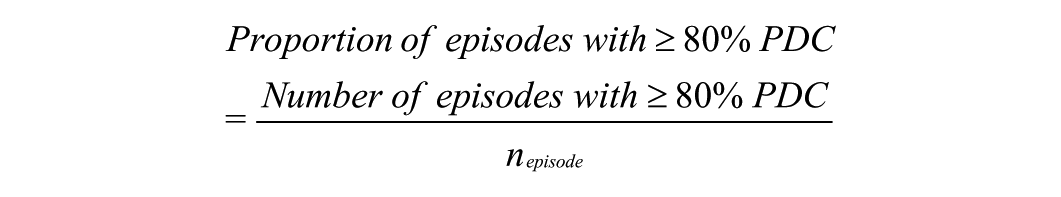Abstract
Objective:
To estimate the adherence of children and adults to oral stimulant ADHD treatments in Sweden during the period 2015 to 2020.
Methods:
This retrospective, nationwide, register-based study evaluated all patients who were dispensed oral formulation stimulants (Anatomical Therapeutic Chemical [ATC] codes: N06BA01, N06BA02, N06BA04, N06BA12) identified in the Swedish Prescribed Drug Register. Treatment episodes were constructed based on drug dispensation data, and treatment adherence was calculated for each treatment episode using a Proportion of Days Covered approach with a modified one-pill-a-day method.
Results:
Between 2015 and 2020, 158,413 treatment episodes were recorded in 128,366 patients, corresponding to a median 1.23 treatment episodes per patient. The majority of treatment episodes involved methylphenidate (64.0%), followed by lisdexamfetamine (32.2%), dexamfetamine (3.0%), and amphetamine (0.8%). Overall treatment adherence was high (78.2%), and was similar for patients receiving methylphenidate, lisdexamfetamine, and dexamfetamine. The proportion of patients with ≥80% medication adherence was high (69.3%–71.8%) for adults aged ≥25 years, but was low for adolescents aged 12 to 17 years (49.7%–52.4%).
Conclusions:
The overall rates of treatment adherence are high for children and adults receiving ADHD medication in Sweden. We observed lower adherence among adolescents, which warrants further study.
Introduction
Attention deficit hyperactivity disorder (ADHD) is a complex neurodevelopmental condition that is characterized by developmentally inappropriate hyperactive, impulsive, or inattentive behavior (Faraone et al., 2021). Although it is most commonly diagnosed in children, ADHD can occur in individuals of any age group (Ayano et al., 2023), with an estimated prevalence of 5% to 6% in children and adolescents and 2% to 3% in adults (Faraone et al., 2021). In Sweden, the estimated prevalence of ADHD increased from 2.7% to 3.2% in children/adolescents and from 1.2% to 1.4% in adults between 2018 and 2021 (Giacobini et al., 2023).
There has been a well-documented increase in ADHD diagnoses over the past 20 years, which has been accompanied by an increase in prescriptions for ADHD medications (Bachmann et al., 2017; Chan et al., 2023; Giacobini et al., 2023; McKechnie et al., 2023; Sørensen et al., 2023). This upward trend could be explained by an increasing frequency of the condition, greater recognition of its symptoms, the availability of broader diagnostic criteria, or overdiagnosis or misdiagnosis of suspected ADHD (McKechnie et al., 2023). There also appears to be a higher frequency of ADHD diagnoses in males versus females (Faraone et al., 2021). Females are typically diagnosed later than males, which could be because their symptoms are overlooked or misinterpreted as anxiety, borderline personality disorder, or bipolar disorder (Skoglund et al., 2024). Although males and females are prescribed ADHD medication with a similar frequency in the years following their diagnosis, females are significantly more likely than males to take concomitant non-ADHD treatments (anticonvulsants, neuroleptics, sedatives, hypnotics, and psychoanaleptics) after they have been diagnosed (Skoglund et al., 2024).
Treatment of ADHD follows a multimodal approach, involving psychosocial interventions, as well as pharmacological treatments, which should be supported by regular follow-ups (Elliott et al., 2020). These interventions have been shown to have a protective effect against some of the more serious complications associated with ADHD, including depression (Chang et al., 2016), substance abuse (Chang et al., 2014), criminality (Lichtenstein et al., 2012), traffic accidents (Chang et al., 2017), suicide (Chen et al., 2014), and mortality (Li et al., 2024).
Although pharmacotherapy is not indicated for children under the age of 6 years (Ponnou & Thomé, 2022), treatment may commence sooner in some patients, such as those with greater functional impairment or more serious levels of hyperactivity and impulsivity (Young et al., 2021). The median age of first treatment prescription has been shown to be between 16 and 21 years (Brikell et al., 2024). Younger patients are the most likely to adhere to treatment once it has been initiated; however, adherence wanes over time, with less than a quarter of all patients remaining on treatment after 5 years (Brikell et al., 2024). Medication use has been shown to decline sharply after the age of 18 years, which may suggest unmet needs for the successful transition of patients with ADHD from pediatric to adult healthcare services (Giacobini et al., 2025) before increasing again a few years later. Factors that have been shown to negatively impact treatment adherence can be patient related, such as the existence of comorbid conditions and a lower IQ; family related, such as poor family support infrastructure, a family history of ADHD, and lower socioeconomic status; medication related, such as the need for multiple dosing and the occurrence of side effects; and other reasons, such as negative opinions or perceived stigma associated with medication and untrustworthy physician–patient relationships (Ferrin et al., 2025). Lower adherence has also been observed in patients receiving immediate-release medications versus extended-release formulations (Jeun et al., 2024). The most common reasons cited for discontinuation of ADHD treatments are: inadequate management of ADHD symptoms; the patients’ (or their families’) dislike of the treatments; and the occurrence of ADHD- or treatment-related complications (Schein et al., 2022).
Several previous studies have investigated the prevalence and incidence (Polyzoi et al., 2018) of ADHD and the treatment patterns (Giacobini et al., 2023) and transition of care (Giacobini et al., 2025) for patients with the condition in Sweden, utilizing national health registry data; however, information on medication adherence in this population using national prescription data from a large patient sample is lacking. Non-adherence is associated with a range of adverse outcomes, including poor performance at school (Jangmo et al., 2019) and familial problems (Abdallah et al., 2024). Therefore, by gaining a greater understanding of patterns of medication adherence among patients with ADHD, it might be possible to develop tools and strategies to support patients in managing their condition (Kamimura-Nishimura et al., 2019). In this study, we sought to estimate adherence to oral stimulant medications for ADHD in children and adults between 2015 and 2020.
Methods
Data Sources
This was a retrospective, nationwide, register-based study of adherence to ADHD medications that was carried out between 2015 and 2021. All patients who were dispensed oral formulation stimulants (Anatomical Therapeutic Chemical [ATC] codes: N06BA01, N06BA02, N06BA04, N06BA12) were identified in the Swedish Prescribed Drug Register (PDR) between July 1, 2005, and October 31, 2021. Patients were followed until death, treatment discontinuation (defined as a gap in medication dispensations for at least 180 days), or emigration, but for a minimum of 90 (dose-titration period) + 180 (minimum treatment duration) days. This study and waiver of patient consent were approved by the Swedish Ethical Review Agency and the Stockholm Regional Ethical Review Committee (references 2012/1315-31, 2013/2177-32, 2017/1395-32, 2018/1678-32, 2018/1835-32, 2020-06376, 2022-06622-02, and 2023-0700402).
Study Design
All patients who had been prescribed and dispensed oral stimulants between July 1, 2005, and October 31, 2021, were included in the study. Dispensations were converted into treatment episodes as per the following criteria: each treatment episode was defined by one ATC code; the date of treatment initiation was defined as the date of a dispensation without prior dispensations in the previous 180 days with the same ATC code; the first 90 days since treatment initiation was determined to be a dose-titration period; and treatment discontinuation was defined as the last dispensation without any dispensations in the next 180 days with the same ATC code. Patients were only permitted to undergo one treatment episode at a time; if patients were treated with multiple stimulants (with multiple ATC codes), the earliest start date of the next treatment episode would be defined as the day after the discontinuation of the previous treatment episode. An individual could contribute with more than one treatment episode, provided that each individual episode had a duration of at least 180 days during the study period. For each treatment episode, medication adherence was assessed from the first dispensation after the end of the dose-titration period until treatment discontinuation; this period also defined the episode duration, which was to be at least 180 days.
To calculate the number of days covered by a dispensation, a modified one-pill-per-day approach was used, based on prescribing conventions in Sweden with input from local adult and pediatric psychiatrists (Supplemental Appendix). The overall approach was based on the assumption that patients are generally prescribed and take one pill per day of their main stimulant treatment (even if they regularly take more pills pro re nata on certain days). Long-/medium-acting formulations were deemed more likely to be the main stimulant treatment, whereas short-acting formulations were deemed more likely to be pro re nata. As such, we estimated the number of days covered per dispensation as the number of pills dispensed for a combination of dose and long-/medium-acting versus short-acting formulations, with priority given to long-/medium-acting formulations. If more than one dose was dispensed within a formulation type, we used the formulation that had the highest number of pills. The estimated days covered was capped at 90 (based on the upper limit set by the Swedish Dental and Pharmaceutical Benefits Agency: https://www.tlv.se/in-english.html), and extended to 120 days in sensitivity analyses. To validate this methodology, we estimated the average daily dose by dividing the total dosage dispensed by the calculated days covered per dispensation and treatment episode, respectively.
Statistical Analysis
Medication adherence was calculated using a Proportion of Days Covered (PDC) approach without stockpiling, that is, on the assumption that patients consumed the medications that were dispensed, accounting only for dispensations with the same ATC code as that of the treatment episode. The PDC was presented numerically as the mean per episode and separately per calendar year, and in subgroups of sex, age, ATC code of the treatment episode, and concurrent dispensation of guanfacine (ATC: C02AC02) or atomoxetine (ATC: N06BA09; which could indicate more complex symptomology) during the treatment episode. As some patients, particularly those in school, may suspend treatment during the summer (“summer drug holiday”), medication adherence was also separately presented excluding days in June, July, and August, in both the numerator and denominator for the calculation of the PDC. The PDC was calculated as follows:
Note. nepisode = number of episodes; PDC = Proportion of Days Covered.
The proportion of patients with a PDC of 80%, which is a commonly used threshold for defining good adherence (Dalli et al., 2022), was determined. This was calculated as follows:
When presented per calendar year, the PDC was calculated as the weighted mean, weighted by the number of days contributed by the individual patient during the calendar year. This was to account for patients who only contributed to the study for very short periods in a year and would therefore have a smaller impact on the mean. PDC per calendar year was calculated as follows:
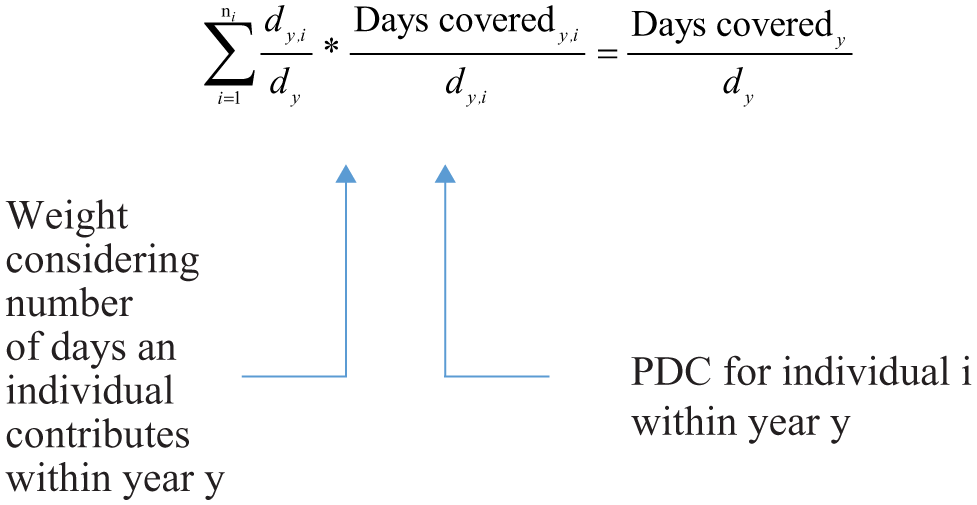
Note. d y ,i = number of days in year y for individual i; d y = sum of the number of days in year y for all individuals; ni = number of individuals.
The PDC per calendar year was also calculated for patients with 80% or above adherence, as follows:
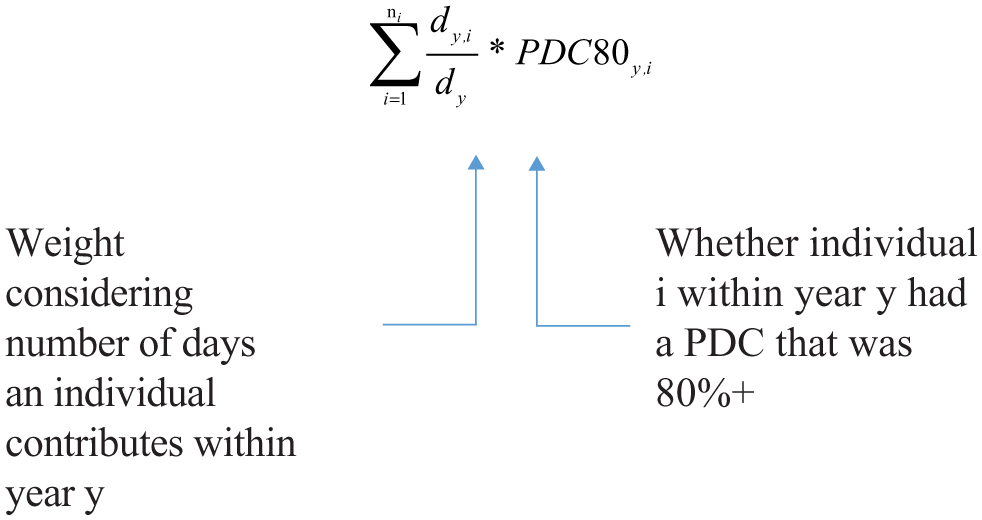
Note. d y ,i = number of days in year y for individual i; d y = sum of the number of days in year y for all individuals; ni = number of individuals.
Statistical analyses were conducted using SAS software, version 9.4 (SAS Institute Inc., Cary, NC, USA). Descriptive statistics were used to summarize data on the type of care and medication use, which was stratified by age.
Results
Study Population
Of more than 8 million stimulant prescriptions recorded in the Swedish PDR from July 1, 2005, to October 31, 2021, the study identified 158,413 treatment episodes for 128,366 patients between 2015 and 2020 after exclusions (Figure 1). This corresponded to a median of 1.23 treatment episodes per patient. The majority of patients were male (59.4%) and the median age at first study entry was 20 years (interquartile range [IQR], 12–34; Table 1). The median length of each treatment episode was 604 (IQR, 339–1,122) days. Treatment episodes most frequently involved methylphenidate (64.0%), followed by lisdexamfetamine (32.2%).
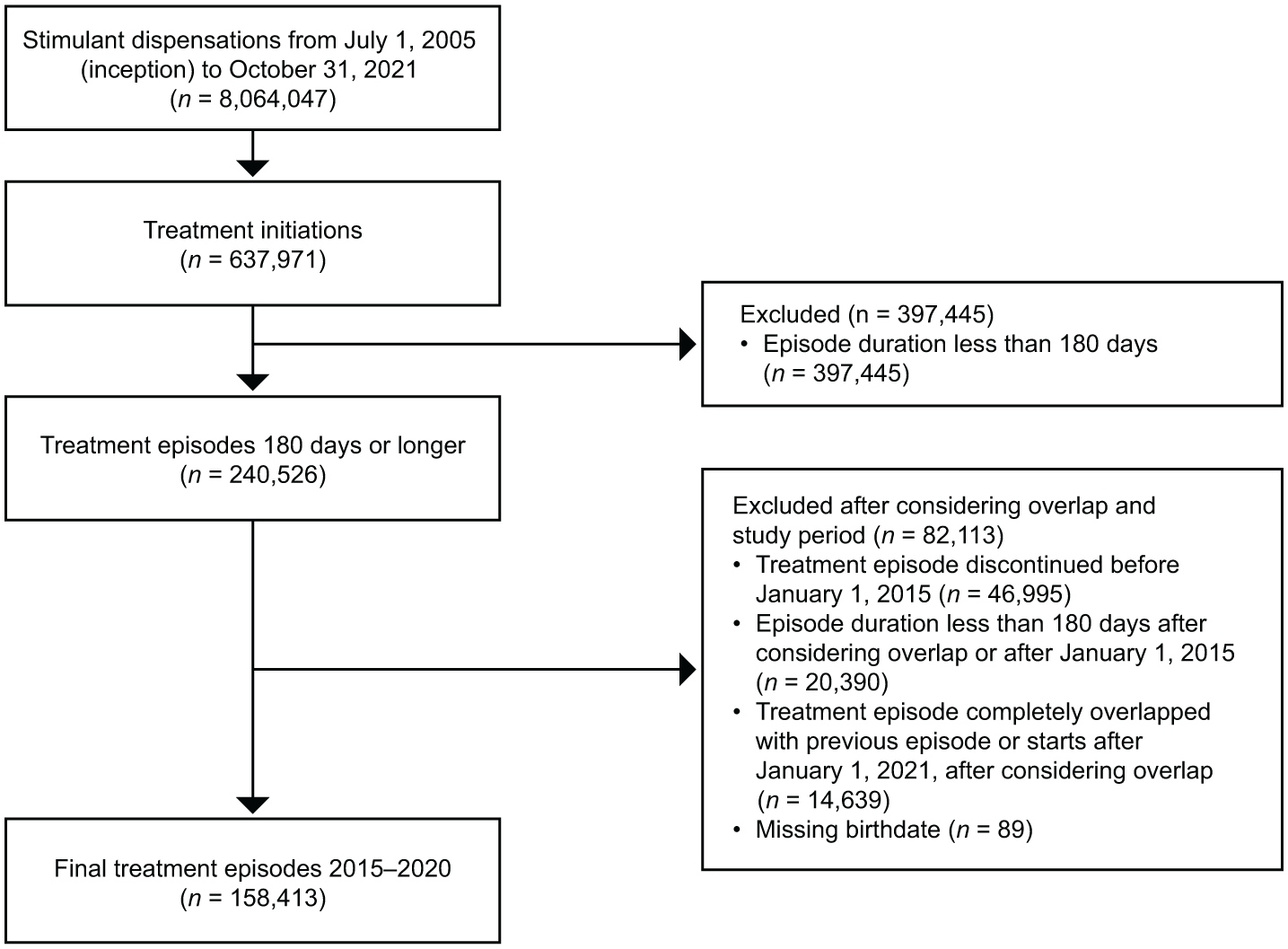
Determination of treatment episodes from all stimulant dispensations recorded in the PDR from July 1, 2005, to October 31, 2021.
Patient Characteristics.
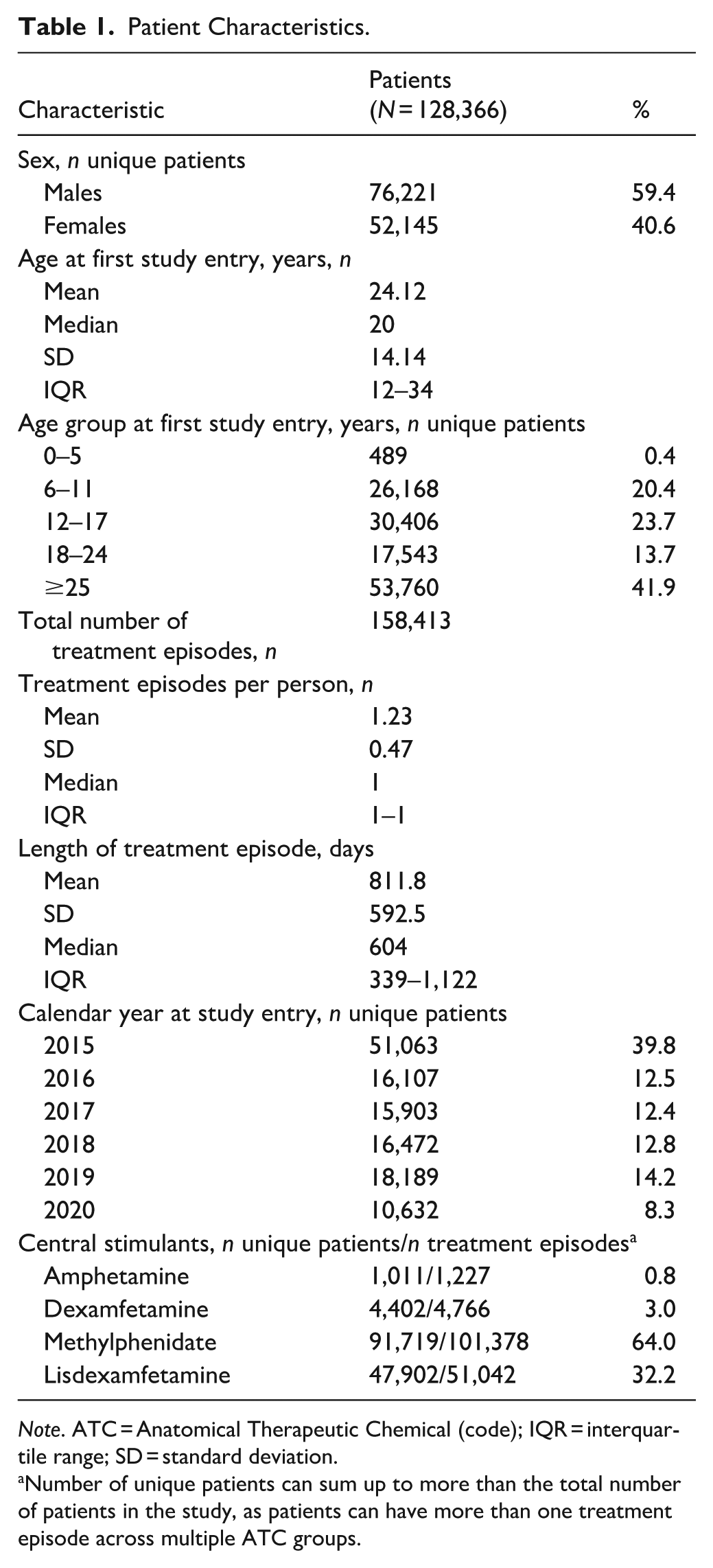
Note. ATC = Anatomical Therapeutic Chemical (code); IQR = interquartile range; SD = standard deviation.
Number of unique patients can sum up to more than the total number of patients in the study, as patients can have more than one treatment episode across multiple ATC groups.
Overall Medication Adherence
Mean medication adherence across the study period was 78.2%, with 55.9% of patients having ≥80% adherence to their medication (Table 2). Average medication adherence was high for methylphenidate (78.2%), lisdexamfetamine (79.5%), and dexamfetamine (82.7%), but was low for amphetamine (7.2%).
ADHD Medication Adherence.
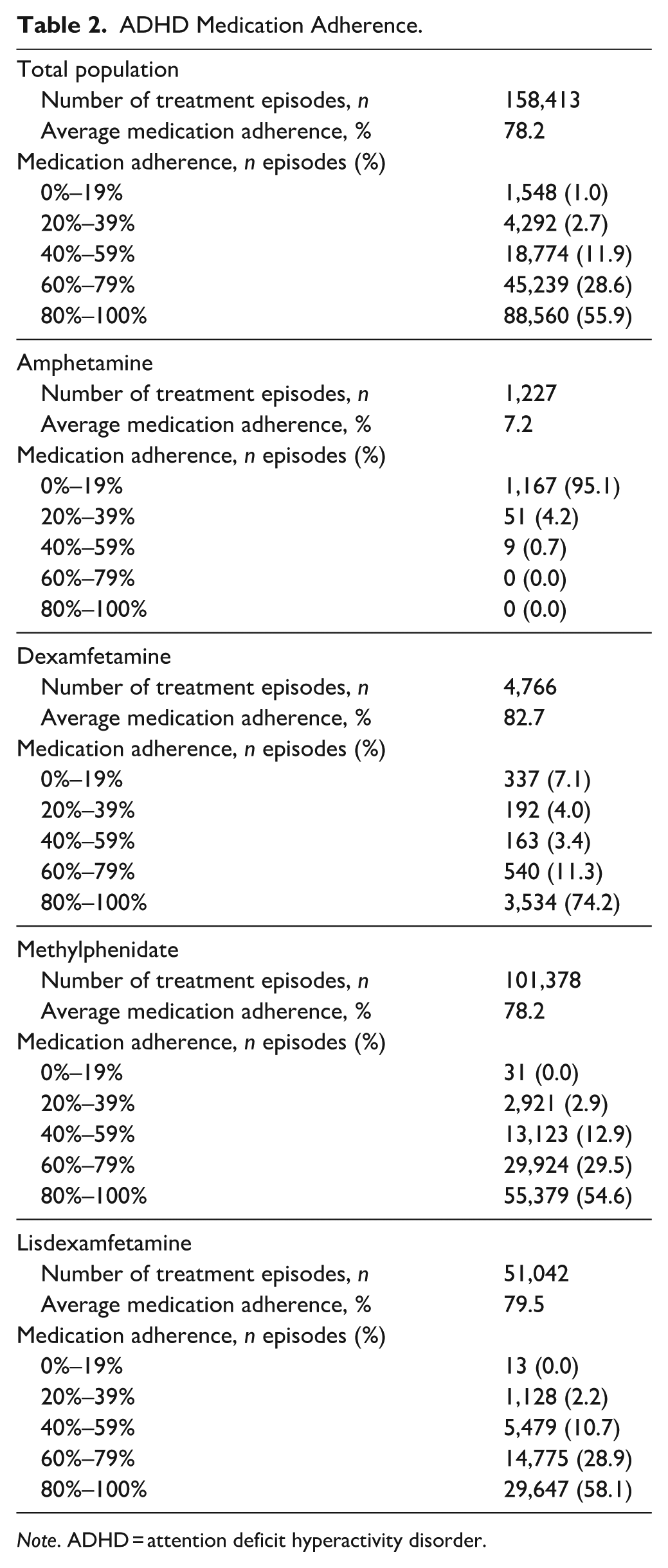
Note. ADHD = attention deficit hyperactivity disorder.
There were no differences over time in medication adherence, which was consistently high, including for the sensitivity analysis that excluded summer months and increased dispensations to up to 120 days. Moreover, there was no difference between males and females in medication adherence (Supplemental Appendix). When analyzed by patient age group, both mean medication adherence and adherence of ≥80% was highest for patients aged 0 to 5 years and lowest for those aged 12 to 17 years over the study period (Figure 2a and b). The proportion of patients with ≥80% medication adherence ranged from 77.8% to 88.3% for patients aged 0 to 5 years, 60.9% to 63.5% for patients aged 6 to 11 years, 49.7% to 52.4% for patients aged 12 to 17 years, 58.0% to 58.9% for patients aged 18 to 24 years, and 69.3% to 71.8% for those aged ≥ 25 years over the study period (Supplemental Appendix). Adherence to methylphenidate, dexamfetamine, and lisdexamfetamine were similar, regardless of whether patients were using concurrent guanfacine or atomoxetine (Figure 3).
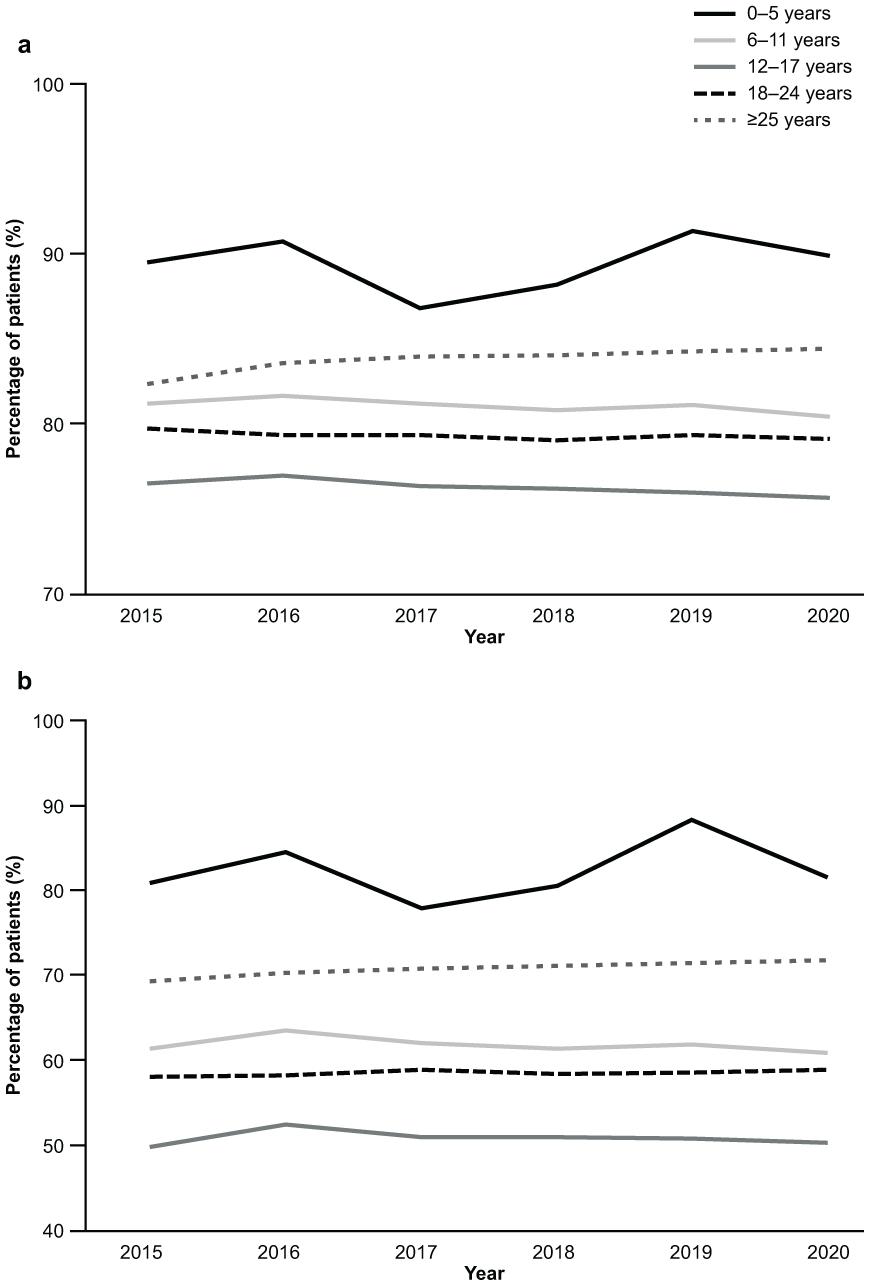
Adherence to ADHD medication by patient age group (2015–2020): (a) mean ADHD medication adherence by patient age group over the study period, and (b) proportion of patients with ≥80% ADHD medication adherence by patient age group over the study period .
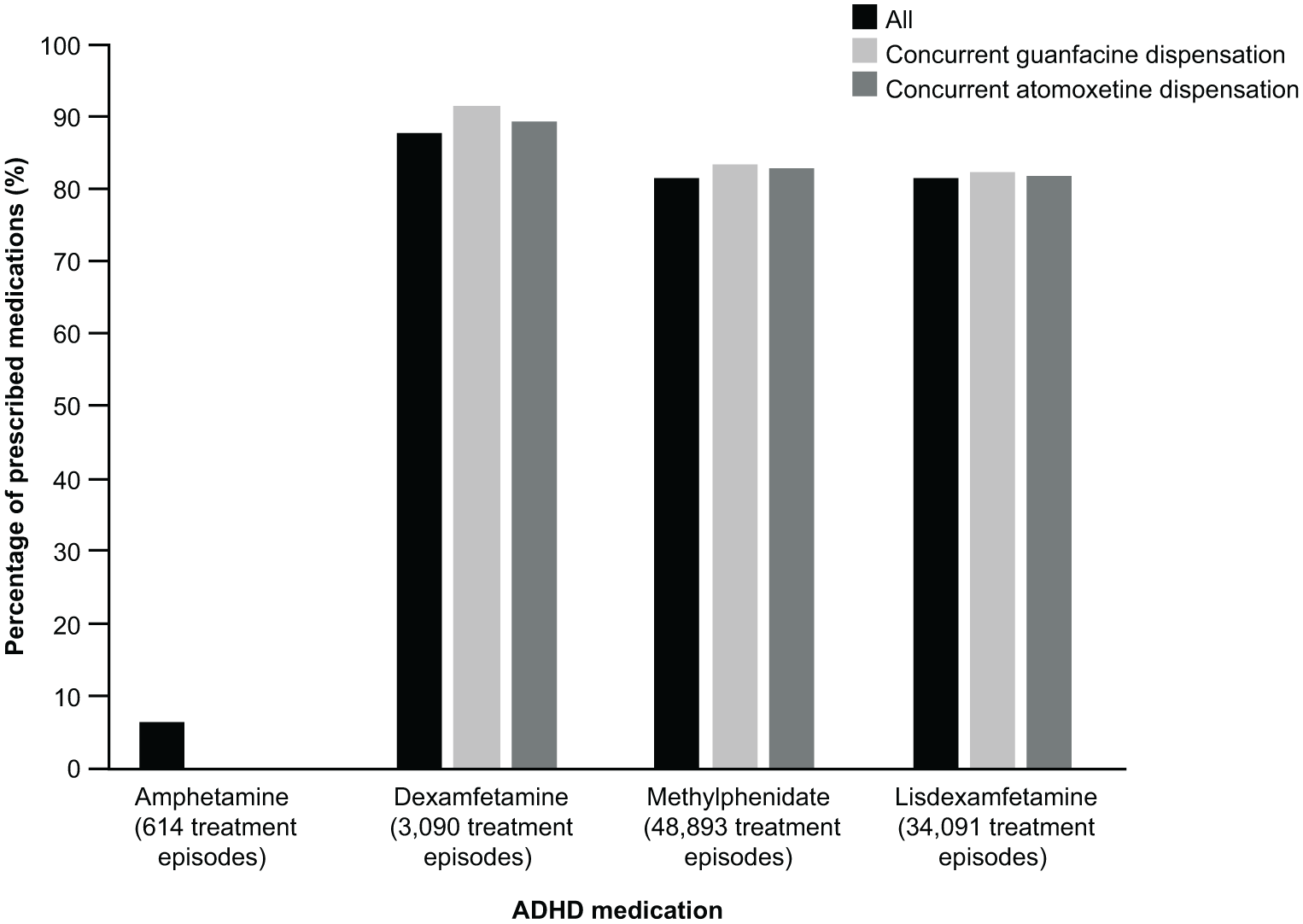
Mean adherence to ADHD medications by stimulant (ATC code), 2020.
Real-World Daily Doses of ADHD Medications
The median estimated daily doses, based on a modified one-pill-per-day algorithm (assuming 90 days’ prescription coverage per dispensation), were 54.7 mg per day for lisdexamfetamine, 40.8 mg for methylphenidate, 20.1 mg for dexamfetamine, and 4.7 mg for amphetamine (Table 3).
Estimated Daily Doses of ADHD Treatments, Based on a Modified One-Pill-Per-Day Algorithm.
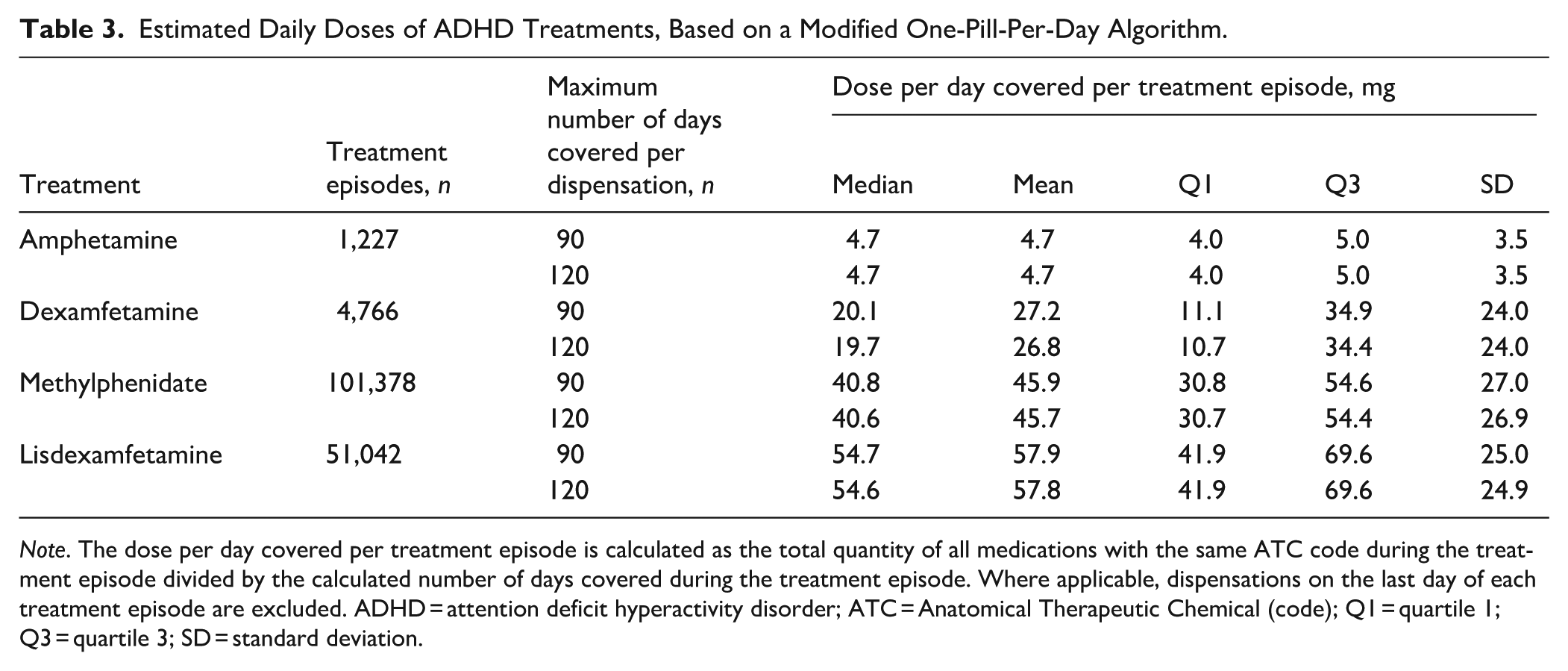
Note. The dose per day covered per treatment episode is calculated as the total quantity of all medications with the same ATC code during the treatment episode divided by the calculated number of days covered during the treatment episode. Where applicable, dispensations on the last day of each treatment episode are excluded. ADHD = attention deficit hyperactivity disorder; ATC = Anatomical Therapeutic Chemical (code); Q1 = quartile 1; Q3 = quartile 3; SD = standard deviation.
Discussion
In this real-world study of medication use, we collected nationwide, individual-level dispensation data from the Swedish PDR to provide an uninterrupted, longitudinal, 6-year follow-up on adherence to ADHD treatments from more than 100,000 patients. Our study demonstrated a high overall level of treatment adherence, which was similar for patients receiving methylphenidate, lisdexamfetamine, and dexamfetamine.
Methylphenidate and amphetamines are considered to be first-line treatments for children aged ≥ 6 years and adolescents with ADHD (Van Vyve et al., 2024). Guidelines recommend that, if these patients do not respond to stimulants, they should switch to an alternative medication such as atomoxetine (a noradrenaline reuptake inhibitor) or guanfacine (an alpha-2 adrenergic agonist), with the latter being indicated only for children and adolescents for whom stimulants are not suitable or are ineffective (Kooij et al., 2019; Van Vyve et al., 2024). Real-world data from Sweden have demonstrated that methylphenidate is used as a first-line treatment in >80% of all patients with ADHD, for a median duration of around 7 months before patients switch to alternative treatments. The most frequent second-line treatment was found to be lisdexamfetamine, a long-acting amphetamine (Giacobini et al., 2023). The duration of treatment with lisdexamfetamine is generally longer than that with methylphenidate and atomoxetine (Giacobini et al., 2023; Siffel et al., 2020).
We developed a robust algorithm for estimating days covered for stimulant dispensations, using a modified one-pill-per-day approach; this was based on clinical experience, local routines, and regulations for local prescription practices. In this study, discontinuation was defined as a gap in medication of ≥180 days, which is twice the duration of a normal prescription in Sweden (Brikell et al., 2024), in order to distinguish clinically meaningful treatment gaps from shorter breaks in treatment coverage. Overall, we found that more than half of the patients were adherent to ADHD medications at least 80% of the time, as determined by the PDC approach. These findings are higher than some earlier studies from different countries that demonstrated medication adherence rates of 40% to 70% (Biederman et al., 2020; Gajria et al., 2014; Wagoner et al., 2020; Zheng et al., 2020). However, direct comparisons between studies from other countries should be interpreted with caution due to differences in study methodology and the definitions of adherence. Furthermore, differences in healthcare systems between countries, including medication reimbursement policies, and continuity of care between child and adult services, may impact access to ADHD treatments and potentially influence adherence rates. The reason for the apparently high rate of adherence shown by our study could be due to the stringent inclusion criteria used, which selected only those patients who had received more than 180 days of treatment. As such, those patients who were more infrequent or shorter-term users of ADHD medications were not included, thereby distinguishing treatment persistence from treatment adherence. Our results were robust to the sensitivity analyses that accounted for patients having a “summer drug holiday” in June, July, and August, and also when extending the assumed maximum drug coverage duration to 120 days.
Medication adherence appeared to be stable over time and consistent between males and females; however, some differences were seen when stratifying by age group and treatment type. Patients aged 12 to 17 years were found to be the least adherent to ADHD treatments in our study. This is similar to the findings of a global study that observed the lowest levels of ADHD treatment persistence in adolescents and young adults (5-year persistence of 9% and 10%, respectively), when compared with younger children and older adults (5-year persistence of 24% and 15%, respectively; Brikell et al., 2024). In another Swedish study, a similar decline in ADHD medication use was observed in young adults, coinciding with patients transitioning from pediatric to adult psychiatric care, which appears to be a critical time point for these patients (Giacobini et al., 2025). Although rates of adherence to ADHD medication were high in our study overall, differences were observed depending on the type of stimulant prescribed. Adherence was highest for dexamfetamine, followed by lisdexamfetamine and methylphenidate. Comparatively, there was a much lower treatment adherence to amphetamine, which was also the least frequently dispensed medication among these patients. In Sweden, amphetamine treatment is not commercially available and requires an import license from the Swedish Medicinal Products Agency in order to be prescribed by a psychiatrist. This likely explains why the number of such prescriptions was low compared with the other stimulant prescriptions recorded in our study (Swedish eHealth Agency, 2024; Swedish Medical Products Agency, 2024). Adherence to methylphenidate, dexamfetamine, and lisdexamfetamine was high, regardless of whether patients were using guanfacine or atomoxetine concurrently.
A key strength of this study was that it used longitudinal data for the entire population of Sweden, limiting the possibility of selection bias. Furthermore, our methodology allowed us to isolate adherence from persistence by defining the end of the observation window as the last dispensation date, as recommended by DeClercq & Choi (2020). Our study relied on data from the Swedish PDR, which we used to infer a diagnosis of ADHD in the cohort studied on account of the treatments that individuals received. We did not exclude individuals if they did not have an ADHD diagnosis, therefore, some patients who had been prescribed stimulants unrelated to ADHD may have been included in the study. However, since the prescription of central stimulants in Sweden is limited to physicians certified in psychiatric or neurological specializations (Swedish Medical Products Agency, 2024), the proportion of patients without ADHD in this study is likely to be low and to have had a minimal impact on the results. Furthermore, we believe that use of the PDR provided a more accurate reflection of the true population in Sweden with ADHD during 2015 to 2020 than the NPR might have. The NPR has been reported to have incomplete coverage of patient data, which could be due to data from the private sector not being included (Everhov et al., 2025). Another study has shown that psychiatric healthcare activities have only an approximate 75% coverage in the NPR (Polyzoi et al., 2018). In contrast, the PDR collects prescription data from all pharmaceutical retailers (i.e., regardless of whether a treatment was prescribed in the public or private sector; Socialstyrelsen, 2024). Therefore, we believe that this study provides national and comprehensive coverage of data for patients with ADHD in Sweden from 2015 to 2020.
Due to the real-world nature of this study, a few study limitations should be noted. Firstly, the timing of when patients were first prescribed ADHD treatment relative to their assumed diagnosis is unknown. This may be important, as adherence may have differed between patients who had undergone a “watchful waiting” period upon receiving a diagnosis and patients who started their treatment immediately. This could be an important research topic to explore in future studies. A further limitation is that it was not possible to differentiate between medically motivated treatment gaps and non-adherence. Patients may have interrupted their treatment for different reasons, such as experiencing a change in their environment or circumstances that allowed them to cope better with their symptoms, adverse effects from medication, or contraindications with other treatments. Patients may have made an active decision to deviate from their treatment regimens, or might have stopped treatment because they forgot to renew their prescriptions or to attend medical appointments. Our study did not assess adherence to stimulants in patients taking concomitant psychotropic treatments, which may be used to control ADHD symptoms or its associated comorbidities (Girand et al., 2020). Furthermore, our study was not designed to estimate adherence to non-pharmacological treatments, such as cognitive behavioral therapy.
Although the Swedish PDR provides details of the types of medicine prescribed, the date of dispensation, and the amount of drug dispensed, it does not have any structured information on the medication dosage or duration of treatment (Zhang et al., 2021). We were not able to capture data for treatments that were provided during in-hospital care. Also, in our study, it was not possible to ascertain whether patients actually took the medications that they were given, nor can we account for patients misplacing pills. Certain special-case treatment regimens may not have been accurately classified in our one-pill-per-day treatment algorithm, such as those that involve patients taking multiple pills of the same strength and formulation every day. This would mainly have affected treatment regimens with dexamfetamine; however, the margin of error in our study was capped by a limit of 90 days’ covered per dispensation. Excluding patients with treatment episodes of <180 days’ duration may have removed a large proportion of patients with low medication adherence; however, those cases would arguably represent low persistence rather than low adherence.
Conclusions
This nationwide longitudinal study in Sweden that was conducted between 2015 and 2020 has demonstrated that patient adherence to ADHD stimulant treatments was generally high, with the exception of amphetamine, which was rarely prescribed. Although some differences were observed across patient age groups, there were no notable differences in treatment adherence over the time period studied nor by patient sex. This study provides important data on patient adherence to ADHD medications in Sweden, including comparative findings for the most commonly used stimulant drugs. Finally, the trends that were observed toward lower adherence among adolescents warrants further study.
Supplemental Material
sj-docx-1-jad-10.1177_10870547251406760 – Supplemental material for Medication Adherence in Children and Adults Receiving Treatment for Attention Deficit Hyperactivity Disorder (ADHD) in Sweden: A Nationwide Study
Supplemental material, sj-docx-1-jad-10.1177_10870547251406760 for Medication Adherence in Children and Adults Receiving Treatment for Attention Deficit Hyperactivity Disorder (ADHD) in Sweden: A Nationwide Study by MaiBritt Giacobini, Jingcheng Zhao, Jonatan Freilich, Carolina Brünner, Niklas Wallin Bernhardsson and Ewa Ahnemark in Journal of Attention Disorders
Footnotes
Acknowledgements
Medical writing support for this manuscript was provided by Kate Bradford, PhD, of Parexel, under the direction of the authors, and was funded by Takeda Pharmaceuticals International AG, Switzerland.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The original research for this study was funded by Shire Development LLC, which is now part of Takeda. The current analysis was funded by Takeda Pharmaceuticals International AG.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MaiBritt Giacobini has served as a consultant for Eli Lilly, Janssen-Cilag, and Shire (a Takeda company); has received speaker fees and reimbursement for travel costs from Nutricia; and has been a principal investigator for Novartis, Shire (a Takeda company), and Takeda. Carolina Brünner and Niklas Wallin Bernhardsson are employees of Takeda Pharma AB, Sweden. Ewa Ahnemark was employed by Shire Sweden AB (a Takeda company) at the time of this research. Jingcheng Zhao and Jonatan Freilich were employed by Parexel International during the study execution, which has received funding from Takeda for conducting the study. This study was funded by Takeda Pharma AB, Sweden, who were involved in the study design, collection, analysis, interpretation of data, and drafting of the manuscript.
Data Availability Statement
The study data cannot be shared due to local data protection regulations.
Supplemental Material
Supplemental material for this article is available online.
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.