Abstract
Objective:
Blood-brain barrier permeability (BBB) has been suggested to be involved in the etiopathogenesis of ADHD. Claudin-5, β-catenin and paxillin are important molecules with different roles in this barrier. Alterations in these molecules may disrupt the neurodevelopmental process by affecting various critical processes in the developing brain. Therefore, this study aimed to evaluate whether the peripheral levels of these molecules differ in children and adolescents with ADHD.
Method:
A total of 90 patients with ADHD aged between 8 and 18 years and 60 healthy controls were included in this study. The severity of ADHD symptoms was determined with the Atilla Turgay Scale. Child Anxiety-Depression Scale-Revised was completed to evaluate additional psychiatric problems of the patients. Serum levels of biochemical parameters were measured using enzyme-linked immunosorbent assay kits.
Results:
Serum claudin-5 levels were significantly lower and β-catenin levels were significantly higher in the ADHD group compared to the control group. However, there was no significant difference in paxillin serum levels between the groups.
Conclusion:
This study suggests that claudin-5 and beta-catenin may play a role in the pathogenesis of ADHD. These proteins may affect the brain by causing a dysregulation in BBB permeability or through other mechanisms.
Introduction
ADHD is a neurodevelopmental disorder that usually initiates in childhood and may continue in adulthood, characterized by inattention, hyperactivity/impulsivity and impairs the functionality of the individual (Thapar & Cooper, 2016). ADHD is a common reason for outpatient clinic visits in childhood and adolescence (Türkoğlu, 2014). It has been reported that the prevalence of ADHD varies between 2% and 7% worldwide with an average of 5% (Polanczyk et al., 2015). Although ADHD is a common disorder, its etiology has not been fully elucidated although genetic, neurobiological and environmental factors are thought to be effective in its etiopathogenesis (Thapar & Cooper, 2016). It is thought that changes in the blood brain barrier (BBB) structure, which have attracted increasing attention in the etiology of psychiatric disorders including schizophrenia, major depressive disorder and bipolar disorder in recent years, may also be effective in the etiopathogenesis of ADHD (Aydoğan Avşar et al., 2020; Najjar et al., 2017). However, data on the relationship between alterations in the structure of the BBB and ADHD are limited in the literature.
The BBB is a structure that prevents the transfer of harmful molecules from the peripheral circulation to the brain and sometimes regulates homeostasis by allowing the transfer of some molecules (Greene et al., 2020). Recent studies seriously point out that there is a relationship between structural changes in the BBB and psychiatric disorders. A recent study has shown that patients with bipolar disorder and schizophrenia have alterations in claudin and metallopeptidase-1 molecules indicating disruptions in the structure of the BBB (Lizano et al., 2023). In autism spectrum disorder (ASD), which is one of the other common neurodevelopmental disorders, there are data showing disruptions in the BBB structure such as neuron-specific enolase and creatine kinase brain isoenzyme changes (Lv et al., 2016). In addition to these data, recent studies suggest that BBB alterations are also observed in ADHD (Aydoğan Avşar et al., 2020; Ferahkaya et al., 2024).
Tight junctions (TJs) form the basis of all barrier systems including the BBB. TJs are dynamic structures formed by configurations of different proteins. The claudin family is one of the main proteins that form TJs. Claudin-5 is a member of this family and has an important role in the regulation of paracellular ionic permeability and is the most common isoform in the BBB. In a recent study, it was found that the levels of claudin-5 were significantly decreased in the hippocampus grey matter of individuals diagnosed with depression and schizophrenia (Greene et al., 2020). In addition to this study, it has been reported that claudin-5 changes could be involved in mechanisms underlying psychiatric disorders including schizophrenia, obsessive-compulsive disorder (OCD) and bipolar disorder (BPD; Greene et al., 2018; Işık et al., 2021; Kılıç et al., 2020). There are only two studies in the literature investigating claudin-5 levels in ADHD sample and although the results of these studies contradict each other, both studies indicate that there may be structural changes in the BBB in ADHD (Aydoğan Avşar et al., 2020; Ferahkaya et al., 2024).
β-catenin is a subunit of the cadherin protein complex and is also an important component of the WNT signaling pathway. In addition to its functions including cell adhesion and gene transcription, it induces endothelial barrier functions and plays an important role in ensuring barrier continuity (MacDonald et al., 2019). Studies indicate that the WNT/β-catenin pathway may play a role in the etiopathogenesis of psychiatric disorders (Santos et al., 2021; Zhang & Li, 2021). In a recent study, increased β-catenin levels were found in ASD (Bilgic et al., 2023). It is thought that the WNT/β-catenin pathway may be involved in the etiology of ADHD at genetic, environmental and pharmacotherapeutic levels (Yde Ohki et al., 2020). There is no study in the literature investigating the relationship between β-catenin levels in the peripheral circulation and ADHD.
Paxillin is a member of the group III LIM domain protein family that binds the integrin-mediated signal to the actin cytoskeleton, which has a very important role in cell adhesion (Ma & Hammes, 2018). In addition to its role in cell adhesion, it plays an important role in the regulation of cell proliferation, migration and dynamic changes required for cell survival in these processes and in cytoskeletal reorganization (Deakin & Turner, 2008). In the light of this information, paxillin may play a role in the etiopathogenesis of ADHD by affecting developmental processes in addition to its possible roles in the formation and maintenance of barrier structures. There is no study in the literature investigating the relationship between serum paxillin levels and ADHD.
Although it is thought that structural changes in the BBB and some proteins involved in the BBB may play a role in the etiopathogenesis of ADHD with this information in the literature, the available information is quite limited. Therefore, evaluation of claudin-5, β-catenin and paxillin levels in children with ADHD may be valuable in terms of clarifying the etiopathogenesis of the disorder. We planned this study based on the hypotheses that these molecules would differ in children with ADHD compared to healthy controls and could be involved in mechanisms underlying ADHD severity.
Methods
Sample
The patient group of our study was consisted of children and adolescents aged between 8 and 18 years who applied to a child and adolescent psychiatry outpatient clinic and who were diagnosed with ADHD according to Diagnostic and Statistical Manual of Mental Disorders Fifth Edition (DSM-5; American Psychiatric Association, 2013). The control group was consisted of healthy children who applied to a pediatric outpatient clinic for routine follow-up and controls. The exclusion criteria for both groups were chronic respiratory, physical, metabolic, genetic, neurological and endocrinological diseases and psychotropic drug use in the last six months. In addition, a diagnosis of any psychiatric disorder was determined as an exclusion criterion for the control group.
Our study included 90 participants with ADHD and 60 healthy controls. Based on an independent two-sample t-test, this sample size provides approximately 80% power to detect a medium effect size of Cohen’s d ≈ .47 at a significance level of α = .05. Given the exploratory nature of our investigation and limited prior data on BBB-related biomarkers in ADHD, this sample size is reasonable to identify moderate group differences. Nonetheless, larger samples will be necessary in future studies to confirm and extend these findings.
All research procedures were conducted in accordance with the Declaration of Helsinki and local laws and regulations. Approval for this study was obtained from Ethics Committee of local university. Verbal and informed written consent was obtained from the parents of all participants.
Diagnosis and Symptom Assessment
The diagnosis of ADHD was made on the basis of DSM-5 criteria with the patient’s history and psychiatric examination using the Schedule for Affective Disorders and Schizophrenia for School-Aged Children, Present and Lifetime Version (K-SADS-PL; American Psychiatric Association, 2013; Kaufman et al., 1997; Ünal et al., 2019). Symptom severity of the patients was determined by Atilla Turgay DSM-IV-Based Screening Scale for Disruptive Behavior Disorders in Children and Adolescents (T-DSM-IV-S). In addition, in order to evaluate the additional psychiatric problems of the patients, the Revised Child Anxiety Depression Scale Child Form (RCADS-CF) version was administered to the patients. The subjects included in the control group were evaluated with the same procedures as the patient group and completed the scale forms. Body mass indexes (BMI) of both groups were calculated and recorded. T-DSM-IV-S is a scale developed by Atilla Turgay which questions inattention, hyperactivity, impulsivity, oppositional defiant disorder and conduct disorder (Ercan et al., 2001). RCADS-CF was developed to screen anxiety disorders and depression in children and adolescents. Turkish validity and reliability study was conducted by Görmez et al. (Gormez et al., 2017).
Blood Samples
Venous blood samples of the participants in the patient and control groups were taken from the antecubital vein in the morning of the psychiatric evaluation in plastic tubes with K3EDTA. The 5 mL venous blood sample was centrifuged at 4°C, 4,000 rpm for 10 min and the serum samples were stored at −80°C until the date of biochemical analyses. Serum levels of claudin-5 (Cloud Clone (USCN)), β-catenin (Cloud Clone (USCN)) and paxillin (Nephente) were measured by double antibody sandwich enzyme-linked immunosorbent assay (ELISA) according to the manufacturer’s instructions. All biochemical parameter results were calculated as ‘ng/mL’ according to absorbance-concentration calibration graphs
Statistical Analysis
SPSS 21.0 was used for statistical analyses in our study. Independent sample t-test was used for two-group comparison for normally distributed parameters, and Mann-Whitney U test was used for two-group comparison for non-normally distributed data. In addition, a multivariate analysis of covariance (MANCOVA) was performed to reduce the risk of type I errors related to the multiple-test effect and to control for potential confounding factors such as age, gender and BMI. After determining that there was a significant difference between the study and control groups by performing MANCOVA, a one-way analysis of covariance (ANCOVA) was performed on the outcome variables. Non-normally distributed variables were log-transformed and log-transformed values were used in MANCOVA and ANCOVA. Pearson chi-square analysis was used to compare categorical parameters. Pearson or Spearman correlation analyses were used for correlation of quantitative parameters. In our study, Type-1 error (α) value .05 (5%) and p significance level <.05 were accepted
Results
A total of 90 children and adolescents (64 boys and 26 girls) diagnosed with ADHD constituted the patient group and 60 healthy children and adolescents (39 boys and 21 girls) constituted the control group. The mean age of the patient group was 11.32 ± 2.79 years and the mean age of the control group was 11.95 ± 2.77 years. No significant difference was found between the groups in terms of sex distribution and age. BMI of the patient group was calculated as 19.6 ± 3.8 and BMI of the control group was calculated as 19.9 ± 4.6 and no statistically significant difference was found between the groups. Demographic data of the ADHD and control groups are given in Table 1. The total and all subscale scores of the T-DSM-IV-S were significantly higher in the ADHD group compared to the control group, whereas no statistically significant difference was found between the groups in the R-CADS scale score.
Demographic Characteristics of ADHD and Controls and Scale Scores of ADHD Group.
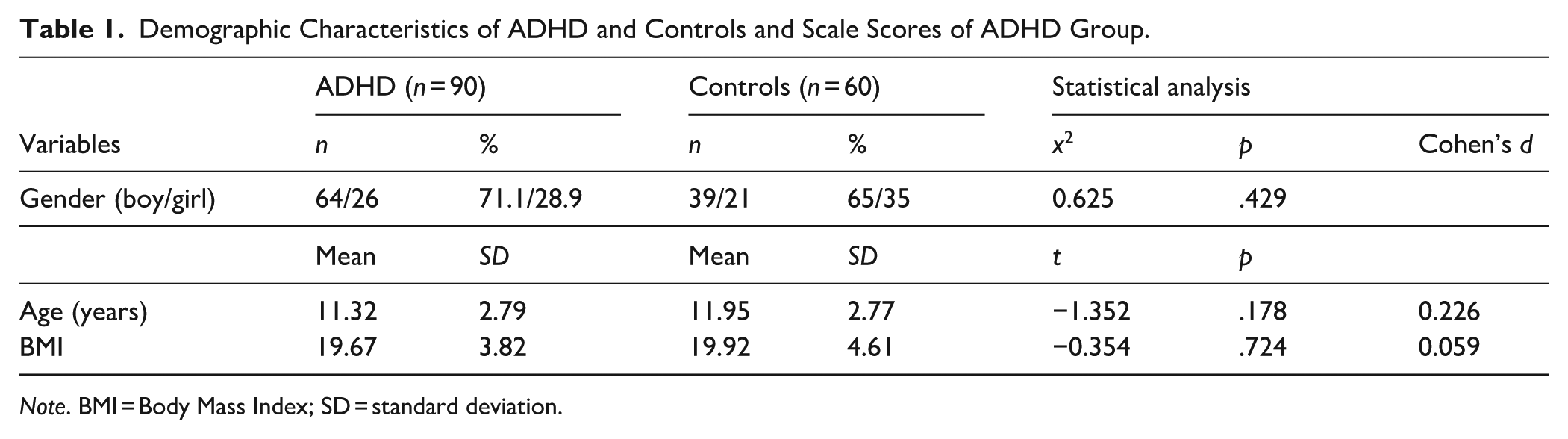
Note. BMI = Body Mass Index; SD = standard deviation.
In the analyses, claudin-5 levels were statistically significantly lower and β-catenin levels were statistically significantly higher in the patient group compared to the control group. However, no significant difference was found between the groups in terms of paxillin levels. The MANCOVA test demonstrated that there were significant differences between the groups for the whole sample (Pillai’s Trace V= 0.084, F = 4.315, p = .006 and η p 2 = .084). Separate univariate ANCOVAs were used after being adjusted confounding factors. After controlling for age, gender and BMI, the statistically significant differences in claudin-5 and β-catenin levels between the groups remained, whereas no significant difference was found in paxillin levels. The results of biochemical parameter levels and analyses are given in Table 2.
Serum Claudin-5, β-catenin and Paxillin Levels of ADHD and Controls.
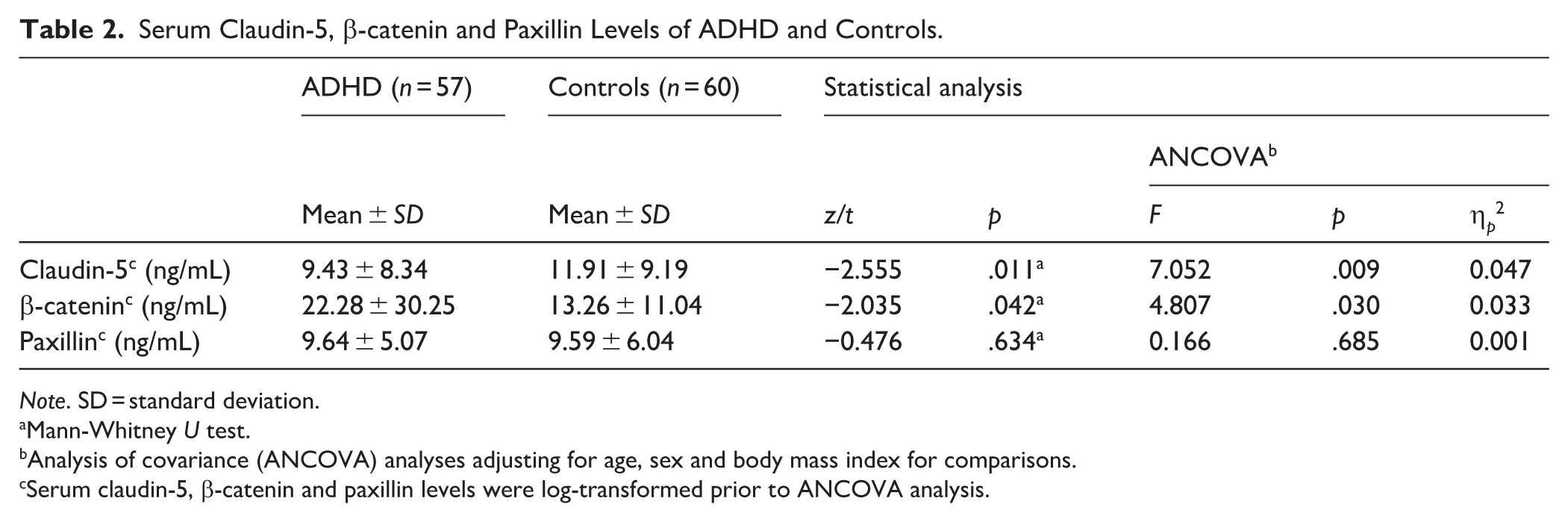
Note. SD = standard deviation.
Mann-Whitney U test.
Analysis of covariance (ANCOVA) analyses adjusting for age, sex and body mass index for comparisons.
Serum claudin-5, β-catenin and paxillin levels were log-transformed prior to ANCOVA analysis.
In the correlation analyses, no statistically significant correlation was found between the serum levels of biochemical parameters and T-DSM-IV-S and RCADS-CF scores.
Discussion
In this study, we investigated whether serum levels of claudin-5, β-catenin and paxillin are could be involved in mechanisms underlying ADHD in childhood and adolescence. We found that serum levels of claudin-5 were statistically significantly lower and β-catenin were statistically significantly higher in children with ADHD compared to healthy controls, and these significant differences persisted after controlling for confounding factors. However, we found no significant difference in serum paxillin levels between ADHD and control groups.
Claudin-5 is an important component of TJs in the BBB. TJs prevent the passage of pathogenic molecules to the central nervous system and the brain is more exposed to adverse environmental factors in claudin-5 dysfunction (Omidinia et al., 2014). The number of studies investigating the relationship between claudin-5 and psychiatric disorders in the literature is not sufficient to elucidate this complex relationship. In a recent study, Green et al. demonstrated that claudin-5 levels decreased in schizophrenia and depression (Greene et al., 2020). In two studies conducted with patients diagnosed with schizophrenia, decreased levels of claudin-5 were found and it was reported that this decrease disrupted the BBB structure and made the brain more sensitive to environmental factors, which may be effective in the development process of schizophrenia (Maes et al., 2019; Usta et al., 2021). In a recent study, an increase in the level of claudin-5 was found in individuals diagnosed with specific learning disorder, which is a neurodevelopmental disorder like ADHD, and it was stated that this may indicate impairment in the integrity of the BBB (Tanir et al., 2023). There are studies reporting that changes in claudin-5 levels in peripheral circulation are observed in ASD, another important neurodevelopmental disorder (Bilgic et al., 2023; McCullumsmith et al., 2007). To the best of our knowledge, only two studies, including the previous study of one of our team, have examined the relationship between ADHD and claudin-5 (Aydoğan Avşar et al., 2020; Ferahkaya et al., 2024). Aydogan et al. found increased claudin-5 levels in the group diagnosed with ADHD and interpreted the change as a compensatory increase secondary to BBB deterioration (Aydoğan Avşar et al., 2020). In our previous study, we found decreased claudin-5 levels in patients with ADHD compared to healthy controls, consistent with the results of the present study (Ferahkaya et al., 2024).
We found that β-catenin levels were increased in the ADHD group, and our study is the first to investigate the relationship between ADHD and β-catenin. β-catenin is a multifunctional protein, playing a central role in the Wnt signaling pathway and cell adhesion (MacDonald et al., 2019), as well as in processes such as cell proliferation, immune regulation and development (Chandrakesan et al., 2013). Therefore, alterations in β-catenin levels in serum may originate from both central and peripheral mechanisms. In the literature, associations between β-catenin and several psychiatric disorders have been reported. For example, increased β-catenin levels and activation of the Wnt/β-catenin pathway have been observed in individuals with ASD (Bilgic et al., 2023; Caracci et al., 2016), whereas decreased β-catenin protein and mRNA expression in the dorsolateral prefrontal cortex and temporal cortex have been reported in postmortem studies of bipolar disorder patients compared to healthy controls (Pandey et al., 2015). The prefrontal cortex is one of the most important anatomical regions implicated in ADHD, and alterations in β-catenin protein or mRNA expression in this area may contribute to the etiopathogenesis of the disorder. Further evidence for the potential involvement of the Wnt/β-catenin pathway in ADHD comes from experimental studies (Abu-Elfotuh et al., 2022; Custodio et al., 2023). For instance in a rat model of ADHD induced by monosodium glutamate, treatment with monosodium glutamate antagonists appeared to act, at least in part, through β-catenin–related mechanisms (Abu-Elfotuh et al., 2022). Based on these data, the elevated β-catenin levels in our ADHD group may reflect compensatory regulation in processes involving this protein. While these findings are consistent with the possible involvement of BBB-related molecular pathways, they should be interpreted cautiously given the multiple roles of β-catenin and the potential contribution of peripheral sources.
Previous studies have reported differing directions of change in claudin-5 and β-catenin levels across neuropsychiatric disorders. For example, Aydogan et al. observed elevated claudin-5 in children with ADHD, whereas we detected reduced levels in our sample (Aydoğan Avşar et al., 2020). In addition, we observed elevated β-catenin levels in individuals with ADHD. In contrast, postmortem brain studies have reported reduced β-catenin levels in the dorsolateral prefrontal and temporal cortices of individuals with bipolar disorder, while no significant alterations were detected in schizophrenia. Nonetheless, evidence suggests that dysregulation of the Wnt/β-catenin pathway may contribute to the pathophysiology of schizophrenia (Pandey et al., 2015; Vallée, 2022). These discrepancies may stem from methodological differences (e.g., plasma vs. serum measurements, assay sensitivity and sample handling), variations in participant characteristics (age, developmental stage, medication exposure and comorbidities) and the influence of acute stress or systemic inflammation on protein expression. Furthermore, claudin-5 and β-catenin regulation is highly context-dependent; changes may reflect compensatory upregulation in some conditions and downregulation in others, even within related neurodevelopmental or psychiatric phenotypes. This complexity suggests that BBB-related alterations may interact with broader systemic, developmental and disorder-specific mechanisms, underscoring the need for longitudinal and multimodal studies to reconcile these differences.
These findings are not directly generalizable to ADHD. We cite them to illustrate that alterations in claudin-5 and β-catenin have been observed across multiple neurodevelopmental and psychiatric disorders, potentially reflecting shared neuroinflammatory or neurovascular mechanisms. For example, altered claudin-5 expression has been demonstrated in postmortem prefrontal cortex tissue in patients with schizophrenia, with concurrent evidence linking its dysregulation to blood–brain barrier dysfunction and immune-related changes (Nishiura et al., 2017). In parallel, there is substantial literature implicating dysregulation of the Wnt/β-catenin pathway in neurodevelopmental and psychiatric conditions, including schizophrenia and autism spectrum disorders (Vallée, 2022; Zhuang et al., 2023). These parallels should be regarded as hypothesis-generating, and direct mechanistic studies in ADHD populations are warranted to clarify their relevance.
There are very few studies on the relationship between paxillin and psychiatric disorders. In a recent study conducted in children with ASD, no significant difference was found between the study groups in paxillin levels (Bilgic et al., 2023). Similarly, no statistically significant difference was found between the ADHD group and the control group in our study. Although these results suggest that paxillin does not play a role in the etiopathogenesis of ADHD, there is a need for further research on this subject which has very little data.
Taken together, while our findings highlight potential alterations in BBB-related proteins in children with ADHD, it is important to consider these results within a broader biological context. In addition to their involvement in blood-brain barrier integrity, claudin-5 and β-catenin have diverse roles beyond the CNS vasculature. Claudin-5 is critical for vascular barrier function in various peripheral tissues and may participate in systemic inflammatory responses that can influence neurodevelopment and psychiatric outcomes (Greene et al., 2019; Kakogiannos et al., 2020). β-catenin, a key mediator of the Wnt signaling pathway, also regulates cell adhesion, synaptic plasticity and immune system functions (Maguschak & Ressler, 2012; Pai et al., 2017). Dysregulation of these pathways has been implicated in multiple neurodevelopmental and psychiatric disorders, including autism spectrum disorder, schizophrenia and mood disorders (Bilgic et al., 2023; Teo et al., 2018; Vallée, 2022). Therefore, the altered serum levels of these proteins observed in our study may reflect a combination of BBB-related alterations and broader systemic or neurodevelopmental processes. This underscores the need to interpret our findings with caution, appreciating the complex and multifaceted nature of these molecular changes.
Limitations/Future Directions
Our study is the first to present data on β-catenin and paxillin levels in individuals with ADHD; however, several limitations should be considered when interpreting these findings. First, the sample size was relatively moderate. Although claudin-5 and β-catenin are highly relevant to BBB structure and function, they are not exclusively expressed in the CNS, and peripheral tissues may also contribute to their serum levels. Consequently, the tissue origin of the observed changes cannot be determined. Moreover, given the multifunctional nature of β-catenin, its elevation in serum could also reflect peripheral Wnt pathway activation, immune processes or developmental changes, and should not be interpreted as a BBB-specific alteration in isolation. Importantly, we did not include direct or surrogate measures of BBB integrity (e.g., albumin quotient, neuroimaging-based permeability assessment and cognitive markers), nor CNS-specific biomarkers (e.g., cerebrospinal fluid analysis and endothelial cell–derived exosome profiling), which limits the ability to confirm BBB involvement. Additionally, no significant correlations were found between biomarker levels and ADHD symptom severity or comorbid symptoms, suggesting that these molecules may reflect peripheral or non-specific processes rather than ADHD-specific mechanisms. Finally, our study does not establish whether serum claudin-5 and β-catenin levels correspond to concentrations at the BBB or in brain tissue, further constraining their interpretation as direct markers of CNS-specific changes.
In conclusion, this study provided important data on the possible roles of claudin-5, β-catenin and paxillin in the etiopathogenesis of ADHD. Changes in these molecules may lead to structural disruptions in the CNS, affecting processes such as cell maturation and adhesion in the central nervous system and disrupting the neuronal development process. These disruptions may also be effective in the etiopathogenesis of ADHD. Future studies to be planned in a longitudinal design with a larger sample by addressing the limitations of our study will provide valuable data on the role of the mentioned parameters in the formation of ADHD.
Footnotes
Ethical Considerations
Approval for this study was obtained from Ethics Committee of Necmettin Erbakan University with the decision number (2021/3225).
Consent to Participate
Verbal and informed written consent was obtained from the parents of all participants.
Author Contributions
Conception or design of the work: Necati Uzun, Ayhan Bilgiç and İbrahim Kılınç. Data collection: Hurşit Ferahkaya, Mehmet Berat Taş, İbrahim Kılınç and Ahmet Osman Kılıç. Data analysis and interpretation: Necati Uzun and Ayhan Bilgiç. Drafting the article: Necati Uzun, Ayhan Bilgiç and Hurşit Ferahkaya. Critical revision of the article: Necati Uzun and Hurşit Ferahkaya. Final approval of the version to be published Necati Uzun and Hurşit Ferahkaya.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided by a grant from the Necmettin Erbakan University Scientific Research Projects Unit within the scope of project number 211218019.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The dataset of the study is available on request from the corresponding author.
