Abstract
Objective:
Susceptibility to visual illusions is a consequence of the adaptation of the visual system, however, their perception or lack of it reflects differences in more general, global cognitive processes. Few studies have focussed on the susceptibility of individuals with attention-deficit/hyperactivity disorder (ADHD), even though visual impairment and percept differences have been thoroughly documented.
Method:
The present study evaluated 75 children (ages 6.09–12.99 years, 72% male) and 37 teenagers (ages 13–16.95 years, 62% male) with ADHD, and a sex-and-age matched sample of typically developing peers. They were tested with 103 pairs of illusory and control images spanning five illusion types.
Results:
We found increased susceptibility to the Müller-Lyer and Kanizsa Subjective Contour and decreased susceptibility to the Ebbinghaus illusion among children with ADHD when compared to typically developing controls. No differences were found for susceptibility to the Simultaneous Contrast and Moving Snake illusions. As for reaction times, we did not find differences between participants with ADHD and their control peers when judging illusions; however, in general participants give answers faster in the illusory trials than in control trials with the same magnitude of difficulties which also confirm the susceptibility of the illusions.
Conclusion:
Our findings point to small but important perceptual alterations, such as slightly reduced or delayed top-down or global processing ability in children with ADHD. Further research can focus on how these alterations may be useful to detect developmental alterations and understand perceptual difficulties in children with neuropathology.
Introduction
Psychiatric disorders involve alterations in an individual’s pattern of behaviour with negative consequences for their well-being and in their capacity to function in many contexts of everyday life. For many decades, research has suggested that various disorders may be associated with alterations in the visual perception, that is, the processing of information from one’s surrounding world obtained ocularly (Wade & Swanston, 2013). This is the case of visual illusions, which are perceptual inconsistencies between the observer’s visual perception and the physical characteristics of the stimulus (Gregory, 1997; Howe & Purves, 2005). Some underlying mechanisms of the perception of illusions have been proposed supporting the idea that different types of illusions do not share a unique common neural path and display different developmental trajectories (Axelrod et al., 2017; Schwarzkopf et al., 2011; von Gal et al., 2023; Xinran et al., 2025; see examples of different type of illusions in Figure 1). Susceptibility to the Ebbinghaus illusion (Figure 1(a)), for instance, develops before 4 years of age and does not change substantially much afterwards during the life spam, while susceptibility to the Simultaneous Contrast (Figure 1(c)), Moving Snake (Figure 1(d)), and Kanizsa (Figure 1(e)) illusions develops only later and increases with age (see Wincza et al., 2024 for a systematic review, Navarrete et al., 2025). However, the susceptibility to some other illusions, for example to the Müller-Lyer, seems that decrease with age (Navarrete et al., 2025; Wincza et al., 2024). In this sense, the study of susceptibility of visual illusions may help to identify perceptual changes across atypical developmental conditions such as ADHD.

(a) Ebbinghaus illusion: circles in the centres are the same size, but the one surrounded by large circles appears smaller. (b) Müller-Lyer illusion: lines are of equal length, but the one with the outward-pointing arrow heads appears longer. (c) Contrast illusion: circles in the centres of rectangle have the same luminosity, but the one with the less luminous background seems brighter (d) Moving Snake illusion: the different luminosity and arrangement of the pieces give the snake the appearance of movement. (e) Kanizsa Subjective Contour illusion (or Kanizsa): evokes the perception of a square while no physical edge is present.
Indeed, the study of illusions represents a powerful window into the neurobiology of visual perception as an inexpensive and non-invasive form of behavioural assessment in medical fields (Eagleman, 2001; Gori et al., 2016). For example, studies measuring atypical responses to visual information in patients with psychopathological disorders, such as schizophrenia (Costa et al., 2023; Grzeczkowski et al., 2018; King et al., 2017; Tibber et al., 2013) or psychosis (Mittal et al., 2015; Sperandio et al., 2023), have provided insights into how these individuals process their environment. For instance in schizophrenia, reviews suggest a weakening of top-down perceptual mechanisms, resulting an abnormal processing, especially, high level illusory displays, which are more dependent on cognitive control mechanisms (Costa et al., 2023; King et al., 2017). Since different illusions are processed in by different neural pathways, evaluating an array of them in the form of a test battery could provide information about certain neurodevelopmental alterations.
One theoretical explanation for the phenomena of the susceptibility of visual illusions is that when the target stimulus is processed globally (considering its environment) rather than locally (considering mainly its own dimensions), the actual physical characteristics of the target may be misperceived (Gori et al., 2016; Nayar et al., 2015; Wincza et al., 2025). The altered local–global visual processing has been suggested to be behind the perceptual abnormalities which are common in individuals with neurodevelopmental disorders, like autism spectrum disorder (ASD; Van der Hallen et al., 2015). Since Frith (1989) first introduced the Weak Central Coherence theory, as a core factor underlying ASD (i.e., altered local – global visual processing; Booth & Happé, 2010), the number of studies addressing this have increased – and so have contradictory findings (see Gori et al., 2016 for a review). A meta-analysis, for example, reports a disrupted temporal pattern of the local–global balance, concluding a slow global processing by individuals with ASD (Van der Hallen et al., 2015).
While studies about ASD and visual illusion perception abound, surprisingly there have been few on individuals with ADHD, even though visual impairment and visual percept differences have been also thoroughly documented for this developmental disorder (Jung et al., 2014; Kim et al., 2014). The altered local–global process has been described and tested by Kalanthroff et al. (2013) in adults with ADHD using the Navon task and found that ADHD participants showed lack of global processing bias. For example, altered contrast sensitivity (degree of contrast in luminosity that is necessary to visually detect differences) compared to healthy controls has been found in children with ADHD (Bartgis et al., 2009), but not in adults (Stevens et al., 2012). Makris et al. (2021) used a wide range of visual illusions and reported that susceptibility of ADHD children differed from neurotypical children for certain types of illusions, suggesting an altered local–global processing. Specifically, they report that children with ADHD were less sensitive to illusory contours (i.e., Kanizsa’s triangle, Letter “E” completion figure), but more sensitive to paradoxical illusions (i.e., Elephant illusion, Blivet illusion, Shelves count illusion). While Makris et al. (2021) focus on testing children with several neurodevelopmental disorders, they grouped illusions according to their perceptual appearance and did not consider participants’ age as an important influential factor in susceptibility either. In contrast, a recent study by Hokken et al. (2024) showed that children with ADHD performed similar to neurotypical children on the Kanizsa Illusory Contours task, with no difference in their gazing and fixation on the task measured by eye-tracker. The above mentioned studies highlight the lack of consensus about visual perception in children with ADHD and present some of the methodological shortcomings such as small sample sizes, narrow age ranges, and a lack of variability in the perceptual tests.
The perception of visual illusion in ADHD children warrants further research, as different levels of evidence suggest they may have altered visual perception. For instance, structural imaging studies examining neuroanatomical differences present in children with ADHD also bring support to the idea that the individuals with this disorder may be more sensitive to some visual illusions. Functional MRI studies showed that the surface area of the primary visual cortex (V1) strongly and negatively correlates with the perception of different visual illusions; participants with a small functionally defined V1 tend to have a stronger perceptual illusion than those individuals with a large V1 (Schwarzkopf & Rees, 2013; Schwarzkopf et al., 2011). Since occipital cortical thinning and significant reduction of grey and white matter volume in the occipital lobe has been found in individuals with ADHD (Brodmann areas 17, 18, and 19; corresponding to visual areas V1–V5), it can be inferred that they could have altered visual perception (Ahrendts et al., 2011; Durston et al., 2004; Proal et al., 2011; Schneider et al., 2006; Wolosin et al., 2009). In addition, a negative correlation has been found between the amount of grey matter in V1 and V2 and ADHD symptoms in childhood (Ahrendts et al., 2011).
In the current work, we aim to compare the illusory perception of children with ADHD with typically developing peers in an attempt to gain insight about their susceptibility to visual illusions. We tested a set of visuall illusions comprising some of the most common types, including geometric, contrast, movement, and contour illusions (Figure 1, Table 1) on children and adolescents with ADHD and sex and age matched neurotypical peers. We opted to present the illusions in a two-alternative forced choice test rather than the method of adjustment, since recent finding shows less reliable results with the latter one (Xinran et al., 2025). Since attention decifits is one of the main traits of ADHD, we implemted an eye-tracker to quantify the time that participants looked away from the screen during the experiment (Hokken et al., 2024; Kofler et al., 2008). We hypotezise that children with ADHD will show an altered susceptibility to visual illusions compared to age-and-sex-matched neurotypical controls and this difference depend on their age, for example, children versus adolescents (Shaw et al., 2007). Differences in susceptibility to a particular illusion could provide insight into the perceptual mechanisms that may be affected by a neural developmental disorder such as ADHD.
The Illusions Used in the Tests and Their Conditions.
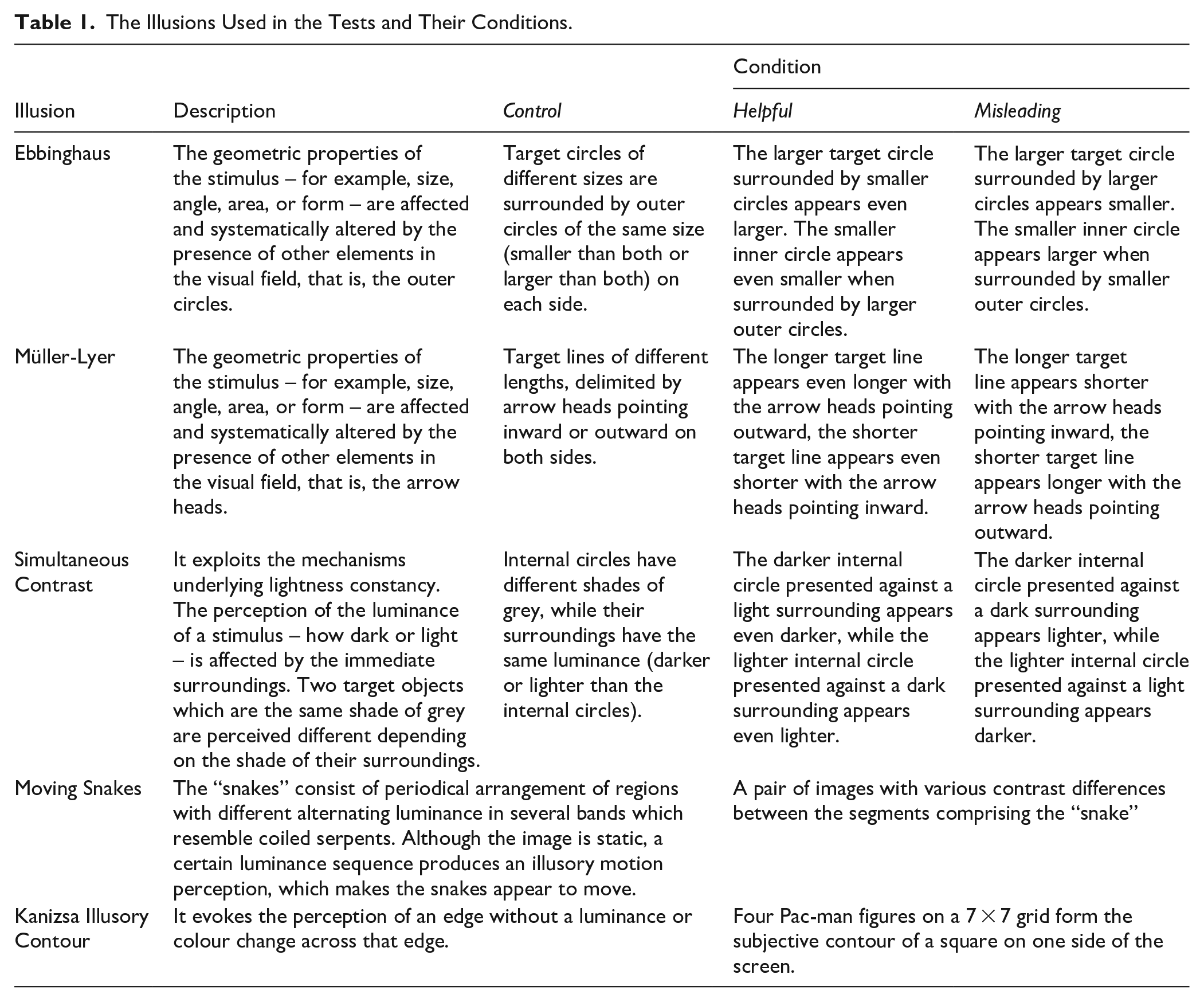
Methods
Participants
We tested a sample of outpatients of the Hospital Psiquiátrico Infantil Juan N. Navarro in Mexico City with a main diagnosis of ADHD and age and sex-paired healthy controls. In total we tested 224 participants (112 participants per group: 75 children [age range = 6.09–12.99 years, 72% male] and 37 teenagers [age range = 13–16.95 years, 62% male]) with ADHD and age and sex-matched neurotypical controls. Inclusion criteria for patients were a confirmatory diagnosis of ADHD based on a structured diagnostic interview with the parents or legal guardians (Mini-International Neuropsychiatric Interview, MINI KID), a intelligence quotient of 80 or above, as measured by the Wechsler Intelligence Scale for Children (WISC-IV, Crawford et al., 2010), normal or corrected vision, and not taking medication 3 months prior. Exclusion criteria were comorbidity with psychosis or ASD. Control children were attending public and private schools in three medium-density suburban cities in the metropolitan area of Mexico City and the neighbouring cities of Huixquilucan, State of Mexico and Cuernavaca, Morelos. Inclusion criteria for controls were normal or corrected vision and a score below 17 on the Strengths and Difficulties Questionnaire (SDQ, Goodman, 1997) in order to discard psychopathologies, such as anxiety and depression, as it has been shown that these may impact the perception of visual illusions (Salmela et al., 2021; Tloczynski et al., 2000).
Instruments and Equipment
Mini International Neuropsychiatric Interview for Children and Adolescents (MINI-KID)
The MINI KID is a structured diagnostic interview that screens for the presence of 23 psychiatric disorders based on DSM-IV and ICD-10 diagnostic criteria. The Spanish version of this interview showed significant concurrent validity and good inter-rater reliability (Sheehan et al., 2010). This interview was administered to patients and their parents by psychiatrist or child psychiatrist, who assessed the frequency of inattention, hyperactivity, and impulsivity in several settings (e.g., school, home, and social).
Wechsler Intelligence Scale for Children Fourth Edition (WISC-IV)
The WISC-IV is a clinical instrument to assess intelligence in children aged 6 to 16 years (Wechsler, 2003). It combines different neuropsychological test scores into an intelligence quotient. In this study the short form was applied (Crawford et al., 2010) in which the final score is extrapolated from the sum of the scaled scores in the Vocabulary, Similarities, Block Design, and Matrix Reasoning subtests.
Strengths and Difficulties Questionnaire (SDQ)
The SDQ is a questionnaire to assess the mental health of individuals aged 3 to 16 years (Goodman, 1997), that can be answered by the parents. It is composed of 25 items divided between five scales: emotional symptoms, conduct problems, hyperactivity/innatention, peer relationship problems and prosocial behaviour. In particular, hyperactivity/innattention is assessed using items 2, 10, 15, 21, and 25. As a screening tool it has been shown to be valid for detecting ADHD symptoms in randomised samples (Hall et al., 2019), good specificity and sensitivity with multi-informants (Goodman et al., 2000).
Visual Illusion Task
This task was built in PsychoPy (Peirce et al., 2019) and consists of a series of forced choice trials between two pictures developed by ; for more example also see Bremner et al., 2016; Doherty et al., 2010; Manning et al., 2017). The battery combines probe trials to evaluate the capacity of participants to judge size, colour, shape, and capacity to move the mouse, which if successful, are followed by illusory and control trials (103 total). We tested two geometric illusions (24 trials each), the Ebbinghaus (1902) and Müller-Lyer (1889), the Simultaneous Contrast illusion (Contrast from here onwards; Chevreul, 1839; 20 trials), a black and white version of the Moving Snakes illusion (10 trials; Kitaoka, 2003), and the Kanizsa Illusory Contour (Kanizsa from here onwards; Kanizsa, 1955; 24 trials; Figure 1). The Ebbinghaus, Müller-Lyer, and Contrast illusions were presented with Control, Misleading, and Helpful contexts (see definitions in Table 1). Stimulus were constructed by a target object in the middle and its surrounding elements to create (or not) the illusion. In the Control conditions the target stimuli were always different in the two pictures (i.e., in the Ebbinghaus they differed in area, in length in the Müller-Lyer, and in luminosity in the Contrast illusion). However, elements other than the target stimulus responsible for the illusory effect (e.g., size of surrounding circles in Ebbinghaus illusion, direction of arrow heads in Müller-Lyer illusion, or shade of the surrounding rectangle in the Contrast illusion, see Figure 1 and Table 1) were identical on both sides. In the Helpful context the elements responsible for the illusory effect were arranged in a manner such that it facilitates correct choices, whereas in the Misleading context the arrangements of the elements created the illusory effect acted against the correct choice. The Moving Snakes and Kanizsa illusions only had the illusion condition (Table 1). All sets of illusions contained trials with incremental difficulty; either the size or luminance of the target objects were modified in a systematic way to create trials with different difficulties in the Ebbinghaus, Müller-Lyer, and Contrast illusions; ratios mark the percentage differences between the target stimuli, larger ratios mean less difference. The difference between the target stimuli was 1%, 5%, 10%, 15%, 20%, or 30%. In the Moving Snakes illusion, the luminosity of the pieces which create the picture were modified to create perceptually more or less movement and in the Kanizsa illusion the size of the Pac-mans was changed. All pairs of pictures and ratios are presented in the Supplemental Material S1.
All images were presented over a white background. Trials were randomised within a set and sets were presented in a random order for each participant. Verbal and text instructions appeared at the beginning of the task, as well as at the beginning of every set of illusions and a brief version repeated after each trial (i.e., “choose the largest”). Participant answered by clicking the image with a mouse; if no click was performed, the next trial was shown after 15 s. No feedback was provided during the trials. A detailed description of the probe trials, the included types and ratios, and the code for the study can be downloaded here: https://github.com/marcosrosetti/visual_illusion_task. All tests were done on a Lenovo ThinkPad P15 15.6″ screen laptops, with 1920 × 1080 screen resolution.
Computer-Mounted Eye Tracker
During all experiments, a non-invasive eye tracker (PCEye, Tobii DynaVox, Danderyd, Sweden) was placed at the bottom of the screen. Eye movements were recorded during visual illusions task. The device provides x/y coordinates with a 33 Hz sampling frequency. Data was used to calculate the duration of lost gaze events (or the total time when the tracker lost connection with the gaze of the participants). Eye tracking allowed us to quantify whether participants in both groups looked at the screen the same amount of time.
Procedure
At the hospital, we first asked for informed consent from parents and assent from the child. After a confirmatory diagnosis with the MINI-KID, they were evaluated with WISC-IV and completed the visual illusion battery. The MINI-KID interview was conducted by a resident child psychiatrist, the WISC-IV was administered by resident clinical psychologists, and the visual illusion task was run by the first author. All tests were administered on the same day, if necessary, with 10 to 15 min breaks when needed. The sessions took place between 9:00 and 14:00; each session lasted about 1 to 2 hr.
At schools, we obtained signed consent and SDQ results from parents. After confirming the participant assented to participate, they were momentarily excused from their class and taken to a quiet classroom to perform the visual illusion trials, which were ran by the first and second authors. All tests were performed during school hours and a session lasted approximately 10 to 25 min.
For the visual illusion task, participants were asked to sit in front of a laptop computer in such a way that the visual angle was relatively constant, at a distance of approximately 60 cm. The information collected in the brief interview, such as handedness and eyesight, was used to accomodate the mouse on the side of the dominant hand. After a brief introduction to the computer and the task at hand, the PsychoPy programme was started. From here on, all vocal instructions came from the computer to homogenise duration and exclude the experimenter’s influence. The instruction at the beginning of the task read: “You are going to be presented with a series of pairs of images in which you have to choose the image according to the task instructions. Try to make your decision as fast as you can.” The brightness and volume of the computer were adjusted to 100% for all participants.
Analysis
We compared performance by means of Generalised Linear Mixed Models, with correct answers or latency as response variables and case (control or ADHD), ratio (1%, 5%, 10%, 15%, 20%, or 30%) and context (when avaliable, Misleading, Helpful, or Control) as predictors and pair id as random factor. For those models with correct answers, we used a Binomial distribution, whereas a Gaussian distribution was used for those with latency. For all models, we report the standarised coeficients (SC) as a form of effect size, along with the 95% Confidence Invtervals (CI). Duration of lost gaze events were analysed in the same way; however, due to equipment malfunctions, this only included 82 pairs. All analysis were done in R Studio (R Core Team, 2024). All comparisons were two-tailed and significance was set at p < .05.
Results
Eye Tracking
We found no differences between ADHD and their control peers regarding the duration of the lost gaze events in either the children or the adolescents, suggesting that they paid the same amount of attention to the task (see Supplemental Material S2).
Ebbinghaus and Müller-Lyer
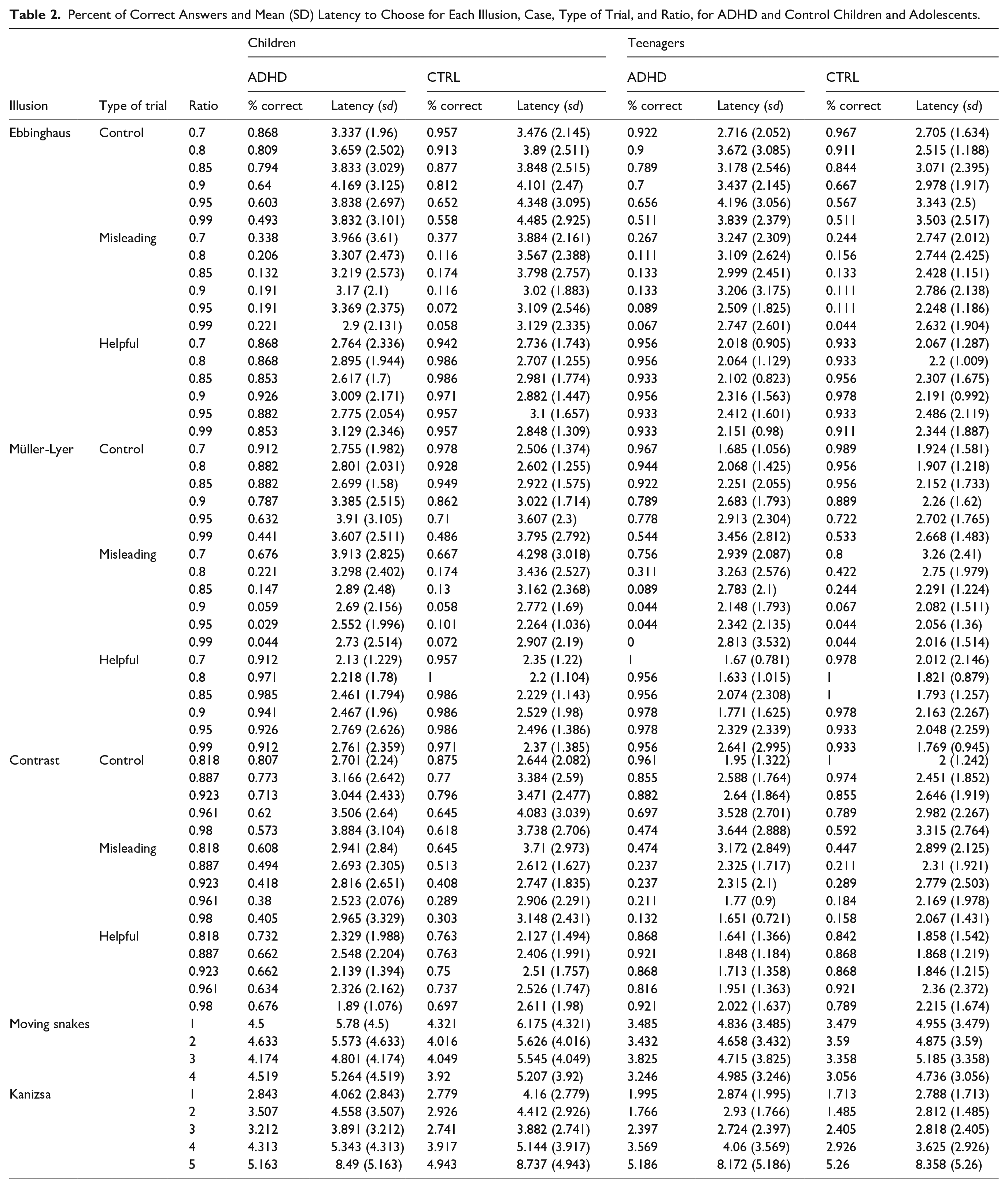
Table 2 shows the descriptive statistics for all illusions classified by age-groups and case. For both geometrical illusions we found significant differences between ADHD and control participants in the number of correct answers in the Misleading trials in case of children, but not for teens. The susceptibility was opposite in the two illusions: during the Ebbinghaus trials children with ADHD gave slightly more correct answers compared to controls which suggests they were less susceptible to this illusion (ADHD: 20.4% vs. control: 16%; SC [95% CI] = −0.511 [−0.907, −0.115], p .007), but we did not find differences between ADHD teens and controls (ADHD: 13.6% vs. control: 11.4%; SC [95% CI] = −0.27 [−0.965, 0.426], p = .905). In the trials of the Müller-Lyer illusion children with ADHD gave slightly less correct answers compared to controls suggesting they are more susceptible to this illusion (ADHD: 19.3% vs. control: 20.4%; SC [95% CI] = 0.332 [−0.145, 0.809], p ≤ .001), however this difference was not present between ADHD teens and their control peers (ADHD: 21.5% vs. control: 27.6%; SC [95% CI] = 0.711 [−0.089, 1.51], p = .785). We did not find differences in the number of correct answers in the Helpful trials in any of the age groups, meaning the size perception was not influenced in this type of trials. However, what we found interesting is that during the Control trials in both illusion: children with ADHD gave fewer correct answers compared to controls (Ebbinghaus: ADHD: 69.3% vs. control: 78.6%; SC [95% CI] = 0.725 [0.465, 0.984], p = .001; Müller-Lyer: ADHD: 75.7% vs. control: 82.1%; SC [95% CI] = 0.768 [0.44, 1.095], p = .001), but not teens. As the ratio between the sizes of the target object increased, so did the difficulty of the task, which became harder for almost everyone, but the correct responses were different for the two illusions. This is illustrated by the significant case × ratio interactions in case of both geometrical illusions in the younger age group (Table 3). Effect sizes for all these results were around the medium range.
Percent of Correct Answers and Mean (SD) Latency to Choose for Each Illusion, Case, Type of Trial, and Ratio, for ADHD and Control Children and Adolescents.
Standardised coefficients with 95% confidence intervals for the models evaluating the regression models of case, ratio, and their interaction on the probability of correct answers and latency to choose. Models were done for the subset of each illusion and type of trial, and for each age group.
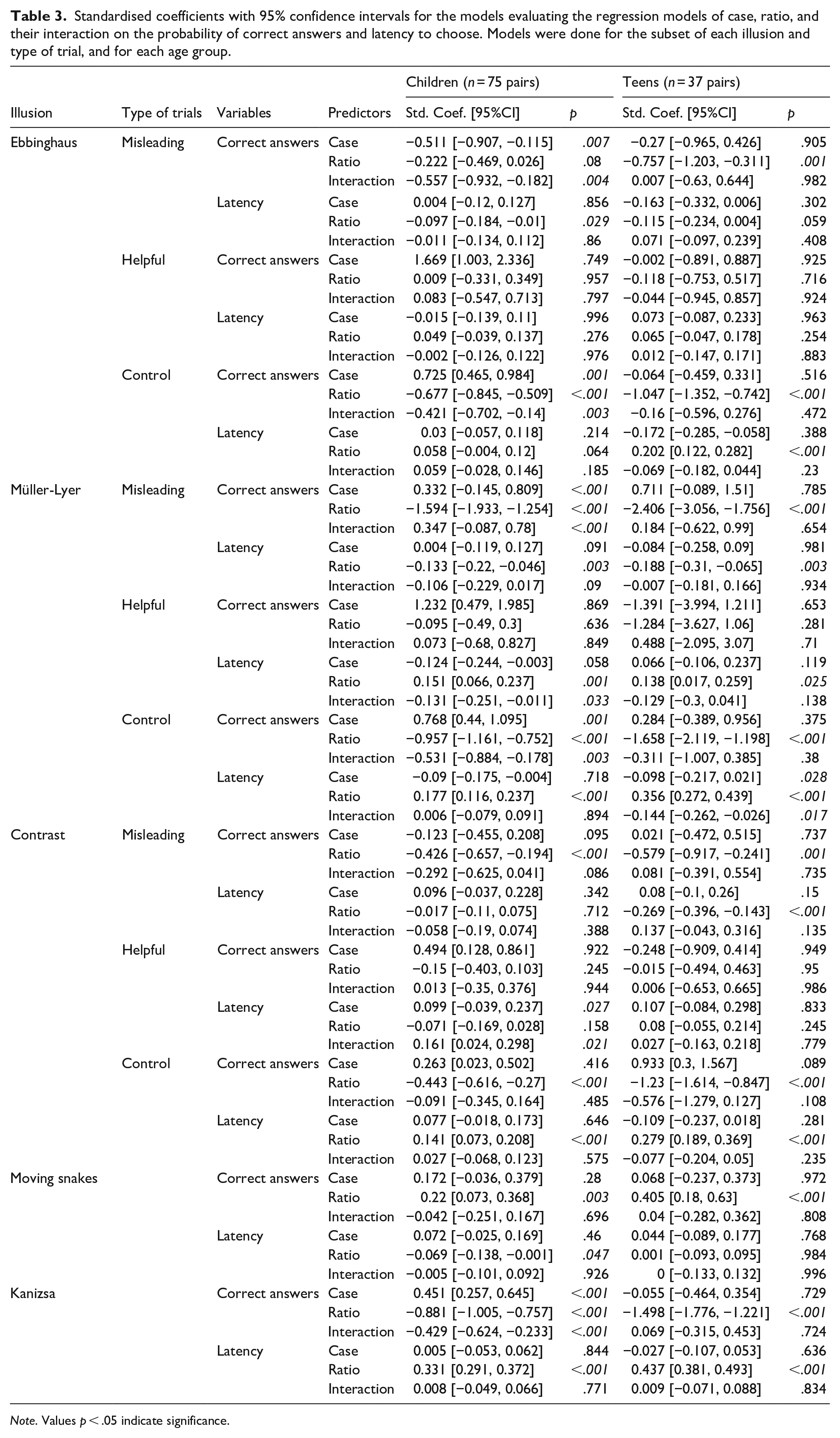
Note. Values p < .05 indicate significance.
The latency to answer mirrored this pattern (Tables 2 and 3). In the Control trials in both, the Ebbinghaus and Müller-Lyer illusion, as expected, response time increased as the ratios increased, as it is easier to distinguish between smaller ratios than larger ones. Also, control teens gave answers faster in the Müller-Lyer illusion control trials than ADHD teens (SC [95% CI] = −0.098 [−0.217, 0.021], p = .028). In the Misleading trials, in both illusions, all participants were faster giving answers on the larger ratios, suggesting they perceived the illusions. The small, non-significant estimate for the Helpful trials of the Ebbinghaus illusion suggests a ceiling effect. However, the significant ratio effect in case of the Müller-Lyer illusion in both, children (SC [95% CI] = 0.151 [0.066, 0.237], p = .001) and teenagers (SC [(95% CI] = 0.138 [0.017, 0.259], p = .025) also confirm that they are more susceptible to this illusion (Tables 2 and 3).
Contrast and Moving Snake Illusions
For both these illusions, no difference was found between the ADHD and control groups in children nor teens in the number of correct responses (Contrast: children: ADHD: 62.9% vs. Control: 66.4%; teens: ADHD: 67.1% vs. Control: 70%; Moving Snake: children: ADHD: 51.8% vs. Control: 55.8%; teens: ADHD: 63.1% vs. Control: 64.5%), suggesting no difference in susceptibility (Tables 2 and 3). However, for the Contrast illusion, ratio had an effect: despite observing no effect for Case in the Control and Misleading conditions, the number of correct answers decreased as the ratio increased in both, children and teenagers. The latency decreased for the Misleading trials, but increased in the Controls as the ratio increased, suggesting more difficulty assessing the difference between the stimuli in both case and age groups (Tables 2 and 3). In the Moving Snake illusion, ratio also had a significant effect, with an increment in the number of correct answers as the contrast differences became more pronounced between the pictures in both age groups, suggesting the presence of an illusory effect. However, in latency this was observed only for the children, but not for the teens (Tables 2 and 3). Effect sizes for the correct answers were in the small to medium range while for latency they were in the small range.
Kanizsa Illusion
We found a difference between ADHD and control children in the number of correct answers in the Kanizsa illusion; controls found the contour of the square more frequently that their ADHD peers, with effect sizes in the small to medium range (ADHD: 74.1% vs. Control: 77.6%; SC [95% CI] = 0.451 [0.257, 0.645], p < .001), suggesting they were more susceptible to the illusion (Tables 2 and 3). More correct answers were registered as the size of the Pac-mans increased, but more so for the controls than ADHD children as shown by the significant interaction (Table 3). Latency to answer mirrored this effect, with responses getting slower as Pac-man shapes became smaller. All additional statistics can be consulted in Tables 2 and 3.
Discussion
We aimed to evaluate children and teenagers with ADHD and age and sex paired healthy controls on a task involving a wide range of illusions, which included various contexts and ratios. We found that children with ADHD were slightly less sensitive to Ebbinghaus, but more so to Müller-Lyer illusion. Such differences were not observed for teenagers. While all participants were shown to be susceptible to Contrast and Moving Snake illusions, no differences were found between ADHD and Controls. Finally, children, but not teens with ADHD performed worse when locating the contour in the Kanizsa illusion. Below we discuss what susceptibility or lack of it may mean for each illusion.
Ebbinghaus and Müller-Lyer illusions have been considered as higher-level visual illusions which trigger complex cognitive and perceptual mechanisms, and their perception is a top-down process and require prior knowledge (King et al., 2017). These illusions used to study susceptibility to the influence of context in samples with psychiatric afflictions, for example, schizophrenia (Costa et al., 2023; Grzeczkowski et al., 2018; King et al., 2017), psychosis (Mittal et al., 2015), ASD (Chouinard et al., 2013; Gori et al., 2016; Manning et al., 2017); they point to a common phenomenon. While susceptibility to the Ebbinghaus illusion of patients with the above-mentioned psychopathologies are often been found decreased compared to the neurotypical population, they are all more sensitive to the Müller-Lyer illusion. Given, for instance, that ADHD and ASD share many cognitive traits (Lau-Zhu et al., 2019; Rau et al., 2020), it could be that they may share traits in regard to local/global processing preferences. Our study supports this idea: we found similar results in the case of the Ebbinghaus and Müller-Lyer illusion: children with ADHD seems less susceptible to the Ebbinghaus and more susceptible to the Müller-Lyer illusion.
This is in contrast with the result of Makris et al. (2021), who reports no susceptibility of ADHD children to the Ebbinghaus illusion. The different results can perhaps be explained by the fact that (1) studies differ in the sample size (the recent study tested almost three times more subjects with ADHD), (2) age categorisation (the current study divided the participants into children and teens) which is important given that susceptibility of the Ebbinghaus illusion develops earlier compared to the Müller-Lyer (and many other, Navarrete et al., 2025) and, (3) methods (number of repetition, different contexts, type of task, and various ratios). Thus, the current study detected that only in children was this difference apparent; the development of susceptibility of ADHD children may be slightly behind than their control counterparts.
One aspect of visual perception is the ability to discern different degrees of luminance. Two of the illusions used in the current study, Contrast and Moving Snake, rely heavily on this ability. In the present study, we found that both ADHD and control participants perceive these illusions in the same way. This is in line with the results of Makris et al. (2021) who also reports no differences in the susceptibility to contrast and motion illusions in children with ADHD and controls. An older study from Bartgis et al. (2009), however, reported that contrast sensitivity was significantly worse in children with ADHD than controls. This difference may be because they used the Functional Acuity Contrast Test and not an illusory image to test contrast luminance sensitivity. Tests involving luminance contrast sensitivity in other neurodevelopmental psychopathologies are few and provide mixed evidence. Grzeczkowski et al. (2018) reported that schizophrenia patients were less susceptible to the Contrast illusion than controls, and Laeng et al. (2018) reports that they did not find any difference in either perceptual or physiological (involuntary pupillary reaction) between ASD and control adults while using the Asahi brightness illusion.
In the current study, we found that children, but not teens with ADHD showed more difficulties to find the square contour than controls, which can be interpreted as less susceptibility to the illusion. This result is in line with Makris et al. (2021), who also reported less sensitivity to subjective contours for ADHD participants than controls, but contrast with the findings of Hokken et al. (2024). The latter study included medicated children and responses were recorded verbally and through eye-tracking. As visual completion of non-visible borders is considered as a top-down neural processes (Murray & Herrmann, 2013), our results further support the idea that there is a specific deficit in this type of attentional control in children with ADHD (Makris et al., 2021; Mazaheri et al., 2010). However, we cannot exclude the possibility that the lower performance of children with ADHD is due to their general poorer visual search. In our task, we present the stimulus hidden within a series of incomplete contours, and the participant needs to perform an extensive visual search to find the complete contour. Several reviews suggest less accuracy and precision and a larger number of errors in the visual search patterns of children with ADHD (Hokken et al., 2023; Mason et al., 2005; Mullane & Klein, 2008). Future studies with another type of visual search test may rule out this possibility and confirm the results.
Susceptibility to certain visual illusions has been reported to change with age (Doherty et al., 2010; Navarrete et al., 2025; Wincza et al., 2024). However, this phenomenon has often been overlooked in previous studies examining perception in individuals with developmental psychopathologies. We observed that the altered perception of children with ADHD – manifested in their responses to the Ebbinghaus, Müller-Lyer, and Kanizsa illusions – appears to diminish during adolescence, as no significant differences were found between adolescents with ADHD and typically developing controls. While the underlying mechanisms remain unclear as to why children but not teens differ, a few possible explanations can be proposed. For instance, differences in attention span might account for the observed effects, as reduced attention might lead to specific strategies affecting the response (Betts et al., 2006; Wincza et al., 2024). However, this explanation is not supported by our eye-tracking data, which showed no differences in the duration of lost-gaze events between children and adolescents with ADHD and their typically developing peers, nor between children and adolescents within each group. An alternative explanation is that these differences reflect heterochronic maturation, whereby visual perceptual development in children with ADHD follows a delayed but converging trajectory. This notion is consistent with findings by Ropar and Mitchell (2001), who reported a decrease in susceptibility to the Müller-Lyer illusion with increasing verbal mental age in children with autism and Asperger syndrome. Finally, Wincza et al. (2024) found it plausible that the differences in the children’s perception may be more likely driven by immature response strategies than by genuine perceptual differences.
Although the study yielded significant results, certain limitations must be acknowledged. The current study does not include any other neurophysiological measurements which might help interpret the findings. Additionally, we did not include the severity of symptoms or intelligence quotients as variables in the analysis as we did not recorded the first and the second was only avaiable for the the patient group. Different assessors conducted testing at school and hospital which could introduce confounding factors, however, as the test was automated, we think such influence might be minimal. Finally, we can only make statements about the illusions included here which represent a subset of all the illusions available. Despite the well documented difference between ADHD and neurotypical participants in attention process registered by eye-tracking measures (Levantini et al., 2020), the current study reports no difference in regards to the duration of lost gaze; further extending the eye tracking metrics may still reveal differences.
Conclusion
Susceptibility to illusions is a consequence of the adaptation of the visual system to a highly complex and dynamic world; manufactured stimuli help them manifest in an experimental context. Despite their artificial nature, the consequences of being highly susceptible or not to certain illusions may reflect differences in more general cognitive processes. The differences found in the present research add to the evidence that there are cognitive and sensorial difficulties associated with ADHD. It seems, however, this perceptual alteration is present only in young children. While it is difficult to relate susceptibility to visual illusions in daily life, we can think of how the underlying processes may influence the daily functioning of individuals with ADHD; for instance, even a slightly reduced or delayed top-down processing ability (as suggested by less susceptibility to Ebbinghaus and Kanizsa and more to the Müller-Lyer) may be reflected in, for instance, taking longer to processes a highly complex scene. Future studies could try to explore the relationship between cognitive tasks such as those evaluating cognitive flexibility or set shifting that require local and global attentional processes with an individual’s susceptibility to visual illusions.
Supplemental Material
sj-docx-1-jad-10.1177_10870547251369665 – Supplemental material for Perception of Visual Illusions in Children and Teenagers With ADHD
Supplemental material, sj-docx-1-jad-10.1177_10870547251369665 for Perception of Visual Illusions in Children and Teenagers With ADHD by Valeria Montiel, Vania Navarrete, Ana M. González-Pérez, Carolina Vázquez de Alba, Ricardo Díaz-Sánchez, Péter Szenczi, Marcos Rosetti, Rosa E. Ulloa and Oxána Bánszegi in Journal of Attention Disorders
Supplemental Material
sj-docx-2-jad-10.1177_10870547251369665 – Supplemental material for Perception of Visual Illusions in Children and Teenagers With ADHD
Supplemental material, sj-docx-2-jad-10.1177_10870547251369665 for Perception of Visual Illusions in Children and Teenagers With ADHD by Valeria Montiel, Vania Navarrete, Ana M. González-Pérez, Carolina Vázquez de Alba, Ricardo Díaz-Sánchez, Péter Szenczi, Marcos Rosetti, Rosa E. Ulloa and Oxána Bánszegi in Journal of Attention Disorders
Footnotes
Acknowledgements
We thank Carolina Rojas for technical and bibliographical assistance and Miriam Alarcon for the help during the data collection process. We also thank the schools, Escuela Nacional Preparatoria número 4 “Vidal Castañeda y Nájera,” Escuela Secundaria Técnica N° 73 “Carlos Vallejo Márquez,” Escuela primaria “Guadalupe Victoria” and the Colegio Montessori de Tepoztlán, to provide us technical and infrastructural help to execute our experiments.
ORCID iDs
Ethical Considerations
All procedures were approved by both Committees of the Instituto de Investigaciones Biomédicas of the Universidad Nacional Autónoma de México and the Hospital Psiquiátrico Infantil (II3/06/0721).
Consent to Participate
Written informed consent was obtained from the children’s parents or guardians.
Author Contributions
O.B., M.R., and R.E.U. served as lead for investigation, methodology, and supervision, served in a supporting role for formal analysis, and contributed equally to conceptualisation, data curation, visualisation, and writing. O.B. received financial support for the project and did the project administration. P.S served as lead for data curation and formal analysis and contributed equally to investigation, methodology, supervision, validation, visualisation, and writing. V.N, V.M., A.M.G.P., C.V.A., and R.D.S. contributed equally to investigation, methodology, writing, and served in a supporting role for visualisation.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Dirección General de Asuntos del Personal Académico, Universidad Nacional Autónoma de México (DGAPA, grant number IA204222).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Dataset can be accessed: dx.doi.org/10.6084/m9.figshare.27046186
Supplemental Material
Supplemental material for this article is available online.
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
