Abstract
Background and Aim:
Crocus sativus also known as saffron, is one of the most popular traditional plants. This study aims to evaluate the efficacy and safety of saffron extracts in ADHD.
Method:
This study includes clinical trial studies that assessed the efficacy and/or safety of saffron in ADHD patients. Non-English papers, review articles, commentaries, letters, observational studies, thesis, animal studies, in-vitro studies, and conference abstracts were not included. The risk of bias in randomized studies was evaluated based on the Cochrane RoB.2, and risk of bias in pre-post intervention studies was assessed using the ROBINS-I tool.
Results:
Four studies met our inclusion criteria with a total of 118 patients. The results manifested an efficient role of saffron as either an adjuvant therapy to MPH or a single therapy against ADHD, without significant safety issues.
Discussion:
Saffron demonstrates promise in improving ADHD symptoms, with an acceptable safety profile. Future well-designed multicentral studies are suggested.
Introduction
ADHD is one of the most common neurodevelopmental disorders among children, with a prevalence of 3% to 7% (Castells et al., 2011), and it is reported to persist during adulthood in 15% to 50% (Faraone et al., 2006; Lara et al., 2009). The prevalence of ADHD in adults is about 2.5% to 5% (Sayal et al., 2018; Thomas et al., 2015).
The symptoms of ADHD in children include inattention, hyperactivity, and impulsivity. Inattention is commonly demonstrated as distractibility, straining in sustaining attention on activities, trouble in organizing tasks and activities as well as forgetfulness. Hyperactivity or impulsivity symptoms mostly include being fidgety, interrupting others’ speech, inability to be still, inappropriate movements, and excessive talking (Posner et al., 2020; Thapar & Cooper, 2016; Wolraich et al., 2019). Patients with ADHD need a comprehensive treatment covering behavioral, psychological, and educational needs (Dalrymple et al., 2020). Pharmacological treatments consist of stimulants such as methylphenidate (MPH/ Ritalin©), and non-stimulant drugs such as atomoxetine and guanfacine (Caye et al., 2019; Toomey et al., 2012).
The medical use of alternative methods, especially herbs, goes back to hundred years ago (Modabbernia & Akhondzadeh, 2013). In this regard, one of the most popular traditional plants, best known for its culinary and coloring use in Asian and European countries, is Crocus sativus also known as saffron. Based on recent studies, saffron as an herbal remedy is believed to have antispasmodic, antitumor, antihypertensive, anticholinergic, anticoagulant, antidepressant, antiseptic, and anticonvulsant effects (Bathaie & Mousavi, 2010; Srivastava et al., 2010). Saffron plays a role as an N-methyl-
Dutta et al. investigated the advancement in the evidence-based use of herbal remedies for ADHD. The usefulness of saffron had only been assessed in one of the included articles of this study (Dutta et al., 2022); however, there are more published articles discussing the effectiveness of saffron (Baziar et al., 2019; Blasco-Fontecilla et al., 2022; Khaksarian et al., 2021; Pazoki et al., 2022). This study aims to systematically evaluate the safety and efficacy of saffron in patients with ADHD.
Methods
This systematic review was conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 statement (Page et al., 2021).
Eligibility Criteria
Interventional studies that investigated the safety and/or efficacy of saffron, as the main remedy or a supplement, in patients with ADHD, with or without a control group, were included in this systematic review. Non-English papers, review articles, commentaries, letters, observational studies, thesis, animal studies, in-vitro studies, and conference abstracts, were not included.
Search
A systematic search was conducted in Medline via PubMed, Embase, Scopus, and Web of Science in November 2022, with the following strategy for PubMed: (Crocus [Mesh] OR Saffron OR Crocus) AND (attention deficit disorder with hyperactivity [Mesh] OR (Attention Deficit) OR ADHD OR Hyperkinetic OR (Minimal Brain Dysfunction) OR ADDH), with no limitations or filters. Also, for the comprehensive inclusion of the published studies, reference lists of the articles were checked.
Study Selection
The results of database searches were imported into EndNote software and after removing the duplicated results, two independent researchers (SS-S and AN) assessed the meeting eligibility criteria in two title/abstract and full-text stages. Disagreements in the study selection process were resolved through consultation or by referring to another author (SS or SF).
Data Collection
Data extraction was conducted using an electronic table in Microsoft Excel by two authors (SS-S and AN) which included the following parameters: the name of the first author of the study, the publication year, study design, setting of the study, inclusion and exclusion criteria, Interventions and the number of patients (Female /male), age (mean, SD), outcomes, and timings of assessing the outcomes and final conclusion.
Risk of Bias Assessment and Certainty of the Evidence
The second version of the Cochrane risk of bias tool (RoB.2) was chosen for the assessment of Randomized Controlled Trial (RCT) studies in five domains. The first domain evaluates the randomization process, the second domain assesses the deviations from intended interventions, the third domain apprises the missing outcome data, the fourth domain is for measurement of the outcome and the fifth domain assesses the selection of the reported results (Sterne et al., 2019). The assessment of the pre-post intervention study was done using The Risk of Bias in Non-randomized Studies—of Interventions (ROBINS-I) assessment tool. This tool assesses the bias due to confounding, the bias in the selection of participants into the study, bias in the classification of interventions, bias due to deviations from intended interventions, bias due to missing data, bias in the measurement of outcomes, bias in the selection of the reported result. In the end, an overall bias shows the risk of bias in the included studies (Sterne et al., 2016). Risk of bias assessments were conducted by two researchers (SS-S and AN) and disagreements were referred to a third researcher (SF or SS). In addition, the Consolidated Standards of Reporting Trials (CONSORT) 2010 checklist was used to assess the quality of reported information in included RCT studies (Schulz et al., 2010). Finally, the overall certainty of the evidence was evaluated using the GRADE approach. This evidence grading system categorized the confidence in the effect estimates as either “high,” “moderate,” “low,” or “very low” for each outcome. This categorization is based on judgment, but the process of making these judgments follows a transparent structure. (Schünemann et al., 2019).
Results
Search Results and Selection Process
The PRISMA flow diagram shows the details of the selection process (Figure 1). In summary, after removing duplications, 32 articles were assessed in the title/abstract stage, and finally, four articles met the inclusion criteria (Baziar et al., 2019; Blasco-Fontecilla et al., 2022; Khaksarian et al., 2021; Pazoki et al., 2022) and two review articles were excluded in the full-text stage (Ozsavci et al., 2019; Ross, 2020).
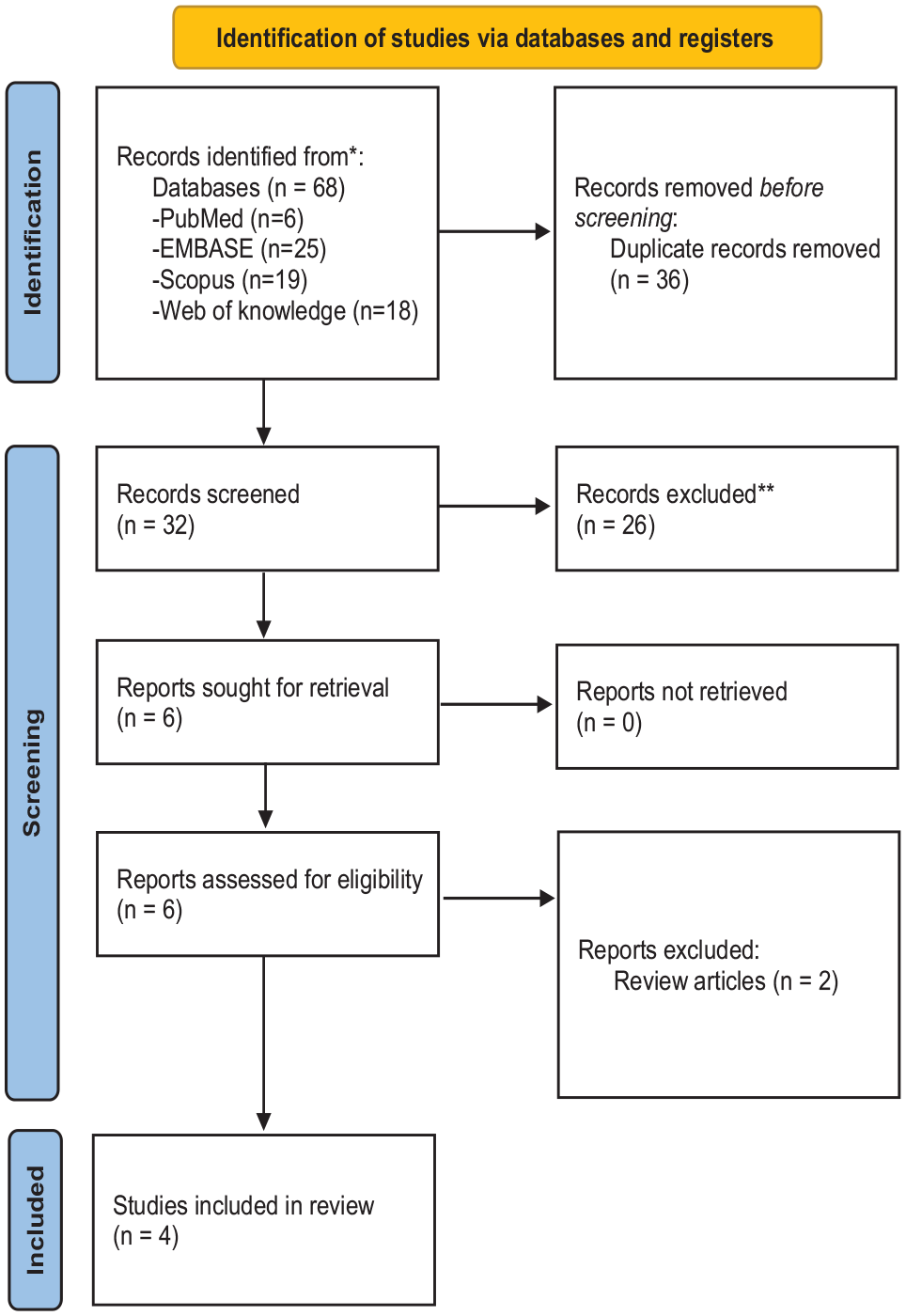
PRISMA 2020 flow diagram for new systematic reviews.
Characteristics of the Included Studies
Out of the four included studies, three were RCTs and the rest was a pre-post-intervention study. Three of the studies were conducted in Iran and the rest was done in Spain. The publication years were from 2019 to 2022. Participants were children and adolescents aged 6 to 16 years in three studies and adults aged 20 to 60 years in one study. The eligibility criteria of the participants in each study are summarized in Table 1. Two of the included studies assessed the efficacy of saffron in comparison with MPH and two studies evaluated the potency of saffron treatment combined with MPH in comparison with MPH alone.
The Characteristics and Findings in the Included Studies.

Note. MPH = Methylphenidate; ADHD-RS-IV = ADHD Rating Scale-IV; DSM-5 = Diagnostic and Statistical Manual of Mental Disorders-5; Hit RT = Hit Reaction time; HRT Block Change = Hit Reaction Time Block Change; HRT ISI = Hit Reaction Time Inter-Stimulus Interval; KSADS = Kiddie Schedule for Affective Disorders and Schizophrenia; ASRS = Adult ADHD Self-Report Scale; CAARS-RS = Adult ADHD Rating Scales-self report: screening version.
Based on ADHD-RS-IV.
Based on SNAP-IV
Based on CPT-3.
Quality Assessment, Risk of Bias, and Certainty of the Evidence
Supplemental Material 1 is the CONSORT checklists for the included RCTs and Figures 2 and 3 present the results of the risk of bias assessments. The most common source of bias in included RCTs was due to the selection of the reported result. Also, in the non-randomized intervention study, dealing with the selection of participants, deviations from intended interventions, outcome measuring, and selection of the reported results were the main sources of bias. Assessing certainty of the evidence indicates a high level of confidence in the results for two studies (Baziar et al., 2019; Khaksarian et al., 2021), while suggests a lower level of confidence for two others (Blasco-Fontecilla et al., 2022; Pazoki et al., 2022).
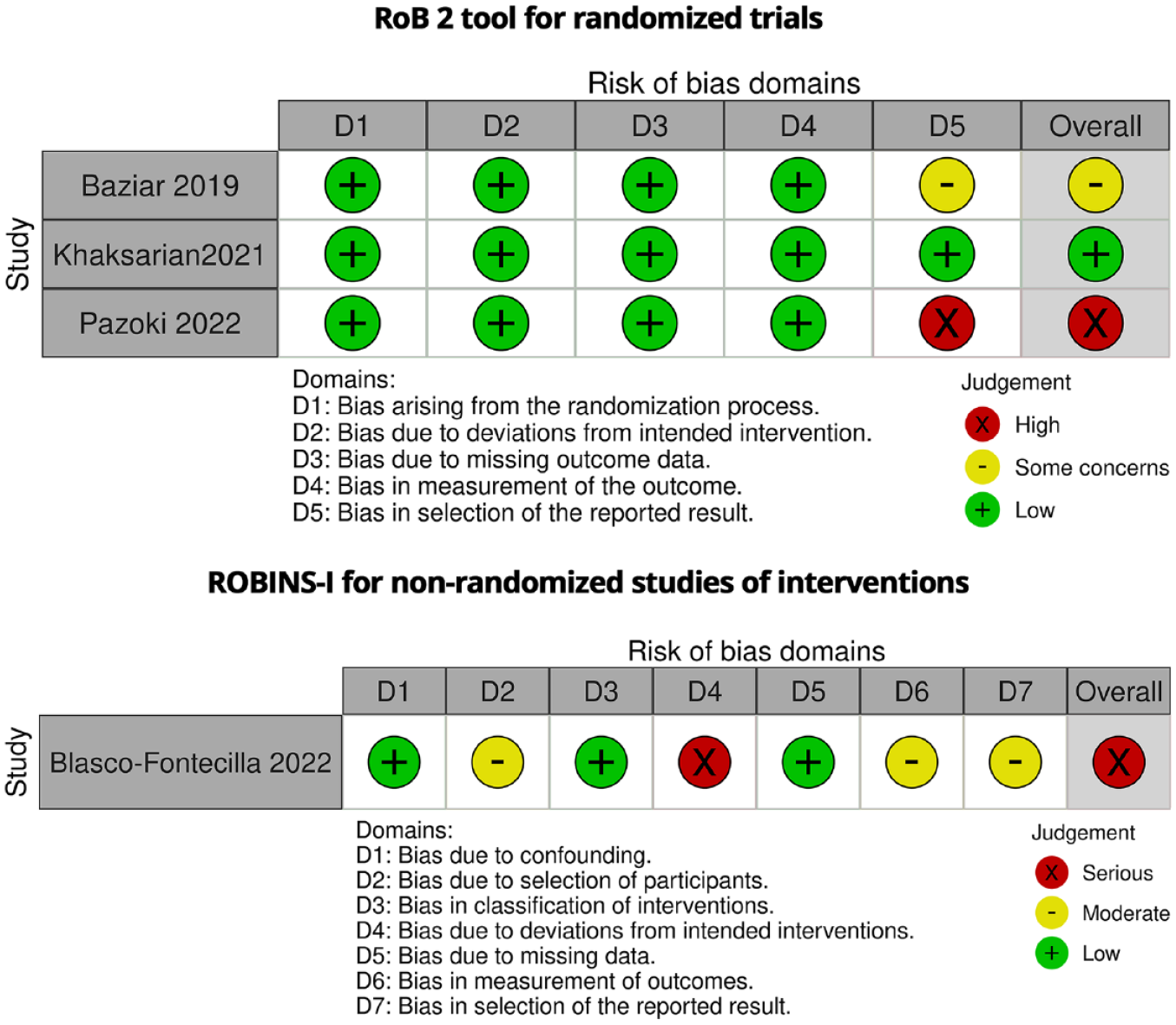
The details of risk of bias assessments in the included studies.
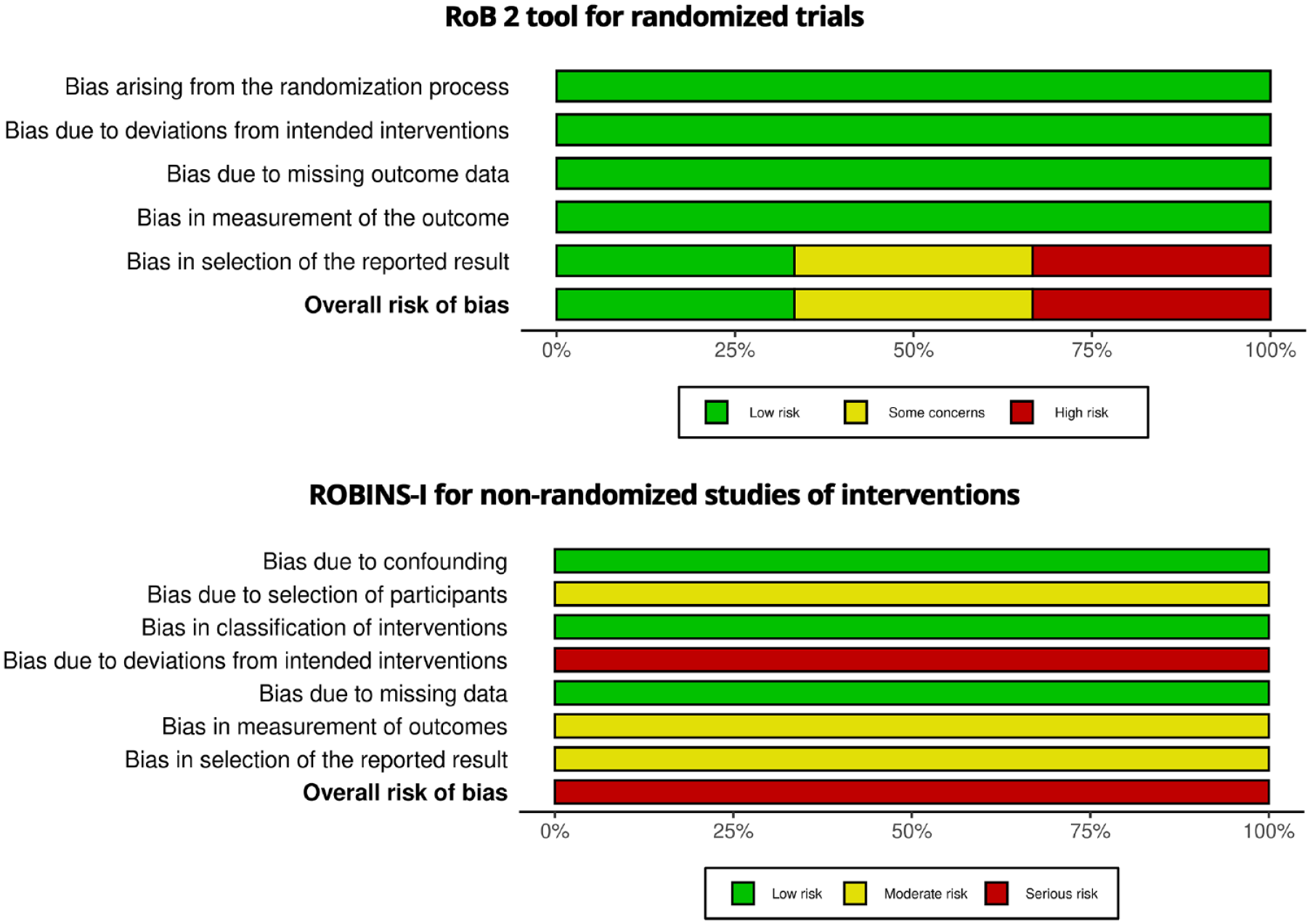
The overall risk of bias in the included studies.
Summary of Findings
Pazoki et al. evaluated the efficacy and safety of saffron as an adjuvant to MPH for improving symptoms in adults with ADHD. In this study, 56 patients, were assigned into two parallel groups and randomly received MPH (30 mg/day) plus placebo or MPH plus saffron (15 mg twice daily) for 6 weeks. The adult ADHD self-report scale change from baseline to the sixth week was significantly different between the saffron and placebo groups. Also, in terms of side effects (including insomnia, headache, abdominal pain, decreased appetite, nausea, itching, and cough), there was no significant difference between the groups (Pazoki et al., 2022).
Baziar et al. compared the efficacy and safety of saffron and MPH in children suffering from ADHD. For this purpose, 54 patients were randomly assigned to receive either 20 to 30 mg/day MPH or 20 to 30 mg/day saffron capsules (depending on weight) for 6 weeks. Teacher and Parent Attention-Deficit/Hyperactivity Disorder Rating Scale-IV (ADHD-RS-IV) was used to assess the symptoms in baseline, third week, and sixth week. This study demonstrated that changes in ADHD-RS-IV score from baseline were not significantly different between the saffron group and the MPH group. In addition, there was no significant difference in terms of the frequency of side effects (Baziar et al., 2019).
Khaksarian et al. evaluated the effectiveness of MPH and its combination with saffron. Seventy children were enrolled in their study and randomly assigned into two groups. Both groups received 20 or 30 mg/day of MPH and one of the groups also received 20 or 30 mg/day of saffron in a capsule based on body mass index. Symptoms of patients were evaluated using the ADHD-RS-IV at baseline and after 4 and 8 weeks of treatment. After 8 weeks, there was no crucial difference between the MPH and MPH plus saffron groups in terms of side effects. Besides, during 8 and 4 weeks of treatment, the symptoms of patients decreased meaningfully. In this study saffron, as a supplementation, was found to add beneficial effects to MPH in patients with ADHD (Khaksarian et al., 2021).
Blasco et al. compared the efficacy of saffron and MPH, using objective and pen-and-paper tests. In this non-randomized clinical trial, patients were divided into two groups. The MPH group (n = 27) received psychoeducation and extended-release MPH, and the saffron group (n = 36) received psychoeducation and saffron for 3 months. The severity of ADHD was measured using SNAP-IV and Conners’ Parent Rating Scale-Revised (CPRS-R) scales and the executive function was evaluated using the Behavioral Rating Inventory of Executive Function—Second Edition (BRIEF-2) test, and sleep quality was measured with the Sleep Disturbance Scale for Children (SDSC). The results showed that there was no significant difference in the SNAP-IV test, in terms of inattention and hyperactivity. Also, there was no difference between the two groups in terms of the CPRS-10 and BRIEF-2 scales (Blasco-Fontecilla et al., 2022).
Discussion
The present study evaluated the efficacy of saffron treatment in patients with ADHD. The results of four included studies manifested an efficient role of saffron as either an adjuvant therapy to MPH or single therapy. The obtained results demonstrated that the combination of MPH and saffron was found to be more effective. In addition, the prescription of saffron can reduce the duration of treatment to 4 weeks (Khaksarian et al., 2021) and saffron combination therapy with MPH could effectively improve the symptoms of adult patients with ADHD (Pazoki et al., 2022). Also, there is no significant difference between saffron and MPH in terms of efficacy and side effects in children and adolescents (Baziar et al., 2019) and even saffron tends to be more effective for hyperactivity improving the number of sleeping hours and making it easier to fall asleep (Blasco-Fontecilla et al., 2022).
There are many different and complicated mechanisms for the pathophysiology of ADHD such as reduced brain volume in the prefrontal cortex, cerebellum, and basal ganglia involvement (Fusar-Poli et al., 2012). Also, there is a failure in neurotransmitter network systems particularly in dopamine and norepinephrine pathways, which indicates the efficacy of MPH as a treatment for ADHD (Curatolo et al., 2009; Sharma & Couture, 2014). Studies show that the combination of medical and behavioral therapy is more effective for treating patients with ADHD (Group, 2004). MPH is the most common pharmacological choice for controlling ADHD symptoms with the mechanism of inhibiting dopamine transporters and increasing the dopamine level in the neuronal synapses (Solanto, 1998; Storebø et al., 2015). Despite the beneficial effect of MPH in increasing the quality of life in patients with ADHD, this medication has some side effects such as nausea, loss of appetite, and problems with sleeping. In addition, studies show that about 30% of ADHD children do not respond to MPH; thus, parents tend to give up their children’s treatment (Spencer et al., 1996). In recent years, non-simulating strategies have been used to manage ADHD symptoms. Among these strategies, antidepressant drugs seem to be effective in improving ADHD symptoms (Banaschewski et al., 2004; Salardini et al., 2016; Zarinara et al., 2010). One of the well-known herbal extracts with anti-depressant activities is Crocus sativus (Butnariu et al., 2022; Siddiqui et al., 2018).
People tend to use herbal medicine due to their cultural learning and confidence about their safety (Modabbernia & Akhondzadeh, 2013). Saffron is an extract obtained from the red stigma of Crocus sativus used as a spice and herbal medicine in many countries such as India, Spain, France, Greece, and especially Iran (Rios et al., 1996). Among 150 different components in saffron, crocetin, crocin (responsible for color), safranal (responsible for odor), and picrocrocin (responsible for taste) are the main components with medical effects (Schmidt et al., 2007). These components play a role by inhibiting dopamine and norepinephrine reuptake, being a GABA-α agonist and NMDA receptor antagonist (Alavizadeh & Hosseinzadeh, 2014; Sarris, 2007). Studies emerged pharmacological effects of saffron such as anti-inflammatory, antidepressant, and antitumor effects. Evidence has also proven the positive effects of this luxurious spice in disorders involving memory and learning like Alzheimer’s Disease (AD) (Akhondzadeh et al., 2010; Moshiri et al., 2015; Rasi Marzabadi et al., 2022; Sanaie et al., 2023). Moreover, animal studies have shown the potential effect of saffron and its products in decreasing locomotor hyperactivity, stereotypic behavioral and ataxia, ameliorating morphine-induced hyperactivity, reducing blood pressure, and enhancing learning and memory in addition to their anti-AD, anti-schizophrenia, anticonvulsant, antidepressant, anti-Parkinson, and neuroprotective effects (Asadpour & Sadeghnia, 2011; Khazdair et al., 2015; Sahraei et al., 2007). The effect of saffron on monoaminergic and glutamatergic systems is one of the clarifications of the usefulness of saffron therapy in combination with MPH for the treatment of ADHD (Pasini et al., 2007). Although the safety of saffron administration has been proven, there are some reports of adverse effects of saffron including bleeding, vomiting, diarrhea, vomiting, and nausea after a high-dose administration (Modaghegh et al., 2008; Schmidt et al., 2007).
To the best of our knowledge, this is the first comprehensive study to be published on the effects of saffron as a supplementation or main remedy for ADHD. This study had some limitations such as the low number of relevant studies, the heterogenicity in the age range of patients, the different assessment scales, and the variation of prescribing treatments in the included studies; therefore, we were not able to do a meta-analysis. In addition, three out of four included studies were conducted in one country (Iran), which may limit the generalizability of the results. According to the mentioned limitations, there is an essential need for more studies to ensure the effectiveness of saffron therapy in the treatment of ADHD.
Conclusion
This study manifested that saffron has an efficient role as either an adjuvant therapy to MPH or a single therapy for ADHD. The limitations of the evidence suggested future well-designed multicentral studies in different settings which can provide more reliable and evidence-based clinical recommendations in this regard.
Supplemental Material
sj-doc-1-jad-10.1177_10870547231203176 – Supplemental material for The Effects of Crocus sativus (Saffron) on ADHD: A Systematic Review
Supplemental material, sj-doc-1-jad-10.1177_10870547231203176 for The Effects of Crocus sativus (Saffron) on ADHD: A Systematic Review by Sepideh Seyedi-Sahebari, Sara Farhang, Mostafa Araj-Khodaei, Shahin Akhondzadeh, Amirreza Naseri, Sarvin Sanaie and Negin Frounchi in Journal of Attention Disorders
Footnotes
Acknowledgements
The research protocol was approved and supported by Student Research Committee, Tabriz University of Medical Sciences (grant number: 71144).
Author’s Note
Sara Farhang is also affiliated to Rob Giel Research Center, University Center for Psychiatry, University Medical Center Groningen, University of Groningen, Groningen, The Netherlands.
Abbreviations
AD: Alzheimer’s Disease; ADHD-RS-IV: ADHD Rating Scale-IV; BRIEF-2: Behavioral Rating Inventory of Executive Function—Second Edition; CONSORT: Consolidated Standards of Reporting Trials; CPRS-R: Conners’ Parent Rating Scale-Revised; GABA: gamma-aminobutyric acid; MPH: methylphenidate; NMDA: N-methyl-D-aspartic acid; PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-Analyses; RCT: Randomized Controlled Trial; ROBINS-I: Risk of Bias in Non-randomized Studies—of Interventions; SDSC: Sleep Disturbance Scale for Children
Author Contributions
S.S-S., S.F., S.S., A.N., and N.F.: systematic search; study selection; data extraction; risk of bias assessment; and preparing the figures, writing the first draft of the manuscript, and revision; S.F, M.A-K, S.A., and S.S.: conceptualization; supervision; and critically editing the manuscript. All authors approved the final version for submission.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The research was supported by Student Research Committee, Tabriz University of Medical Sciences (grant number: 71144).
Ethical Approval
Ethics code: IR.TBZMED.VCR.REC.1401.341.
Availability of Data and Materials
All data generated or analyzed during this study are included in this published article and its supplementary information files.
Supplemental Material
Supplemental material for this article is available online.
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
