Abstract
Objective
Children with attention-deficit/hyperactivity disorder (ADHD) show hypoactivity of the hypothalamic-pituitary-adrenal (HPA) axis. Whether the association between hyperactivity/inattention symptoms with HPA axis dysfunction holds in the general child population too is not clear.
Method
We assessed associations between longitudinal trajectories of hyperactivity/inattention symptoms during ages 4 to 13 years and basal cortisol profiles at age 15 in a British general population cohort.
Results
Adolescents with persistently high levels of hyperactivity/inattention symptoms since childhood showed lower total morning cortisol and a smaller diurnal decline, even after adjusting for confounders. No associations were found between any of the symptom trajectories and cortisol awakening response, diurnal slope or daily output of cortisol.
Conclusion
This study provides evidence for hypocortisolism among adolescents with chronic hyperactivity/inattention symptoms in the general population.
Introduction
Hyperactivity/inattention symptoms are common among children and adolescents (Smalley et al., 2007; Smidts & Oosterlaan, 2007; Warner-Rogers et al., 2000), ranging between 7% and 16% (Faraone et al., 2003). They have been shown to be associated with future adverse outcomes, such as depression (Humphreys et al., 2013), nicotine dependence (Pingault et al., 2013), and suicidal behaviors (Galéra et al., 2008). Research on children with attention-deficit/hyperactivity disorder (ADHD) suggests that one reason potentially underlying symptom onset might be an impaired hypothalamic-pituitary-adrenal (HPA) axis (Angeli et al., 2018; Blomqvist et al., 2007; Isaksson et al., 2012).
The HPA axis is one of the body’s critical stress–response systems, producing a cascade of hormones including cortisol. A natural diurnal cortisol rhythm is characterized by rapid rise after awakening and decline throughout the day (Vining et al., 1983). Deviations from the rhythm, such as hypercortisolism, hypocortisolism, or a flattened diurnal cortisol slope, are strong correlates of mental and physical health problems (Adam et al., 2017; McEwen, 2008). Empirical studies on individuals diagnosed with ADHD have found down-regulated basal HPA axis activity.
However, the relation between hyperactivity/inattention symptoms and HPA dysfunction in community samples is not clear (Hatzinger et al., 2007; Pesonen et al., 2011; Sondeijker et al., 2007). With the exception of two studies (Scerbo & Kolko, 1994; Susman et al., 2007), most research with community samples has found normal activity of the HPA axis (i.e., a normal diurnal cortisol rhythm) in children with hyperactivity/inattention symptoms (Klimes-Dougan et al., 2001; Pesonen et al., 2011; Saridjan et al., 2014). A large-scale study with over 1,700 children found no association between such symptoms and cortisol levels at wake-up or 30 minutes post-wake-up, but only a small association with higher evening salivary cortisol levels (Sondeijker et al., 2007). Another study (Hatzinger et al., 2007) reported higher mean morning cortisol levels in boys, but not girls, with such symptoms.
Three reasons may contribute to the inconsistency in findings from clinical and community samples. First, most studies using community samples used linear regressions which assume the relationship between hyperactivity/inattention symptoms and HPA axis function to be linear. However, there might be a threshold in the severity of symptoms, above which the HPA axis may show hypoactivity as found in clinical samples. Second, most studies using community samples were cross-sectional and thus unable to investigate potential effects of chronicity of hyperactivity/inattention symptoms on HPA axis function. Though symptoms of hyperactivity and inattention tend to decline over time, a significant number of children follow persistently high or increasing symptom trajectories (Arnold et al., 2014; Jester et al., 2008; Malone et al., 2010). Previous research suggests that compared to children with decreasing numbers of such symptoms over time, those with symptoms that persist through to adolescence are more likely to exhibit a blunted cortisol response to stressors (Campbell, 1994; King et al., 1998). It is therefore possible that only persistently high levels of hyperactivity may be related to impaired HPA axis activity. Finally, several community studies measured cortisol using a single measure, thus neglecting to capture diurnal cortisol activity (Scerbo & Kolko, 1994). Comprehensive rhythm parameters of cortisol are needed to thoroughly investigate HPA axis function.
The aim of the present study was to explore for the first time the role of chronicity and severity of hyperactivity/inattention symptoms across childhood in HPA axis function in adolescence in a British general-population cohort. We used growth mixture models to identify classes with distinct developmental trajectories of hyperactivity/inattention symptoms and we captured HPA axis function by diurnal cortisol output. We hypothesized that persistently high level of such symptoms across childhood and adolescence would be associated with hypoactivity of the HPA axis (hypocortisolism, i.e., lower cortisol awakening response [CAR], lower cortisol levels throughout the day, and lower total daily output as well as flatter diurnal cortisol slope).
Method
Participants
The Avon Longitudinal Study of Parents and Children (ALSPAC) is an ongoing transgenerational longitudinal cohort study (Boyd et al., 2013) that enrolled 14,541 pregnant women with expected delivery dates between 1 April 1991 and 31 December 1992 (for details of all the data that is available http://www.bristol.ac.uk/alspac/researchers/our-data/). Children were invited to attend annual assessment clinics, including face-to-face interviews and psychological and physical tests from age 7 years onwards (Fraser et al., 2013). Additional children were recruited using the original enrolment definition from the participating children’s age 7 years onwards, increasing the number to 15,454 pregnancies, resulting in 15,589 fetuses to date. Of these, 14,901 were alive at 1 year of age. Ethical approval for all measures was gained from the ALSPAC Ethics and Law Committee and the Local Research Ethics Committees. All participants provided written informed consent and there was no financial compensation (details at www.alspac.bris.ac.uk).
At age 15, 5,501 of the ALSPAC cohort children were recruited for a clinical assessment. Of those, 3,020 were invited at random to participate in the cortisol data collection and 1,845 agreed. For 968 participants who provided at least one valid saliva sample, we applied the following exclusion criteria: (1) second born in case of a twin birth; (2) gestation at birth ≤32 weeks; (3) birth weight ≤1500 g; and (4) current exposure to steroid medication. We further excluded 10 participants due to the lack of hyperactivity/inattention data, resulting in a total sample of 939 individuals (Figure 1).
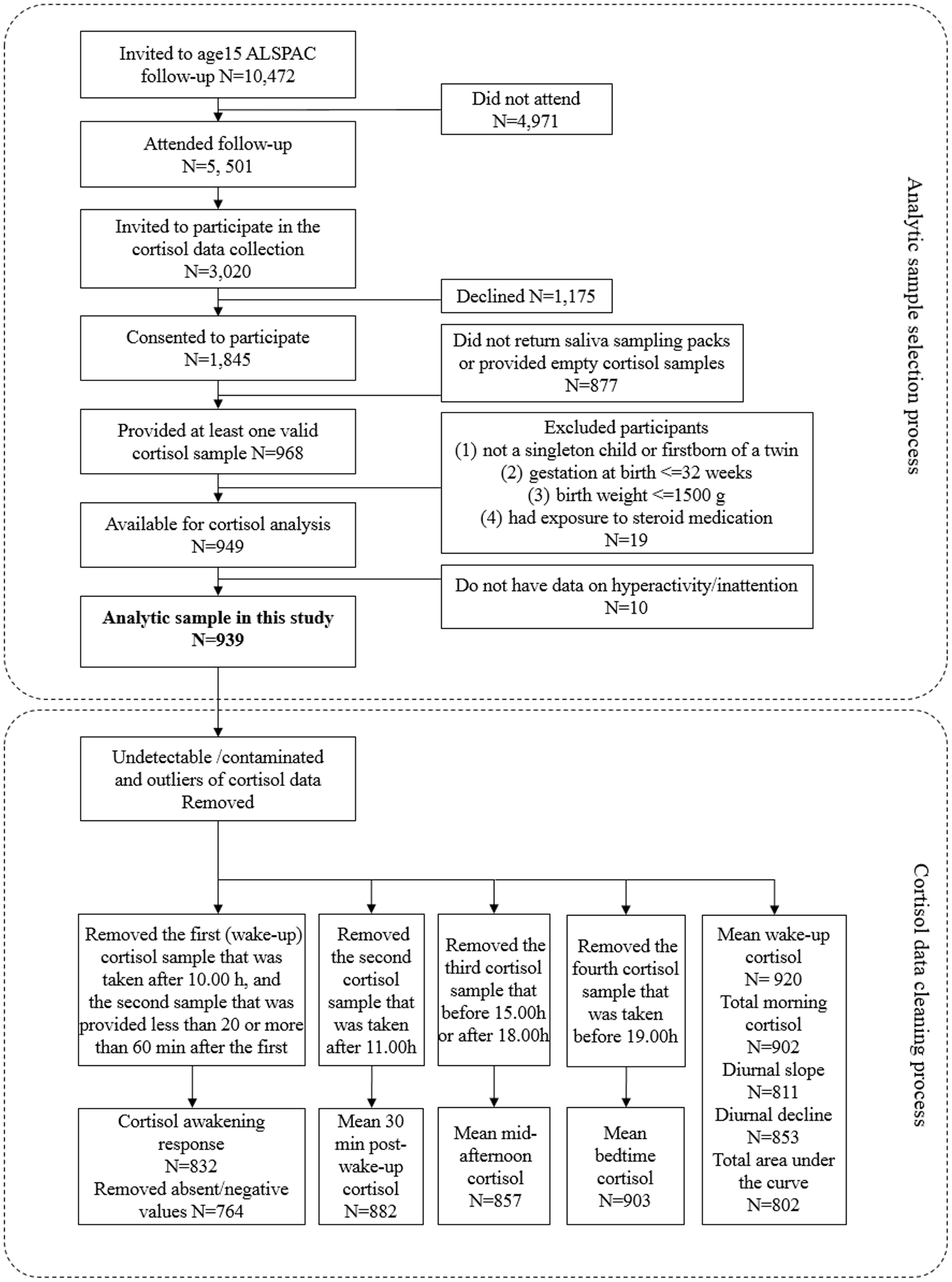
Flowchart of sample selection process.
Cortisol Sampling
As described by O’Donnell et al. (2013) and Carnegie et al. (2014), each participant was given a cortisol sampling pack during their clinic visit. Each pack contained detailed sampling instructions, 12 Salivate collection devices (Sarstedt, Germany), a sample collection sheet and a prepaid envelope in which to return the samples to the laboratory. For each participant, four measurements were taken daily over three consecutive school days: wake-up, 30 minutes post-wake-up, mid-afternoon, and before bedtime. Participants were shown how to collect saliva and were instructed to avoid eating or brushing their teeth before the collection of each sample, to record collection times, and to place samples in their freezers until they returned the samples to the laboratory. Samples were mailed back and stored at −20°C until they were assayed. Cortisol samples, expressed as nmol/L, were assayed using a commercially available enzyme immunoassay (Salimetrics, UK). Inter-assay and intra-assay coefficients of variation were 7.9% and 8.9%, respectively (O’Donnell et al., 2013).
Cortisol data were treated as missing (Figure 1) if: (1) values were undetectable; (2) values > 82 nmol/L; (3) values >4 SD above the sample mean for that time point; (4) for CAR, the first sample was taken after 10.00 hours, or the second sample was provided less than 20 or more than 60 minutes after the first; (5) for mean cortisol level, the second time point was after 11.00 hours; (6) for mean cortisol level, the third time point was before 15.00 hours or after 18.00 hours; (7) for mean cortisol level, the fourth time point was before 19.00 hours. Following previous practice (Carnegie et al., 2014), we replaced absent or negative CAR as missing, in case this was the result of sampling error or non-adherence (Kudielka et al., 2003).
We derived five between-person parameters for cortisol: (1) diurnal cortisol decline, taken as the difference between mean morning and bedtime cortisol levels; (2) diurnal cortisol slope, calculated using diurnal cortisol decline divided by time in hours; (3) CAR, calculated by subtracting the wake-up cortisol from the post-awakening cortisol; (4) total morning cortisol output, estimated as the sum of the two morning samples; (5) total daily cortisol output, derived from the area under the curve with respect to ground (AUCg) of four time points based on the trapezoid formula (Pruessner et al., 2003). Each cortisol variable was calculated as a mean of the available samples over the study period. The mean cortisol concentration at each time point was also computed and analyzed separately.
Hyperactivity/Inattention Symptoms
These symptoms were measured with the hyperactivity/inattention symptoms subscale of the mother-rated Strengths and Difficulties Questionnaire (SDQ; Goodman, 1997) at ages 4, 7, 8, 9, 11, and 13 years. The SDQ is a behavioral screening questionnaire with sound psychometric properties (Goodman et al., 2010). The subscale assesses hyperactivity/inattention using five items, scored 0 to 2 (“not true,” “somewhat true,” and “certainly true”). Questions include three items assessing hyperactive-impulsive behaviors (“Restless, overactive. Cannot stay still for long,” “Constantly fidgeting or squirming,” “Thinks things out before acting”) and two questions measuring symptoms of inattention (“Easily distracted, concentration wanders,” “Sees tasks through to the end. Good attention span”). Symptom scores showed moderate temporal stability across the years (0.44 < Spearman’s Rho < .73; Supplemental Table S1).
Covariates
We controlled for a number of covariates which are known to be associated with children’s hyperactivity/inattention symptoms and HPA axis activity. These included sex (Goel et al., 2014; Hermens et al., 2005), ethnicity (White, non-White) (Bax et al., 2019; DeSantis et al., 2007), socioeconomic disadvantage (Marsman et al., 2012; Russell et al., 2016), and stressful life events (Elzinga et al., 2008; Vrijsen et al., 2018). Socioeconomic disadvantage was derived from a principal components analysis of maternal education (below O-level, O-level only, A-level only, university degree, or more), social class at 32 weeks of pregnancy (I, II, III—non-manual, III—manual, IV, V), and the mean score of financial difficulties at 8, 21 and 33 months (a sum score of dichotomous items about being unable to afford the cost of food, clothes, heating, housing, and things for the baby, ranging 0 to 20 for each time point). Stressful life events were assessed as the sum of events at child’s age 8, 21, 33, 47, 61, 73, 110, and 134 months. At each time-point, a stressful events checklist with 43 dichotomous items was completed by the mother (Flouri et al., 2020).
Additional covariates measured at participants’ age 15 years included exact age (Kudielka & Kirschbaum, 2003), waking time (the mean during the study period) (Dahlgren et al., 2009; Stalder et al., 2009), overweight (“yes,” “no”); (Yu et al., 2020), vigorous physical activity (such as running, football, swimming, athletics; “less than once a week,” “1–3 times a week,” “more than three times a week”) (Anderson & Wideman, 2017), daily smoker (“yes,” “no”) (Eiden et al., 2020), contraceptive and psychotropic medication use (“yes,” “no”) (O’Donnell et al., 2013), and alcohol use (drinking on more than 2 days a week) (Ruttle et al., 2015). Overweight was defined using the International Obesity Task Force (IOTF; Cole et al., 2000) age- and sex-specific cut-offs for body mass index (males: 23.29 kg/m2; females: 23.94 kg/m2 at age 15 years). As cognitive function has been linked to both basal responsive cortisol levels (Lupien et al., 2001) and inattention/hyperactivity symptoms (Polderman et al., 2009), intelligence quotient (IQ) at age 15 years was also included as a covariate. It was assessed using the Wechsler abbreviated scale of intelligence (Wechsler et al., 1992). We also adjusted for presence of ADHD, oppositional defiant disorder (ODD) or conduct disorder (CD) at age 15, ascertained using the parent version of the Development and Well-being Assessment (DAWBA, Goodman et al., 2000). Answers to the questions of the DAWBA (e.g., “Over the last 6 months, compared with other children of his/her own age, does s/he often not finish a job properly?”) were coded according to ICD-10 and DSM-IV criteria by two experienced psychiatrists to generate diagnoses of CD, ODD, or ADHD. Symptoms of ODD and ADHD were reported for the previous 6 months and symptoms of CD for the year preceding the assessment. DAWBA is a validated instrument with good discriminant ability for behavioral disorders in both community and clinical samples (Goodman et al., 2000). As at the time of measurement of cortisol none of the individuals in the sample were pre-pubertal (taken as a Tanner stage < 2), pubertal stage was not included as a covariate. In analyses involving CAR only, we additionally controlled for wake-up cortisol levels, as it is frequently reported that high levels of salivary cortisol at wake-up are associated with an attenuated CAR (Adam et al., 2006).
Statistical Analysis
To correct for skewed distributions, the values of cortisol measures were log-transformed in all analyses. The statistical analysis was carried out in two parts. First, we fitted growth mixture models (GMMs) to identify classes with distinct developmental trajectories of hyperactivity/inattention symptoms from ages 4 to 13 years in the sample. 1 A Robust maximum likelihood estimator (MLR) was used to account for skewed data distributions in hyperactivity/inattention scores and the covariates. Full information maximum likelihood (FIML) was used to account for missing data in hyperactivity/inattention scores and covariates. The optimal number of classes was determined using the following fit indices (Jung & Wickrama, 2008): (a) The Bayesian information criterion (BIC), the sample size adjusted BIC (SSA-BIC) and the Akaike information criterion (AIC). Lower values in all three indices indicate better fit; (b) the Vuong-Lo-Medell-Rubin likelihood ratio test (LRT), which compares a model with K classes to a model with (K–1) classes. Significant p-values (<.05) indicate significantly better model fit compared to the more parsimonious model; and (c) the entropy of the model, which evaluates the accuracy of model classification, with values closer to 1 indicating less uncertainty in class allocation. 1 Upon selection of the best-fitting solution, participants were assigned to the class for which they had the highest posterior probability of belonging. We tested for differences in sociodemographic characteristics and cortisol measures between classes using analyses of variance and chi-square tests. Next, we computed the average predicted levels (marginal means) of cortisol based on a linear regression model using as predictor the interaction between sampling time and class membership. By doing so we were able to graphically illustrate the diurnal cortisol profiles of adolescents across classes and to compare predicted mean cortisol levels across classes at the four time points.
In the second part of the analysis, 2 multiple linear regression models were fitted to investigate the predictive effect of class membership on CAR, total morning cortisol, diurnal cortisol decline/slope, and AUCg. For each cortisol measure, unadjusted regression models were initially run followed by models adjusted for confounders (sex, ethnicity, overweight, socioeconomic disadvantage, age, smoking, alcohol use, medication use, vigorous physical activity, wake-up time, IQ, stressful life events, and behavioral disorders). We weighted all regression models using the conditional probabilities of individuals’ group membership, thereby giving more weight to participants with higher certainty of class assignment. All analyses were conducted in Mplus 8.1 (Muthén & Muthén, 1998–2017) and Stata 16 (StataCorp, 2019).
Results
Identification of Developmental Trajectories of Hyperactivity/Inattention Symptoms
GMM was conducted on symptom data from 939 children with available data in at least one assessment at ages 4, 7, 8, 9, 11, and 13 years. Starting with a one-class model, stepwise addition of classes resulted in lower BIC, SSA-BIC, and AIC values, suggesting a better model fit to the data for higher-class solutions (Table 1). However, the 4- and 5-class solutions had non-significant LRT p-values while changes in BIC values became negligible compared to the 3-class model. Moreover, the 3-class solution had the highest entropy value of the five competing models, indicating the least ambiguity in class assignment. The 3-class solution was therefore selected for further testing. Based on the developmental patterns of hyperactivity/inattention problems from 4 to 13 years observed in the classes we termed them as follows. (a) “Low and decreasing” (71.0% of the sample). Children in this class showed decreasing number of symptoms with a low score at baseline (intercept = 3.04, p < .01; slope = −0.18, p < .01). (b) “Intermediate” (24.5% of the sample). Children in this class had a moderate and stable number of symptoms throughout the study period (intercept = 4.81, p < .01; slope = −0.04, p = .20). (c) “High” (4.5%). Children in this class were characterized by high levels of symptoms throughout the study period (intercept = 7.17, p < .01; slope = 0.10, p = .12). The trajectories of symptoms characterizing each of the three classes are illustrated in Figure S1.
Fit indices of 1- to 5-Class Solutions of Growth Mixture Models Examining the Developmental Trajectories of Hyperactivity/Inattention From Ages 4 to 13 years.
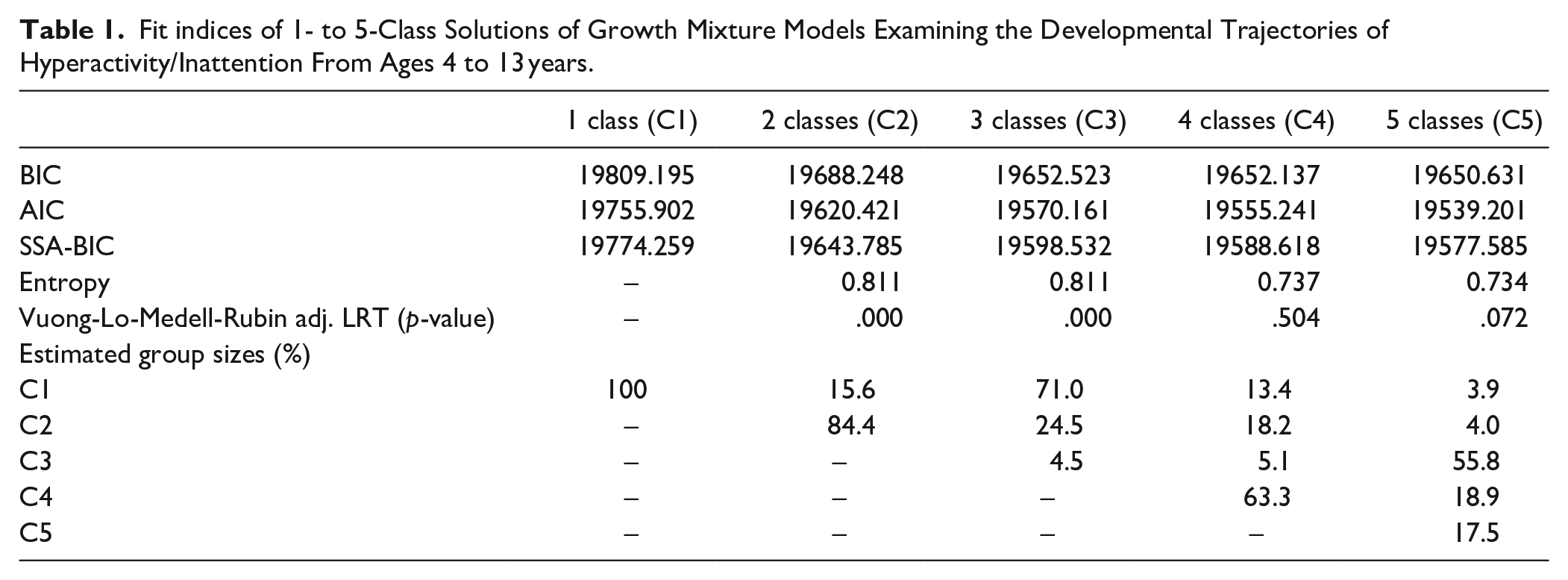
Table 2 summarizes the characteristics of participants and all cortisol measures by class membership. Compared to the “low and decreasing” class, participants assigned to the “intermediate” class were more likely to be male (p < .01), regular smokers (p = .024), in socioeconomic disadvantage, and to have lower IQ and more adverse life events in childhood (all p < .01). Participants allocated in the “high” group were also more likely to be male (p < .01), have lower average IQ (p < .01) and higher rates of behavioral disorders (i.e., ODD, CD or ADHD; p < .01), compared to the “low and decreasing” class. There were no significant differences in age, ethnicity, obesity rates or any of the other lifestyle factors considered (i.e., vigorous physical activity and alcohol use) at age 15 years between classes. The four daily samples displayed the typical diurnal cortisol rhythm, that is, the cortisol level was highest at 30 minutes post-wake-up, and declined over the day. The estimated marginal means of the “high” group was 0.334 log(nmol/L) lower compared to the “low and decreasing” group (p = .02) at wake-up, whereas cortisol levels at the remaining three time points did not differ between classes (Table 3). Figure 2 illustrates the predicted diurnal cortisol rhythm for the three classes.
Characteristics of Participants by Class Membership of Hyperactivity/Inattention Symptom Trajectories.
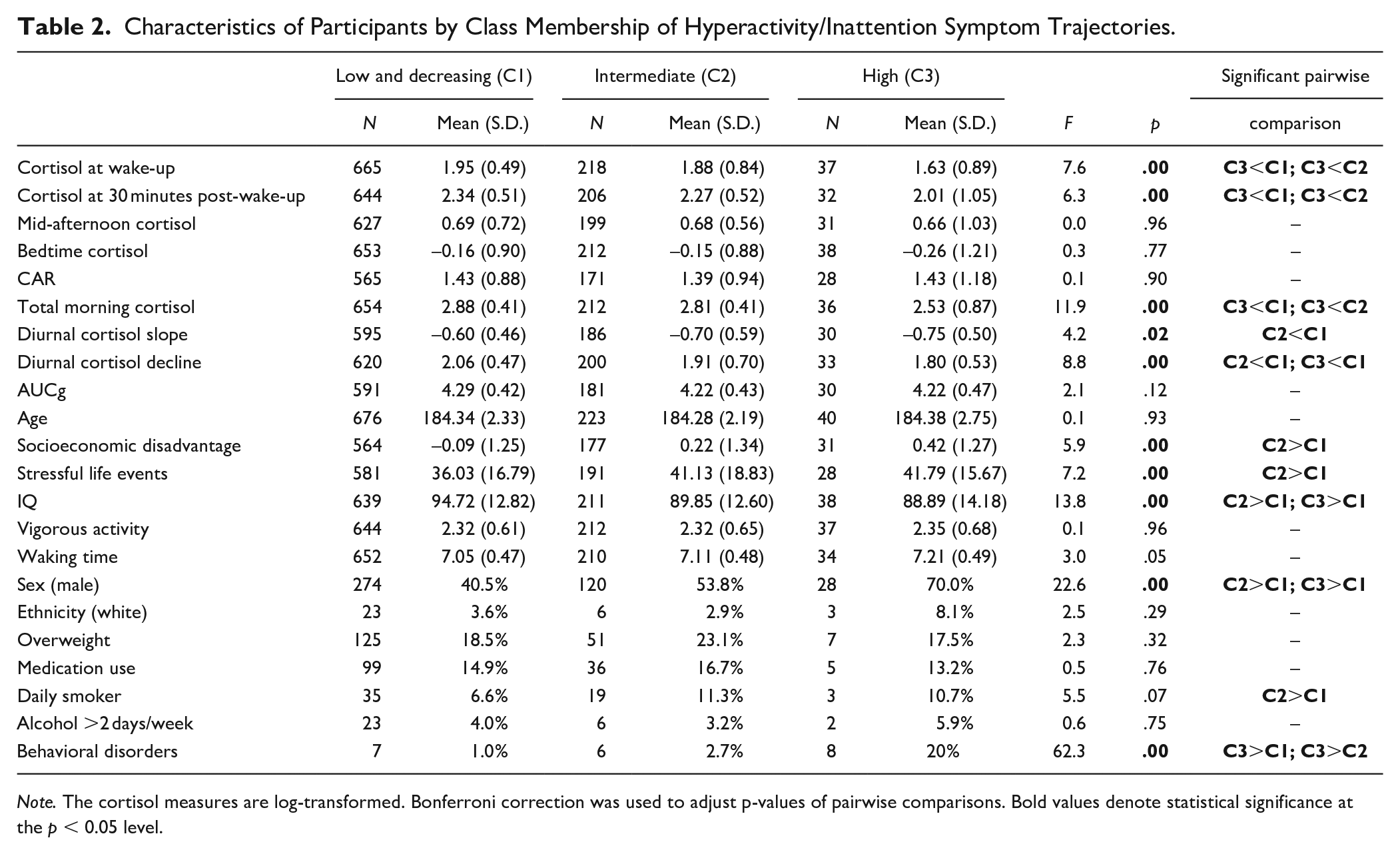
Note. The cortisol measures are log-transformed. Bonferroni correction was used to adjust p-values of pairwise comparisons. Bold values denote statistical significance at the p < 0.05 level.
Predicted Mean Differences of Cortisol Levels Across Classes at Four Time Points (Weighted by the Conditional Probabilities of Individuals’ Group Membership).
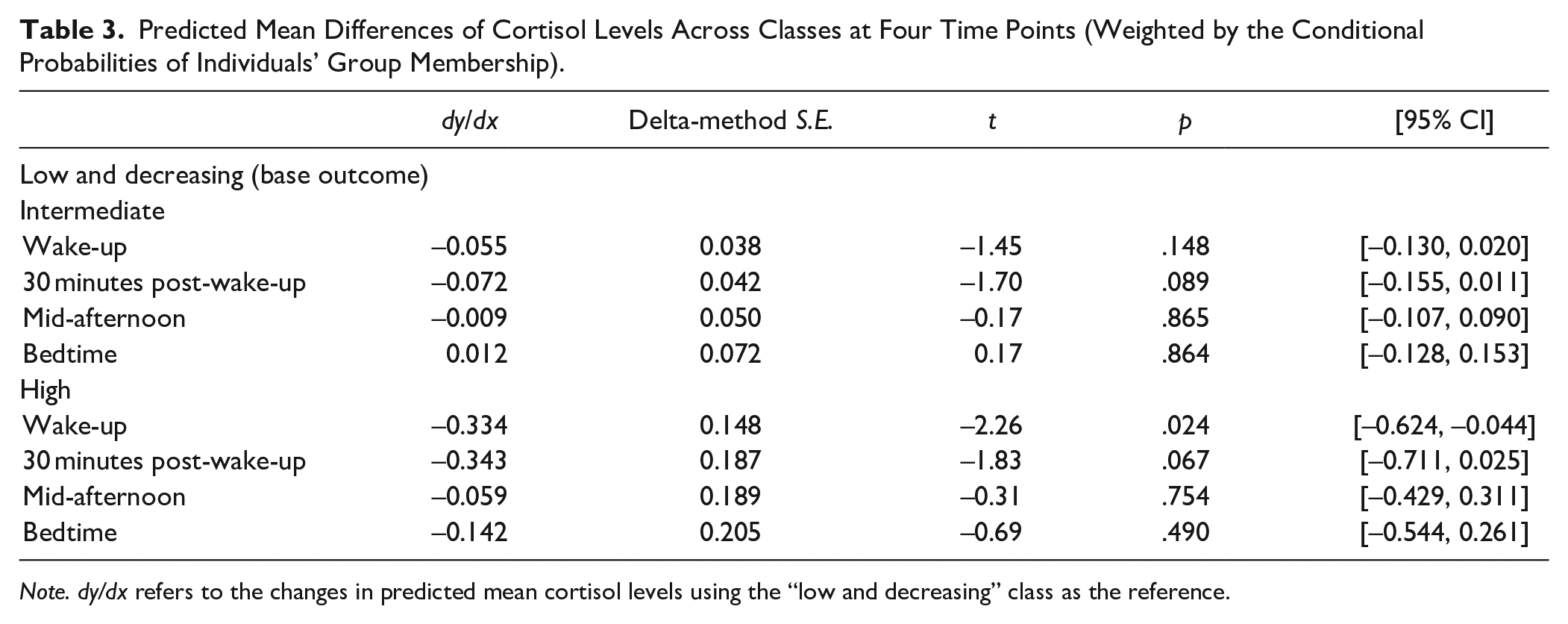
Note. dy/dx refers to the changes in predicted mean cortisol levels using the “low and decreasing” class as the reference.
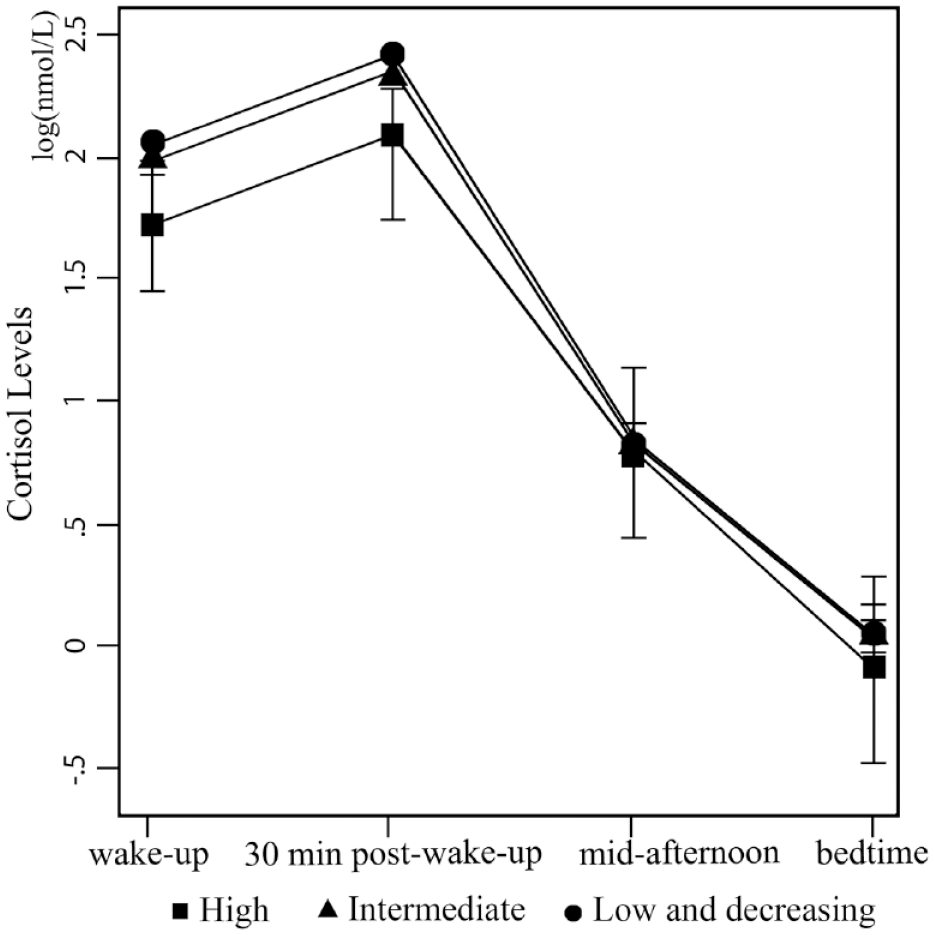
Predicted diurnal cortisol rhythm across classes (95% CI).
Predictive Value of Class Membership
The results of linear regression models for the cortisol parameters are shown in Table 4. Compared to the “low and decreasing” class, total morning cortisol was found to be significantly lower among the adolescents in the “high” group in the unadjusted model (b = −0.37, p = .01) but also after adjusting for covariates (Model A: b = −0.30, p = .03; Model B: b = −0.31, p = .03; and Model C: b = −0.32, p = .03). Compared to the “low and decreasing” class, diurnal cortisol decline was shown to be significantly larger among adolescents in the “high” (Model C: b = −0.21, p = .03) and “intermediate” classes (Model C: b = −0.11, p = .03) after adjustments for confounding. There was no difference of diurnal cortisol slope between adolescents in the “high” or “low and decreasing” classes. The diurnal cortisol slope of adolescents in the “intermediate” class was only found to be significantly flatter than that in the “low and decreasing” class in the unadjusted model (b = −0.10, p = .04), but this finding did not survive adjustments for confounding. There was no evidence for an association of CAR or AUCg with class membership. As sex was a significant predictor in all fully adjusted models, we explored if the associations of class membership with cortisol measures varied by sex. Of all interaction terms examined, only the interaction between sex and “intermediate” class was a significant predictor of AUCg, indicating that males in the intermediate group had more total daily output of cortisol than females in this group. The results of the remaining regression analyses examining the effects of the interaction terms between sex and class membership on all cortisol measures are summarized in Table S3 (Supplemental Material).
Unadjusted and Adjusted Coefficients (SE) of Regression Models Examining the Association of Class Membership with Cortisol Measures.
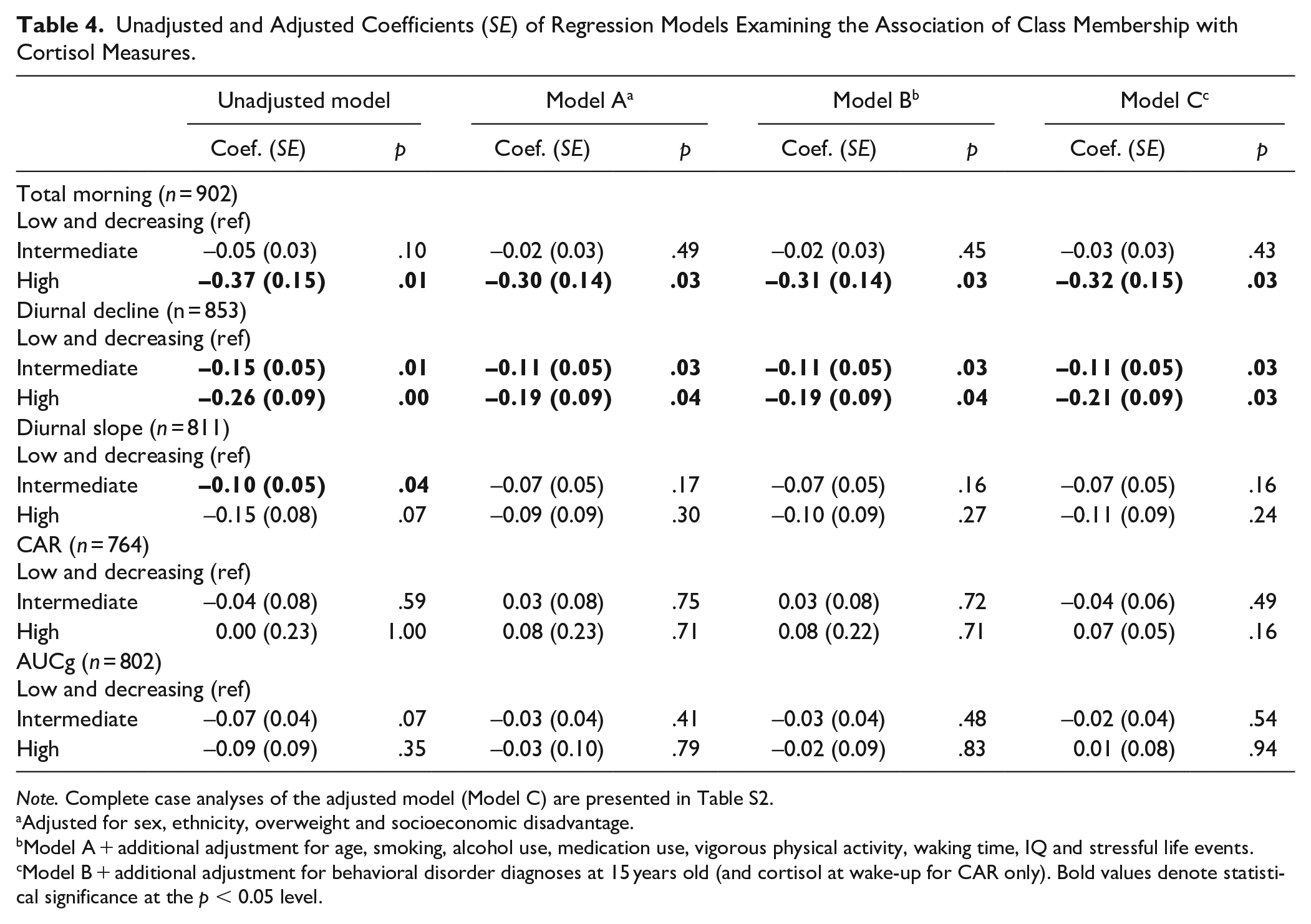
Note. Complete case analyses of the adjusted model (Model C) are presented in Table S2.
Adjusted for sex, ethnicity, overweight and socioeconomic disadvantage.
Model A + additional adjustment for age, smoking, alcohol use, medication use, vigorous physical activity, waking time, IQ and stressful life events.
Model B + additional adjustment for behavioral disorder diagnoses at 15 years old (and cortisol at wake-up for CAR only). Bold values denote statistical significance at the p < 0.05 level.
Discussion
We investigated in a large prospective general-population cohort the association between the course of hyperactivity/inattention symptoms since the preschool years and HPA axis function in adolescence. We identified three distinct hyperactivity/inattention trajectory classes (low and decreasing, intermediate, high) in line with previous research (Sasser et al., 2016). We then tested the associations between class membership and diurnal cortisol output, a robust indicator of basal HPA axis activity. Our findings partially support the hypothesis that chronically high levels of hyperactivity/inattention symptoms relate to hypoactivity of the HPA axis. We found that children with high levels of symptoms during childhood were characterized by a lower total morning cortisol level and a smaller diurnal cortisol decline by mid-adolescence compared to those with low levels of symptoms. These findings suggests that hypocortisolism might be a key characteristic of adolescents with a history of significant hyperactivity/inattention. However, we did not find robust links between any of the atypical developmental trajectories of hyperactivity/inattention symptoms and other cortisol parameters at 15 years old, including CAR, diurnal cortisol slope, and AUCg.
As we expected, only adolescents with high, but not intermediate, levels of hyperactivity/inattention symptoms since early childhood showed lower morning cortisol, compared to those with low and decreasing levels. This finding is consistent with previous evidence of attenuated morning cortisol profiles in adolescents diagnosed with ADHD (Angeli et al., 2018; El-Moez et al., 2016; Fortier et al., 2013; Imeraj et al., 2012; Isaksson et al., 2012). Hypocortisolism in the general child population can be considered a marker of allostatic load. A low, flat diurnal rhythm is indicative of chronic mild (e.g., following exposure to adverse life events) or high stress (e.g., post-traumatic stress or burnout) (Anda et al., 2006; Miller et al., 2007). Compared to their typically developing peers, children with hyperactivity/inattention tend to experience more adverse events or accidents, as they tend to take more risks and seek novelty, resulting in higher exposure to chronic stressors (Brown et al., 2017).
We also found evidence for smaller diurnal cortisol declines in the classes of adolescents with high and intermediate levels of hyperactivity/inattention symptoms after adjustment for cofounders. It is likely that this decline was driven by the lower morning cortisol levels seen in these two groups as there were no differences in afternoon or evening cortisol levels between classes. However, we found no evidence for an association between the different trajectories of hyperactivity/inattention and diurnal cortisol slope which is a more robust measure of basal cortisol activity since it is adjusted for differences in the total awake time (Adam & Kumari, 2009). The association between diurnal cortisol slope and symptoms disappeared when we controlled for sex. Hyperactivity/inattention symptoms are more common in males than females, and there are pronounced sex differences in diurnal cortisol profiles in the general population (Barkley, 2003). Hence, this finding might be an artifact of the higher proportion of males in the high and intermediate symptom groups. However, there was no moderating effect of sex on the association between hyperactivity/inattention and diurnal slope. Taken together, these results suggest that even if there is a true difference in diurnal cortisol change between adolescents with hyperactivity/inattention symptoms and their typically developing peers, it is very small and markedly attenuated by confounders.
CAR did not differ between classes; thus, we did not find evidence in support of the hypothesis that adolescents with persistently high levels of symptoms are characterized by smaller CAR (Angeli et al., 2018). CAR does not represent total daily cortisol exposure or diurnal cortisol rhythm and captures a different aspect of HPA axis function (Golden et al., 2013). CAR measures a response to awakening rather than the circadian increase in HPA axis activity during the early morning hours (Wilhelm et al., 2007). Therefore, our finding may indicate that chronic hyperactivity/inattention symptoms do not impair one’s ability to react to a natural stimulus (i.e., awakening).
The finding that adolescents with persistently high levels of hyperactivity/inattention symptoms have low morning cortisol levels raises clinical possibilities. First, low morning cortisol concentration may be a biological marker for chronic and significant hyperactivity/inattention and could improve the accuracy of targeting adolescents with constantly high levels of such symptoms. Second, low morning cortisol concentration may partly explain the poor adult health of those with hyperactivity/inattention symptoms in adolescence (Agnew-Blais et al., 2018; Brook et al., 2013) and their increased risk for inflammation, autoimmune diseases and atopic illnesses (Buske-Kirschbaum et al., 1997; Heim et al., 2000). Nonetheless, we note that the effect size found for total morning cortisol was small in magnitude.
Our study has a number of strengths. First, this was the largest prospective study to date to investigate HPA axis function in adolescents with different types of hyperactivity/inattention symptom profiles since the preschool period. Second, we controlled for a wealth of confounders, including behavioral disorders, measures of lifestyle and psychosocial risk, and current indicators of adolescent health and behaviors. Third, we used a four-sample protocol for cortisol assessment, which was more sensitive and enabled us to capture diurnal cortisol rhythm, compared to a “minimum” two-sample protocol which would include using only one sample in the morning and one at bedtime to estimate diurnal cortisol slopes (Adam & Kumari, 2009). Nevertheless, it would be preferable to measure CAR using samples from more than two time points. For example, females may peak later than males and show a more delayed decrease in cortisol (Saxbe, 2008), thus the assessments at a fixed time-point (30 minutes post-wake-up) across the sample might not provide sensitive enough measurements.
This study also comes with certain limitations. First, the assessment of the HPA axis function was limited to basal salivary cortisol. Salivary cortisol alone may be too crude a measure for prospective prediction and a more comprehensive approach may be required. Other aspects of HPA axis function, such as the upstream release of CRH and ACTH, were not directly assessed. Cortisol levels may look identical in participants who have different levels of glucocorticoid sensitivity, cortisol-evoked genomic activity, and glucocorticoid receptor expression (Saxbe, 2008). Second, we did not explore hyperactivity/inattention symptom subtype differences in basal cortisol. For example, in adults with ADHD, inattentive and combined subtypes show differences in the association with cortisol levels in the morning (Ramos-Quiroga et al., 2016). Our five-item scale capturing both types did not allow us to explore symptom specificity in detail. Third, symptoms were reported by mothers, which may raise concerns about potential reporter bias. Fourth, though this study included a large and socioeconomically diverse sample, it did not vary widely across other demographic characteristics, such as ethnicity. Thus, the external validity of the study might be somewhat compromised. Finally, the absence of longitudinal data on cortisol does not allow having a clear picture of the link between hyperactivity/inattention and long-term HPA axis habituation, which may be particularly relevant in the general population. Given that a stronger HPA axis response to acute stress is related to steeper CAR and steeper diurnal cortisol decline (Chen et al., 2017), while chronic stress is related to relative glucocorticoid resistance (Rohleder, 2012), it would be useful to track within-individual change in HPA axis response to stress in children and adolescents with high levels of hyperactivity/inattention symptoms, a group widely considered at risk for increased exposure to stressors. This group may show initially adaptive responses to stress which then become maladaptive as stress becomes chronic.
Conclusion
We observed lower morning cortisol in adolescents with persistently high levels of hyperactivity/inattention symptoms since early childhood compared to those with low levels of such symptoms. This finding is in line with evidence suggesting that adolescents with chronic hyperactivity/inattention symptoms may be characterized by hypoactivity of the HPA axis. We did not find abnormal CAR, diurnal cortisol slope or daily output of cortisol in these adolescents. The results suggest that low morning cortisol may be a biomarker for chronic hyperactivity/inattention problems in community samples of youth.
Supplemental Material
sj-pdf-1-jad-10.1177_10870547211036755 – Supplemental material for Childhood Trajectories of Hyperactivity/Inattention Symptoms and Diurnal Cortisol in Middle Adolescence: Results from a UK Birth Cohort
Supplemental material, sj-pdf-1-jad-10.1177_10870547211036755 for Childhood Trajectories of Hyperactivity/Inattention Symptoms and Diurnal Cortisol in Middle Adolescence: Results from a UK Birth Cohort by Dongying Ji, Eirini Flouri, Efstathios Papachristou and Marta Francesconi in Journal of Attention Disorders
Footnotes
Acknowledgements
We are extremely grateful to all the families who took part in this study, the midwives for their help in recruiting them, and the whole ALSPAC team, including interviewers, computer and laboratory technicians, clerical workers, research scientists, statisticians, volunteers, managers, receptionists, and nurses.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The UK Medical Research Council and Wellcome (Grant ref: 217065/Z/19/Z) and the University of Bristol provide core support for ALSPAC. A comprehensive list of grants funding is available on the ALSPAC website (![]() ). This research was specifically funded by the UK Economic and Social Research Council (Grant ref.: ES/P001742/1; PI Eirini Flouri). This publication is the work of the authors and they will serve as guarantors for the contents of this paper. The authors report no biomedical financial interests or potential conflicts of interest.
). This research was specifically funded by the UK Economic and Social Research Council (Grant ref.: ES/P001742/1; PI Eirini Flouri). This publication is the work of the authors and they will serve as guarantors for the contents of this paper. The authors report no biomedical financial interests or potential conflicts of interest.
Supplemental Material
Supplemental material for this article is available online.
Notes
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
