Abstract
Objective:
Assess executive function (EF) improvement with SHP465 mixed amphetamine salts (MAS) extended-release in adults with attention-deficit/hyperactivity disorder (ADHD) using responder analyses of the Brown Attention-Deficit Disorder Scale (BADDS).
Methods:
Post hoc analyses examined data from placebo-controlled SHP465 MAS dose-optimization (12.5–75 mg) and fixed-dose (25–75 mg) studies. Treatment response was assessed using two definitions (BADDS total score at endpoint <50 [no EF impairment] vs. ≥50 [impaired]; BADDS total score at endpoint relative to the in-treatment 90% CI range for baseline total score [below the range = improved]).
Results:
Response rates (SHP465 MAS vs. placebo) favored SHP465 MAS (all nominal p < .0001) in the dose-optimization (BADDS <50: 41.9% vs. 19.2%; below 90% CI range: 57.4% vs. 29.6%) and fixed-dose (BADDS <50: 51.9% vs. 16.7%; below 90% CI range: 70.6% vs. 32.3%) studies.
Conclusion:
Improvement in EF measured by BADDS response rates was approximately 2-fold greater with SHP465 MAS than placebo.
Keywords
Introduction
“Executive function” (EF), which is a component of the self-management system of the brain, is a term used to describe operations of brain circuits that prioritize, integrate, and regulate other cognitive functions (Brown, 2006; Gioia et al., 2000); EF provides a mechanism for “self-regulation” (Vohs & Baumeister, 2004). Questions related to EF ask how or whether a person goes about doing something (e.g., Will you do it and, if so, how and when?) (Lezak et al., 2004).
The diagnosis of attention-deficit/hyperactivity disorder (ADHD) is currently based on criteria provided by the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) (American Psychiatric Association, 2013). Those diagnostic criteria, which are based primarily on a collection of inattentive and/or hyperactive/impulsive symptoms (American Psychiatric Association, 2013), describe many, but not all, of the aspects of EF that are important components of ADHD (Barkley, 1997a; Brown, 2000; Kessler et al., 2010; Ustun et al., 2017). As the DSM criteria for ADHD were originally developed using data from research in children and adolescents rather than adults (Lahey et al., 1994), normed rating scales can provide information that is useful for assessing the impact of EF impairment in adults diagnosed with ADHD beyond the DSM criteria alone.
Evidence for impaired EF function in youth and adults with ADHD has been observed using batteries of neuropsychologic tests (Doyle et al., 2018; Nigg et al., 2005; Willcutt et al., 2005) and normed self-report rating scales (Adler et al., 2013; Barkley & Fischer, 2011; Brown & Landgraf, 2010; Brown et al., 2010; Brown et al., 2011), with the latter providing an index of how impaired EF can influence daily activities (Barkley, 2011; Brown, 1996; Gioia et al., 2000). However, a meta-analysis of neuropsychologic EF tests administered to adults with ADHD showed that only about 30% of those with ADHD showed significant impairment of EF when assessed with these tests (Hervey et al., 2004). In contrast, >70% of adults diagnosed with ADHD based on the DSM, Fourth Edition (DSM-IV) criteria were reported to exhibit some level of EF deficit when assessed using a range of normed EF measures that included three index scores from standardized tests of memory and cognitive abilities and five subscales of a normed self-report measure of EF impairments in daily life (Brown et al., 2009).
Furthermore, studies have shown that when specific types of EF impairment are combined with DSM–based ADHD adult diagnostic criteria a more robust assessment of ADHD is obtained compared with when DSM criteria alone are used (Kessler et al., 2010; Silverstein et al., 2020; Ustun et al., 2017). For example, an analysis of the physician-administered Adult ADHD Clinical Diagnostic Scale showed that non-DSM symptoms of EF were found to be specific predictors of narrowly defined ADHD (difficulty prioritizing work, trouble planning ahead) and broadly defined ADHD (difficulty prioritizing work, cannot complete tasks on time, makes careless mistakes) in adults (Kessler et al., 2010). In the Adult ADHD Self-Report Scale for the DSM-5 (ASRS-5), two non–DSM-5 EF symptoms (puts things off to last minute, depends on others to keep life in order) are included in the 6-item screener (Ustun et al., 2017). In an analysis of the relationships between ADHD DSM-5–defined ADHD symptoms and EF, both the inattentive and hyperactive/impulsive symptoms of ADHD were observed to be significantly correlated with and highly predictive of EF deficits (Silverstein et al., 2020). Detailed information describing the assessment of impaired EF is provided in the Discussion section of this paper.
The Brown Attention-Deficit Disorder Scale (BADDS) has been used to measure the effects of ADHD pharmacotherapy on EF in multiple studies of adults diagnosed with ADHD (Brown & Landgraf, 2010; Brown et al., 2010; Brown et al., 2011; Rivkin et al., 2012; Spencer et al., 2008). In a crossover study of adults diagnosed with ADHD, lisdexamfetamine dimesylate (LDX) produced statistically significant reductions in BADDS total and cluster scores at endpoint relative to baseline (Brown et al., 2010). Furthermore, LDX treatment was associated with improvements in EF relative to baseline as measured by the percentage of participants with BADDS total scores <50 at endpoint and BADDS total scores below the baseline 90% CI range at endpoint (Brown et al., 2010). In a placebo-controlled study of atomoxetine in adults diagnosed with ADHD, treatment with atomoxetine resulted in significantly greater reductions in BADDS total and cluster scores than placebo (Brown et al., 2011).
SHP465 mixed amphetamine salts (MAS) extended-release (SHP465 MAS [Mydayis®; Shire, Lexington, MA, a member of the Takeda group of companies]) is a once-daily, single-entity MAS product for oral administration approved in the United States for the treatment of ADHD in individuals aged ≥13 years (Mydayis®, 2019). Each SHP465 MAS capsule contains three types of drug-releasing beads (one immediate-release bead and two different types of delayed-release beads) that contain equal amounts (by weight) of four salts (dextroamphetamine sulfate, amphetamine sulfate, dextroamphetamine saccharate, and amphetamine aspartate monohydrate), resulting in a 3:1 mixture of dextroamphetamine-to-levoamphetamine base equivalent.
The efficacy and safety of SHP465 MAS were examined in a series of phase 3 studies in adults diagnosed with ADHD. In these studies, SHP465 MAS significantly reduced ADHD Rating Scale, Fourth Edition (ADHD-RS-IV) total scores versus placebo (Frick et al., 2020; Spencer et al., 2008; Weisler et al., 2017). These studies also reported that the safety and tolerability profile of SHP465 MAS was generally consistent with the profiles of other long-acting stimulants (Adler et al., 2008, 2009; Spencer et al., 2007; Weisler et al., 2006), with the most frequently reported treatment-emergent adverse events being insomnia, dry mouth, and decreased appetite (Frick et al., 2020; Spencer et al., 2008; Weisler et al., 2017).
In two of the aforementioned SHP465 MAS studies (Frick et al., 2020; Spencer et al., 2008), the BADDS was included as a secondary efficacy endpoint. In these studies, treatment with SHP465 MAS produced greater reductions in BADDS total and cluster scores than placebo (Brown & Landgraf, 2010; Spencer et al., 2008). However, to date, there are no published reports of SHP465 MAS treatment response rates based on BADDS total score in adults diagnosed with ADHD. Therefore, the objective of the current post hoc analyses was to examine SHP465 MAS treatment response rates using two previously described definitions (Brown et al., 2010). The first definition examined BADDS total score at endpoint, with BADDS total score ≥50 being used as an index of EF impairment. The second definition examined BADDS total score at endpoint relative to the in-treatment 90% CI range for baseline total score, with total scores below the 90% CI range indicating improvement from baseline.
Methods
Study Design, Treatment, and Participants
These post hoc analyses included data from two studies of SHP465 MAS in adults with ADHD. The methods have previously been described in detail elsewhere (Frick et al., 2020; Spencer et al., 2008) and are summarized here.
The dose-optimization study (ClinicalTrials.gov, NCT00150579) was a 7-week, phase 3, randomized, double-blind, placebo-controlled study in which participants were randomized to SHP465 MAS (12.5–75 mg) or placebo (Spencer et al., 2008). For each participant randomized to SHP465 MAS, treatment was initiated at 12.5 mg SHP465 MAS and titrated to 25 mg at Week 2, 50 mg at Week 3, and 75 mg at Week 4 until an optimal dose was reached. An optimal dose was defined as one that produced a ≥30% ADHD-RS-IV total score decrease from baseline and was well tolerated. Dose reductions were permitted only from 50 to 37.5 mg SHP465 MAS at the end of Week 3 and from 75 to 62.5 mg at the end of Week 4. Once an optimal dose was reached, it was maintained until the end of the study.
The fixed-dose study (ClinicalTrials.gov, NCT00152022) was a 6-week, phase 3, randomized, placebo-controlled, double-blind study in which participants were randomized to SHP465 MAS (25, 50, or 75 mg) or placebo (Frick et al., 2020). All SHP465 MAS groups initiated treatment at 25 mg during Week 1. The 25 mg SHP465 MAS group received 25 mg throughout the study; the 50 mg SHP465 group received 37.5 mg during Week 2 and 50 mg during Weeks 3 to 6; the 75 mg SHP465 MAS group received 37.5 mg during Week 2, 50 mg during Week 3, and 75 mg during Weeks 4 to 6. Modifications to the dosing regimens were not allowed.
In both studies, eligible participants were men and nonpregnant/nonlactating women (aged 18–55 years) who had satisfactory medical assessments, with no clinically significant or relevant abnormalities as determined by medical history, physical examinations, or clinical and laboratory evaluations. Participants were also required to meet DSM-IV, Text Revision (DSM-IV-TR) criteria for ADHD and to have ADHD-RS-IV baseline total scores ≥24 (for the dose-optimization study) or ≥32 (for the fixed-dose study).
Participants were excluded if they were underweight (body mass index <18.5 kg/m2) or morbidly obese; had a comorbid psychiatric diagnosis controlled with a prohibited medication or uncontrolled and associated with clinically relevant symptoms that contraindicate SHP465 MAS use or could confound study assessments; had any concurrent chronic or acute illness or unstable medical condition that could confound the safety assessments, increase participant risk, or lead to difficulty complying with the protocol; had cardiac structural anomalies, electrocardiogram anomalies, or laboratory anomalies at screening or baseline; history of hypertension; resting systolic blood pressure >139 mm Hg, or resting diastolic blood pressure >89 mm Hg; had a history (within 6 months before screening) of drug dependence or substance use disorder according to DSM-IV-TR criteria (excluding nicotine); and had a documented allergy to, intolerance of, or history of nonresponse to methylphenidate or amphetamines.
Both study protocols were approved by institutional review boards and conducted in accordance with the principles of the World Medical Association Declaration of Helsinki, including amendments of the 29th, 35th, 41st, and 48th World Medical Assemblies. All participants provided written informed consent before any study-related procedures were conducted; informed consent documents were written in accordance with Good Clinical Practice and guidelines of the 1996 Health Insurance Portability and Accountability Act.
Endpoint
The primary efficacy endpoint in both studies was change from baseline on the ADHD-RS-IV. The findings for the primary efficacy analyses for both studies have been reported (Frick et al., 2020; Spencer et al., 2008). The BADDS (Brown, 1996) was included as a secondary efficacy endpoint in these studies and was assessed at baseline and at the final study visit. Findings for the prespecified analyses for the BADDS have been reported (Brown & Landgraf, 2010; Spencer et al., 2008). This report focuses on the results of post hoc responder analyses.
The BADDS is a 40-item, validated, self-report scale administered by a clinician or appropriately trained individual that assesses aspects of EF that are conceptually related to ADHD (Brown & Landgraf, 2010). The items assessed in the BADDS (Figure 1) include all of the symptoms of inattention for ADHD listed in the DSM-IV, along with wide coverage of symptoms of EF not included in the DSM-IV ADHD criteria (Brown, 1996; Brown & Landgraf, 2010). BADDS items are grouped into five clusters (organizing and activating to work, sustaining attention and concentration, sustaining energy and effort, managing affective interference, utilizing working memory and accessing recall). Cluster scores are generated by summing item scores within each cluster, and total score is generated by summing all item scores; higher cluster scores and total scores indicate greater levels of impairment in EF (Brown & Landgraf, 2010). The items of the BADDS are scored on a 4-point Likert scale (0 = never; 1 = once a week or less; 2 = twice a week; 3 = almost daily), with total score ranging from 0 to 120. Based on psychometric analyses, a BADDS total score ≥50 has been suggested to be indicative of impaired EF, and posttreatment total score shifts below the pretreatment 90% CI range are considered an index of improved EF (Brown, 1996).
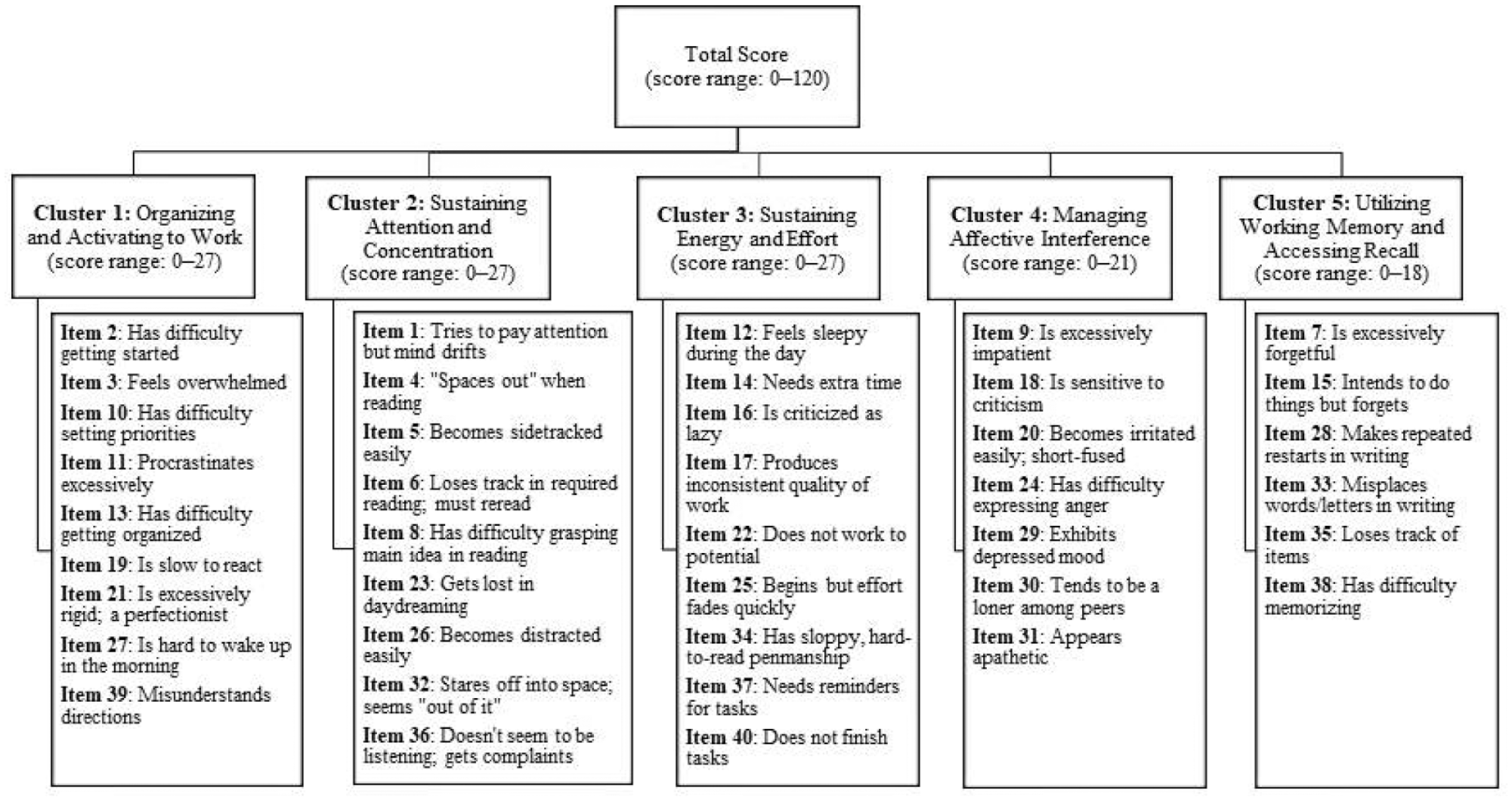
BADDS structure and scoring.
Statistical Analysis and Data Presentation
All analyses were conducted in the overall intent-to-treat (ITT) population (all randomized participants who received ≥1 study drug dose and who had a baseline assessment and ≥1 postbaseline primary efficacy assessment). These post hoc analyses assessed SHP465 MAS treatment response rates at study endpoint (defined as the last nonmissing postbaseline assessment) using two previously described definitions (Brown et al., 2010). First, BADDS total score at endpoint (<50 [no clinically impaired EF] vs. ≥50 [some level of EF impairment]) was used to assess the percentage of participants in each treatment group exhibiting EF impairment. To more fully examine the level of EF impairment across treatment groups, participants with BADDS total score ≥50 were subdivided into three additional groups (scores 50–59, 60–69, and ≥70). Second, BADDS total score at endpoint relative to the in-treatment 90% CI range for baseline total score was used to assess the percentage of participants exhibiting improved EF (total score at endpoint below the 90% CI range), no change in EF (total score within the 90% CI range), or worsening of EF (total score at endpoint above the 90% CI range).
Between-group differences were examined with chi-square tests for dichotomized assessments (BADDS total score at endpoint <50 vs. ≥50) and Mantel-Haenszel row mean score chi-square tests for assessments between more than two groups (BADDS total score at endpoint <50 vs. 50–59 vs. 60–69 vs. ≥70; BADDS total score at baseline relative to the in-treatment 90% CI range [below 90% CI range vs. within 90% CI range and vs. above 90% CI range). The studies were not powered for these analyses. Therefore, all reported P values are nominal and descriptive.
Results
Participant Disposition and Demographics
The ITT population consisted of 268 participants in the dose-optimization study (SHP465 MAS, n = 136; placebo, n = 132) and 405 participants in the fixed-dose study (all SHP465 MAS, n = 302 [25 mg, n = 103; 50 mg, n = 101; 75 mg, n = 98]; placebo, n = 103). Baseline demographic and clinical characteristics for the ITT population of each study are summarized in Table 1. Most participants in each study were white and had the combined ADHD subtype. In both studies, the SHP465 MAS and placebo groups had comparable ADHD-RS-IV total scores and BADDS total scores at baseline.
Participant Demographic and Clinical Characteristics, ITT Population.
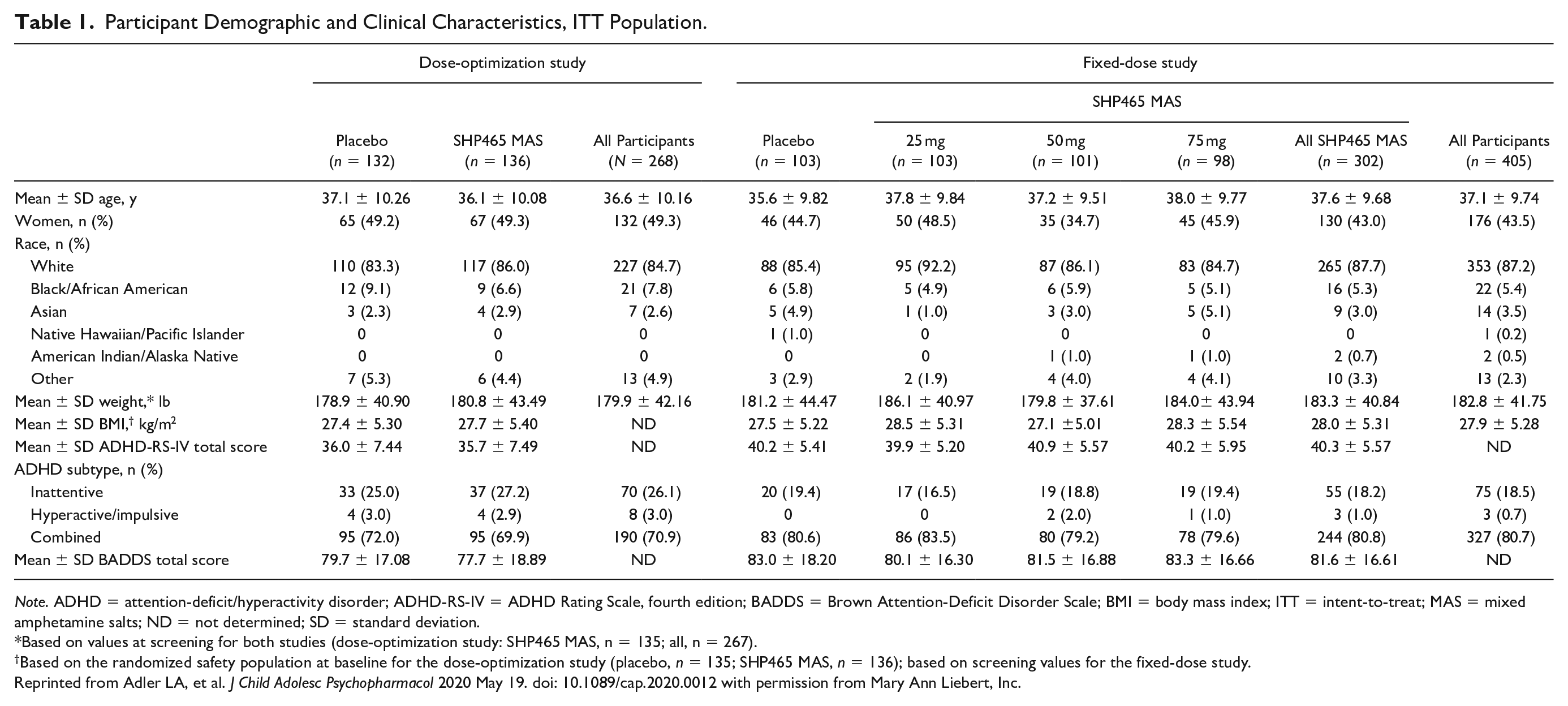
Note. ADHD = attention-deficit/hyperactivity disorder; ADHD-RS-IV = ADHD Rating Scale, fourth edition; BADDS = Brown Attention-Deficit Disorder Scale; BMI = body mass index; ITT = intent-to-treat; MAS = mixed amphetamine salts; ND = not determined; SD = standard deviation.
Based on values at screening for both studies (dose-optimization study: SHP465 MAS, n = 135; all, n = 267).
Based on the randomized safety population at baseline for the dose-optimization study (placebo, n = 135; SHP465 MAS, n = 136); based on screening values for the fixed-dose study.
Reprinted from Adler LA, et al. J Child Adolesc Psychopharmacol 2020 May 19. doi: 10.1089/cap.2020.0012 with permission from Mary Ann Liebert, Inc.
BADDS Responder Analyses
BADDS Total score at endpoint
The percentage of participants with BADDS total scores <50 at endpoint was greater and the percentage with BADDS total score ≥50 at endpoint was smaller with SHP465 MAS compared with placebo in the dose-optimization study (Figure 2a; nominal p < .0001) and the fixed-dose study (Figure 2b; nominal p < .0001). In the fixed-dose study, assessment of individual SHP465 MAS doses indicated that the percentage of participants with BADDS total score <50 was greater for all SHP465 MAS doses (42.4% [42/99] for 25 mg; 57.1% [56/98] for 50 mg; 56.3% [54/96] for 75 mg) and the percentage with BADDS total score ≥50 was smaller for all SHP465 MAS doses (57.6% [57/99]; 42.9% [42/98]; 43.8% [42/96], respectively) compared with placebo (<50: 16.7% [16/96]; ≥50: 83.3% [80/96]; all nominal p < .0001 vs placebo for each SHP465 MAS dose). Moreover, participants with BADDS total scores ≥50 in the SHP465 MAS groups exhibited lower levels of EF impairment at endpoint versus placebo (Table 2). In both studies, greater percentages of participants in the SHP465 MAS treatment groups had BADDS total scores between 50 and 59 at endpoint (lower levels of EF impairments) compared with placebo, and smaller percentages of participants in the SHP465 MAS treatment groups had BADDS total scores ≥70 at endpoint (greater levels of EF impairment) compared with placebo (all nominal p < .0001 vs. placebo).
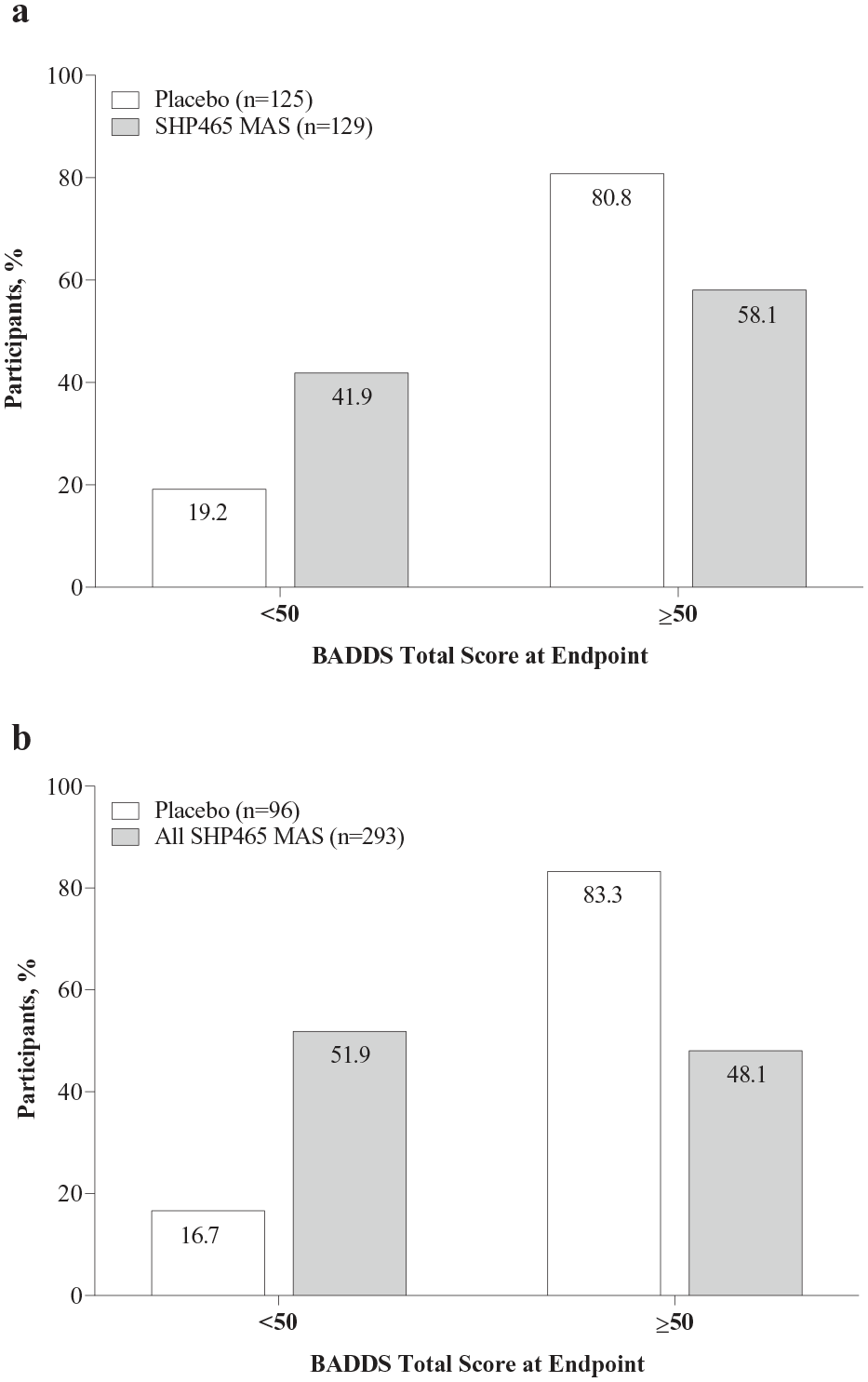
Percentage of participants with BADDS total score <50 versus ≥50 at endpoint in the (a) dose-optimization study and (b) fixed-dose study, ITT population.
Level of EF Impairment in Participants With BADDS Total Score ≥50 at Endpoint, ITT Population.
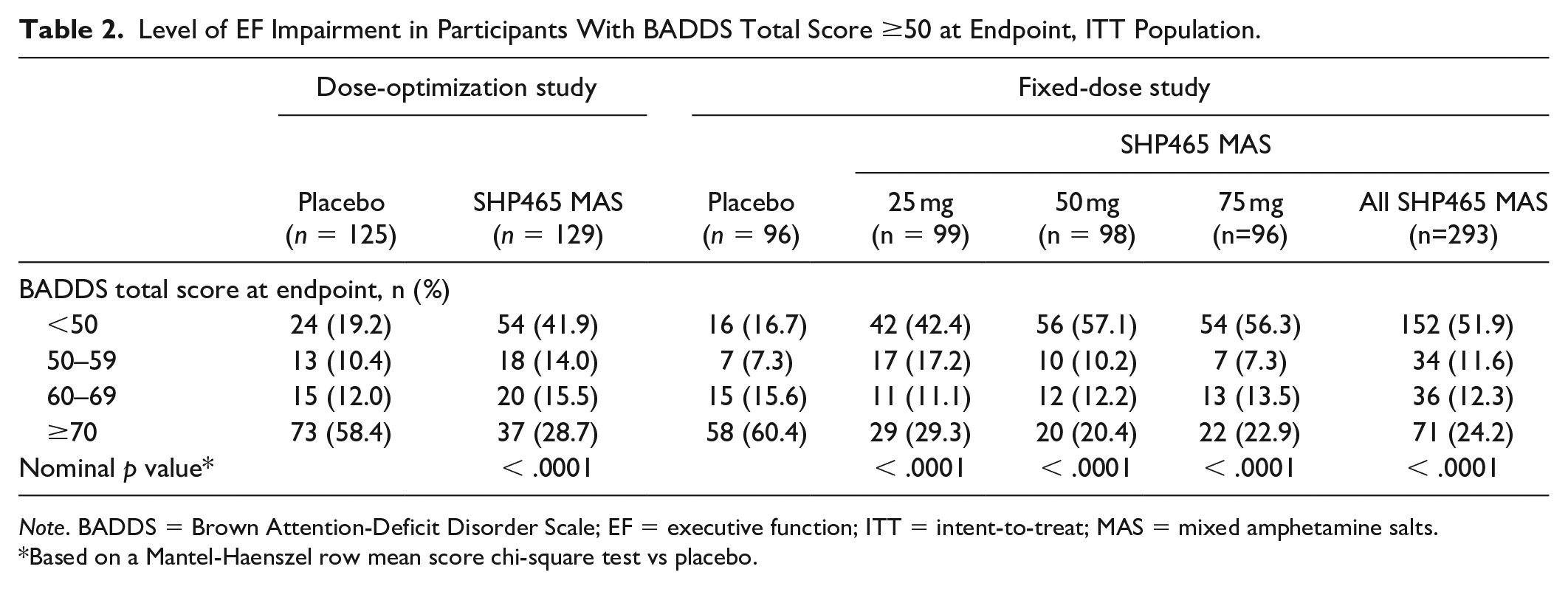
Note. BADDS = Brown Attention-Deficit Disorder Scale; EF = executive function; ITT = intent-to-treat; MAS = mixed amphetamine salts.
Based on a Mantel-Haenszel row mean score chi-square test vs placebo.
BADDS Total Score at Endpoint Relative to the Baseline In-Treatment 90% CI Range
The percentage of participants with BADDS total scores at endpoint that were below the baseline in-treatment 90% CI range (an index of improved EF from baseline) was greater and the percentage of participants with BADDS total scores at endpoint that were within or above the baseline in-treatment 90% CI range (indices of no change or worsening of EF, respectively) was smaller with SHP465 MAS than placebo in the dose-optimization study (Figure 3a; nominal p < .0001) and the fixed-dose study (Figure 3b; nominal p <.0001). Examination of the individual SHP465 MAS dose data in the fixed-dose study revealed the same pattern of effects. Greater percentages of participants in the SHP465 MAS groups had BADDS total scores at endpoint below the baseline in-treatment 90% CI range (61.6% [61/99] for 25 mg; 77.6% [76/98] for 50 mg; 72.9% [70/96] for 75 mg; all nominal p < .0001) compared with the placebo group (32.3% [31/96]). Smaller percentages of participants in the SHP465 MAS groups had BADDS total scores at endpoint that were within the baseline in-treatment 90% CI range (26.3% [26/99]; 18.4% [18/98]; 14.6% [14/96], respectively; all nominal p < .0001) compared with the placebo group (52.1% [50/96]) and that were above the baseline in-treatment 90% CI range (12.1% [12/99]; 4.1% [4/98]; 12.5% [12/96]; all nominal p < .0001) compared with the placebo group (15.6% [15/96]).
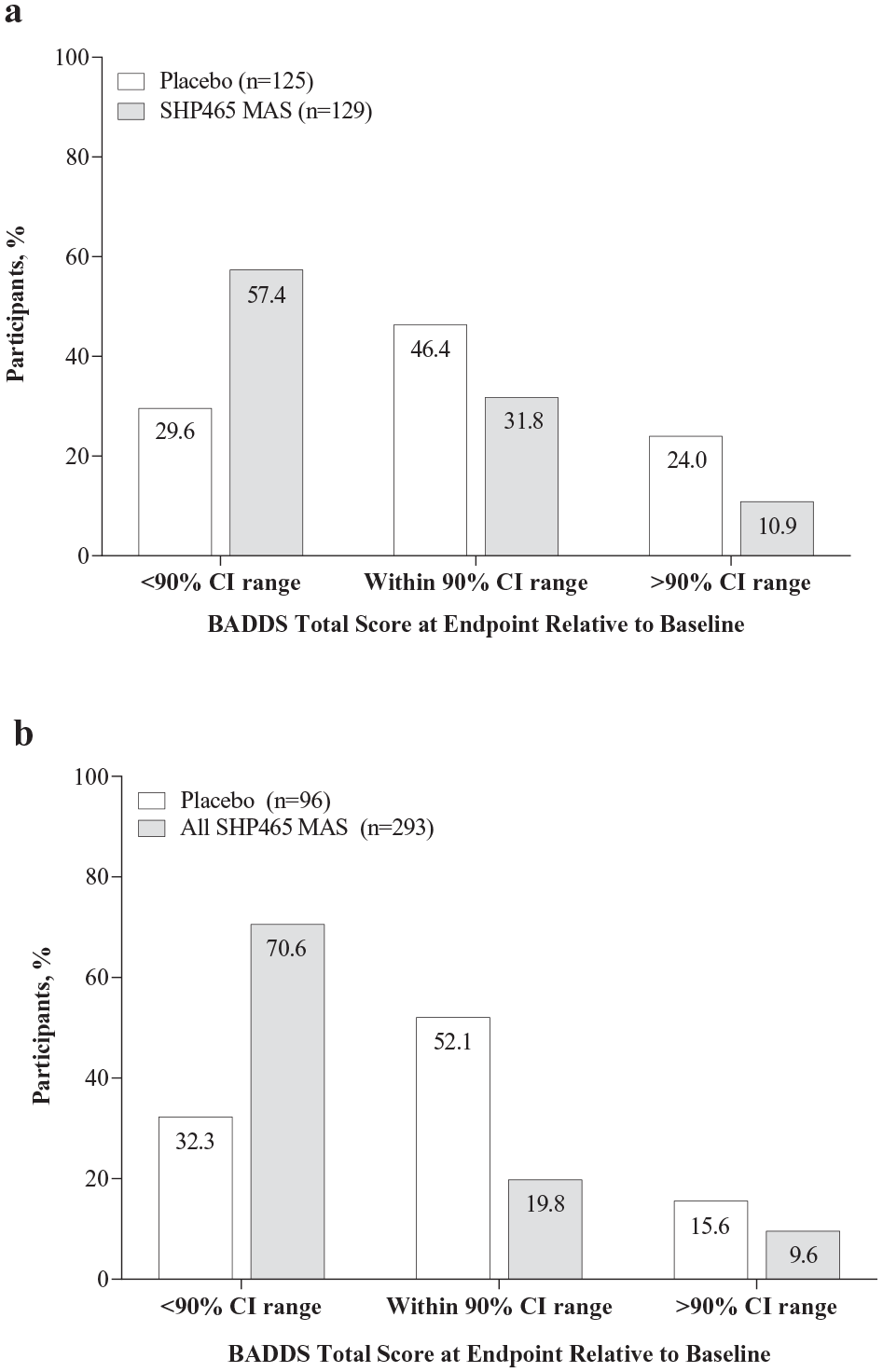
Percentage of participants with BADDS total score above, within, or below the in-treatment 90% CI range at endpoint in the (a) dose-optimization study and (b) fixed-dose study, ITT population.
Discussion
This paper describes improvement of EF in adults diagnosed with ADHD following treatment with SHP465 MAS, as measured by the BADDS. Normed rating scales for ADHD are one well-recognized way of assessing EF impairments (Barkley, 2019; Brown, 2006), which many researchers consider the underlying core problem in ADHD (Barkley, 1997b, 2006, 2008, 2012, 2019; Brown, 2000, 2005, 2006, 2013, 2017; Castellanos, 1999; Kessler et al., 2010; Ustun et al., 2017). For example, Castellanos (1999) observed that “the unifying abstraction that best encompasses the faculties principally affected in ADHD has been termed executive function. . .there is now impressive empirical support for its importance in ADHD.”
As has been described by Brown (2006) there are two conflicting ways of describing and measuring EF. One view sees EF as being defined by low scores on a battery of neuropsychological tests administered by a neuropsychologist, a view supported in part by studies that have reported impaired performance on tests of EF in youth and adults diagnosed with ADHD (Doyle et al., 2018; Nigg et al., 2005; Willcutt et al., 2005). However, a meta-analysis of such EF tests administered to adults with ADHD showed that only about 30% of those with ADHD showed significant impairments of EF functions when assessed with these tests (Hervey et al., 2004). The alternative view of EF has been advocated by Barkley (1997b, 2006, 2008, 2012, 2019) and Brown (2000, 2005, 2006, 2013, 2017). Their view is that EF impairments associated with ADHD are best assessed by use of normed rating scales for ADHD utilized in combination with clinical interviews and DSM criteria for ADHD. They claim that rating scales are more valid ecologically because they gather data on the individual’s functioning in a variety of settings and situations of daily life over a more protracted time frame than can be assessed by neuropsychological tests of EF (Barkley, 2019). In support of this view, it has been reported >70% of adults diagnosed with ADHD based on the DSM-IV criteria exhibit some level of EF deficit when assessed using a range of normed EF measures that included three index scores from standardized tests of memory and cognitive abilities and five subscales of a normed self-report measure of EF impairments in daily life (Brown et al., 2009).
The post hoc analyses described in this paper examined the effect of SHP465 MAS treatment on EF as measured by BADDS total score response rates at study endpoint. One key finding of these analyses is that nominally greater percentages of participants treated with SHP465 MAS compared with placebo had BADDS total scores <50, which is indicative of the absence of significantly impaired EF. The second key finding is that nominally greater percentages of participants treated with SHP465 MAS compared with placebo had BADDS total scores at endpoint that were below the baseline in-treatment 90% CI range, which is indicative of improved EF from baseline.
The current findings support the previously published prespecified analyses from these studies, which reported that SHP465 MAS treatment resulted in greater reductions from baseline in BADDS total score than placebo (Brown & Landgraf, 2010; Spencer et al., 2008). Moreover, these findings expand upon previously published data by providing insight into the percentage of participants who exhibit a response to SHP465 MAS as measured by BADDS total score at endpoint. Because it has been suggested that a BADDS total score <50 is indicative of a lack of significantly impaired EF (Brown, 1996), these post hoc analyses indicate that a substantial proportion (approximately 40%–50%) of participants who received SHP465 MAS did not exhibit significantly impaired EF at endpoint compared with <20% of participants who received placebo. Additionally, among participants who still exhibited some level of EF impairment at endpoint, less severe levels of EF impairment (i.e., greater percentages of participants with BADDS total scores 50–59) were observed with SHP465 MAS compared with placebo. Taken together, these findings provide further support for the potential of SHP465 MAS to reduce EF impairment in adults diagnosed with ADHD.
Multiple studies have used the BADDS to assess the effects of ADHD pharmacotherapy on EF (Brown & Landgraf, 2010; Brown et al., 2010; Brown et al., 2011; Rivkin et al., 2012; Spencer et al., 2008). However, to the best of our knowledge, only one other published study used the same definitions of treatment response employed in this report (Brown et al., 2010). Brown et al (2010) reported that after 4 weeks of LDX treatment, 62.7% of participants had BADDS total scores <50, and 78.9% of participants had BADDS total scores at endpoint that were below the baseline in-treatment 90% CI range. Further use of these definitions based on BADDS total scores may provide a deeper understanding of how approved pharmacotherapies affect impaired EF in adults diagnosed with ADHD.
In a post hoc analysis of the relationship between changes in ADHD symptoms and EF following treatment with SHP465 MAS (Brown et al., 2019), it was reported that changes in BADDS total scores and BADDS cluster scores after treatment with SHP465 MAS were positively correlated with changes in ADHD-RS-IV total and subscale scores. Although the BADDS includes all of the ADHD symptoms of inattention listed in DSM-IV, it also includes wide coverage of EF symptoms that are not included in the DSM-IV ADHD criteria. Therefore, these correlations are not likely to be solely related to improvement in the inattentive and/or hyperactive/impulsive symptoms of ADHD. Furthermore, although there are no published reports describing the effects of ADHD treatment on the individual items of the BADDS to the best of our knowledge, previously published reports indicate that treatment with ADHD medications are associated with reductions across all BADDS cluster scores (Brown & Landgraf, 2010; Brown et al., 2010). Taken together, these findings suggest that treatment with ADHD medications is associated with broad improvement in EF, as measured by the BADDS.
The current findings have broad clinical relevance because EF impairment is a predictor of adult ADHD (Kessler et al., 2010). As described by Kessler et al (2010), non-DSM symptoms of EF have been shown to be specific predictors of narrowly defined ADHD (difficulty prioritizing work, trouble planning ahead) and broadly defined ADHD (difficulty prioritizing work, cannot complete tasks on time, makes careless mistakes) in adults. Because the individual items and domains of the BADDS capture a wide array of EF behaviors that are conceptually related to ADHD (Brown & Landgraf, 2010), use of the BADDS in the assessment of the effects of ADHD pharmacotherapy can provide valuable information regarding the wide-ranging effects of ADHD treatment on EF. Although the current analyses focus solely on BADDS total score, and hence global EF, previous publications based on the same studies used in the current analyses have described the effects of SHP465 MAS on BADDS domain scores (Brown & Landgraf, 2010; Spencer et al., 2008). In both studies, SHP465 MAS treatment was associated with reductions from baseline across all BADDS domains (Brown & Landgraf, 2010; Spencer et al., 2008), suggesting that SHP465 MAS can reduce impairment across a wide range of behaviors related to EF in adults diagnosed with ADHD.
These findings should be considered in light of certain limitations. First, the study population was primarily white and had the combined ADHD subtype. Therefore, these findings may not be representative of a more heterogeneous population of adults diagnosed with ADHD. Second, the study was not powered for these post hoc analyses. Therefore, all reported P values are nominal and included for descriptive purposes only. Last, the current analyses focus only on BADDS total score, so it is not known if treatment responses to SHP465 MAS differed across specific domains or items.
Conclusion
These post hoc analyses support the efficacy of SHP465 MAS compared with placebo for reducing EF impairment in adults diagnosed with ADHD. The percentages of participants with BADDS total scores <50 at study endpoint, which is indicative of the absence of significantly impaired EF, were approximately 2 to 3 times greater with SHP465 MAS than placebo. In addition, the percentages of participants with BADDS total scores at endpoint that were below the baseline in-treatment 90% CI range, which is indicative of improved EF relative to baseline, were approximately 2 times greater with SHP465 MAS than placebo. Given the growing amount of evidence indicating that impaired EF is an important component of ADHD (Barkley 1997b, 2006, 2008, 2012, 2019; Brown 2000, 2005, 2006, 2013, 2017; Castellanos, 1999; Kessler et al., 2010; Ustun et al., 2017) and the fact that approximately 30% to >70% of adults diagnosed with ADHD may exhibit some level of EF deficit (Biederman et al., 2006; Brown et al., 2009; Silverstein et al., 2020), understanding how currently approved pharmacotherapies for ADHD affect EF is critically important. Taken together, these findings indicate that in addition to improving the core DSM-based symptoms of ADHD in adults diagnosed with ADHD (Frick et al., 2020; Spencer et al., 2008; Weisler et al., 2017), SHP465 MAS also reduced EF impairment versus placebo.
Footnotes
Acknowledgements
Under the direction of the authors, Craig Slawecki, PhD, an employee of ICON (North Wales, PA), provided writing assistance for this manuscript.
Authors’ Note
Brigitte Robertson was an employee of Shire, a member of the Takeda group of companies, at the time this research was conducted, and holds Takeda stock. She is currently employed by Yumanity Therapeutics Inc. (Cambridge, MA).
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article:
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Shire Development LLC, a member of the Takeda group of companies, Lexington, MA, funded this clinical research and provided funding to ICON for support in writing and editing this manuscript.
