Abstract
Frailty is associated with an increase in mortality and unplanned hospitalization. We aimed to test the ability of a novel home healthcare program to reverse frailty in a geriatric home health population. We designed a home healthcare exercise program including aerobic, resistance, balance, and flexibility components (Vitality Therapy™, “VT”). We assessed physical performance measures in a frail cohort at baseline, and at multiple follow-up points. Between August 27, 2023 and May 31, 2024, 98 frail adults (35 males, 63 females; mean age 80 ± 10 years) participated in physical therapist-led exercise. The Short Physical Performance Battery (SPPB), the clinical frailty scale and level of ambulation assistance were assessed. Mixed linear modeling was used to perform statistical significance testing. There were notable improvements in all measured physical performance outcomes among program participants. Pairwise comparisons showed that the SPPB was significantly greater at 120 days (M = 9.03, SE = 0.48), 90 days (M = 7.66, SE = 0.41), 60 days (M = 7.45, SE = 0.31) and at 30 days (M = 6.39, SE = 0.29) compared to baseline (M = 4.28, SE = 0.28). Significant improvement was also observed on the clinical frailty scale (F3, 77.37 = 18,24, p < .01) with patients improving their frailty score from 5.67 (SE = 0.17) at baseline to 4.43 (SE = 0.28) after 90 days in the program. The VT program improved physical performance measures and decreased frailty in older adults. Expansion of home healthcare programs targeted to reverse frailty could provide increased access to an impactful intervention for this vulnerable population.
Introduction
Frailty, a state of physical vulnerability independent of age, is associated with an increase in mortality, unplanned hospitalization, 1 as well a greater risk of dependency and falling. 2 Both frail Medicare beneficiaries 3 and frail Medicare Advantage members 4 utilize inpatient and emergency settings at a significantly higher rate than non-frail beneficiaries and have a higher total cost of care. 3 Despite the impact of frailty on the geriatric population and on healthcare systems, there are few programs currently in widescale use built to reverse it. Multifaceted, structured exercise interventions have been shown to reverse frailty in the geriatric population.5 -7 Programs including 4 components of exercise focused on strength, balance, flexibility, and endurance training have been shown to improve measures of fitness even in those with severe frailty, such as geriatric populations living in long-term care. 8
Home healthcare is recognized to be a safe and cost-effective method of delivering care that has been shown to be associated with decreased mortality and fewer readmissions to the hospital after discharge. 9 It is also a segment of the healthcare system that frequently provides care to older adults. Typical areas of focus for home healthcare programs include providing skilled nursing to homebound patients requiring treatments for conditions such as congestive heart failure and diabetes mellitus, and providing physical, occupational, and speech therapy to patients with conditions such as gait instability, stroke, and neurodegenerative disease. Despite the increasing utilization of home healthcare in the U.S., 10 a structured approach utilizing a primary home healthcare intervention specifically built to reverse frailty has not been previously reported. In this study, we aimed to test the ability of a novel home healthcare program to reverse frailty in a geriatric home health population.
Methods
We developed a new, structured home healthcare program (Vitality Therapy™, “VT”) that included aerobic, resistance, balance, and flexibility components (Figure 1) with an aim of reversing frailty in a geriatric home healthcare population. Due to the observational design, and the use of HIPAA-compliant, de-identified data, included patients did not meet the criteria for human subjects, and as such, the study was exempt from institutional review board review. Frailty was determined using the frailty phenotype as described by Fried et al. 11 Ninety-eight frail adults (35 males, 63 females; mean age 80 ± 10 years) participated in physical therapist-led exercise for 45 minutes 3 times a week during home health episodes of care between August 27, 2023 and May 31, 2024. Participant progress in the program was assessed at baseline and at 30-day intervals up to a total period of 120 days using the Short Physical Performance Battery (SPPB), the Clinical Frailty Scale and a measure of ambulation assistance. Complete baseline data included into the analyses was available for 83 enrolled participants. Seventy-one participants underwent their 30-day assessment, and 53 participants completed the 60-day assessment. Twenty participants completed the Short Physical Performance Battery (SPPB) at 90 days with 13 participants undergoing assessments at 120 days following program enrollment. By the time of data analysis, 57 participants completed the program with 6 patients still listed as current participants. Other sources of missing data included hospitalizations of 17 participants as well as incomplete assessments (not able to be performed as per the judgment of the treating physical therapist).
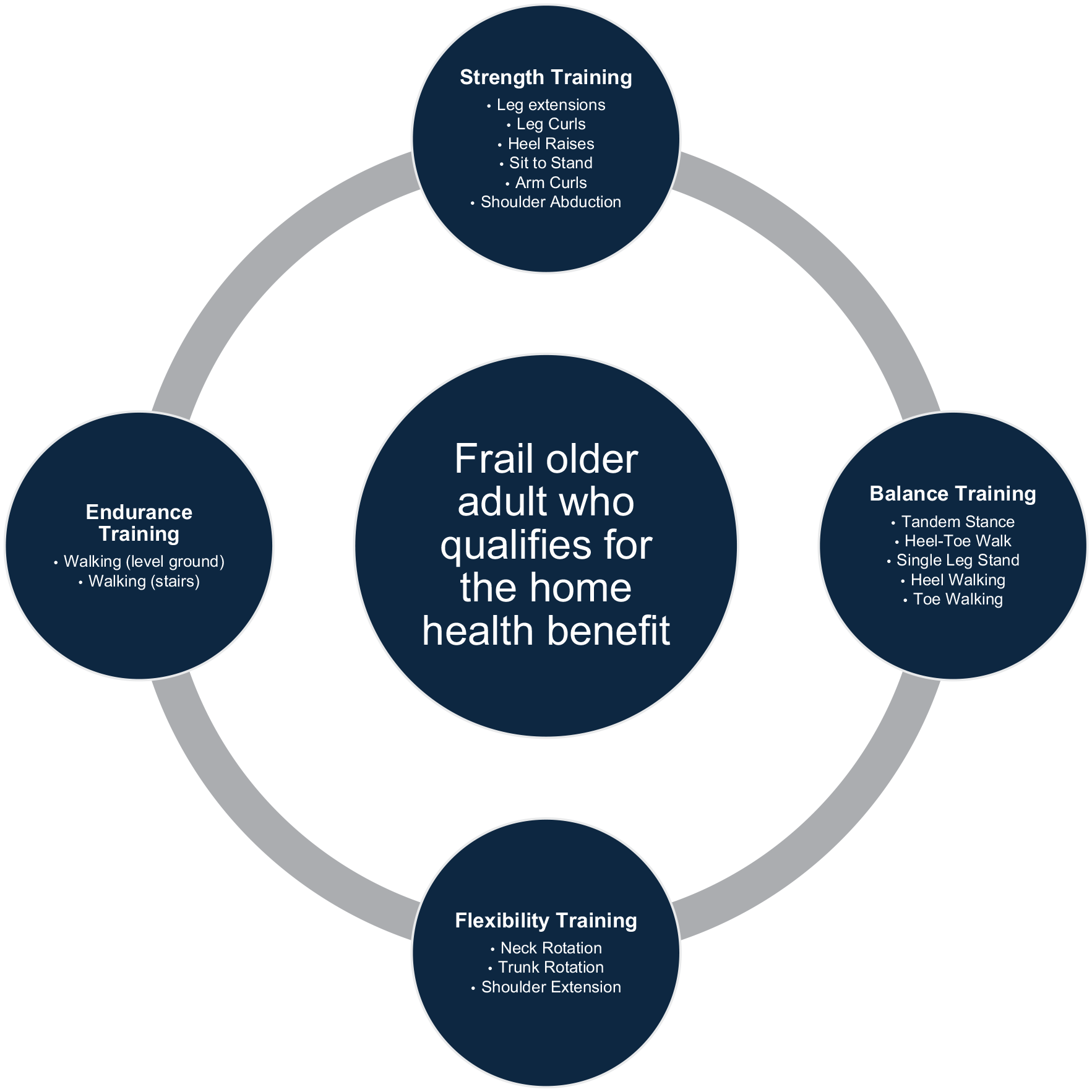
Four components of the Vitality Therapy™ home health program, led by a physical therapist, and conducted for 45 minutes, 3 times per week. Examples of exercises in each of the 4 components are included.
The SPPB assesses physical function through 3 key tests, each scored on a scale of 0 to 4: Balance Test (evaluates the ability to maintain 3 standing positions for up to 10 seconds each: feet side-by-side, semi-tandem stance, full tandem stance); Gait Speed Test (measures the time taken to walk 4 m at a normal pace) and Chair Stand Test (measures the time it takes to stand up from a seated position 5 times without using arms). The scores from these 3 tests are added together to produce a total SPPB score, ranging from 0 to 12. A higher total score indicates better physical function, while a lower score suggests reduced functional capacity. This assessment helps gage functional status and physical performance in older adults. 12 Research has demonstrated that lower SPPB scores are associated with negative outcomes including heightened fall risk, reduced independence in daily activities, decreased mobility, increased disability, declining overall health, more frequent hospital readmissions, longer hospital stays, cognitive decline, higher likelihood of nursing home placement, and increased mortality. 13 The SPPB has been validated as a highly reliable and responsive tool for assessing physical function in community-dwelling older adults. 14 In terms of clinical significance, a 0.5-point change on the SPPB is considered a minor meaningful difference, while a 1-point change represents a substantial meaningful difference. 15 Finally, a SPPB score of less than 8 has been shown to be associated with frailty. 16
The Clinical Frailty Scale (CFS), developed as part of the Canadian Study of Health and Aging, is a widely used tool to assess frailty in older adults.17 -19 The CFS relies on clinical judgment to evaluate key aspects of a person’s health, including existing medical conditions, functional capabilities, and cognitive status. It assigns a score ranging from 1 to 9, with lower scores indicating increased vitality and higher scores representing increased frailty or terminal illness. Originally used primarily in geriatric medicine, the CFS has expanded its reach to specialties such as cardiology, intensive care, and emergency medicine. As the medical community has increasingly recognized the importance of frailty as a health indicator, the use of the CFS in research has grown substantially. Researchers frequently employ the scale to predict various health outcomes, with studies showing significant correlations between CFS scores and mortality rates, comorbidities, functional and cognitive decline, and mobility issues. 18 This versatility and predictive power have cemented the CFS’s position as a valuable instrument in both clinical practice and medical research for assessing the overall health status of older adults. Finally, functional mobility was assessed using the following categories: 0—non-ambulatory, 1—maximal assistance, 2—moderate assistance, 3—minimal assistance, 4—contact guard, 5—stand-by-assistance, 6—supervision, 7—modified independent, 8—independent.
To determine whether the intervention affected these physical performance measures, we performed statistical comparisons between baseline scores and follow-up scores measured at 30-day intervals of up to 120 days after enrollment into the VT program. To account for correlated subject data over multiple measurement points we used Mixed Linear Modeling (MLM) with Restricted Maximum Likelihood (REML) of estimation available in SPSS 29.0. Patient ID was specified to model the random effect of correlated subject data. Pairwise comparisons for the main fixed effect of time of assessment were conducted with the Sidak correction. MLM is preferred to Repeated Measures Analysis of Variance as it correctly models the random effect of data point clustering within an individual as well as the correlation matrix structures of repeated observations and is robust to missing data (MCAR). It uses the Restricted Maximum Likelihood method of parameter estimation which handles missing data by using all available information in the observed data, without the need for imputation or deletion of incomplete cases (Twisk, 2013). In our study we used an Autoregressive Type I (AR1) correlation structure for repeated measures that assumes an exponential decrease in correlations between measurements the further apart the time points are. To preserve minimal statistical power of 0.80 at alpha level of 0.05 and to be able to detect a medium-to-large effect size (f = 0.35) we varied the number of time points included into the MLM between 90 days for CFS and 120 days for SPPB measures with the minimum number of subjects per time point no less than 13 (13).
Results
Individual differences accounted for a significant proportion of variance in all dependent measures (p < .01), as indicated by significant random intercept effects. After accounting for this between-subject variability, significant residual variance remained in all dependent measures (p < .01), suggesting substantial within-subject variability that could not be explained by the fixed effect of time alone. This unexplained within-subject variability likely represents individual differences in response to the intervention as well as other unmeasured time-varying factors (see Table 1). Specifically, participants significantly increased their total SPPB score (F4,158.74 = 59.366, p < .01) between baseline and all 4 follow-up points. Pairwise comparisons showed that the SPPB was significantly greater at 120 days (M = 9.03, SE = 0.48), 90 days (M = 7.66, SE = 0.41 ), 60 days (M = 7.45, SE = 0.31) and at 30 days (M = 6.39, SE = 0.29) compared to baseline (M = 4.28, SE = 0.28). These results are presented in Figure 2a. Each of the 3 components of the SPPB composite score also showed improvement among participants in the VT program. Participants significantly increased their gait velocity (F3,137.18 = 21.31, p < .01) from 0.51 m/s (SE = 0.025) at baseline to 0.71 m/s (SE = 0.042) after 90 days of program participation. In fact, the gait speed showed significant improvement from baseline at each 30-day interval (M = 0.63, SE = 0.027 at 30 days and M = 0.72, SE = 0.03 at 60 days). These results are summarized in Figure 2b. After 3 months of program participation, the patients also significantly (F3,128.40 = 20.97, p < .01) increased their lower body strength, which was reflected in a significantly shorter duration to complete the chair stand test at 90 days (M = 16.31, SE = 1.91), 60 days (M = 16.76, SE = 1.33), and 30 days (M = 19.01, SE = 1.94) compared to the baseline time of 26.52 seconds (SE = 1.15). These results are summarized in Figure 2c. Out of the 3 balance tests, the effect of the program was most notable in the improved ability of the participants to maintain a tandem stance (F4,169.80 = 22.88, p < .01). While the patients were only able to maintain the stance for an average of 2.3 seconds (SE = 0.86) at baseline, they progressed to 5.42 seconds (SE = 0.94) after 30 days in the program and continued to significantly improve at each subsequent assessment point (M@60 days = 8.98, SE = 1.10 ; M@90 days = 11.50, SE = 1.69) reaching 20.95 seconds (SE = 2.19) at 120 days (Figure 2d).
Variance in Dependent Measures Explained by Intraclass Correlation Coefficients (ICC).
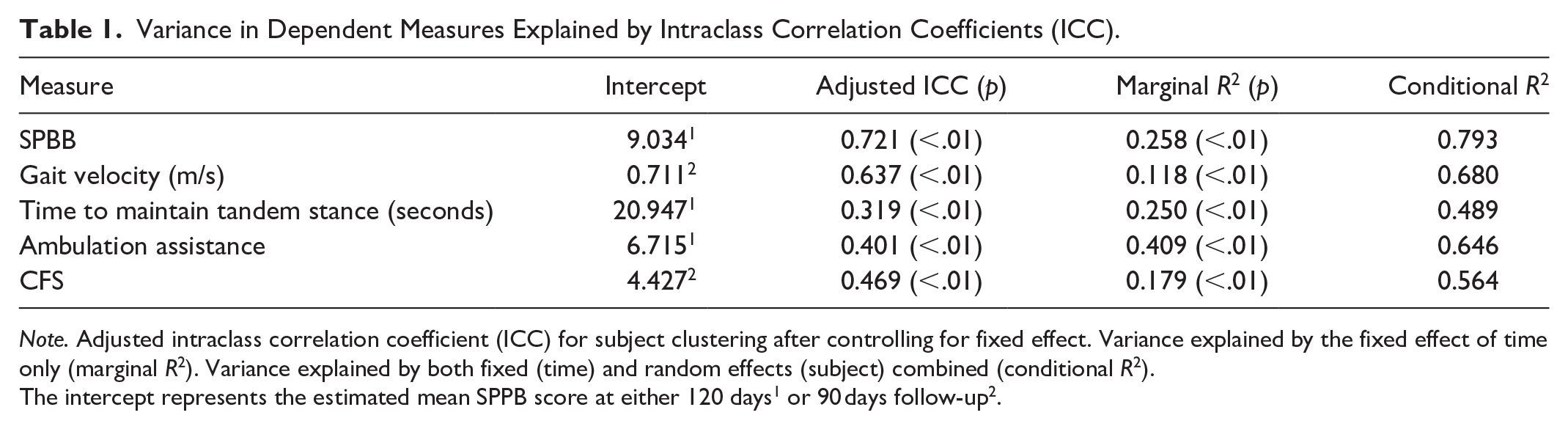
Note. Adjusted intraclass correlation coefficient (ICC) for subject clustering after controlling for fixed effect. Variance explained by the fixed effect of time only (marginal R2). Variance explained by both fixed (time) and random effects (subject) combined (conditional R2).
The intercept represents the estimated mean SPPB score at either 120 days1 or 90 days follow-up2.
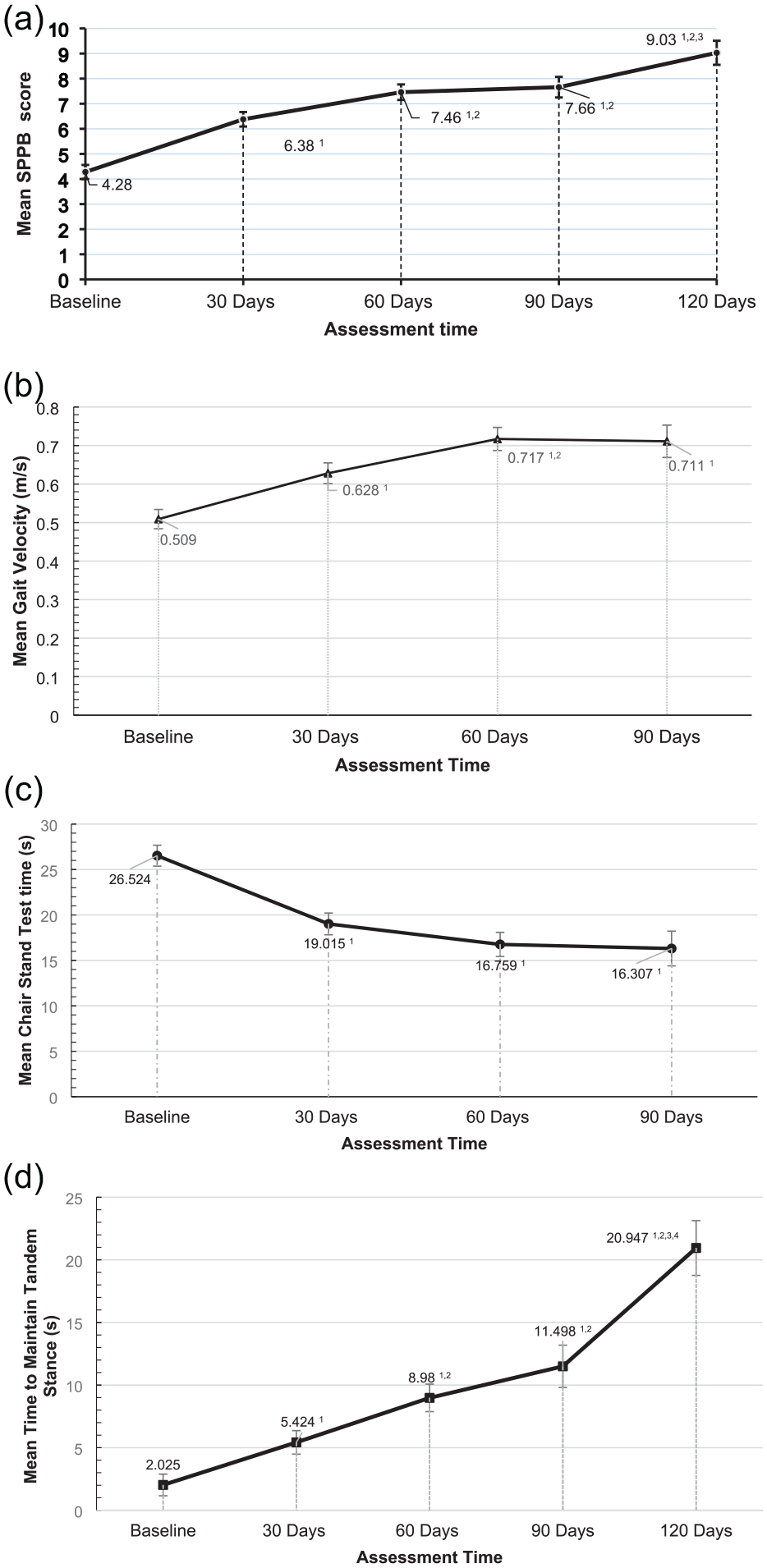
Change in physical performance scores as a function of participation in the Vitality Therapy™ program intervention. (a) Change in short physical performance battery scores compared to baseline at 30, 60, 90, and 120 days of the program. (b) Change in gait velocity (m/s) compared to baseline at 30, 60, and 90 days of the program. (c) Change in the time to complete the chair stand test (s) compared to baseline at 30, 60, and 90 days of the program. (d) Change in time to maintain a tandem stance on the balance test (s) compared to baseline at 30, 60, and 90 days of the program.
There was also a significant beneficial effect of the program (F4,139.13 = 49.98, p < .01) on ambulation assistance, as patients gained significant mobility and independence after 30 (M = 5.32, SE = 0.13), 60 (M = 6.06, SE = 0.15), 90 (M = 5.85, SE = 0.23), and 120 days (M = 6.72, SE = 0.26) respectively, compared to baseline (M = 4.11, SE = 0.12). These results are summarized in Figure 3.
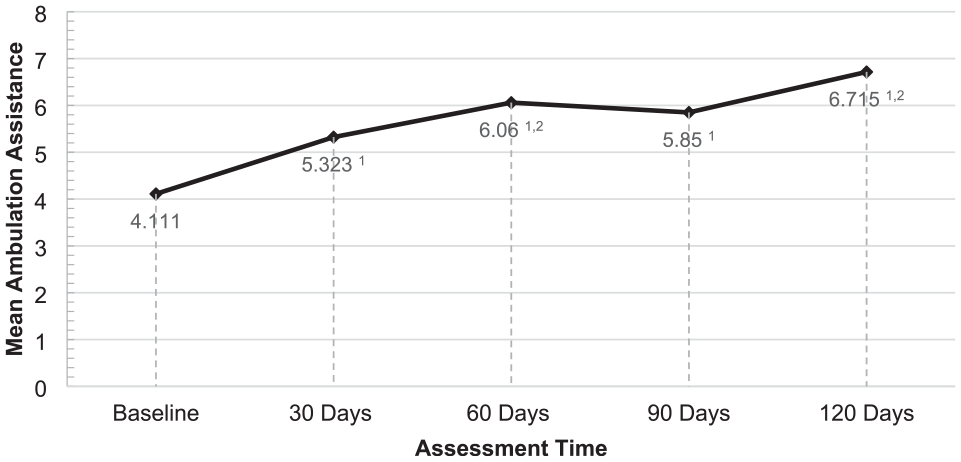
Change in ambulation assistance scores compared to baseline at 30, 60, 90, and 120 days of the Vitality Therapy program intervention.
A significant improvement was also observed on the Clinical Frailty scale (F3,77.37 = 18.24, p < .01) with patients improving their frailty score from 5.67 (SE = 0.17) at baseline to 4.43 (SE = 0.28) after 90 days in the program (see Figure 4).
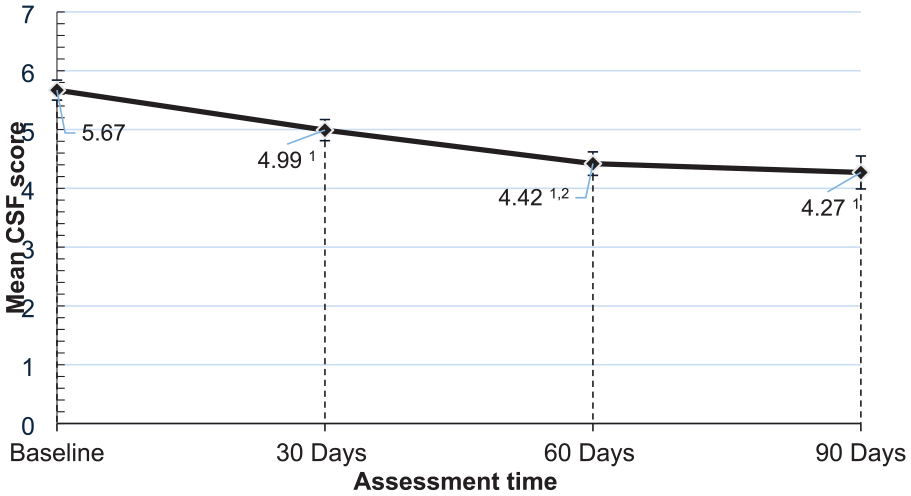
Change in clinical frailty scale (CFS) score as a function of the Vitality Therapy program intervention.
Discussion
We designed a home healthcare program aimed at reversing frailty in older adults, and we found that the program improved the SPPB composite score (as well as the gait velocity, chair-stand and balance test components of the SPPB), improved ambulation independence and decreased frailty. To our knowledge, a frailty-specific rehabilitation program fitting within the Medicare home health benefit has not previously been reported. These improvements are clinically important. At baseline, this geriatric cohort had a clinical frailty score of 5.67 (SD = 0.81, range: 4-7), which means that these patients are living with moderate frailty, and typically need help with all outside activities and with keeping house. Inside, they often have problems with stairs and need help with bathing, and they might need minimal help with dressing. At this level, falls risk is high—moderately frail patients frequently fall, and sometimes have a catastrophic fall—that may result in serious and life-altering injuries such as a fracture or an intracranial bleed. After the intervention, this cohort had a CFS of 4.27 (SD = 0.57, range: 4-5)—a score associated with people who are living with very mild frailty and whom are not typically dependent on others for daily help. Similarly, we found that before the VT intervention, patients had a mean 4.11 (SD = 0.84; range: 2-6) functional mobility score, requiring contact guard assistance. After the intervention at 120 days, this group had an average functional mobility score of 6.72 (SD = 0.98; range: 5-8)—which means that on average, patients only required minor modifications in their activities to be functionally independent. An improvement of this magnitude could mean the difference between needing a family member or paid caregiver to provide continuous (at least daytime) supervision and assistance, versus only occasional assistance, and can possibly be associated with a cost savings of approximately $3000 per month (based on an estimated baseline caregiver need of 8 hours per day, 7 days per week at $18/hour compared with a post-VT program caregiver need of 2 hours per day, 7 days per week at $18/hour). Finally, the nearly five-point increase in the SPPB that patients experienced was also clinically significant. A patient with a score of 4 on the SPPB typically has diminished muscle strength impairing the ability to transfer, significantly impaired balance increasing the risk for falls, and diminished gait speed. In contrast, a patient with a score of 9 typically has adequate functional strength and balance to support safe transfers, and a gait speed that supports home ambulation. 20 The increase observed in gait velocity from baseline to 90 days of 0.2 m/s is clinically significant. Middleton et al showed that such a change in gait velocity is associated with a significantly lower risk of death, hospitalization, falls and cognitive decline, and can also predict an older adult’s ability to be more independent with self-care following hospital discharge. 21
As frail older adults experience high rates of mortality, acute care utilization, dependency, long-term care institutionalization, and total cost of care over $71,000 per year, 3 a simple and effective home-based program that utilizes the existing Medicare home health benefit would be a welcome strategy to improve the lives and well-being of older adults, while potentially decreasing hospitalizations and cost. This study was limited by its observational nature, and future studies on whether the VT intervention reduces hospitalizations or total cost of care are needed.
Conclusions and Implications
A novel anti-frailty home health program improved physical performance measures and increased independence in frail older adults. Expansion of home healthcare programs targeted at reversing frailty could provide increased access to an impactful intervention for this vulnerable population.
Footnotes
Acknowledgements
None.
Brief Summary
A novel home health program targeted at frail older adults improved physical performance measures and decreased frailty.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: WM, RLS, STB, SAB, AB, JDN, JS are employees, and stockholders in organization a. DP is an employee of organization c.
Declaration of Generative AI and AI-assisted Technologies in the Writing Process
No forms of AI were used in the preparation of this manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
