Abstract
Hearing loss affects approximately 30 million people in the United States. It has been estimated that only approximately 20% of people with hearing loss significant enough to warrant amplification actually seek assistance for amplification. A significant interest in middle ear implants has emerged over the years to facilitate patients who are noncompliant with conventional hearing aides, do not receive significant benefit from conventional aides, or are not candidates for cochlear implants. From the initial studies in the 1930s, the technology has greatly evolved over the years with a wide array of devices and mechanisms employed in the development of implantable middle ear hearing devices. Currently, these devices are generally available in two broad categories: partially or totally implantable using either piezoelectric or electromagnetic systems. The authors present an up-to-date overview of the major implantable middle ear devices. Although the current devices are largely in their infancy, indications for middle ear implants are ever evolving as promising studies show good results. The totally implantable devices provide the user freedom from the social and practical difficulties of using conventional amplification.
Background
Hearing loss affects approximately 30 million people in the United States. The prevalence increases with age, and more than one third of people older than 65 years have clinically significant hearing loss. It has been estimated that only approximately 20% of people with hearing loss significant enough to warrant amplification actually seek assistance for amplification. Of these, as many as 15% do not wear their devices (Kochkin, 2005). Within the past decade, technological improvements have greatly improved the usability of conventional aids; however, many limitations remain. Among the numerous reasons for low compliance are acoustic feedback, occlusion effect, frequent battery changes/maintenance, discomfort, infection, irritation, lifestyle restrictions, and repair, loss, or damage. Faulty circuitry, suboptimal fit, or intrinsic external auditory canal issues can lead to intolerable feedback. Poor sound quality and difficulty hearing in crowded situations are frequent complaints of those who wear conventional hearing aids.
The social stigma of wearing hearing aids may preclude some from using or even considering the devices. In today's society, where appearance and youth are socially valued, the stigmata of appearing older than their stated age with hearing aids may further deter potential users. In addition to the mild to moderate hearing loss from 20 to 60 dB that can be ameliorated by hearing aids, there is the group of patients with hearing loss of approximately 50 to 90 dB who may receive greater benefit from other technologies such as middle ear implants. As such, a significant interest in middle ear implants has emerged over the years to facilitate patients who are noncompliant with conventional hearing aids, do not receive significant benefit from conventional aids, or are not candidates for cochlear implants.
Brief History
Many credit the evolution of middle ear implants with Wilska's (1935) experiment with iron particles placed on the tympanic membrane. Wilska was able to generate a magnetic field from an electromagnetic coil inside an earphone, which caused the iron particles to vibrate with the magnetic field. The subsequent vibration was transmitted to the tympanic membrane, which subsequently transduced sound to the inner ear in the physiologic manner. Further significant studies were largely stalled until Rutschmann (1959) documented stimulation of the ossicles with alternating magnetic fields acting on a magnet affixed to the tympanic membrane. Other developments were documented in the 1970s when devices were created to be surgically placed into the middle ear. These early studies, albeit far from successful, helped lay the foundation for subsequent clinical trials evaluating the feasibility of middle ear implants. This eventually led to trials in humans that looked at piezoelectric-type devices attached to the stapes and used an external microphone and sound processor (Yanagihara, Sato, Hinohira, Gyo, & Hori, 2001). Needless to say, the technology has greatly evolved over the years with a wide array of devices and mechanisms employed in the development of implantable middle ear hearing devices. Currently, these devices are generally available in two broad categories: partially or totally implantable using either piezoelectric or electromagnetic systems.
Rion Device
Historically, it is worthwhile to mention one of the first piezoelectric devices, the Rion Device E-type, which has been used for both conductive and sensorineural losses. The device was partially implanted with the vibratory portion attached to the squamous region of the temporal bone. This device worked by passing an electric current into a piezoceramic crystal attached to the incus or stapes capitulum. Yanagihara first reported implantation of the device in 1987 and later reported long-term results showing that the device can safely run for 10 years with natural sound quality and high patient satisfaction (Yanagihara et al., 2001). However, decreased efficacy of the device from 36 to 21 dB was attributed to the sensitivity of the ossicular vibrator caused by aging and to tissue reaction around the vibrator element impeding its function (Yanagihara et al., 2001). Limited additional data are available on the performance of the Rion device.
Vibrant Soundbridge (VSB) Device
One example of a partially implanted electromagnetic device is the VSB (see Figures 1 and 2). Symphonix Devices, Inc. (San Jose, CA) developed the Soundbridge, which was the first FDA-approved implantable middle ear hearing aid to treat sensorineural hearing loss, receiving the designation in 2000. The product has since been acquired by Vibrant Med-El (Med-El Corporation, Innsbruck, Austria). The VSB is unique in that it takes an innovative approach to make sound audible: It mechanically causes the ossicles to vibrate, and the amplified vibrations may be adjusted to optimally compensate for hearing loss. Specifically, vibration of the device is transmitted to the ossicles, thereby delivering stimuli to the middle and, subsequently, inner ear.

Soundbridge Device.
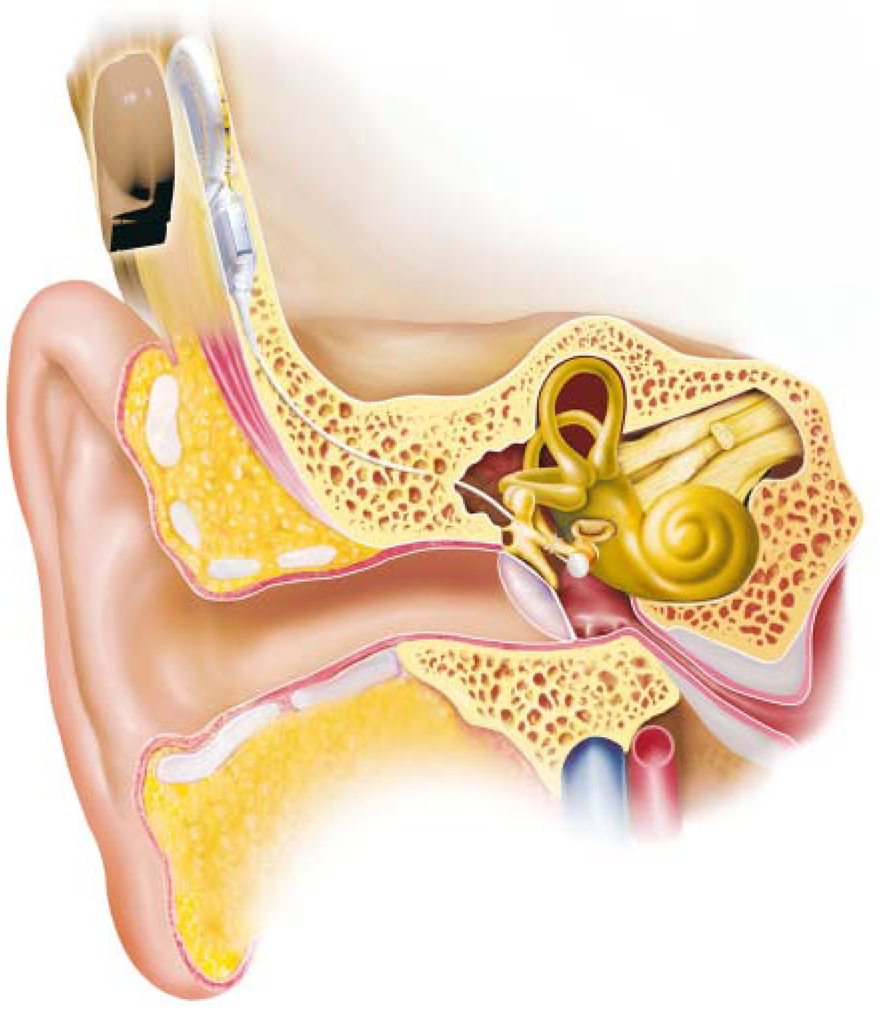
Soundbridge Device Implanted.
The Soundbridge is semi-implantable and is composed of an external microphone, sound processor and amplifier, an audio processor, and an internal vibrating ossicular prosthesis. Sound waves are detected by the microphone located behind the ear and then processed by an implanted receiver. The vibrating ossicular prosthesis conducts the sound to a magnet surrounded by a coil called the floating mass transducer. The transducer is attached to the long process of the incus, and the magnet abuts the long axis of the stapes, which causes it to vibrate. The device uses a battery, and the life depends on usage and device settings. A typical patient who uses his or her device for 12 to 16 hours a day may need to change the battery about once a week.
Phase III trial results noted that the device was safe, and 94% of patients stated an improvement in overall sound quality over conventional hearing aids (Luetje et al., 2002). Another study concluded that the VSB remained a highly reliable middle ear implant system both surgically and audiologically (Labassi & Beliaeff, 2005). This has been further corroborated by a study demonstrating that the performance of the Soundbridge does not deteriorate with more than 5 years of follow-up. These results confirm the safety and the effectiveness of the VSB with a longer term follow-up (Mosnier et al., 2008). In comparison with conventional hearing aids, a prospective study demonstrated that direct-drive stimulation provided by the VSB allows better speech performances than acoustic stimulation for patients with steeply sloping high-frequency hearing losses (Truy, Philibert, Vesson, Labassi, & Collet, 2008).
Additionally, Cremers, Verhaegen, and Snik (2009) recently concluded that when the incus is absent, placement of the floating mass transducer directly on to the stapes is an acceptable solution. This finding led to further studies designed at using a middle ear implant capable of directly driving the cochlear fluids by placement of the device on the round window. The initial studies, however, showed significant variability in hearing gain, likely reflecting variability in responsiveness of the cochlea associated with chronic pathologic findings and, possibly, variability in how the VSB effector interfaces with the round window. The authors further suggested that modifying the shape of the VSB effector can improve the mechanical coupling to the round window to better exploit the device's amplification power (Beltrame, Martini, Prosser, Giarbini, & Streitberger, 2009).
More recently, in cases of atresia patients, it is proposed that by circumventing the malformed middle ear and directly stimulating the cochlea, the VSB may provide a new rehabilitation option for these patients. The mean threshold with the VSB activated in the free field warble tone audiometry was 23.8 dB hearing level (HL). Mean functional gain was 45.5 dB HL. Mean aided free field speech discrimination in quiet was 64% at 50 dB, 99% at 65 dB, and 100% at 80 dB. The authors concluded that the use of this middle ear implant is both safe and effective and can be implemented in combination with outer ear reconstruction (Frenzel et al., 2009). Another indication could potentially be with patients possessing combined hearing loss after undergoing subtotal petrosectomy with complete fat obliteration of the middle ear and mastoid area. A recent study concluded that the VSB can be used to safely rehabilitate these patients by placing the floating mass transducer of a VSB device onto the round window membrane either at the time of primary surgery or as a staged secondary procedure (Linder, Schlegel, DeMin, & van der Westhuizen, 2009).
Soundtec Direct System
The Soundtec system (Soundtec, Inc., Oklahoma City, OK; see Figure 3) was initially introduced in the United States in 2001. This semi-implantable device converts sound waves to electromagnetic energy, which in turn directly stimulates the ossicular chain. A surgically placed neodymium–iron–boron magnet is placed around the neck of the stapes. An earmold with an embedded electromagnetic coil stimulates the magnet. The coil assembly is placed into the ear canal approximately 2 mm away from the tympanic membrane. A sound processor is fit typically either posterior to the auricle or in the external auditory canal.

Soundtec.
Of note, this system requires temporary separation of the incudostapedial joint for placement of the magnet. Theoretically, this maneuver could result in mobility of the stapes footplate with resultant senorineural hearing loss. Earlier studies indicated positive outcomes regarding safety and efficacy. Results also demonstrated improvement in performance compared with the subjects' optimally fit hearing aids and an improvement in quality of life as demonstrated by objective and subjective tests and measures (Hough, Matthews, Wood, & Dyer, 2002). Additionally, physical and mechanical testing of the Soundtec implant indicates that the structural integrity of the ossicles will be maintained during 0.3-T magnetic resonance imaging (MRI) of the human head (Dyer, Dormer, Hough, Nakmali, & Wickersham, 2002).
Other studies with the Soundtec direct system suggested that the device produced an average functional gain of 26 dB (Silverstein, Atkins, Thompson, & Gilman, 2005). Fifty-five percent of patients complained of magnet movement; this was eliminated in 80% of Silverstein Institute patients when the external processor was worn, and fat was used to stabilize the magnet. Furthermore, the Soundtec direct device was well tolerated in the majority of patients, with a significant increase in functional gain. Magnet instability and noise were the most frequent complaints, which improved with processor placement and anchoring the magnet with fat. This electromagnetic semi-implantable hearing aid confers greater functional gain over conventional hearing aids and reduces occlusion effect and feedback (Silverstein et al., 2005). Further studies on electromagnetic devices led to an FDA trial with Soundtec in 2001.
Studies then began to emerge comparing conventional technology with the more recent implantable devices. Soundtec reported that patients had improved speech discrimination in crowded situations/background noise and higher functional gain. Furthermore, patients stated a preference to the device in comparison with traditional hearing aids (Matthews, Wood, & Dyer, 2002). The device was voluntarily withdrawn from the market in 2004 when the company decided to identify ways to improve it and to eliminate the distortion some patients experienced. To date the release of the updated product has not occurred.
Totally Integrated Cochlear Amplifier (TICA)
One of the first totally implanted devices, the TICA (Implex American Hearing Systems), is now owned by Cochlear Corporation, Sydney, Australia (Figure 4). The TICA has the greatest use in Europe and was first implanted there in 1998. In addition to the transducer, the microphone is placed beneath the skin of the auditory canal, allowing the skin to cover the microphone membrane completely. A digitally programmable processor located subcutaneously on the mastoid bone processes the signal, which is then translated to a piezoelectric coupler in direct contact with the body of the incus. The device is implanted through a mastoidectomy, and the device can be charged transcutaneously within 90 minutes. The battery is estimated to last for approximately 5 years. In one study, the authors concluded that the TICA implantation may be indicated when a patient fulfills three primary criteria: lack of benefit from conventional hearing aids, moderate to severe high-frequency sensorineural hearing loss, and adequate space in the mastoid for implantation (Zenner & Leysieffer, 2001). To the knowledge of the authors, no further clinical trials have been completed in the United States to date.

Totally integrated cochlear amplifier.
Otologics
The Otologics device (Otologics, LLC, Boulder, CO; Figures 5 and 6) was initially developed as a semi-implantable middle ear transducer (MET) that was available in Europe by 2000; subsequently, a totally implantable device (marketed as Carina) has been available since 2006. The device consists of the implant, the programming system, the charger, and the remote control. The implant itself has the electronics, which contains the microphone, battery, magnet, digital signal processor, and connector. The system uses a microphone located under the skin that picks up sounds that are amplified and converted into an electrical signal. The signal is sent down the lead and into the transducer, and the MET ossicular stimulator is coupled directly to the ossicular chain. While the wearer performs normal daily activities, the device can be charged, the implant can be turned on and off, and the volume adjusted. To charge the implant, the patient removes the charger body from the base station and places the coil on the skin, over the implant site. The charger body contains a clip that allows the charger to be attached to the belt of the wearer during charging. Typically, charging time will be about 60 minutes and must be performed daily. Early preclinical research demonstrated that the MET ossicular stimulator can effectively benefit patients with moderately severe to severe sensorineural hearing loss. The MET ossicular stimulator is now in an FDA-approved clinical study. To demonstrate benefit over conventional hearing aids, the MET ossicular stimulator is compared with state-of-the-art digital conventional hearings aids using the same signal processing programs. The device program and performance are then verified in the clinic using referenced and calibrated measurement tools (Kasic & Fredrickson, 2001).

Otologics Device Implanted.

Otologics Device.
The U.S. Phase I trial results yielded a 15 to 20 dB functional gain across audiometric frequencies in 20 patients. The monaural word recognition scores and pure-tone averages were better with the hearing aid in the same ear preoperatively, whereas the patients generally perceived a more substantial benefit in the postoperatively implant-aided conditions (Jenkins, Niparko, Slattery, Neely, & Fredrickson, 2004). Fully implantable clinical trial results were 10 to 20 dB better than the semi-implantable trial results, with less variability at all frequencies. These findings indicate more consistent and better coupling efficiency of the transducer to the ossicles (Jenkins et al., 2004). Use of the recently introduced intraoperative loading instrumentation during surgery improves the coupling efficiency and consistency of a fully implantable hearing device to the ossicular chain and ultimately leads to better patient performance with the device (Jenkins, Pergola, & Kasic, 2007).
In another study, audiometric results demonstrate that the device can be implanted without affecting residual cochlear hearing levels. Based on preliminary experience, the device serves as a viable treatment alternative for moderate to severe sensorineural hearing loss (Jenkins, Atkins, et al., 2007). In a recent study, the safety and performance of the fully implantable otologics device was performed in a small group of patients with mixed conductive and sensorineural hearing loss. Preliminary results of this trial yielded evidence that the device is capable of efficiently transferring the sound to the inner ear through the round window membrane in patients with mixed hearing loss (Lefebvre et al., 2008).
Envoy Esteem
Another totally implantable device is the Esteem (Envoy Medical, St. Paul, MN; Figure 7). The original development began in late 1980s when the patent was owned by Medical Devices Group (Madison, WI). Subsequently, in 1995, the patent was acquired by St. Croix Medical Corporation based in Minneapolis, MN. In 2000, European surgeons began to implant patients in a feasibility study. Since then, the company has developed a totally implantable device. A Phase I U.S. FDA study was started in 2002 (Kroll, Grant, & Javel, 2002). This device uses the eardrum as the microphone, taking advantage of the natural acoustics of the ear canal without obstruction, interference, or any external devices. The device uses a sensor placed at the body of the incus that sends an electrical current to a sound processor. This in turn is amplified, filtered, compressed, and converted back to a mechanical stimulus. The mechanical stimulus/vibratory signal is then delivered to the stapes capitulum. Of note, 3 mm of the lenticular process of the incus must be removed surgically for the device to work properly. Once the device is programmed, the user can adjust the volume and filter settings. The Envoy Esteem I is totally implantable with no external components and allows 24/7 hearing. Other added benefits include no microphone and no recharging of battery. The battery is estimated to last for approximately 3 to 5 years and can be replaced by a relatively simple outpatient procedure that takes 30 to 45 minutes.

Envoy.
The Esteem may provide for a more “natural” sound by using the body's own hearing mechanisms. Furthermore, the ear canal resonance and pinna effect for total pinna and resonance effect are important for maximum acoustic gain.
The success of the Esteem I has led to the further development of the Esteem II. This device employs an advanced processor and a broader fitting range. Battery life has been expanded from 5 to 8 years. The Esteem II Phase II clinical trial began in early 2008. Esteem II technology can be regarded as a safe and reliable system for hearing restoration in the case of moderate to severe sensorineural hearing loss. In one study, the authors concluded that the Envoy middle ear device may, in the near future, well play an important role in helping people affected by moderate to severe sensorineural hearing loss, both from the functional benefit and the evident cosmetic advantage (Barbara, Manni, & Monini, 2009).
Baha
The Baha (Cochlear Corporation, Sydney, Australia; Figures 8 and 9) is an implantable device indicated for both conductive and unilateral mixed or sensorineural hearing losses. Although the device is not a conventional implantable hearing device in the true sense of the phrase, the topic will be briefly broached here as it is a common device that is well represented in the literature. The device has been in use since the late 1970s in Europe and employs a percutaneous osseointegrated titanium post implanted in the post auricular area and an external sound processor attached to the post. The microphone of the sound processor picks up vibrations and transfers them to the functioning cochlea by bone conduction. Once the cochlea receives these sound vibrations, the organ “hears” in the same manner as through air conduction; in the cochlea, the sound is converted into neural signals and is transferred to the brain. Approximately 3 to 4 months are required after surgical placement to allow for osseointegration before the external processor can be attached and its benefits actualized. A major advantage of the Baha is the elimination of the head-shadow effect, improved speech recognition in noise, and elimination of any occlusion effect from a hearing aid or a CROS aid. Recent studies confirm both the short- and long-term efficacy of the Baha in adults with single-sided deafness for recognition of speech in noise (noise in front, speech lateralized to the bad ear) and the subjective measures of benefit (Linstrom, Silverman, & Yu, 2009). Improvement in speech understanding occurred when the primary signal was spatially separated from background noise. Given these overall improvements in speech understanding, patients were found to be satisfied with their Baha and would still elect to have this procedure if given a second chance (Newman, Sandridge, & Wodzisz, 2008). Additionally, quality-of-life studies have demonstrated that the use of a Baha in children significantly enhanced general well-being and improved quality of life (McDermott, Williams, Kuo, Reid, & Proops, 2009).

Baha Device.

Baha Device Implanted.
The complication rate is generally low but typically involves soft tissue reaction and/or osseointegration. One series reported that 4.5% of Baha users required revision surgery for soft tissue complications and 1.3% needed revision for failure of osseointegration. Eight percent required local care and their complications resolved within 2 to 3 weeks of treatment (Wazen et al., 2008). Five percent of Baha users required in-office procedures. Ninety-two percent reported using the device regularly, and 77% were happy with the device (Falcone, Kaylie, Labadie, & Haynes, 2008). The authors concluded that the Baha system is safe and effective in the rehabilitation of patients with conductive or mixed hearing losses and with single-sided deafness. Newer studies indicate that site reactions and skin overgrowth can be fairly easily remedied with the use of clobetasol. The authors concluded that clobetasol allows patients to resume Baha use and obviates the need for scar revision (Wazen et al., 2008). Furthermore, safety in MRI scanners was confirmed in a recent study showing that the bone-mounted post resisted any magnetic forces trying to displace it up to 9.4 T. Two patients underwent scanning at 1.5 and 3.0 T with no adverse effects. MRI image quality was excellent starting at 1 cm from the post. The study findings lend strong evidence that patients can be safely scanned up to 9.4 T without adverse effects (Fritsch, Naumann, & Mosier, 2008). The high success rate, patient satisfaction rate, and predictable auditory outcome place the Baha among the leading choices for auditory rehabilitation when clinically warranted (Falcone et al., 2008).
Summary
Indications for middle ear implants are ever evolving as promising studies show good results. Implanted devices act by directly stimulating the ossicles (and more recently directly stimulating the round window), thereby bypassing the external auditory canal. Further studies such as these, with head-to-head comparisons of the current gold standard, conventional hearing aids, and middle ear implants, are warranted in the future. As the technology advances, devices that are totally implanted may play an important role by using the eardrum as the microphone and using the pinna and external auditory canal to collect sound in a natural manner.
The initial goals of development included increased efficacy, quality-of-life issues, and transparency to the patient and others. For efficacy, the goal would be improved gain, sound quality, hearing-in noise, and the elimination of acoustic feed back. In terms of quality of life, the ideal middle ear implant would allow hearing 24 hours a day and 7 days a week in all environments. Specifically, one would be able to use the device while showering and/or participating in aquatic activities. The transparency, though not the most important aspect in terms of technology, cannot be understated. The device should be invisible or virtually so to the casual observer with minimal maintenance/cleaning. Ideally, there should be a long battery life (or ease with recharging) and long-term reliability.
Methods of achieving the above stated goals centered on extensive clinical research trials to treat conductive and sensorineural hearing loss. Most implantable middle ear devices are capable of eliminating feedback—a critical feature upgrade compared to conventional hearing aids. Improved cosmesis is also noted by means of concealing and miniaturizing most of the device components. Equally important is the elimination of the occlusion effect.
Most studies indicate that the sound quality of implantable hearing devices is at least comparable to conventional hearing aids though patient satisfaction and quality of life tend to be substantially improved. Furthermore, one study suggests that patients fitted with a VSB or an Otologics MET middle ear implant had hearing results comparable to those of patients fitted with today's conventional hearing aids, though not clearly better. However, the study suggested that both the VSB and Otologics MET are good options in patients with moderate (VSB) to severe (Otologics MET) sensorineural hearing loss and external otitis (Verhaegen, Mylanus, Cremers, & Snik, 2008).
Currently, the cost of the devices and surgical implantation, with concomitant limited insurance coverage, prohibits the widespread use of the devices. Additional cost analysis studies are warranted, though a recent study demonstrated cost effectiveness of implantable hearing devices (Snik, Moulder, Cremers, & Noten, 2006). Furthermore, there may be some surgeon bias in regard to maintenance of such a device; however, one may look to the success of bone-anchored hearing aids and cochlear implants to dispel some of those concerns. There is a huge patient base that could potentially benefit from middle ear implants—easily exceeding the number for those who would qualify for cochlear implantation. The key point is that conventional hearing aids have been available for many years and most patients who would benefit do not use the devices. To meet the needs and concerns of this patient population would likely result in an increased compliance. Implantable middle ear devices are taking the appropriate steps in the right direction to provide the user freedom from the social and practical difficulties of using conventional amplification.
