Abstract
In this study, collagen and inulin were incorporated in soft candy formulations with different ratios. The candies were subjected to textural, chemical, sensory, and correlation analyses in order to evaluate the effects of single or combined use of inulin and collagen on the freshly produced and stored products. Single use of inulin caused decrease in springiness contrary to collagen. Addition of collagen hydrolysate to soft candy formulations increased the pH and softness of the final product directly proportional to the collagen rate. The addition of inulin and collagen (20%) resulted in noticeable lightning in the color of the candies. Combined use with collagen was found to be more effective in promoting the prebiotic activity compared to the use of inulin alone. For Bacillus clausii, the combination of 10% collagen + 50% inulin provided a significantly higher colony growth. While collagen did not negatively affect the taste of confectionery; at 20% ratio the candy was perceived softer and stickier to the teeth. The inclusion of collagen and inulin together in the formulation decreased the sensory scores and slightly lower relative frequency rate was obtained for the “I would definitely buy” category (2–12% less than that of the other candies). The study revealed that if collagen and inulin are included in the formulation together, the confectionery can provide a significant amount of collagen by strengthening the structure with the support of an extra polysaccharide addition, and at the same time, inulin will be beneficial in terms of calorie reduction.
Introduction
The consumption volume of confectionery products has increased continuously around the world. It is thought that the world market for confectionery products was $210.3 billion in 2019 and will reach $270.5 billion in 2027, with an annual development share of 3.6% (Global Confectionery Market Report, 2019). Nowadays, sucrose or sucrose-containing foods are increasingly consumed; since it is associated with some health problems such as obesity, tooth decay, heart diseases, and some types of cancer and diabetes (Grembecka, 2015), interest in functional confectionery is increasing and these products are also desired not only to contain bioactive compounds but also to be “low calorie” and/or or “sugar-free” (Dorn et al., 2015).
Collagen is the most abundant structural protein in animals, accounting for approximately 30% of the protein content in mammals. By thermal partial hydrolysis of collagen polypeptides, small peptides of low-molecular weight, called hydrolyzed collagen, are formed. Among the 28 different types of collagen, the most abundant types include fibril-forming collagens and types I, II, and III, which are related to tissue strength, mechanical integrity, elasticity, and water retention capacity (Kuivaniemi and Tromp, 2019). Collagen peptides have chemotactic properties in the blood circulation, as well as helping the skin, hair, and restoration process (Lupu et al., 2020). These proteins are structural components of human vascular and joint tissues (Dzyuba et al., 2019). With age and poor nutrition, collagen production in the body decreases, tissues become weaker and less flexible. Collagen supplements are intended to protect users’ skin, nails, hair, and body tissues. Collagen metabolites increase the thickness and elasticity of tissues and increase hydration in tissues. Collagen supplementation can increase lean muscle gain, reduce tissue recovery time, reconstruct damaged joint structure, and improve cardiovascular performance (Hashim, 2015). Hydrolyzed collagen peptides have attracted great attention in the food, cosmetic, pharmaceutical, biomedical, and leather industries due to their properties such as bioavailability, biocompatibility, and poor antigenicity (Bhadra et al., 2021). Collagen is used in juices and soups to increase nutritional value and add functionality. In the United States, bone broth has been recognized as a healthy alternative to coffee. Malaysia Dairy Industries reported that adding collagen peptides to probiotic drinks containing prebiotic fiber along with vitamin C affects the growth of beneficial intestinal flora (Hashim, 2015). The hydrolyzed form of collagen is water-soluble, has high absorption in the body, and is preferred for cold-stored foodstuffs during low-temperature storage without affecting the sensory properties of the product (Wang et al., 2015).
Prebiotics are fermentable components that come to the large intestine without being digested by enzymes in the small intestine and enable the metabolism to benefit from the microorganisms in the best way by selectively developing and/or increasing the activities of some beneficial bacteria in the intestinal flora (Rolim, 2015). While prebiotics inhibit the development of pathogenic microorganisms by regulating the activity and/or development of beneficial microflora in the intestine, which aids digestion, strengthens the immune system, and positively affects nutrient absorption. Prebiotics are used in the food industry due to their functional properties, nutritional fiber content, prebiotic effects, and low-calorie value. It stimulates the development of bifidobacteria in the intestines. Inulin is a “fructan”—that is, it is a complex carbohydrate composed of chains of fructose molecules. Inulin is found mostly in endive, Jerusalem artichoke, leek, garlic, and onion (Ilievska et al., 2020). Inulin polymer consists of a long chain of 2–60 fructose molecules linked together by β-(2-1) bonds. The terminated fructose molecule is connected to a glucose molecule via an α-(1-2) bond. As a result of enzymatic hydrolysis, oligofructose is obtained from inulin (Shoaib et al., 2016). The calories of inulin, with a value of 1.39 kcal/g, are only 40–50% of digestible carbohydrates (Kaur and Gupta, 2002). It is suitable for diabetics because it has low calories and a low glycemic index and is beneficial to diabetic patients in high doses (40–100 g/day) (Delgado and Bañón, 2018). Inulin, which passes into the small intestine without being digested and ferments in the large intestine, is therefore used in functional foods with its prebiotic properties (Franck, 2002).
The studies on the use of hydrolyzed collagen peptide in candy production are scarce. A recently published study by Blinnikova et al. (2020) could be found which was concerned with the development of a mathematical method in order to design recipes including fruit puree, fruit powder, and collagen hydrolysate solution enabling partial replacement of sucrose. The study does not include the effects of collagen on product properties. In a study where an okara (soybean pulp) protein extract was used in the production of gummy candy containing gelatin, modified starch, and xylitol, it was reported that candies with a high okara protein content had a better taste than others and received the highest praise in terms of chewiness and sensory properties (Liu et al., 2023). Musika et al. (2022) reported that the physicochemical properties of jelly candies enriched with fish collagen (max. 2%) and calcium (1%) were not affected at a statistically significant level. However, it should be taken into consideration that the collagen level used was much lower than in the present study.
In food legislation, for the expression calorie/energy/nutrients to be less/reduced, the amount of nutrients/energy must be reduced by at least 30% compared to the same product. In the energy value declaration, it must be stated which feature of the food results from the total decrease in energy value. With this study, by incorporating inulin and collagen hydrolysate to the jelly-type soft candy formulation, it was aimed to reduce the total sugar in the content by 60% and produce a candy that provides functional benefits and is low in energy. It is possible to come across confectionery products with added collagen in the market, but the amount of collagen in these products is below 10%. Based on the idea that the consumer may prefer products containing higher amounts of collagen in order to approach the daily collagen intake recommendation of 10 g (Adam, 1991), in this study, candy with inulin and collagen added was produced. The candies were produced by using sucrose, corn syrup, inulin, pectin, and gelatin in an appropriate combination with pure collagen hydrolysate. Changes in texture, color, physicochemical parameters, and sensory properties of candies were analyzed. Prebiotic activity and “consumer willingness to buy” were also determined.
Materials and methods
Materials
Corn syrup (dextrose equivalent 40) (Sunar Mısır, Adana, Turkiye), sucrose (general purpose, crystal form, Torku, Konya Turkiye), low set high methoxyl pectin (E440) (Andre pectin APA165B, Yantai Andre Pectin, China), beef gelatin Bloom 220 (Seljel, Gönen, Turkiye), green food coloring (Amoretti, Oxnard, USA), natural pineapple flavor (Symrise, Holzminden, Germany), citric acid and trisodium citrate (Smart Kimya, İzmir, Turkiye), chicory inulin (ORAFTI HSI, Beneo-Orafti, Belgium), and hydrolyzed collagen peptide powder (Collasel, Seljel, Gönen, Turkiye) were used in the production of gummy soft candies. All ingredients were of food grade, chemicals, and reagents were of analytical grade.
Preparation of soft jelly candies
The formulations containing collagen and inulin were designed to partially replace sucrose and corn syrup with these functional ingredients in order to reduce the total carbohydrate and energy content of the candies. Therefore, the observed differences in sucrose and corn syrup levels among samples were intentional and based on the targeted formulation strategy rather than random variation. As a result of the preliminary trials, the formulations obtained by reducing both corn syrup and sucrose at the determined substitution rates are given in Table 1. The method of Tireki (2017) was followed for jelly production with slight modifications. In the study, as an initial step, jelly-type control candy containing sugar, glucose syrup, pectin, and citric acid was produced. Many preliminary tests were carried out with the compositions used in the production of soft candies. In the jelly-type soft candies produced, varying amounts of glucose syrup and sucrose were replaced with inulin and collagen. The sugar ratios in the formulations were reduced by certain amounts of inulin and collagen. However, when the collagen hydrolysate rate exceeded 7%, the candy was too soft to come out of the mold, so the use of gelatin along with pectin was necessary to use a higher amount of collagen. With experiments carried out in this way, the collagen rate could be increased by up to 20%. For this reason, various acid ratios were tried according to collagen increase and the acid ratio that provided the most appropriate texture was applied.
Composition and calorie values of the soft candy samples on dry matter basis.
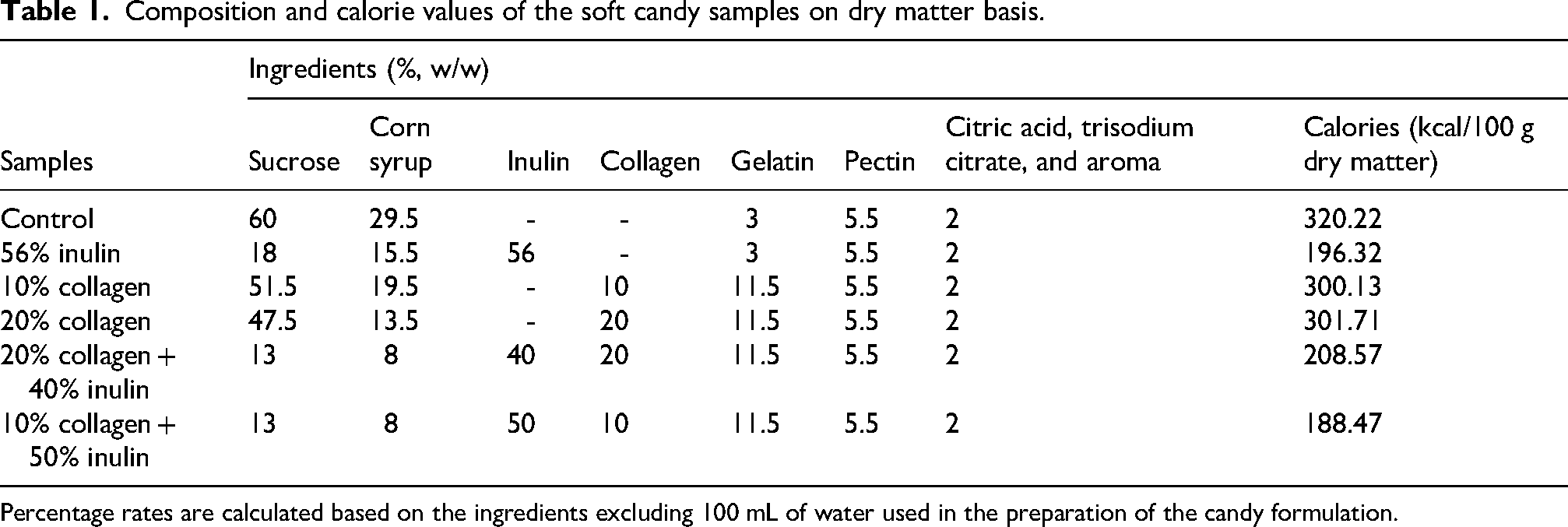
Percentage rates are calculated based on the ingredients excluding 100 mL of water used in the preparation of the candy formulation.
For preparation of sugar mix, corn syrup, sucrose, and water were first heated at 120 °C for 5 min under stirring. The gelatin solution was prepared by dispersing 11.5 g gelatin in 20 mL water, shaking at room temperature for 30 min and stirred at 77 °C. Collagen solution of 50% w/w was prepared by dissolving collagen hydrolysate powder in water at 70 °C. Pectin, inulin (for inulin-contained formulations), and gelatin solution were added in this mix and homogenized by vigorous mixing until a complete dissolution achieved. The hot viscous mixture was cooked at ∼100 °C. For collagen-contained formulations, collagen solution was added and mixed evenly at this step. The cooking process was continued until the Brix value reached 77 by controlling with a refractometer (Atago-Pal Alpha, Japan). Afterward, while the mixture was at approximately 70 °C, citric acid–trisodium citrate solutions and flavoring and coloring agents were added. The jelly mass obtained was immediately poured into silicone molds (16 mm diameter and 10 mm depth), left to cool and solidify at room temperature for 24 h before analysis. The candies were stored at 25 °C in polypropylene pouches for 60 days for storage analysis. Analyses were performed on the 1st and 60th days of storage (Miranda et al., 2020). Soft candies without added inulin and/or collagen hydrolysate were prepared as the control sample. All the samples were prepared in triplicate.
Color analysis
Color indices (L*, a*, and b*) of candy samples were measured using the Minolta Chromameter CR 400 (Minolta Co., Osaka, Japan). The instrument was calibrated by reading the white plate according to CIE Standard Illuminant C. Color values were measured by touching the surface of 10 different points of the candy samples and average values were calculated.
Physicochemical properties of soft candies
The water activity (aw) of the candies was measured by using water activity meter (LabTouch-aw, Novasina, Switzerland). Moisture contents of jellies were determined using the method of Moura et al. (2019). To measure pH, candy samples were shaken well with distilled water (candy:water = 1:9, w/w), stirred until dissolved completely while keeping in a water bath. This solution was cooled at room temperature before the measurements were performed by a pH meter (WTW 3110, Germany) (Delgado and Bañón, 2018). The titratable acidity was determined by titrating the candy samples with 0.05 N NaOH to pH 8.0 (Bhat et al., 2011).
The caloric values of the jelly samples were calculated on a dry matter basis using the theoretical energy contribution of each ingredient. The total caloric value (kcal/100 g dry matter) was determined by summing the caloric values of the individual components, weighted by their proportions in the formulation. The caloric values of the ingredients were obtained from standard food composition databases (sucrose: 386 kcal/100 g; corn syrup: 283 kcal/100 g; gelatin: 61 kcal/100 g; collagen: 348 kcal/100 g; inulin: 139 kcal/100 g; pectin: 60 kcal/100 g).
Texture analysis and determination of glass transition temperature
Firmness and springiness of the candies were determined using a texture analyzer (TA-XT Plus, Stable Micro Systems Ltd, Surrey, UK) equipped with a cylindrical stainless-steel probe (P/35R). Candy samples were prepared in cylindrical geometry with a diameter of approximately 20 mm and a height of 10 mm. Texture analysis was performed at room temperature using a pre-test speed of 1 mm/s, test speed of 1 mm/s, and post-test speed of 10 mm/s, with a deformation of 20%, a probe–sample distance of 10 mm, and a dwell time of 60 s between compressions (Kurt et al., 2022).
Differential scanning calorimetry (DSC) was used to determine the glass transition temperature (Tg) of the candy samples using a DSC instrument (e.g. DSC 214 Polyma, NETZSCH, Germany). Approximately 5–8 mg of sample was weighed into aluminum pans and hermetically sealed, with an empty sealed aluminum pan used as a reference. The samples were initially cooled from 25 °C to −50 °C at a cooling rate of 10 °C/min and held at −50 °C for 5 min to ensure thermal equilibrium. Subsequently, the samples were heated to 100 °C at a rate of 10 °C/min under a nitrogen atmosphere. The Tg was determined as the midpoint of the step change in the heat flow curve. The remaining analysis procedures were conducted according to the method described by Wang et al. (2024). All measurements were performed in triplicate.
Determination of prebiotic activity
The method of Huebner et al. (2007) was followed for the determination of prebiotic activity. Two commercial probiotic supplements Bacillus clausii (Enterogermina, Sanofi, France) and Lactobacillus acidophilus (Natrol LLC, Chatsworth, CA, USA) strains were used in the assay. B. clausii and L. acidophilus strains were added in different ratios (1, 1.5, and 2 capsules and vials) to the test tubes containing 15 mL of Nutrient Broth (Oxoid England) and MRS broth (De Man Rogosa and Sharpe, Merck, Darmstadt, Germany), respectively. Test tubes were incubated at 35 °C for 72 h. Tubes were identified from which aliquots containing 1 × 106 cells were obtained by enumerating on nutrient agar for B. clausii and MRS agar for L. acidophilus. Accordingly, test tubes inoculated with 1.5 capsules (0.42 mg) of L. acidophilus and one vial (5 mL) of B. clausii strain were found suitable for prebiotic activity assays. Test tubes containing 1% (wt/vol) sample were prepared in broth medium. Broth inoculated with 1% glucose (wt/vol) as a non-prebiotic substrate was prepared as positive control; broth without glucose or candy sample inoculation was prepared as negative control. Analysis was performed by adding 0.1 mL of each probiotic strain to the test tubes of the three different media prepared: negative control (regular MRS) (1), positive control (2), sample (3). Incubation was performed at 35 °C for 72 h under anaerobic conditions (85% N2, 10% CO2, and 5% H2). The absorbance of the incubated test tubes was recorded by a spectrophotometer at 600 nm (Biochrom Libra S22, England) on the 7th day of incubation. On the other hand, each test tube was cultivated on agar by the spread plate method with three parallels and colony counts were also enumerated.
Sensory analysis and consumer willingness to buy
The hedonic sensory panel comprised of 108 volunteers with ages in the range of 22–53 years. The age distribution of the panelists was as follows: 55.5% (20–35) and 44.5% (37–49), besides being 48.15% male and 51.85% female. Candies were served on white plates, randomly identified with codes. The service order was also randomized. Panelists were asked to score appearance, color, taste, tooth adhesiveness, and softness/hardness properties of the candies. Panelists also evaluated the candies in terms of “willingness to buy.” Sensory features were evaluated on a 9-point hedonic scale (1 = dislike extremely, 9 = like extremely) (Pashazadehkelisakandi, 2014). The evaluation of “willingness to buy” was performed on a 5-point scale (1 = I would definitely not buy, 5 = I would definitely buy) was applied (Meilgaard et al., 2006). The responses of “willingness to buy” consumer test were presented as a function of frequency. The experiment was approved by the Research Ethics Approval Committee and registered under the EKOR number 2024.00018.0001003.
Statistical analysis
The data obtained were the mean of three replicates (n = 3) and were expressed as the mean ± standard deviation of the obtained values. The means obtained were subjected to statistical analysis by applying “General linear model multivariate analysis” with a significance level of p ≤ 0.05 (SPSS 17.0). Significant differences between the means were determined with Duncan and Least Significant Difference tests. Pearson's linear correlation analysis was evaluated, and a heat map analysis was generated using Microsoft Office 365.
Results and discussion
We can state that the use of inulin enabled significant calorie reduction in candies (Table 1). The calories of collagen (348 kcal/100 g) are approximately twice that of inulin (150 kcal/100 g). Therefore, high amounts of inulin containing formulations (inulin candy and 10% collagen + 50% inulin candy) provided the lowest calorie values.
Physicochemical properties of the candies
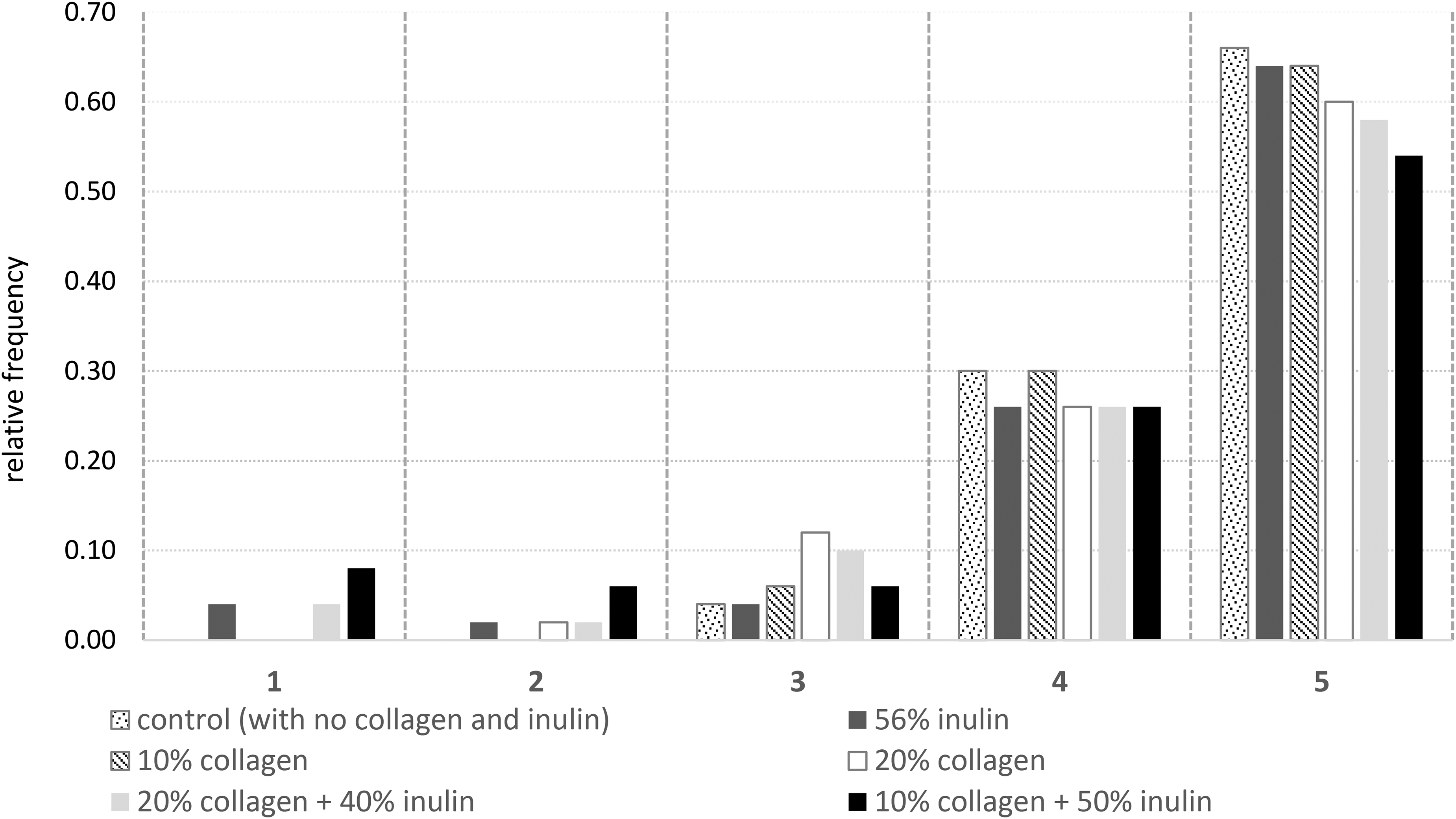
Moisture content directly affects the quality of the final product. It is an important factor for forming the texture and is one of the limiting parameters affecting the shelf life during storage (Ergun et al., 2015). The highest dry matter content before storage was found in control candy (81.17%), and the lowest was measured in samples containing 20% collagen + 40% inulin and 20% collagen. The high moisture content in samples containing 20% collagen can be attributed to water-holding capacity of proteins (Tanaka et al., 2009). Immediately after production, the moisture retained by collagen approached the levels of other collagen-free samples with storage, and there was no significant difference between the moisture values of the stored samples. Demircan's (2019) study on jelly production with high-fiber content showed that the effect of sugar and inulin ratios on the moisture of the final product was similar, but the gelatin solution affected the moisture as the main parameter. The reason for this was explained by the high moisture content of gelatin solution along with the water retention capacity of gelatin.
Hygroscopicity was observed in control and inulin candies with storage, while moisture loss occurred in collagen-added formulations. By storage, moisture values increased approximately around 3% in the control and inulin jellies. The remaining type of jellies lost moisture at the levels changing between 2.8% and 6.9%. Moisture loss was higher in samples containing high percentages of collagen. The initial structures of pectin and gelatin gels are tightly bound to sugar molecules in the fresh sample; the gel structure looseness with the dissolution of sugar during storage. This situation caused more water to enter the structure. The increase in humidity during storage is also caused by corn syrup, as it absorbs moisture easily due to its hygroscopic properties.
Before storage, the highest aw values were determined in the high collagen candy sample (0.75), while the lowest was determined in the control sample (0.61). Components with low-molecular weight and high solubility are effective in reducing water activity (Ergun et al., 2015). At the end of storage, aw of the samples ranged between 0.67 and 0.78, and the lowest aw values were shown by the inulin candy and control candy samples, and the highest aw was detected in the 20% collagen + 40% inulin sample. It was observed that aw increased as the amount of collagen increased. Proteins are compounds that hydrate. Collagen turns into gelatin when heated at 65 °C. Collagen gelation provides significant water retention capacity, which affects the texture and some other quality characteristics of the food. Hydration of collagen is high on the acidic side of the isoelectric point. Between pH 6 and 9, 1 g of collagen can immobilize 10 g of water (Zayas, 1997). In our study, a positive correlation was detected between pH and aw (r = 0.824), of course, it should not be forgotten that our pH values were at most 4.93. Dry matter content is also a parameter that significantly affected the aw of candies (r = −0.817) (Figure 1). Collagen triple helices retain water with aliphatic surfaces cross-linked by sugars (Kudo and Nakashima, 2020). Before storage, in samples with high collagen ratios (20%), aw decreased after storage to levels similar to other samples but use with inulin kept aw high. Inulin is known to increase water-binding ability of myosin which is also a structural protein like collagen (Zhang et al., 2020) and emulsions (Silva-Vazquez et al., 2018).
Heat map of the correlation analysis results of the studied parameters.
The pH values of candies before storage are in the range of 3.76–4.57 (Table 2). The pH value was higher in samples where collagen was included in the formulation. After storage, the pH values of the candies were determined to be between 4.15 and 4.93 with an increase in all the samples. Again, high pH values were detected in samples with collagen, while the control candy sample showed the lowest value. In confectionery products, pH is important for the stability of the gel during baking and before molding. For gelatin-based products, the pH of the solution should be between 4.4 and 5.0 (Lees and Jackson, 1973).
Physicochemical properties of soft candies formulated with inulin and collagen.
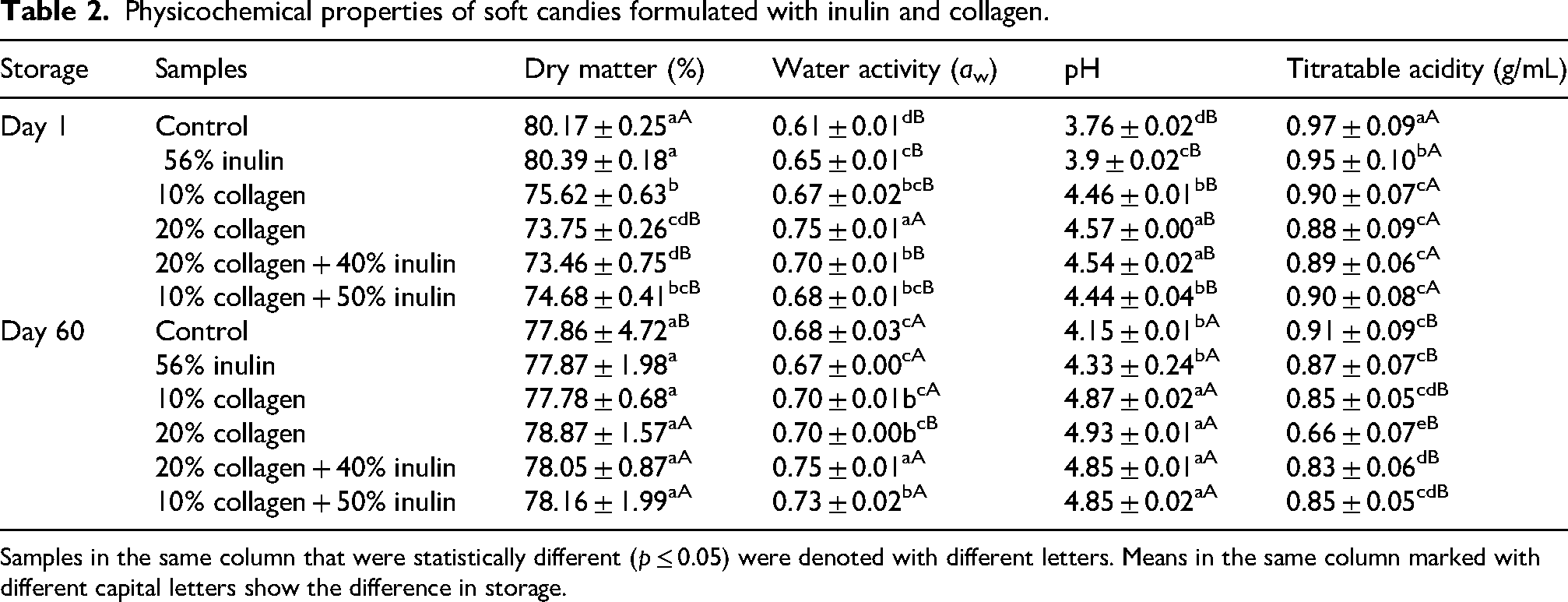
Samples in the same column that were statistically different (p ≤ 0.05) were denoted with different letters. Means in the same column marked with different capital letters show the difference in storage.
Titratable acidity values of candy samples before storage were in the range of 0.66–0.97 (Table 2). Titratable acidity values were lower in collagen-fortified samples. These values decreased during storage. The lowest titratable acidity value belonged to the high collagen candy, according to both pre- and post-storage results. It can be interpreted that the increase in pH values and the decrease in titratable acidity is related to the basic character of amino acids in the collagen.
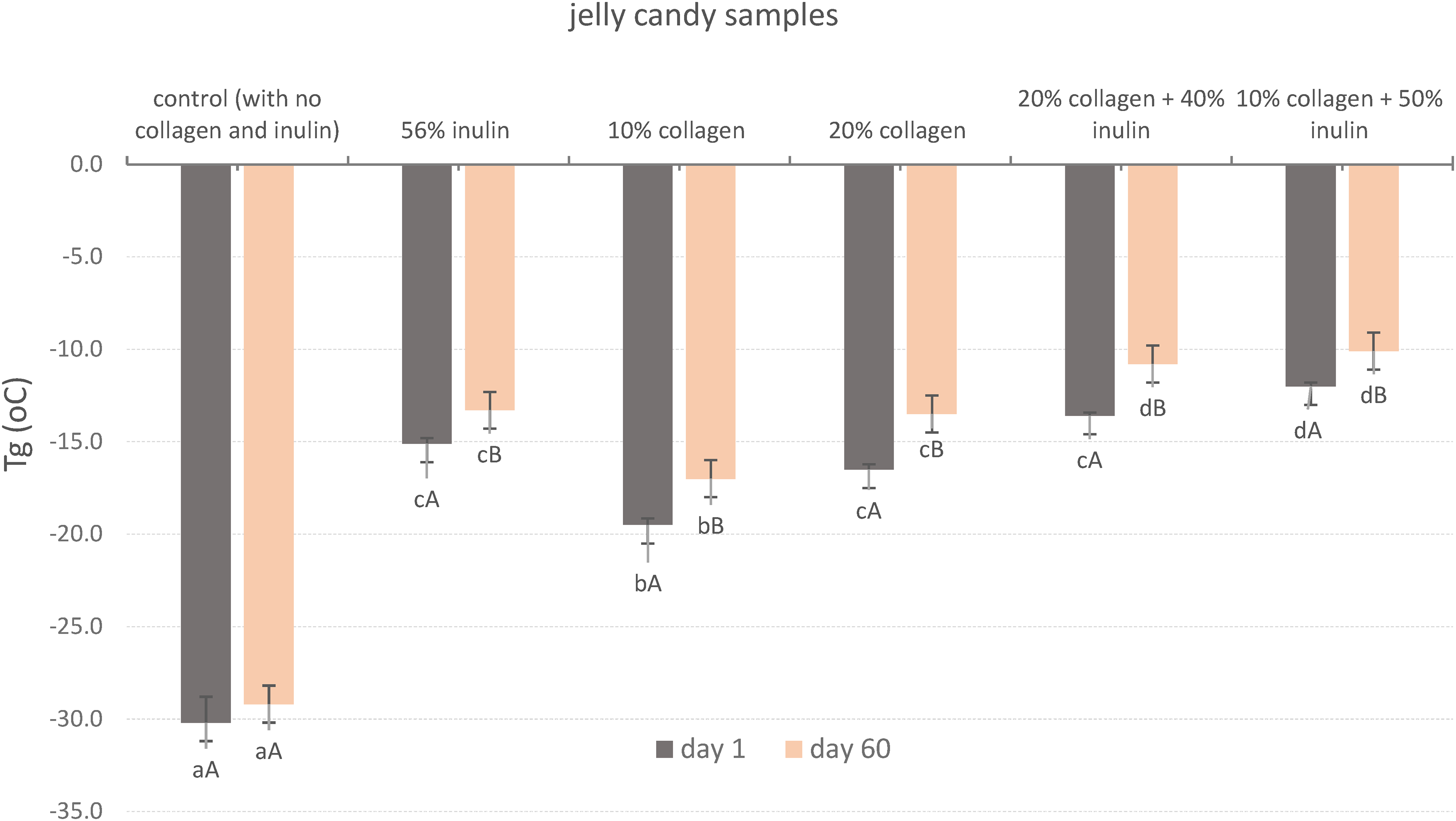
Tg of candy samples
The results of Tg were very low below 0 °C (Figure 2) since a small amount of water was known to end up with a large decrease in Tg of the candy as an indicator of the softness of the gummy candies (Ergun et al., 2015). The addition of inulin and collagen increased the Tg of the candies (from −30 °C to −19.5 to −12 °C). As the protein content increased (from 10% to 20%), increases in Tg temperatures were also observed. These increases were expected considering the individual Tg of the inulin and collagen components. Tg of protein is 150–220 °C, inulin 111–145 °C, bovine collagen 188 °C, isomalt 63 °C, sucrose 62 °C, and corn syrup is 100 °C (Kawai et al., 2011; Nowakowski and Hartel, 2002). The Tg of a solution depends on Tg of dissolved dry protein and water (Monkos, 2015). It was also reported in the study of Buitink et al. (2000) that Tg increased in the order of sucrose < trehalose < raffinose < poly-
Glass transition (Tg) temperatures of the soft candy samples after production and 60 days of storage.
Textural properties (firmness and springiness) of the candies
The firmness (force, N) analysis results of candy samples immediately after production were in the range of 0.97–2.43 and 1.36–2.94 for the stored candies at 4 °C for 60 days (Table 3). Inulin candy had the highest firmness value with 2.43 N. Firmness of all the samples increased after storage. Analyses also showed that collagen hydrolysate added to soft candy formulation increased the softness of the final product. As the amount of collagen increased, the firmness value of the candy decreased. In samples containing high collagen ratio (20%), the high moisture content and water-binding effect caused increased softness (Tanaka et al., 2009). In literature, there are studies that reported decreases in hardness with the use of inulin in various products (Delgado and Bañón, 2018). In the study of Shourideh et al. (2012), investigating dark chocolate formulations consisting of a mixture of inulin and
Textural properties of soft candies formulated with inulin and collagen.
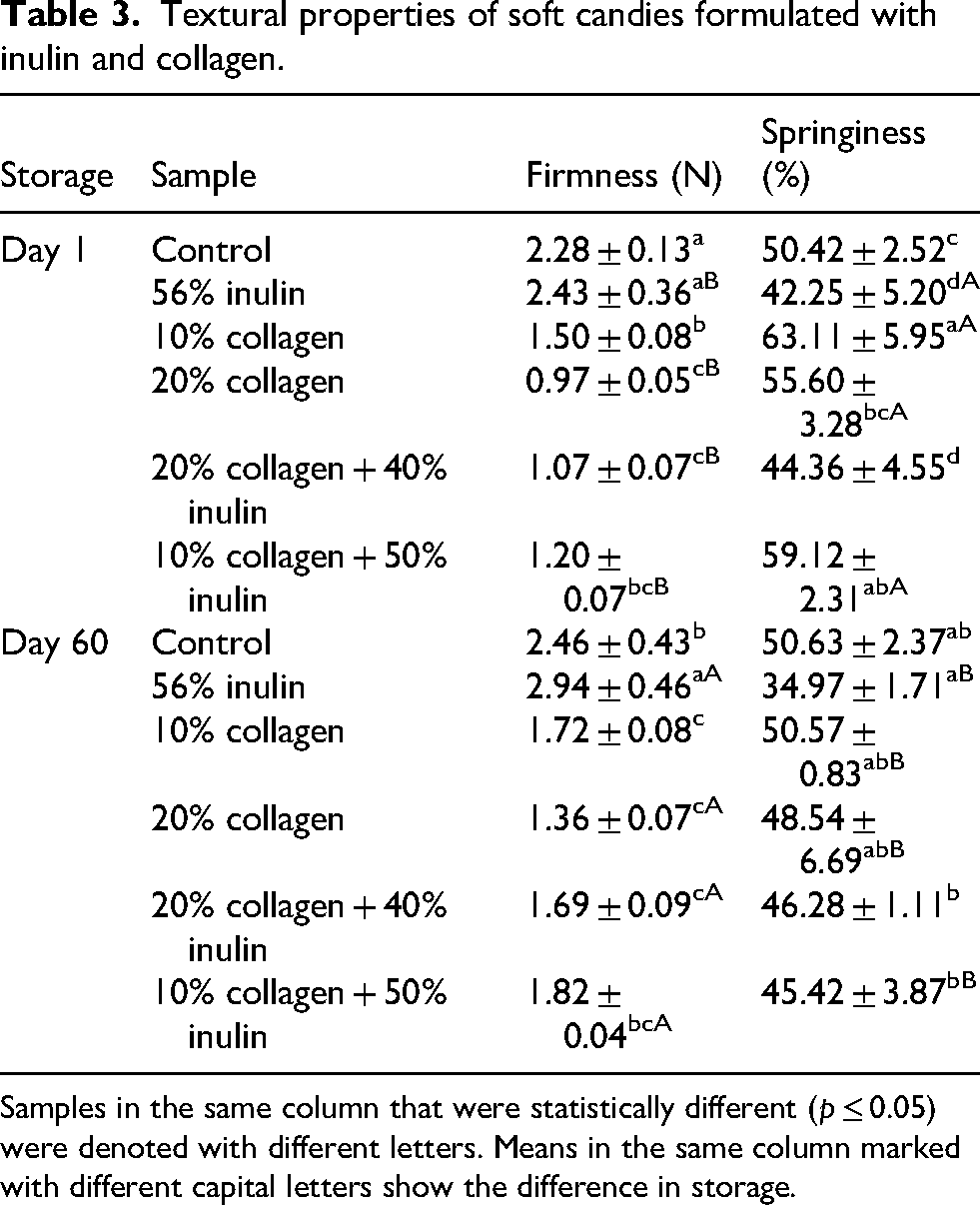
Samples in the same column that were statistically different (p ≤ 0.05) were denoted with different letters. Means in the same column marked with different capital letters show the difference in storage.
The springiness of the candies right after production varied between 42.25% and 63.11%, and the highest value belonged to the candy sample containing 10% collagen, while the inulin candy had the lowest value. Use of only inulin in the candy formulation caused a decrease in springiness, while collagen caused an increase in springiness. Springiness also increased when collagen was used together with inulin compared to solely inulin-included candies. After 60 days of storage, springiness results were found to be in the range of 45.42–53.25%. There was no statistically significant change in the springiness of control and 20% collagen + 40% inulin candies after storage, and the springiness of the remaining samples decreased. Unlike our findings, Ge et al. (2021) reported that the incorporation of inulin into gelatin-based soft candy strengthened the gel network through hydrogen bonding between inulin and gelatin. The springiness of inulin-containing candy was reported to be 10 times higher than that of candy produced with gelatin only. However, it is difficult to make a comparison since the study did not specify the amount of inulin used. A sugar content of 40–60% tends to increase the chain–chain association in gelatin gel networks (Kasapis et al., 2004). Kasapis et al. (2003) recommended sugar contents above 40% to reduce the contact surface between gelatin and solvent and thus stabilize the system. The authors stated that when the sugar concentration was high in gelatin gel network, there were heterogeneous domains rich in gelatin and sugar, and the gelatin chain association was promoted. The collagen + inulin candies we produced in this study had 40% sugar content. This might have reduced the stabilization of the gel network.
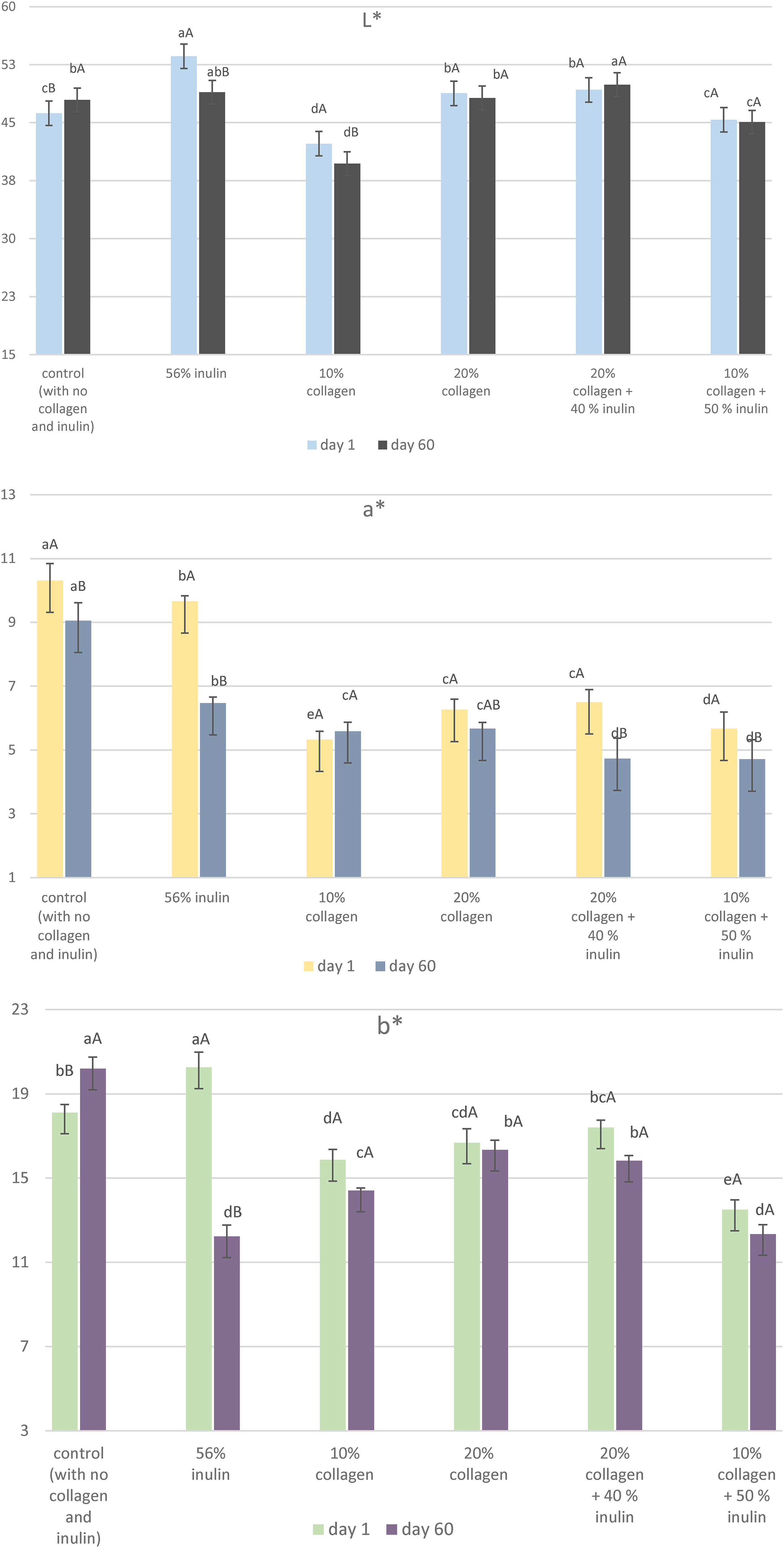
Color indices of soft candies
As for the L* values of the candies before storage, the highest brightness belonged to the inulin candy (Figure 3). The brightness of the low-collagen candy was at the lowest value. It was observed that the addition of inulin and collagen (20%) resulted in a visible lightening in the colors of the candies. After 60 days of storage, the L* value of the candy containing 20% collagen + 40% inulin had the highest value, while the low-collagen candy showed the lowest L* value. The a* (greenness-redness) values of the control (10.31) and the inulin candy (9.66) before storage were higher, while there was no significant difference between the a* values of the remaining samples. The 60-day storage caused decreases in the a* values of the candies. We could see that candies with higher a* values were scored with higher color and appearance scores in the sensory test (Table 4). It should be considered that non-enzymatic browning reactions occur during cooking and storage might have an impact on the candy color.
Color indices of the soft candy samples after production and 60 days of storage.
Sensory evaluation scores for the soft candy samples after production and 60 days of storage.
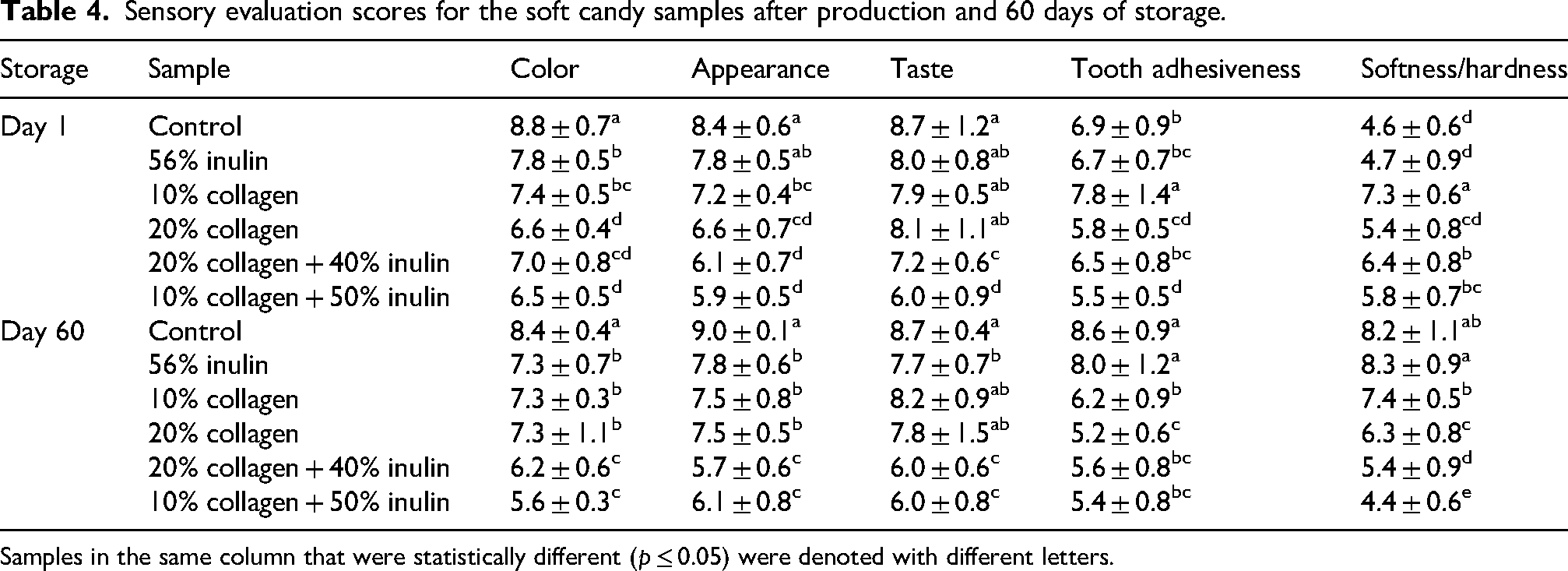
Samples in the same column that were statistically different (p ≤ 0.05) were denoted with different letters.
The highest b* (blueness-yellowness) values before and after storage were measured for the inulin candy (20.24) and for the control (20.18) candy, respectively. The b* value of the inulin candy decreased significantly by storage with a much greater decline than seen in other examples. In the overview, it was seen that co-addition of inulin and collagen reduced a* and b* values (decrease in redness and yellowness, respectively). This result might be explained by binding of the reactive group (reducing end) in inulin molecule to the amino group of the protein which can promote the reactivity. Similarly, Wang et al. (2019) notified that dry heating of whey protein + inulin complex had significantly higher absorbance values than untreated complex, indicating an increased browning intensity.
Prebiotic activity
Inulin, fructooligosaccharides, galactooligosaccharides, and other related prebiotic carbohydrates possess prebiotic activity (Huebner et al., 2007). Two different strains L. acidophilus and B. clausii were used in determining the prebiotic activity of the soft candies (Table 5). The control candy and no sample control had the lowest absorbance for both bacteria. These were followed by inulin sample. While the glucose sample was one of the samples showing the highest absorbance in B. clausii, it fell behind the samples containing 20% collagen for L. acidophilus. Prebiotic activity was also calculated by determining colony growth. Formulations containing collagen alone or with inulin provided the highest values in terms of both bacterial growths. High-collagen candy showed the highest growth for the L. acidophilus strain. Regardless of the inulin content, it can be said that B. clausii exhibited higher absorbance and colony development in candy with 10% collagen and L. acidophilus in candy with 20% collagen. The promoting effect of collagen in terms of prebiotic activity was evident for both bacteria. When used alone, inulin lags behind in this respect compared to use with collagen.
Prebiotic activity in soft candies formulated with inulin and collagen.

Samples in the same column that were statistically different (p ≤ 0.05) were denoted with different letters.
Mean ± standard deviation.
*Glucose: prepared by adding only 1% (v/v) glucose to a test tube containing MRS broth.
**Negative control: prepared by adding 1% (vol/vol) of each probiotic strain to a test tube containing only MRS broth.
Consumer willingness to buy
No major differences in consumer test results were seen between the scores given to the candies (Figure 4). While 66% of the participants scored 5 points (the category “I would definitely buy”) for the control sample, 64% for inulin candy and 10% collagen candy, 60% for 20% collagen and 58% for 20% collagen + 40% inulin candy. This category had the lowest rate of 54% for 10% collagen + 50% inulin candy. Only 8 out of 100 people graded this candy with the category 1 (“I would not buy”). Inulin candy ranked second among the samples evaluated with the highest scores, which can be attributed to the positive effect of inulin on the perception of sweetness intensity (Mandura et al., 2020; Ünal and Arslan, 2022, Villegas et al., 2010). For inulin and 20% collagen + 40% inulin candy, for each four people responded in the “I would not buy” category, and no one responded in the “I would not buy” category for the control and 10% and 20% collagen candies. When enriched with the combination of inulin and collagen, as much as 54–58% of the subjects responded in the “I would definitely buy” category which were slightly lower rates than that of the other candies (2–12% less).
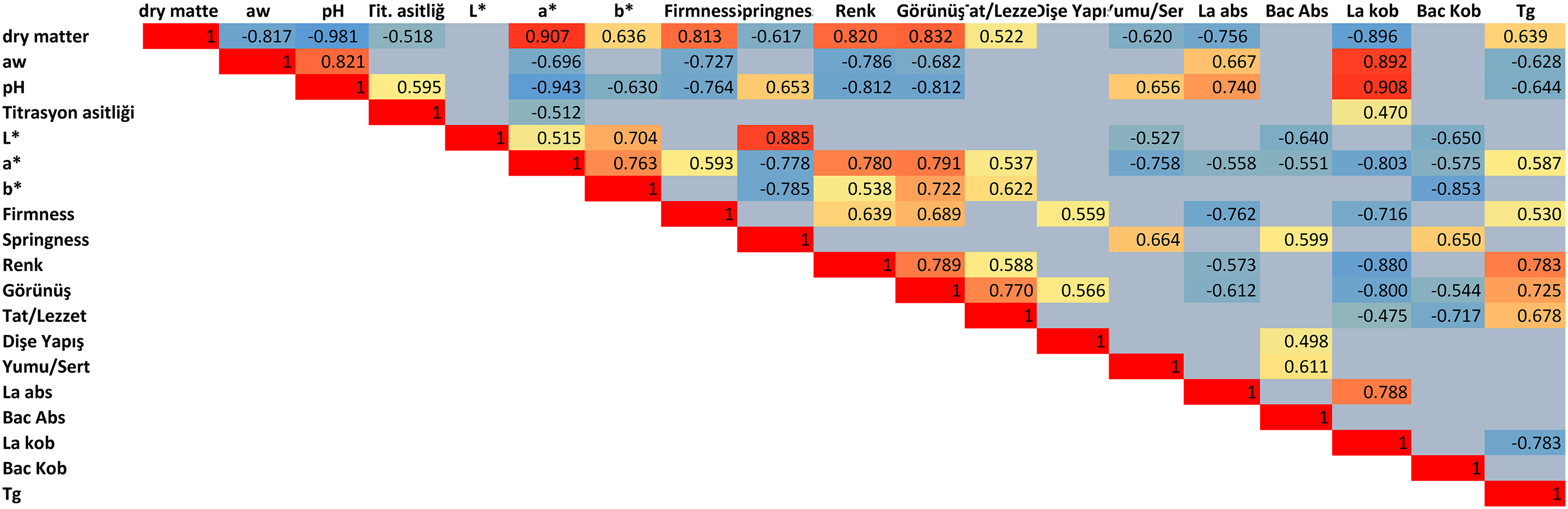
Consumer willingness to buy soft candies containing collagen and inulin. Relative frequency: number of panelists who chose a category/number of panelists in total (50). Category 1: I would definitely not buy, 2: I would probably not buy, 3: I am not sure, 4: I would probably buy, 5: I would definitely buy.
Consumer hedonic sensory test results
Considering the sensory evaluation of the candies before and after storage, control sample received the highest scores in terms of color and appearance, while the 10% collagen + 50% inulin and 20% collagen + 40% inulin candies received the lowest scores (Table 4). Addition of inulin and collagen to the soft candy reduced color scores. While the color of the control candy after storage was evaluated with the highest scores, the least preferred sample was the candy with 10% collagen + 50% inulin. A decrease in color scores was observed after storage. However, appearance appreciation increased, except for the sample containing 20% collagen + 40% inulin.
Regarding the taste evaluation, both for the before and after storage samples, inulin, 10% and 20% collagen, and control candies, presented the highest scores, while the least preferred was the sample with 10% collagen + 50% inulin. The samples that were found stickier to the teeth before storage were 20% collagen and 10% collagen + 50% inulin. Among the freshly produced samples, 10% collagen candy was evaluated as less sticky to the teeth. After storage, control and inulin candies were found significantly less sticky than the others. Similarly, in terms of softness/hardness parameter, before storage 10% collagen and after storage besides this candy, control and inulin candies perceived as harder than the others. The results were in agreement with Narala et al. (2022), who announced that replacing fat with inulin in ice cream increased its adhesiveness. Qaziyani et al. (2019) reported an increase in adhesion to teeth in chewing gum by the addition of inulin. And the same trend was observed for a traditional spread with jam-like consistency increased adhesiveness ratings with the amount of inulin addition (Phang and Chan, 2009). The adhesiveness of tamales (a Mexican local food) presented a positive correlation with the inulin content. El-Nagar et al. (2002) reported that the increase of adhesiveness observed within inulin-enriched samples could be due to the formation of a viscous inulin gel matrix.
Overall, the candy evaluated with the highest scores before storage was 10% collagen, post-storage were inulin, control, and 10% collagen (except for stickiness) candies. The combination of inulin and collagen was not appreciated in terms of sensory parameters. Approximately 10% inulin addition into gelatin was reported to generate slightly softer, springier, and stickier jellies than starch (Delgado and Bañón, 2018). In the study of Wang et al. (2021), addition of inulin alone to gelatin did not provide the desired mouthfeel; therefore, a polysaccharide was required to enhance the texture and sensory qualities of a mixed gel system. Inulin was found inadequate to form direct interactions with gelatin and was only physically embedded in the gelatin matrix.
The use of 10% and 20% collagen in jelly candy did not impart any negative effects in terms of taste. However, incorporation of collagen at 20% caused the candy to become softer and stickier to the teeth. The inclusion of collagen and inulin decreased the sensory scores compared to other formulations which might be associated with the insufficient formation of a matrix between inulin and other system molecules.
Heat map and correlation analysis
The heat map (Figure 1) revealed that firmness showed a positive correlation with dry matter content and a negative correlation with aw. There was also a negative correlation between pH and firmness. A low pH facilitates the interaction between pectin molecules, promotes gel formation, and improves the formation of connection sites, resulting in stronger gel networks (Thakur et al., 1997). A negative correlation was detected between pH and dry matter, and a positive correlation between aw. As with firmness, positive and negative correlations were observed between Tg and dry matter and aw, respectively. This was an expected result, as water behaves as a plasticizer in amorphous sugars and as water content and water activity increases Tg decreases (Mathlouthi and Reiser, 1995). The a* value was also correlated with dry matter, aw, firmness, and Tg. As the dry matter increases, the caramelization and Maillard reaction products are expected to be released more. It was also noteworthy that the a* value and with relatively less coefficient b* value were compatible with the color and appearance scores determined in the sensory analysis. Correlation analysis also revealed positive correlations between taste scores and a* and b* values, which again supported that the relation with increased dry matter was significant. As for the sensory test, color, and appearance scores showed positive correlations with dry matter and firmness and negative correlations with pH.
Although variations in sucrose content can influence the texture, moisture, and glass transition behavior of sugar-based confections, the observed differences in hardness, adhesiveness, water activity, and thermal properties were primarily associated with the specific functional characteristics of collagen and inulin. Collagen, due to its protein-based water-binding capacity, and inulin, through its fiber structure and hydrogen-bonding potential, directly affected the gel matrix and moisture distribution of the candies. Therefore, the variations in physical and sensory properties among the samples reflect the combined influence of sugar reduction and the distinct physicochemical actions of these added biopolymers, rather than being solely attributable to sucrose content differences.
Conclusions
Addition of inulin to the soft candy formulation, thanks to the strengthening of the gel network with the hydrogen bonding effect, possessed positive effects in terms of hardness and adhesiveness to the teeth along with decreases in aw and hygroscopicity. Additionally, it caused lightening of the candy color and decrease in elasticity. The presence of collagen in the formulation resulted in an increase in pH, opaqueness of the color, and higher moisture retention before storage. Although prebiotic activity was promoted when the amount of collagen increased from 10% to 20%, due to the protein structure-based high water-holding capacity, the aw, softness, and tooth adhesiveness of candy increased. The presence of inulin and collagen together in the formulation caused combined effects such as an increase in Tg, a reduction in redness and yellowness, and a decrease in sensory appreciation and consumer willingness to buy, which may be attributed to the softer texture, higher tooth adhesiveness, and lighter color of the candies. In these samples, the sucrose and corn syrup ratio being below 40% suppressed gel network formation. It was clear that formulations containing high amounts of collagen and inulin required the use of a polysaccharide such as pectin, agar, or starch to promote gel network formation through enhanced interactions and to decrease the softness and tooth adhesiveness of the product.
Footnotes
Acknowledgments
The authors would like to acknowledge Artisan Gida A.Ş. for kindly providing inulin and pectin for this research.
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was financially supported by the Office of Scientific Research Projects in Necmettin Erbakan University (NEÜ-BAP, Project number: 211319022).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data that support the findings of this study are available from the corresponding author upon request.
