Abstract
Resistant maltodextrin (RMD) was added at increasing concentrations (0%, 2.5%, 5% and 7.5%) before pasteurisation to orange juice to analyse its potential protective effect on the health-related bioactive compounds of pasteurised orange juice throughout its storage time. Samples were characterised in terms of basic physico-chemical properties and bioactive compounds at the beginning of the storage. Higher concentrations of RMD proved to better preserve the bioactive compounds of orange juice, thus obtaining a higher antioxidant capacity (AC). Stability of all samples was determined by measuring the same parameters at days 0, 15, 45, 75, 105, 136 and 170 of storage. °Brix and pH were very stable in all samples along storage, while all bioactive compouds had negative variations. However, RMD addition slightly improved ascorbic acid, vitamin C, total phenols, and total carotenoids retention, improving then its AC. This effect was greater in the 5% RMD-added samples. All bioactive compounds showed a positive Pearson's correlation coefficient with AC. Colour variations were also measured at days 105 and 170. All samples had a positive variation of all colour parameters, being this clearer at day 170. This work enlights the potential functionality of RMD to better preserve the health-related compounds of pasteurised orange juice.
INTRODUCTION
Prebiotics are a group of non-digestible carbohydrates, mainly dietary fibers, that can selectively influence gut microbiota resulting in desirable effects for human health (Singla and Chakkaravarthi, 2017). To our knowledge, inulin, fructo-oligosaccharides (FOS), isomalto-oligosaccharides, gluco-oligosaccharides, or human milk oligosaccharides, among others, are the most studied substances because of their potential prebiotic activity (Corzo et al., 2015). However, the identification of fiber as the potential cause of healthy intestinal function led to the search for substances with prebiotic activity, including resistant maltodextrin (RMD) in recent years (Fuentes-Zaragoza et al., 2011).
RMD is a water-soluble fiber produced from the heat treatment of corn starch. It is indigestible in the small intestine but fermentable by the colonic bacteria and, therefore, enhancing the production of short-chain fatty acid (Lockyer and Nugent, 2017). Up to now, RMD has shown to exert a wide variety of positive health effects. For example, the intake of 10 grams of RMD with a meal stimulated the production of satiety hormones, hence decreasing hunger (Ye et al., 2015). In a meta-analysis of randomized controlled trials, RMD consumption attenuated the insulin and triacylglycerol response to meal, being this effect stronger for RMD in drinks than in solid foods (Livesey and Tagami, 2009). The suppresive effect on insulin and triacylglycerol levels was also found when consuming a meal with a beverage containing 5 or 10 grams of RMD (Kishimoto et al., 2007). Also, in a double-blind placebo-controlled randomized crossover study, a beverage containing 25 or 50 grams of RMD added to water and consumed for 24 days increased fecal bulk (Baer et al., 2014). According to these cited articles, liquid matrices seem to facilitate the RMD transport through the gastrointestinal tract, compared to solid foods. In fact, fruit juices have been suggested as an ideal vehicle for prebiotic delivery because their appealing organoleptical properties that makes them well accepted by population of all age groups (Valero-Cases et al., 2020). In terms of flavor, citrus juices are popular. Specifically, orange juice is the most consumed fruit juice worldwide, playing an important role at the nutritional and economic level. Orange juice is well known by antioxidant properties thanks to its intrinsic bioactive compounds content, mainly total phenols (TP), total carotenoids (TC), ascorbic acid (AA) and vitamin C. Antioxidants emerge as desirable compounds because their effect against oxidative stress (Adwas et al., 2019), as they inhibit reactive oxygen species production and scavenging of free radicals. These reactive oxygen species have been shown to be involved in the development of cancer, cardiovascular and neurodegenerative diseases, in addition to aging (Yang et al., 2018). Therefore, preserving the intrinsic bioactive compounds of the orange juice is a challenge worth it because its potential health impact, both at the time of juice processing and along its storage during its shelf life.
Moreover, in plant-derived products there is a relationship between the physico-chemical properties and the bioactive compounds (Sánchez-Moreno et al., 2003). For instance, the natural orange colour of the oranges is mostly due to its carotenoid content, so the chemical transformation of such compounds may imply colour changes. Deeply understanding this relationship between the physico-chemical properties and the bioactive compounds of orange juice could also be of great interest to develop healthier and higher quality food products. This is especially interesting when applying thermal preservation technologies as pasteurisation, which is the most widely technology used as it is the most cost-effective method to reduce microbial populations and enzyme activity (Perez-Cacho and Rousell, 2008). Nevertheless, it is well known that applying thermal processing causes irreversible losses of bioactive compounds and antioxidant properties (Lu et al., 2018). In our previous study, RMD addition before the pasteurisation juice process showed a protective effect on the bioactive compounds of orange juice, being this effect greater with higher RMD concentrations (Arilla et al., 2021). However, the effects of RMD addition on the stability of the bioactive compounds and its antioxidant capacity (AC) along storage needs to be elucidated. Therefore, the aim of the present study is to analyse if the protective effect of RMD addition on orange juice bioactive compounds extends throughout the shelf life of the finished product.
MATERIALS AND METHODS
Raw materials
This study was conducted with freshly squeezed orange juice supplied by Refresco Iberia S.A.U. (Valencia, Spain). All oranges were from Spanish origin. RMD (Fibersol-2) added to the juice was purchased from ADM/Matsutani, LLC (Decatur, IL, USA). Frozen pasteurized orange pulp was provided by a local fruit processing company (Zumos Valencianos del Mediterráneo, Valencia, Spain).
Sample preparation and pasteurisation
Eight samples of orange juice were prepared. Four were orange juice with pulp (OJP) and the other four were orange juice without pulp (OJWP). Fresh orange juice was directly collected from the industrial squeezed lines. Orange pulp (2.5%) was added to the OJP samples. Pulp content was homogenised using a stirrer (LH Overhead Stirrer, VELP Scientifica, Italy), by applying 200 rpm for 5 min. Increasing RMD concentrations (2.5, 5, and 7.5%) were mixed into both OJP and OJWP samples. Thus, for a finished beverage portion of 200 mL, 5, 10, or 15 g of RMD would be ingested, enough to display functional effects according to other studies (Livesey and Tagami, 2009; Ye et al., 2015). Control samples without RMD addition (OJP0 and OJWP0) were also prepared, and they complied with the European Fruit Juice Association orange juice guidelines (AIJN), so no adulteration or deviation occurred during the juice extraction. To properly dissolve RMD in the fresh orange juice, the same stirrer at 200 rpm for 15 min was used. Finally, all samples were pasteurised (Fruchtsaftdispenser, Mabo Steuerungselemente GmbH, Germany) at 85°C for 10 s, and were hot filled into 250 mL polyethylene terephthalate bottles. All bottles were immersed in a cold-water bath (<10°C) for 30 min to cool down their temperature after the heat treatment. Then, samples were stored at 25°C in darkness for 170 days.
Determinations
°Brix, pH, AA, vitamin C, TP, TC and AC were determined at 0, 15, 45, 75, 105, 136 and 170 days of storage.
°Brix and pH
Measurement of total soluble solids (°Brix) was conducted using refractometry (Abbemat 200, Anton Paar, Austria). Determination of pH was made using a Basic 20 pH meter (Crison, Spain). All determinations were performed in triplicate in accordance with AOAC guidelines (Latimer, 2012).
Ascorbic acid and Vitamin C
AA and vitamin C, which involves AA and dehydroascorbic acid (DHAA), were determined using a HPLC-UV detector (Jasco equipment, Italy) in triplicate. The method proposed by Xu et al. (2008) was used to determine the ascorbic acid with some modifications made by Igual et al. (2016). To determine the AA, 1 g sample was extracted with 9 mL 0.1% oxalic acid for 3 min and immediately filtered (0.45 µm) before injection. The procedure employed to determine total vitamin C was the reduction of DHAA acid to AA, using DL-dithiothreitol as the reductant reagent. A 0.5 mL aliquot sample was taken to react with 2 mL of a 20 g/L dithiothreitol solution for 2 h at room temperature and in darkness. Afterwards, the same procedure as that used for the AA method was performed. The HPLC method and instrumentation was: Ultrabase-C18, 5 µm (4.6 × 250 mm) column (Scharlab, Barcelona, Spain); mobile phase 0.1% oxalic acid, volume injection 20 µL, flow rate 1 mL/min, detection at 243 nm and at 25 °C. AA standard solution (Sigma-Aldrich, Steinheim, Germany) was prepared.
Total phenols
Determining TP was based on the Folin-Ciocalteu method. The extraction procedure comprised mixing sample with methanol. The mixture was centrifuged (12,857xg, 10 min, 4 °C) to obtain the supernatant (Igual et al., 2016). Absorbance was measured at 765 mm in a UV-3100PC spectrophotometer (VWR, Leuven, Belgium). The total phenolic content was expressed as mg of gallic acid (Sigma-Aldrich, Steinheim, Germany) equivalents per 100 g of orange juice to compare all the samples.
Total carotenoids
The TC in the samples were extracted with a solvent hexane/acetone/ethanol mixture following the Olives Barba et al. (2006) method in triplicate. Sample absorbance was measured at 446 nm in a UV-visible spectrophotometer (Thermo Electron Corporation). The TC content was expressed as mg of β-carotene (Fluka-Biochemika) per 100 g of orange juice to compare all the samples.
Antioxidant capacity
AC was assessed using the free radical scavenging activity of the samples evaluated with the stable radical 2,2-diphenyl-1-picryl-hydrazyl-hydrate (DPPH) following Igual et al. (2019) methodology in triplicate. Samples were mixed with methanol. The homogenate was centrifuged (10.000 rpm, 10 min, 4 °C) to obtain the supernatant. 0.1 mL of supernatant was added to 3.9 mL of DPPH (0.030 g/L, Sigma-Aldrich, Steinheim, Germany) in methanol. A UV-visible spectrophotometer (Thermo Electron Corporation) was used at the absorbance at 515 nm. The results were expressed as milligram Trolox equivalents (TE) per 100 g of orange juice to compare all the samples.
Colour measurement
Sample colour was measured using a colourimeter (Konica Minolta CM-700d/600d series, Tokyo, Japan) with a standard illuminant D65 and a visual angle of 10°. Measurements were realized at 0, 105 and 170 days of storage when visual changes were observed. Results were obtained in terms of L* (brightness: L* = 0 (black), L* = 100 (white)), a* (−a* = greenness, + a* = redness), and b* (−b* = blueness, + b* = yellowness), according to the CIELab system (CIE, 1986). Differences in L*, a* and b* because of storage time were calculated (ΔL*, Δa* and Δb*). The total colour difference (ΔE) was calculated with respect to the sample at the beginning of storage to evaluate the storage effect.
Statistical analysis
Analysis of variance (ANOVA) was applied with a confidence level of 95% (p < 0.05), to evaluate the differences among samples. Furthermore, a correlation analysis among studied bioactive compounds and antioxidant capacity of juices, with a 95% significance level was conducted. Statgraphics Centurion XVII Software, version 17.2.04 (Statgraphics Technologies, Inc., The Plains, VA, USA) was used.
RESULTS AND DISCUSSION
Orange juice characterisation
Table 1 shows the mean values with standard deviations of °Brix, pH and colour parameters because they are usually measured in the quality control processes of the citrus fruit industry (Kimball, 2012). Control samples without RMD (OJP0 and OJWP0) obtained comparable physico-chemical values than those reported in other orange juice-based studies (Mennah-Govela and Bornhorst, 2017; Sánchez-Moreno et al., 2003; Wibowo et al., 2015). Orange pulp did not display a significant (p > 0.05) effect on the soluble solids fraction. Adding a water-soluble fiber as RMD proportionally increased significantly (p < 0.05) °Brix values. The same behavior was also observed in other prebiotic fibers with similar water-dissolving properties (Igual et al., 2019; Pimentel et al., 2015). Increasing °Brix with RMD addition could improve the sweet profile and mouthfeel of orange juice, thus exhibiting interesting food technology applications together with its prebiotic function. For instance, prebiotics have been proposed as sugar replacers (Pimentel et al., 2015; Singla and Chakkaravarthi, 2017). Besides, pH was not altered (p > 0.05) either by RMD addition nor presence of orange pulp.
Mean values (and standard deviations) of °Brix, pH, colour coordinates (L*, a* and b*) and total colour differences (ΔE) of pasteurised orange juice.
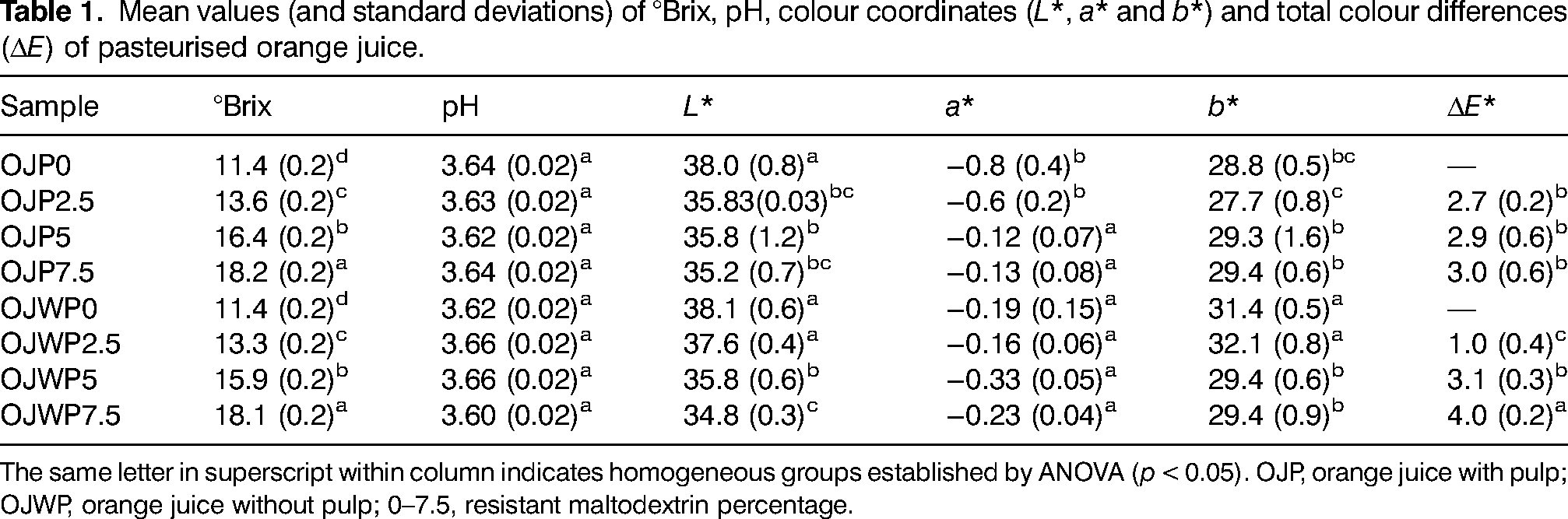
The same letter in superscript within column indicates homogeneous groups established by ANOVA (p < 0.05). OJP, orange juice with pulp; OJWP, orange juice without pulp; 0–7.5, resistant maltodextrin percentage.
Regarding colour, which is one of the major attributes that affect the consumer perception of food quality (Sant’Anna et al., 2013), L*, a*, b* and ΔE* values are also shown in Table 1. Orange pulp did not show a significant effect (p > 0.05) on the L* values as OJP0 and OJWP0 got similar results. However, L* values significantly decreased (p < 0.05) due to RMD addition, meaning that orange juice turned slightly darker as higher RMD concentrations were added. In addition, OJWP0 marked a higher (p < 0.05) a* value than OJP0, meaning that pulp-free orange juice was slightly reddish than pulp-added orange juice. RMD addition on the OJWP samples did not have any significant effect (p < 0.05) on the a* values. However, it did have a protective effect (p < 0.05) on the reddish tones, as higher RMD concentrations led to higher a* values, especially from 5% RMD. This could be due to a protective effect on the carotenoids content of the orange pulp, which was found in our previous study (Arilla et al., 2021). For the b* values, OJWP0 samples obtained a higher (p < 0.05) value than OJP0, thus achieving a more yellowish colour. RMD addition to OJWP samples slightly decreased (p < 0.05) b* values from 5%. On the other hand, RMD addition did not show any effect (p > 0.05) on the b* values of OJP samples. Thus, orange pulp could also interact with RMD to maintain yellowish tones. Colour differences were higher (p < 0.05) as higher RMD concentrations were added in all OJP and OJWP samples.
Table 2 shows the mean values with standard deviations of AA, vitamin C, TP, TC and AC for all OJP and OJWP samples at the beginning of the storage period. Control samples (OJP0 and OJWP0) had almost the same vitamin C content. OJP and OJWP RMD-added samples had a similar behaviour, as higher RMD concentrations slightly but not significantly (p > 0.05) increased vitamin C content. However, in the 7.5% RMD-added samples, the pulp-added sample obtained a significant (p < 0.05) higher vitamin C content than the rest. A similar behaviour was also observed in our previous study (Arilla et al., 2021). Besides, the protective effect of other prebiotic fibers such as inulin or gluco-oligosaccharides on the vitamin C content of heat-treated juices has also been suggested by Alves Filho et al. (2018). In terms of AA content, OJWP0 got a higher (p < 0.05) value than OJP0. RMD addition maintained the AA content in the OJP samples (p > 0.05), while it did have a clear protective effect (p < 0.05) in the OJWP samples, especially at higher RMD concentrations (p < 0.05). Therefore, considering the vitamin C results, RMD addition seems to prevent DHAA degradation to other forms that no longer have a vitamin function.
Mean values (and standard deviations) of ascorbic acid (AA), vitamin C, total phenols (TP), total carotenoids (TC) content and antioxidant capacity (AC) of pasteurised orange juice, expressed in mg/100gorange juice.
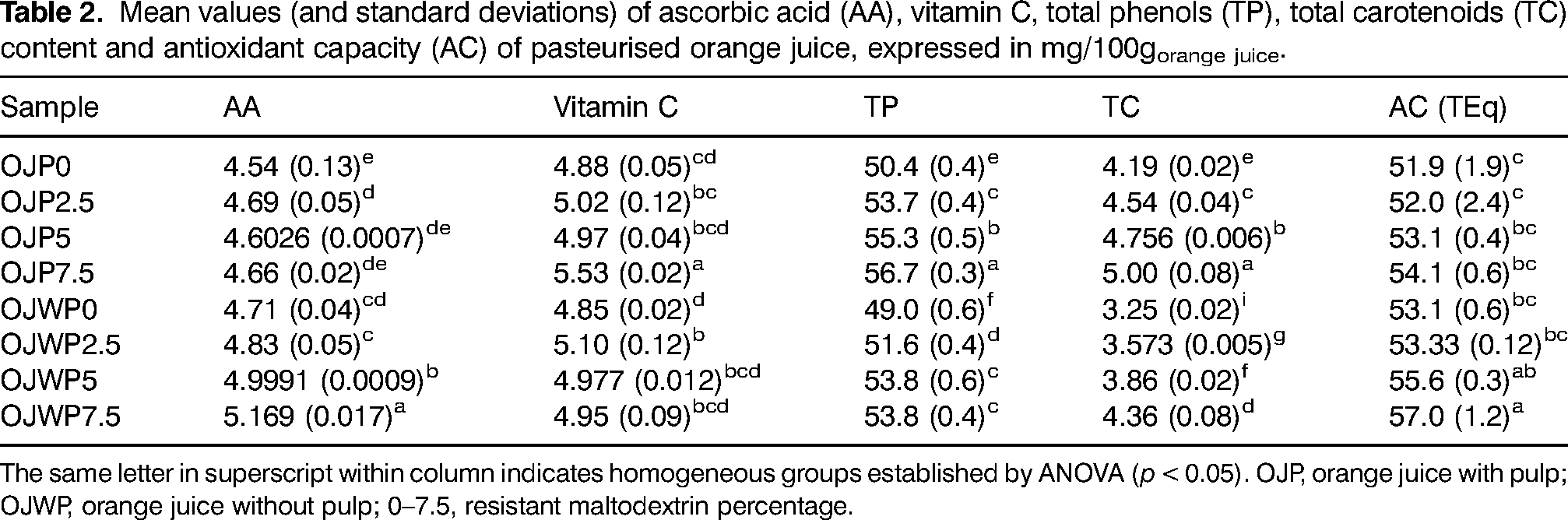
The same letter in superscript within column indicates homogeneous groups established by ANOVA (p < 0.05). OJP, orange juice with pulp; OJWP, orange juice without pulp; 0–7.5, resistant maltodextrin percentage.
OJP0 marked higher (p < 0.05) TP content than OJWP0, probably because orange pulp contains hesperidin, which is the main phenolic compound in oranges (Iglesias-Carres et al., 2019). Besides, orange pulp has been reported to contain 1.6 times higher TP concentration than orange juice (De Ancos et al., 2017), so its addition to orange juice is expected to increase TP content. This difference was even more noticeable in terms of TC, where OJP0 also marked higher (p < 0.05) TC content than OJWP0. Rodrigo et al. (2015) also found an increase close to 40% in the TC content of orange pulp in comparison to freshly prepared orange juice. This enlights the potential application of citrus by-products as an economic and natural way to increase health-related compounds content, as it has been previously suggested (Kaur et al., 2021). Also, RMD addition had a clearer impact on the TP and TC content that on the AA and vitamin C content of orange juice. It showed a protective effect (p < 0.05) of the TP and TC content of all OJP and OJWP samples, being this effect greater (p < 0.05) with higher RMD concentrations. Protecting TP from process degradation is of interest because their potential prebiotic-like effect (Dueñas et al., 2015; Lima et al., 2019). In addition, the protective effect of RMD on TP and TC content seems to be slightly greater in OJP samples than in OJWP samples, suggesting that orange pulp presence might also reinforce TP and TC protection. This agrees with the results in the a* and b* values (Table 1), where RMD addition showed a higher protective effect in the reddish and yellowish tones of OJP samples.
The AC is the result of each bioactive compound (AA, vitamin C, TP, and TC) contribution. OJPW0 showed a slight but not significant (p > 0.05) higher value than OJP0. RMD addition also sightly but not significantly (p > 0.05) increased the AC in OJP samples. However, it did exert a significant (p < 0.05) protective effect on the AC of OJWP samples, especially at 7.5% RMD concentration. A similar behaviour was also observed in our previous study (Arilla et al., 2021), where OJWP7.5 achieved the highest AC variation. It has been also reported that adding other prebiotic fiber such as inulin to acerola juice had a protective effect from thermal degradation on vitamin C and TP content, thus leading to higher AC (Fonteles et al., 2021). Consequently, adding prebiotic fibers to pasteurised fruit-based beverages could be beneficial to better preserve health-related compounds from thermal degradation.
Orange juice stability
Figure 1 shows the mean values and standard deviation of pH and °Brix of all OJP and OJWP samples along storange. pH of all OJP and OJWP samples moved in the narrow range between 3.55 and 3.65 along the 170 days of storage. Consequently, not significant (p > 0.05) changes on the pH are appreciable during storage period due to orange pulp presence nor RMD addition. °Brix were also very stable over the course of the whole storage period, with no significant (p > 0.05) changes in any sample.
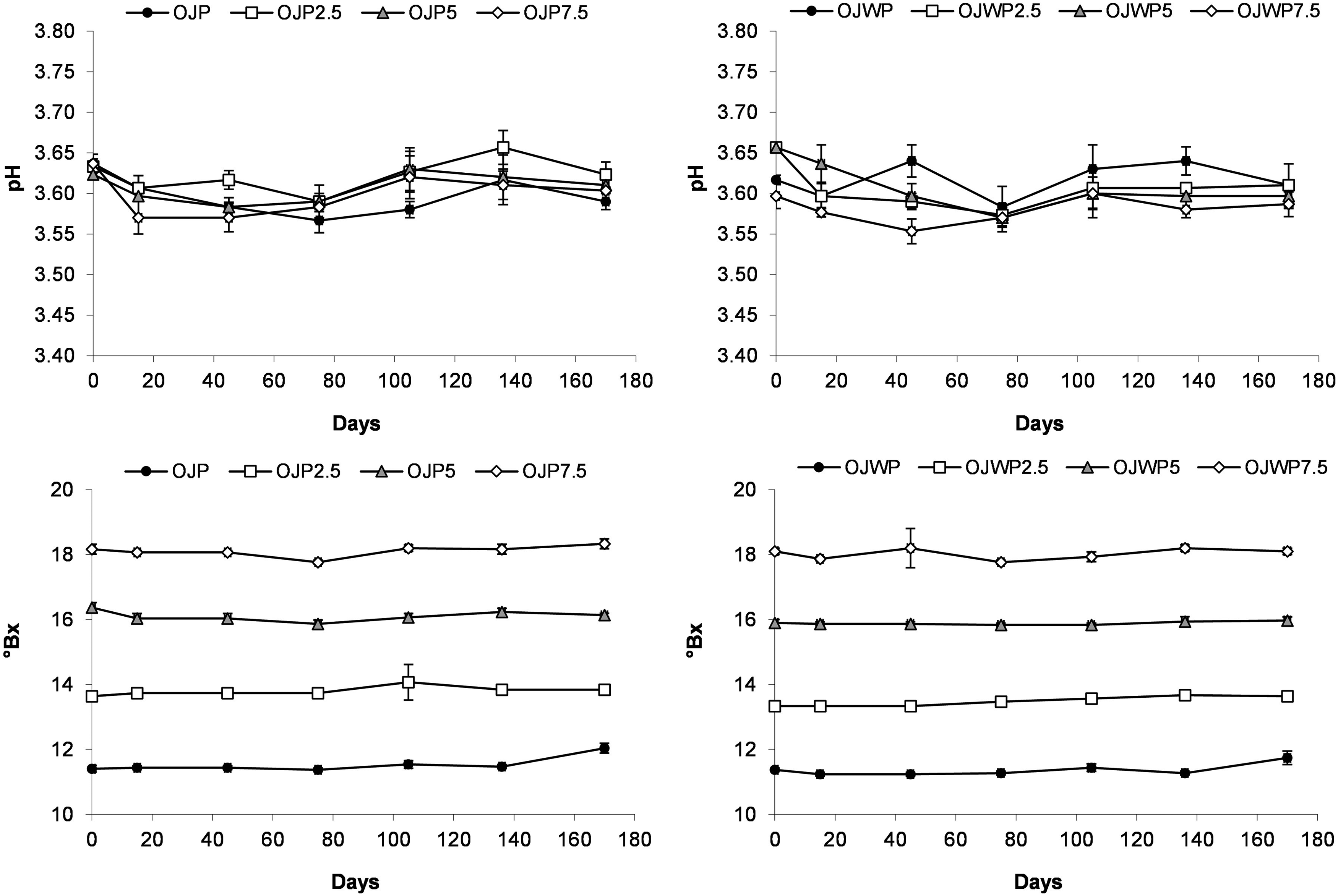
Mean values and standard deviation of pH and °Bx of studied juices during storage. OJP, orange juice with pulp; OJWP, orange juice without pulp; 0–7.5, resistant maltodextrin percentage.
The stability of °Brix and pH in citrus fruit juices along storage was also reported by another authors (Agcam et al., 2016; Elez-Martínez et al., 2006; Igual et al., 2010). A similar behaviour was observed in fruit juice beverages fortified with another prebiotic fiber such as FOS, where pH and °Brix hardly changed in storage periods of 4–6 months (Cascales et al., 2021; Renuka et al., 2009). As explained before, °Brix and pH were measured as quality control parameters. Therefore, the stability of these parameters along the whole storage period indicates that no degradation reactions that altered the results of the study occurred, hence retaining the desirable physico-chemical properties.
To evaluate the effect of the storage time to all samples, the variation of each component at each storage time (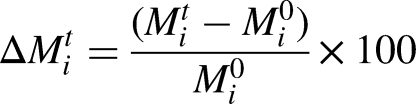
Figure 2 shows the mean values and standard deviation of AA and vitamin C variations of all OJP and OJWP samples along storage. Despite both OJP0 and OJWP0 samples got similar AA variations along storage (p > 0.05), the initial drop in the AA content in OJWP0 is more noticeable than in OJP0. This suggests that orange pulp could play a role in better preserving the AA of pasteurised orange juice at the first stage of storage time. Also, RMD addition in OJWP samples, especially at higher doses, resulted in lower (p < 0.05) AA loss being this protective effect clearer at the first stages (days 15 to 75) of storage. On the contrary, all RMD-added OJP samples were losing AA at almost the same magnitude as OJP0, indicating that RMD addition did not exert a protective effect (p > 0.05) on the AA content of OJP samples.
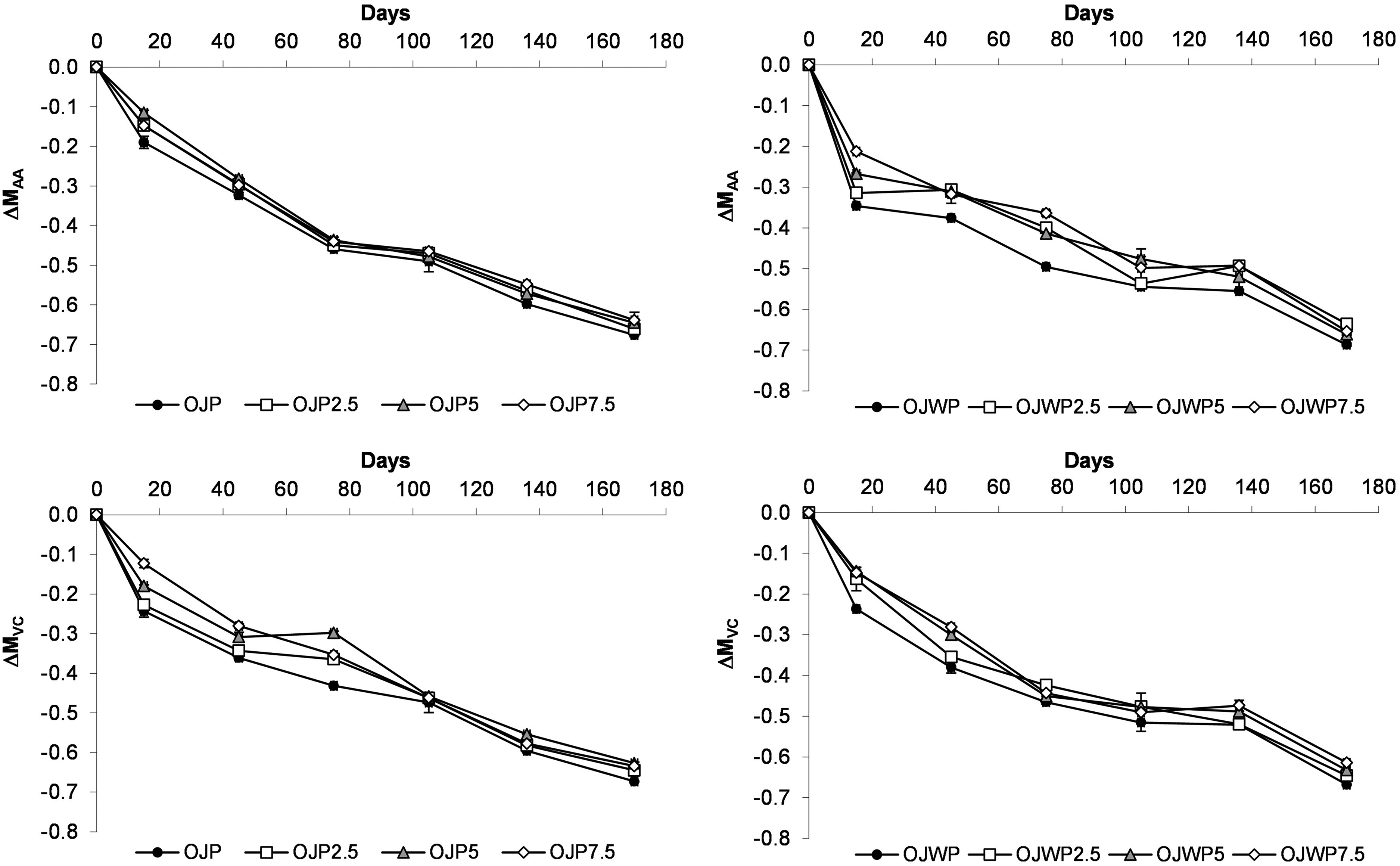
Mean values and standard deviation of ascorbic acid variation (ΔMAA) and vitamin C (ΔMVC) during storage. OJP, orange juice with pulp; OJWP, orange juice without pulp; 0–7.5, resistant maltodextrin percentage.
Regarding vitamin C variations, OJP0 and OJWP0 had the same behaviour, meaning that orange pulp did not display a significant (p > 0.05) impact on the vitamin C loss along storage. Besides, RMD addition to orange juice slightly improved (p < 0.05) vitamin C retention at the first stages of storage, especially in the OJP samples. However, from day 105 all samples had similar vitamin C loss. Other orange juice-based studies have also reported AA and vitamin C loss along storage (Elez-Martínez et al., 2006; Esteve and Frigola, 2008; Islam et al., 2014; Spira et al., 2018; Torres et al., 2011). Moreover, it has been suggested that AA and vitamin C degradation because dissolved oxygen presence may cause great colour changes in heat-treated juices stored at room temperature (Elez-Martínez et al., 2006; Ros-Chumillas et al., 2007). Ascorbic acid acts as an oxygen scavenger for the removal of molecular oxygen. For this reason, deaeration has been suggested as a recommended process for juice industrial producers to improve juice quality along storage, as this process diminishes the oxygen consumption via oxidative reactions of ascorbic acid (Remini et al., 2015).
Concerning TP variation along storage (Figure 3), OJP0 and OJWP0 samples followed the same trend: both were losing TP by a similar magnitude over time. Thus, orange pulp did not have a significant (p > 0.05) impact on TP variation throughout storage. Contrarily, higher RMD concentrations led to lower (p < 0.05) TP variations. However, this protective effect of RMD on TP variations was limited, as the most noticeable differences were found on days 45, 75 and 105, from which time the differences narrowed. Other pasteurised orange-juice based studies also reported slight TP degradation during the shelf life of pasteurised orange juice (Ros-Chumillas et al., 2007; Spira et al., 2018). Pasteurisation has been reported to generally reduce TP content in comparison to fresh juice, while non-thermal preservation methods as high hydrostatic pressure may even improve TP content, probably due to changes in the structure of vesicles in the orange juice that enables greater extraction of flavanones (Esteve and Frigola, 2008; Sánchez-Moreno et al., 2005)
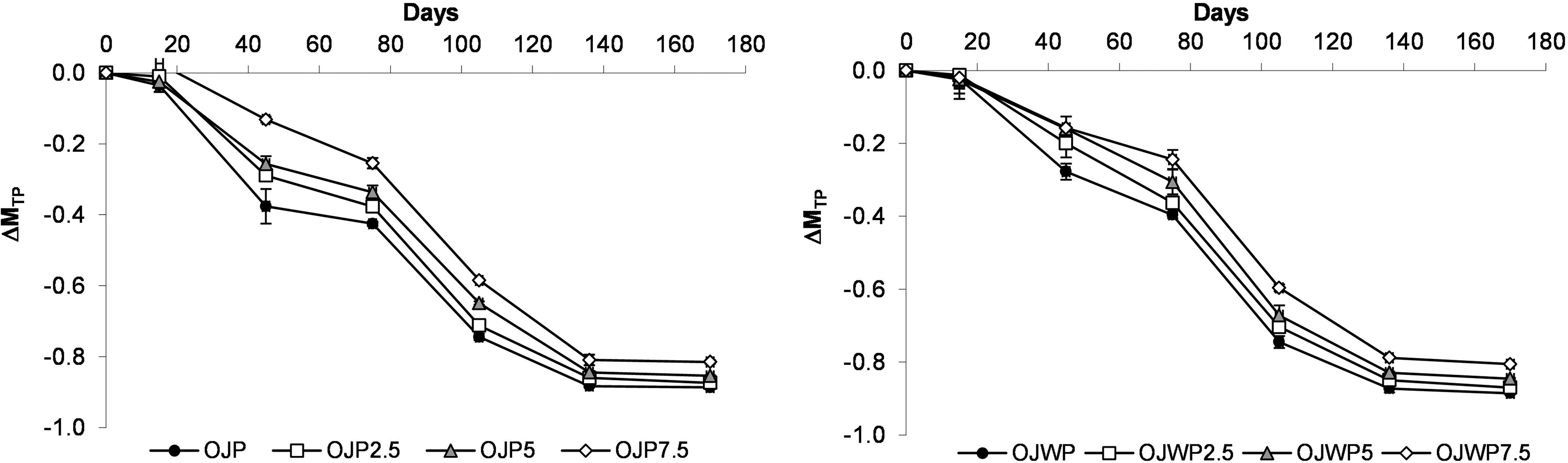
Mean values and standard deviation of total phenols variation (ΔMTP) during storage. OJP, orange juice with pulp; OJWP, orange juice without pulp; 0–7.5, resistant maltodextrin percentage.
Although the loss of TC during the first 15 days is greater in the OJP samples, from day 45 the evolution of TC variations was the same in both OJP and OJWP samples (Figure 4). Thus, the role of orange pulp in TC loss along storage is not significant (p > 0.05). RMD addition improved (p < 0.05) TC protection in all OJP and OJWP samples, being this effect clearer in the OJP samples, as all RMD-added OJP samples ended up with lower TC loss than the RMD-added OJWP samples. A slight decrease of TC content along storage period in pasteurised orange juice has also been reported (Esteve and Frigola, 2008).
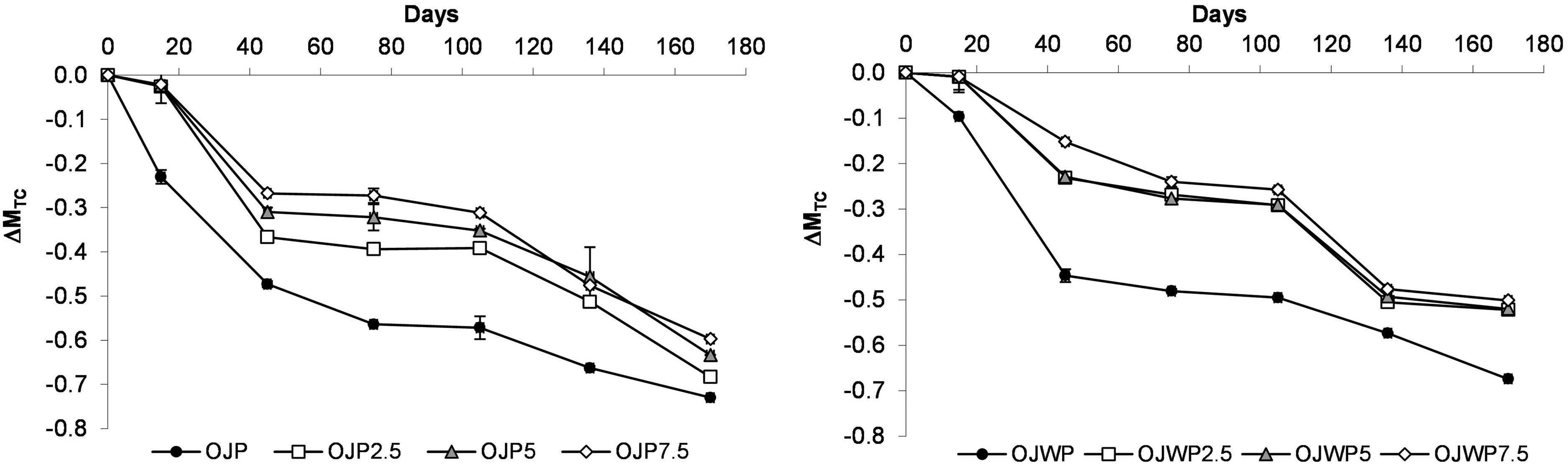
Mean values and standard deviation of total carotenoids variation (ΔMTC) during storage. OJP, orange juice with pulp; OJWP, orange juice without pulp; 0–7.5, resistant maltodextrin percentage.
Figure 5 shows the mean values and standard deviations of AC variation throughout the storage period. OJP0 had less (p < 0.05) AC variations along storage than OJWP0, meaning that orange pulp contributes to improve AC protection of orange juice. Also, all RMD-added samples marked lower (p < 0.05) AC variations in comparison to control samples. In the OJP samples, all RMD-added samples marked similar AC variations (except from day 75), indicating then that higher RMD concentrations in OJP samples did not lead to a significant higher (p > 0.05) protective effect along storage period. In the OJWP samples, however, higher RMD concentrarions led to higher (p < 0.05) protective effect at the first stages of the storage time. At the end of storage time (from days 105 to 170), all RMD-added OJWP samples exhibited also similar AC variations.
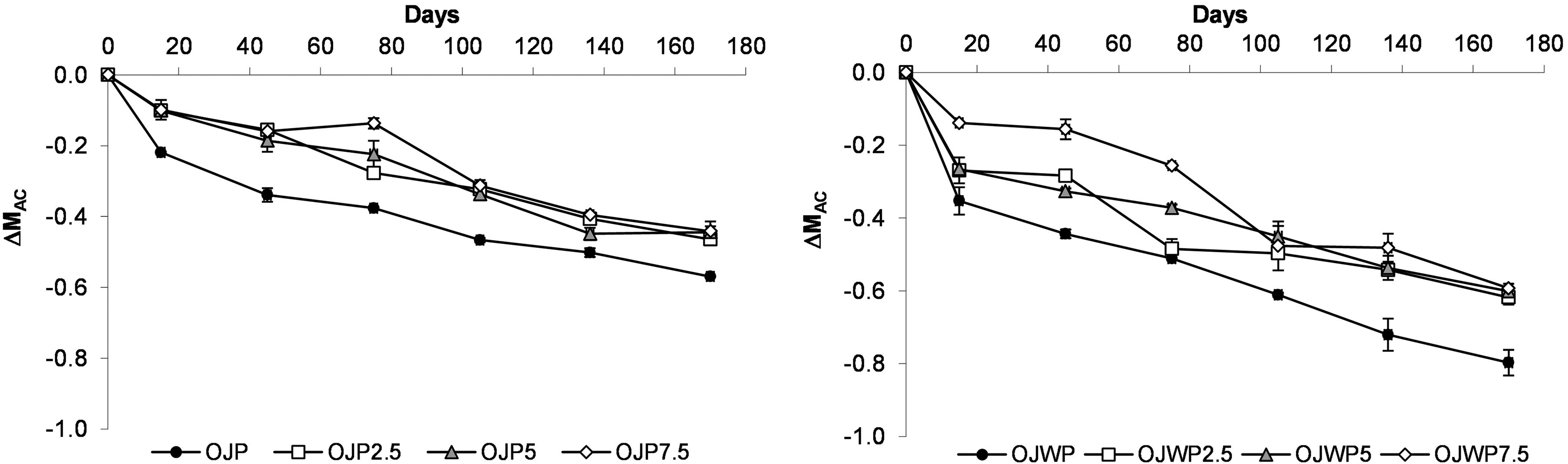
Mean values and standard deviation of antioxidant capacity variation (ΔMAC) during storage. OJP, orange juice with pulp; OJWP, orange juice without pulp; 0–7.5, resistant maltodextrin percentage.
To summarise, all samples were very stable along the whole storage period in terms of °Brix and pH. RMD addition to OJWP samples slightly improved AA loss at the first stages of storage, while it did not have any effect in the OJP samples. In addition, both OJP and OJWP RMD-added samples also increased vitamin C protection at the first stages of storage. Orange pulp did not play any effect on the TP variations along storage, while RMD addition improved TP retention. Regarding TC variations, OJP and OJWP samples had also similar behaviours, and RMD addition better preserved TC along storage in all samples. Finally, orange pulp helped to retain the AC of orange juice, and RMD addition increased AC retention in all samples.
Table 3 shows the mean values (and standard deviations) of studied bioactive compound and the AC at the end of storage period. OJP0 and OJWP0 marked almost the same AA (p > 0.05) at the end of storage time, indicating that orange pulp presence did not dampen AA loss over time. This contrasts to the results at the beginning of the study, where OJWP0 had a higher AA content than OJP0 (Table 2). RMD addition slightly improved (p < 0.05) AA retention, especially in the pulp-free samples. Probably this could be because RMD addition to OJWP samples before pasteurisation process helped to maintain higher AA levels than in the OJP samples. Moreover, OJWP0 and OJP0 also marked similar (p > 0.05) vitamin C content at the end of the storage period. RMD addition improved (p < 0.05) vitamin C retention in all OJP and OJWP samples, being this effect higher with higher RMD concentrations.
Mean values (and standard deviations) of ascorbic acid (AA), vitamin C, total phenols (TP), total carotenoids (TC) content and antioxidant capacity (AC) of pasteurised orange juice after storage, expressed in mg/100 gorange juice.
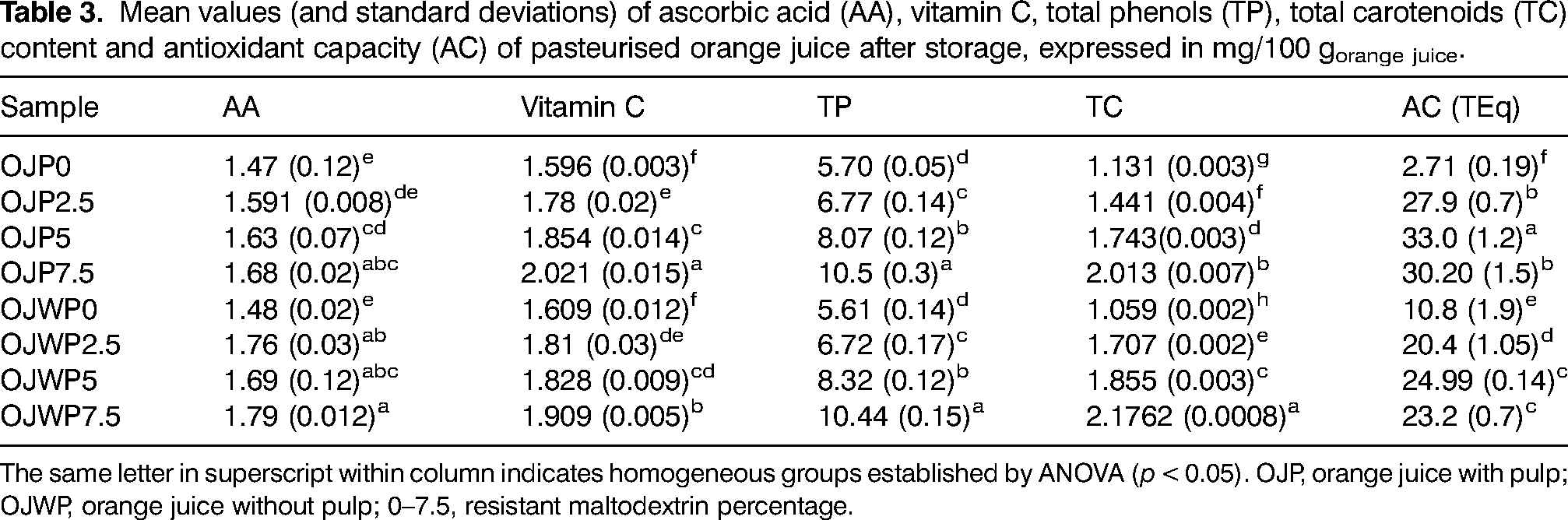
The same letter in superscript within column indicates homogeneous groups established by ANOVA (p < 0.05). OJP, orange juice with pulp; OJWP, orange juice without pulp; 0–7.5, resistant maltodextrin percentage.
In terms of TP content, orange pulp did not play a significant role (p > 0.05) at the end of the storage period, and higher RMD doses helped to maintain higher (p < 0.05) TP levels of all samples. This behaviour agrees with the results at the beginning of the storage time shown in Table 2. Also, OJP0 had a higher TC content than OJWP0 at the firs stage of the storage period. However, despite still showing significance (p < 0.05), this difference narrowed at the end of storage. RMD addition significantly (p < 0.05) improved TC retention in all OJP and OJWP samples, being this effect greater as higher RMD concentrations were added. At the beginning of the storage time, the difference between OJP0 and OJWP0 regarding AC was not significant. However, at the end of the study, OJWP0 marked a significant (p < 0.05) higher AC than OJP0. Therefore, although orange pulp has been suggested as an economic and natural way to add health-related compounds, is not enough to improve the AC of pasteurised orange juice over storage time by itself. However, RMD addition significantly improved (p < 0.05) the AC of all OJP and OJWP samples, being this effect higher in the pulp-added samples. Therefore, it seems that RMD addition could have better preserved the AC of orange pulp over storage time, so the combined addition of RMD and orange pulp resulted in higher AC retention. In addition, the highest AC were obtained at 5% RMD in both OJP and OJWP samples, suggesting that higher RMD concentrations does not necessarily help to maintain higher AC levels.
To explain the influence of the different compounds quantified in this study on the AC of the samples, correlation statistical analyses were performed. All Pearson's correlation coefficient with AC were positives. TC played a major role in the antioxidant capacity of orange pasteurised juices (0.8949, p < 0.05), followed by the vitamin C (0.8410, p < 0.05), AA (0.8333, p < 0.05) and TP (0.8237, p < 0.05).
Figure 6 shows the mean values and standard deviations of L*, a*, b* and total colour variations at days 105 and 170 with respect to the beginning of storage. All samples had a positive variation of all colour parameters, being this clearer at the end of the storage time (day 170). L* values of OJP0 were lower (p < 0.05) than OJWP0, so orange pulp seems to help retaining the original brightness of orange juice. However, RMD addition to OJP samples significantly (p < 0.05) increased L* variations from a concentration of 5%, indicating that OJP5 and OJP7.5 obtained a lighter colouration at the end of storage. On the contrary, RMD did not play a clear role (p > 0.05) on the OJWP samples. Furthermore, OJP0 got a significant (p < 0.05) higher a* variation than OJWP0, meaning that orange pulp presence increased reddish tones of orange juice over storage time. RMD addition to OJP samples slightly decreased a* variations (except from OJP2.5), being this effect clearer with higher doses of RMD and especially at the end of storage (day 170). Therefore, RMD presence at high concentrations seems to dampen the effect of orange pulp on the reddish tones of orange juice. Conversely, RMD increased a* variations in the pulp-free samples. In fact, at the the highest RMD concentration (7.5%), both OJP and OJWP samples at both storage times (days 105 and 170) did not show a significant (p > 0.05) difference in terms of a* values. Also, a* variations were relatively small. For example, at day 105 all samples except from OJP2.5 marked an a* variation lower than 1 unit, indicating that this colour parameter is the most stable one in orange juice. Moreover, OJWP0 presented a higher (p < 0.05) b* variation than OJP0 at the end of storage. This indicates that orange pulp may help to maintain the original yellowness of orange juice. RMD addition to OJP samples increased (p < 0.05) b* variations over time, while in the OJWP samples it did not have a significant (p > 0.05) effect. In fact, the pulp-added samples with higher RMD concentrations (OJP5 and OJP7.5) obtained comparable b* variations to all OJWP samples. Therefore, RMD played a role in turning pulp-added orange juice more yellowish. Regarding total colour differences, all samples marked variations larger than 8 units at days 105 and 170. This indicates that colour difference at the end of storage was clearly perceptible to human eye in comparison to the colour of the samples at the beginning of the study, as it needs at least a colour difference of 3 units to be distinguished (Bodart et al., 2008). OJP0 had a lower total colour variation that OJWP0, indicating that orange pulp helped to better preserve (p < 0.05) the original colour of orange juice. RMD addition to OJP samples at higher concentrations caused greater (p < 0.05) colour variations. This also led to the fact that OJP5 and OJP7.5 presented a clear colour difference in comparison to OJP0 and OJP2.5 at day 170. However, in the pulp-free samples RMD did not show a clear impact (p > 0.05) on the colour differences, as all OJWP samples obtained similar total colour variations.
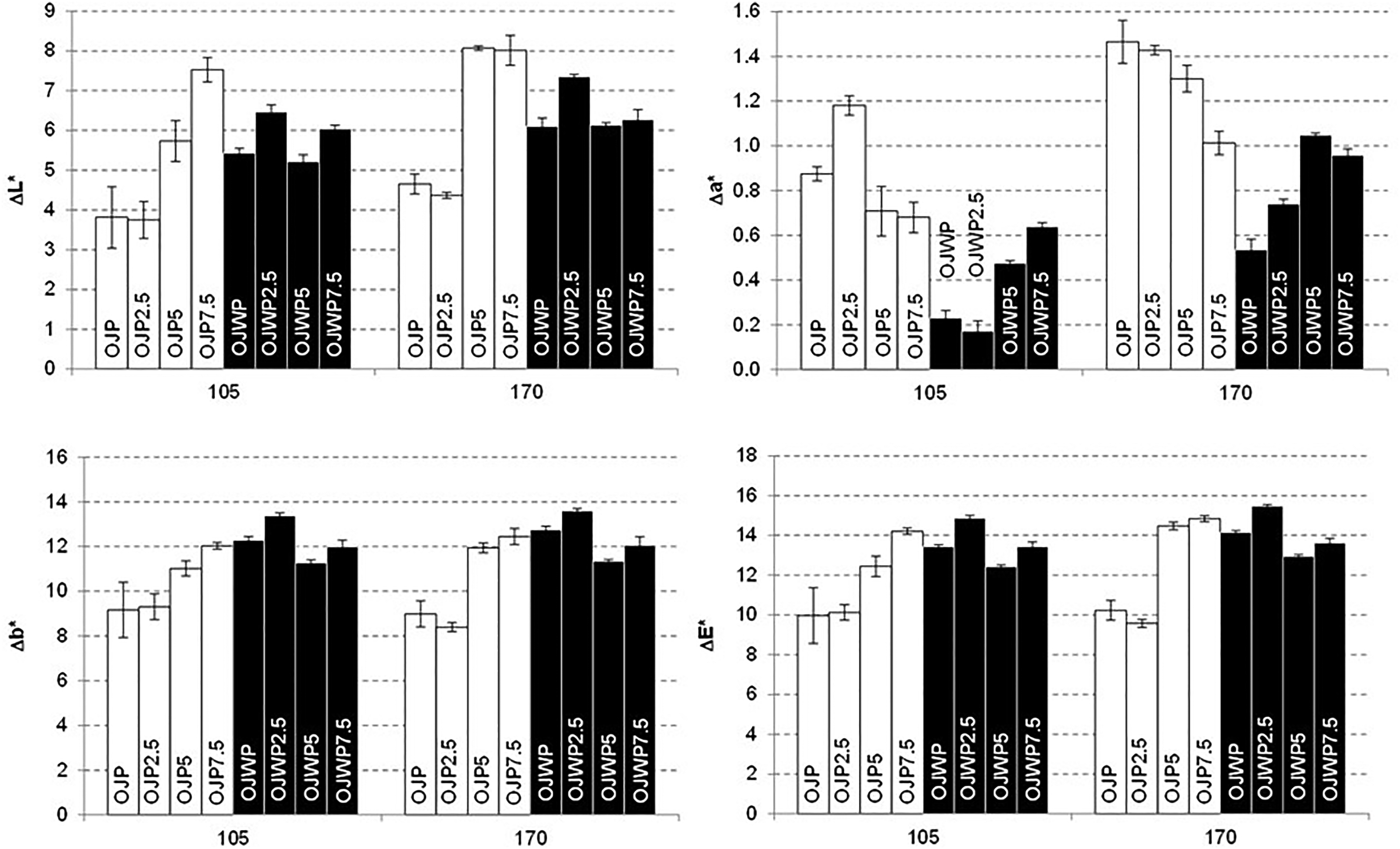
Mean values and standard deviation of L*, a*, b* variation in orange juices (ΔL*, Δa*, and Δb*) and total colour differences (ΔE) at 105 and 170 days of storage. OJP, orange juice with pulp; OJWP, orange juice without pulp; 0–7.5, resistant maltodextrin percentage.
Establishing Pearson correlations between the colour coordinates and the bioactive compounds on the days tested, a high correlation was observed between the AA content and the colour coordinates. Pearson coefficients were for AA-L* 0.8012 (p < 0.05), AA-a* −0.7899 (p < 0.05) and AA-b* 0.9449 (p < 0.05). According to these results, the colour changes are probably due to oxidation reactions of AA.
CONCLUSIONS
RMD addition to orange juice before being pasteurised had a protective effect in all bioactive compounds at the beginning of the storage period, especially in the TP and TC content, which led to higher AC, especially in the OJWP samples. Despite orange pulp has been suggested as a natural way to increase bioactive compounds of food products, its effect over storage time was limited in the control samples, as OJWP0 achieved a higher AC than OJP0. However, RMD addition helped to improve all bioactive compounds retention in all OJP and OJWP samples, especially TP and TC. This led to higher AC in all samples, being this even clearer in the OJP. Therefore, this study enlights the potential use of RMD to better preserve the health-related compounds of pasteurised orange juice along storage time, especially when orange pulp is added.
Footnotes
Author Contributions
Elías Arilla: conceptualization, methodology, formal analysis, investigation, data curation, writing – original draft preparation and editing. Marta Igual: conceptualization, methodology, investigation, data curation, writing – review, supervision. Purificación García Segovia: conceptualization, resources, data curation, writing – review, supervision, funding acquisition. Javier Martínez-Monzó: conceptualization, resources, writing – review, project administration, funding acquisition. Pilar Codoñer-Franch: conceptualization, data curation, writing – review, supervision. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article
