Abstract
The biomedical discovery journey from laboratory to population often encompasses meandering paths, iterative cycles, replication and validation efforts, complex trade-offs between adaptability and fidelity, long lag times and often high costs. The path is sometimes described as a translational spectrum or continuum, with linear or serial-parallel segments and specific transitions. The starting point is usually represented by the inception, ideation or funding opportunity (T0), progressing to laboratory studies (T1), toward human specimen-based studies (T2) and clinic or hospital-based trials (T3), then to community (T4), and populations at large (T5). The latter translation is often accomplished through new policies, laws, regulations or societal normative changes. An innovation-friendly ecosystem requires powerful, effective, and efficient platforms to accelerate the adoption of innovations and discoveries not only in academic organizations that generate new knowledge or provide cutting-edge education, but also in organizations trying to maintain their relevance and competitive advantage by embracing novelty, incremental or breakthrough discoveries, swiftly and efficiently. Local research and development hubs that provide nurture and upkeep of traditional, ‘secular’ innovation trees often must create de novo, ‘engineered’ trees to compensate for gaps in the investigative capability portfolio, and sometimes need to graft ‘engineered’ branches to create ‘hybrid’ trees, able to fulfill successfully and completely the discovery journey. This type of innovation orchard framework may be a powerful environment, ecosystem or milieu, regulating the growth and development of all these types of innovation trees, all with the purpose of agile functionality in implementing research and discovery in population health and clinical care.
Keywords
First movement (Largo): Initium
Since the turn of the 21st century, biomedical research has made tremendous and very fast progress, in parallel with an explosion in computational capabilities, impressive artificial intelligence tools and unprecedented technological advances. Even in very agile and functionally mature organizations, these high-speed advances represent significant barriers to nimble adaptation, successful implementation, adequate upscaling or cultural integration. All these processes are means to accomplish commercial go-to-market or to achieve the desired specific outcomes – epistemologically, in revenue, in health, in overall quality of life of their stakeholders, etc. These fast and unexpected developments may also induce organizational confusion or decision paralysis related to optimal resource allocation for research and discovery departments and innovation-related operations, or for best implementation strategies.
The article proposes a model of integration of the traditional organic growth in the translational research continuum (presented as ‘secular’ innovation trees), with strategically guided, complementary growth (‘engineered’ and ‘hybrid’ trees), all in a highly functional ecosystem conceptualized as innovation orchards (see Box). The proposed model invites leaders, managers and strategic planners to use this type of functional design for better alignment of current and future resources to ensure that more innovations, inventions, scientific discoveries, incremental improvements or breakthrough ideas make it to the real world, faster and with more palpable health benefits to the real end-beneficiary, the member of the society.
Second movement (Andante): Translation
The early and standard meaning of the term
In contrast,
Third movement (Allegro): Pomarium
In 2011, the National Institutes of Health (NIH) created the National Center for Advancing Clinical and Translational Science (NCATS), with the specific aims to pursue, encourage, catalyze, and grow funding opportunities for disruptive translational innovation, using both intra- and extramural mechanisms. 10 Over the past decade and a half, the NCATS, through Clinical and Translational Science Award (CTSA) mechanisms, funded more than 60 hubs in order to spur the development of local and loco-regional translational research partnerships and to establish robust local translational science capabilities. The CTSA hubs and other local translational science engine models have been serving well the research stakeholders’ support needs and end-users’ necessities by creating these advanced translational research functionalities.3,6 Irrespective of the mechanism employed, an organization should consider core infrastructures and functions such as local CTSA hubs as powerful models of trans-disciplinary collaboration and translational science engines able to build and strengthen necessary investigative platforms, catalyze their functions and optimally calibrate long-term sustainment.
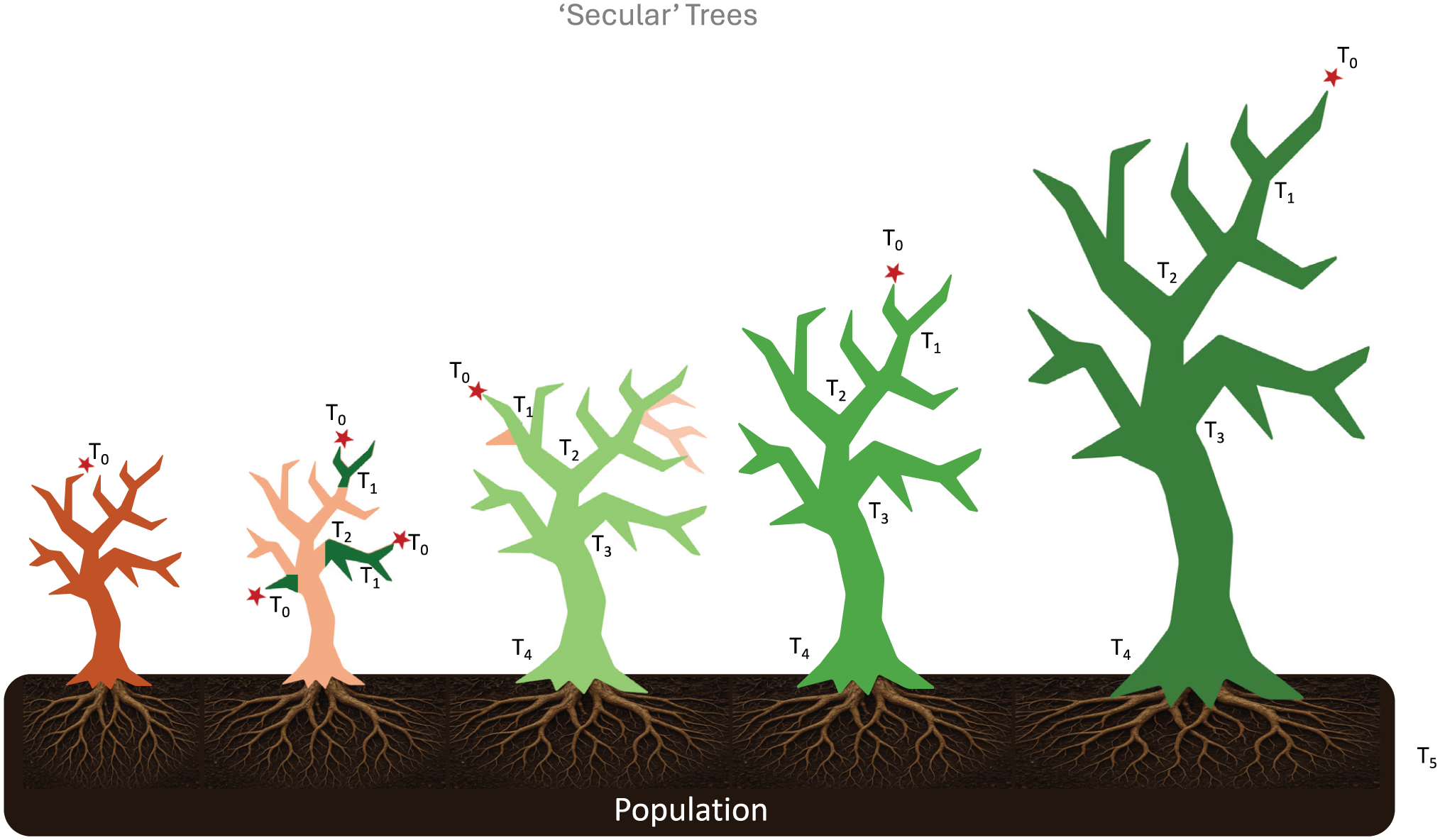
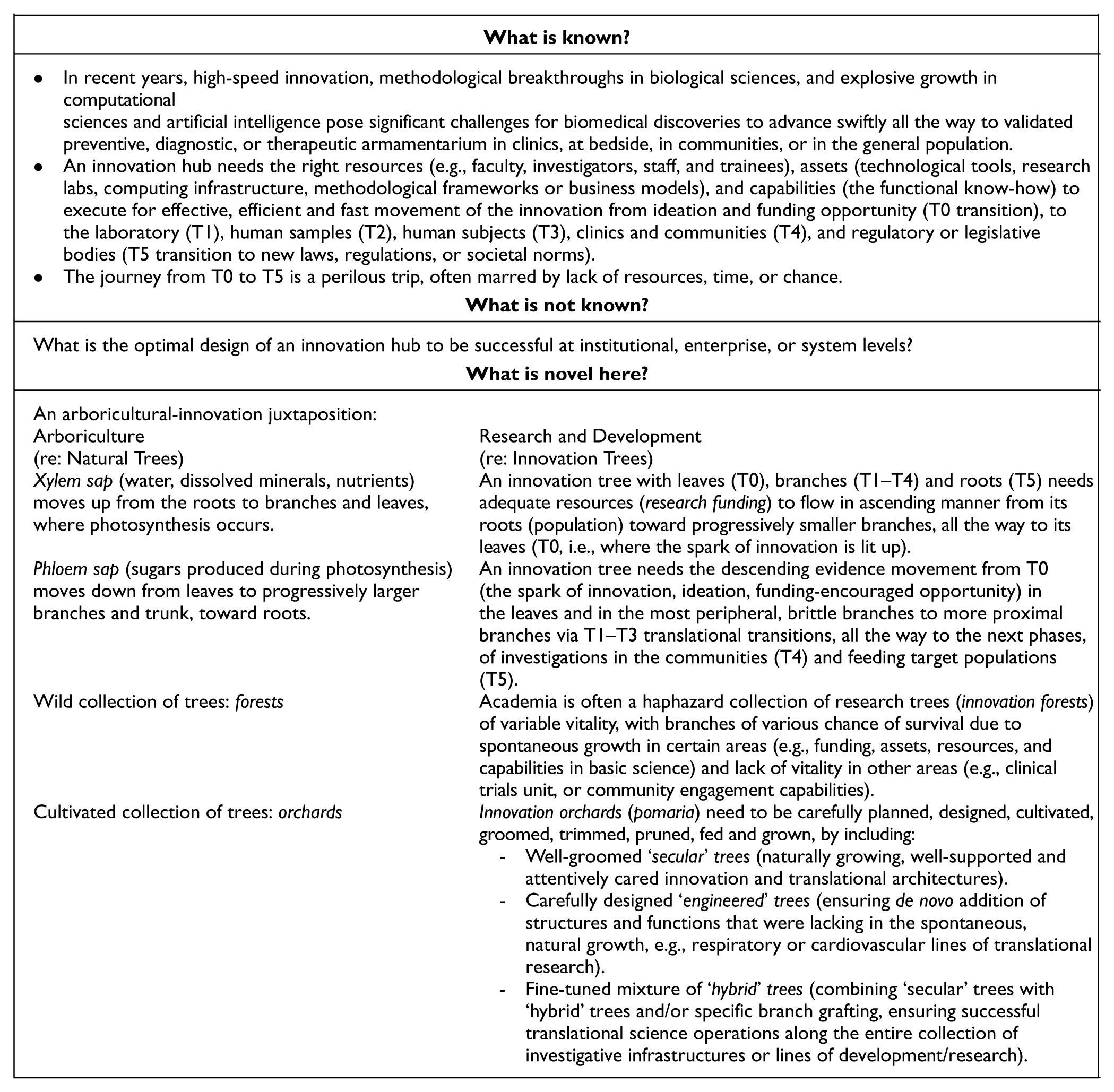
Organic growth patterns of the ‘secular’ trees of research and discovery, guided by spontaneous questions, organizational areas of strength or induced by specific funding opportunities.
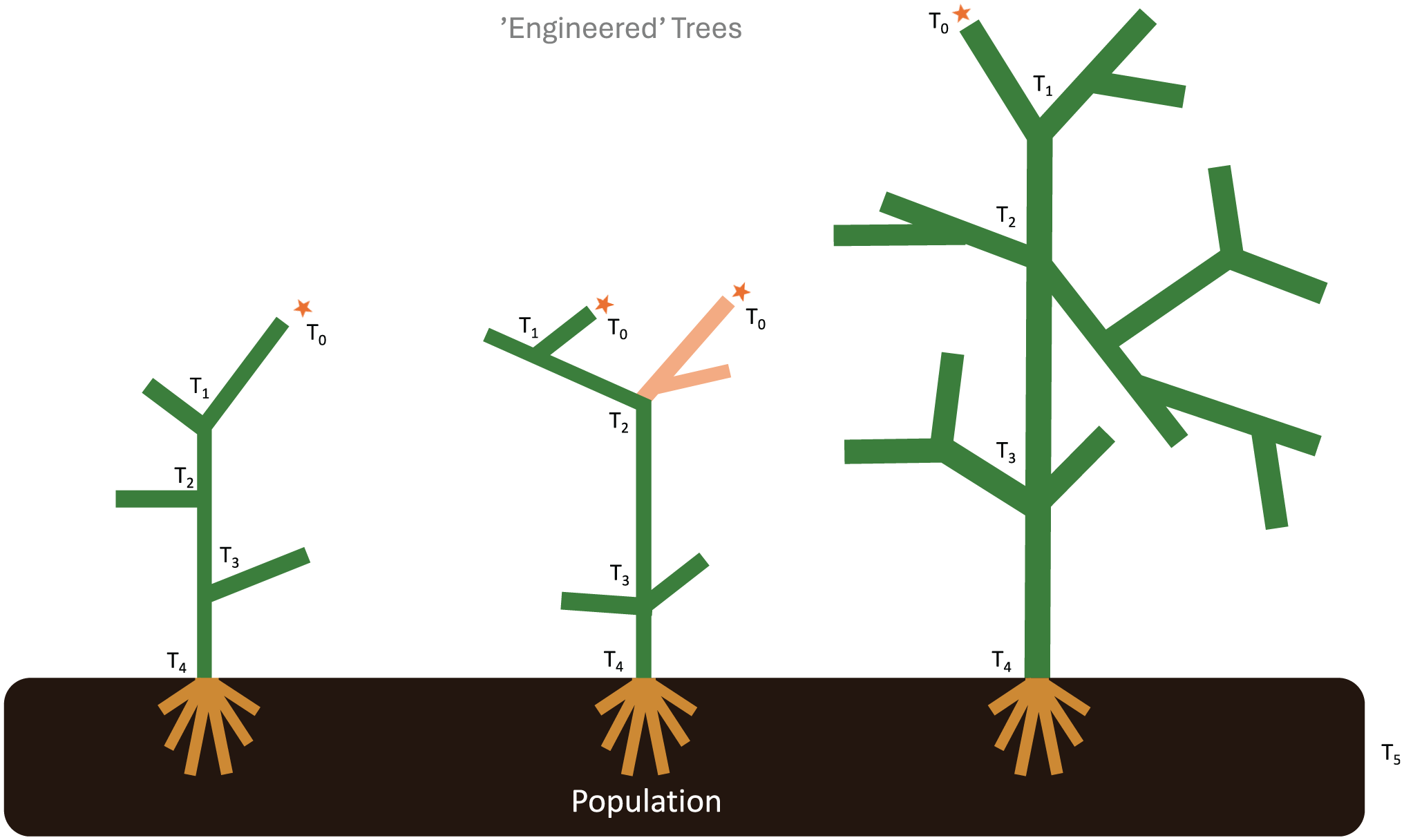
Strategically guided research and development capabilities (‘engineered’ trees), fulfilling known infrastructural gaps, weaknesses or needs.
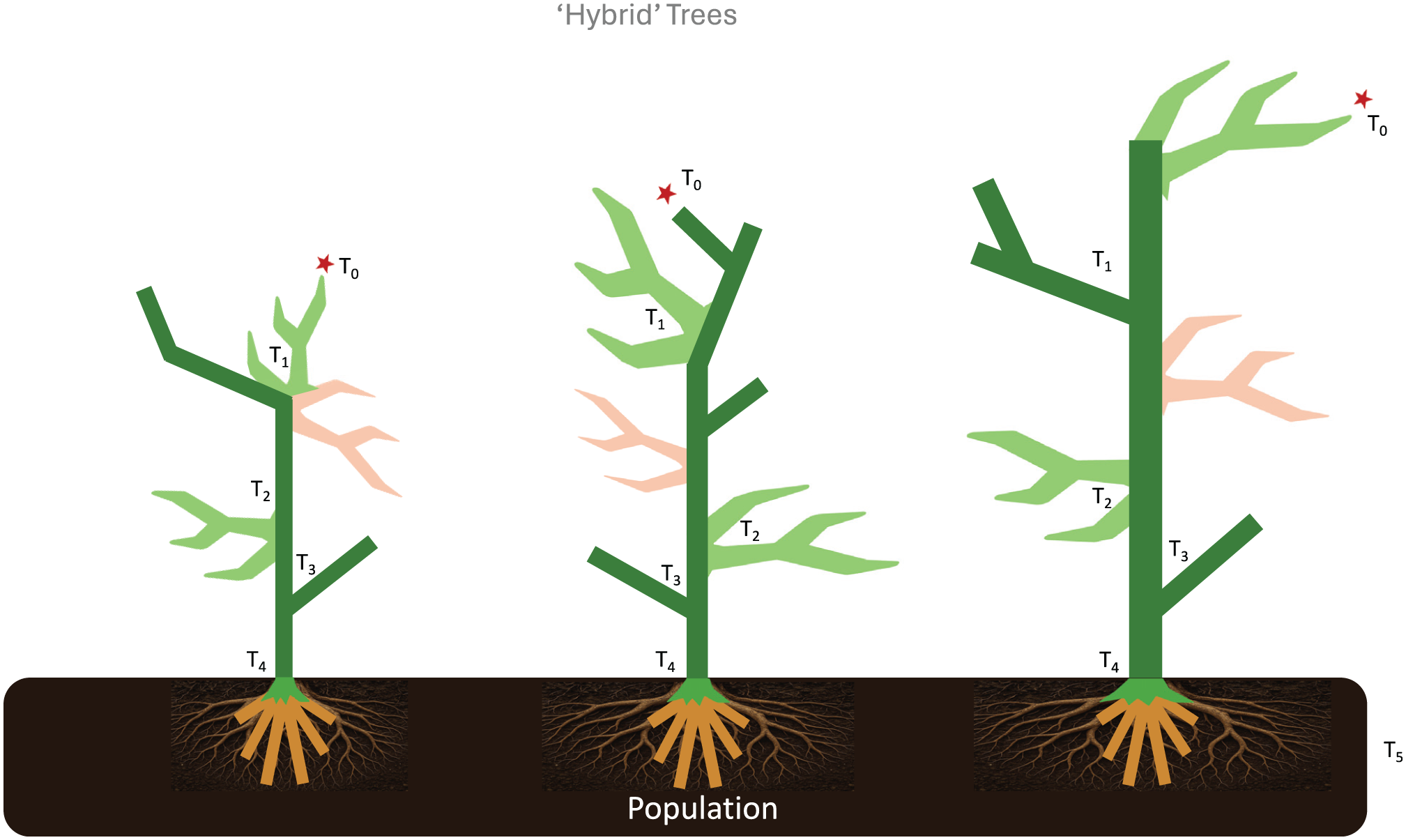
Grafted ‘hybrid’ trees, combining organic growth of the ‘secular’ trees with strategically ‘engineered’ trees to accomplish a more robust, comprehensive and sustainable investigative platform.
For example, organization X has a strong T0–T2 discovery segment in investigating airway disorders such as asthma (i.e., great external and internal funded opportunities, great basic and translational research infrastructures and capabilities, even capacity to perform well in early phase asthma clinical trials or first-in-human studies), but lacks the T3–T5 segment of the TS spectrum (i.e., it lacks the capacity to conduct larger phase II/III asthma clinical trials, comparative effectiveness, implementation or hybrid studies, population health studies or to get involved in health policy advocacy). ‘Engineering’ the novo a T0–T5 full spectrum under its new strategic priority of developing neurosciences allows organization X’s ‘controlled’ and capability-wide growth of a clinical trial unit (CTU), developing a disease-agnostic implementation science core and a data science support unit. This allows easy ‘grafting’ of a subunit of the new CTU for future asthma studies, effective intervention of the dissemination and implementation (D&I) consultative groups in the design and conduct of late-phase clinical trials, comparative effectiveness or hybrid trials and population-based studies. Furthermore, organization A has a strong T3–T4 arm of studies in the specific area of chronic obstructive pulmonary disease, which can easily be ‘grafted’ on the asthma tree trunk, creating an effective ‘hybrid’ tree.
In another example, organization Y has a full complement of ‘secular’ trees in its innovation orchard, covering most of the TS spectrum except perhaps in leading and coordinating large-scale phase III and IV clinical trials. Its approach in assuming the role of the coordinating center for a large multicentric CT was to reach out to the Trial Innovation Network (TIN) of the CTSA Consortium, which provided the necessary resources and the
Example of a working activity matrix for an innovation orchard development in pulmonology (e.g., research in asthma) at an academic medical center level.
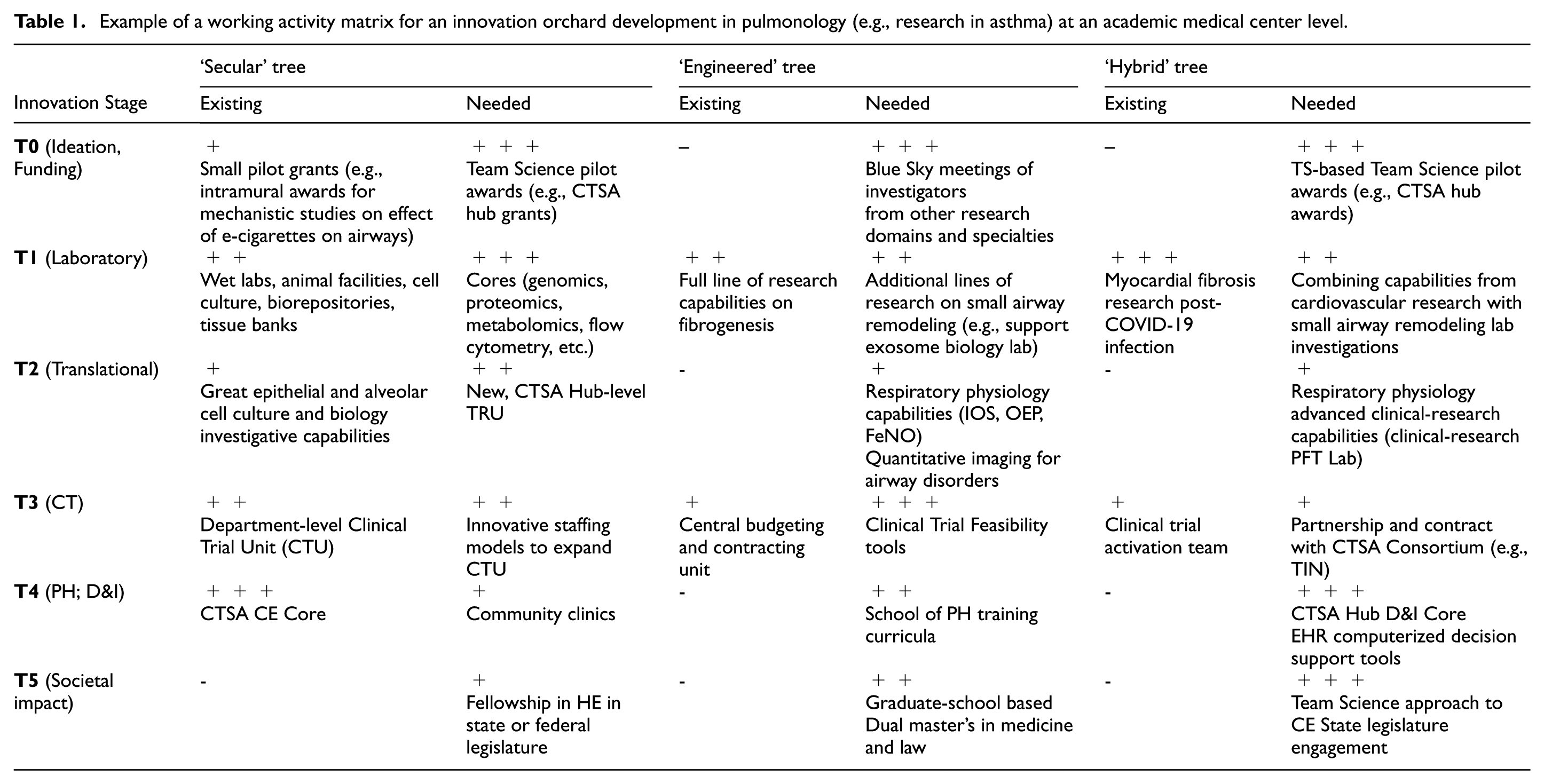
Example of a working activity matrix for an innovation orchard development in respiratory medicine or pulmonology (e.g., research in asthma) in a state or federal research funding agency.
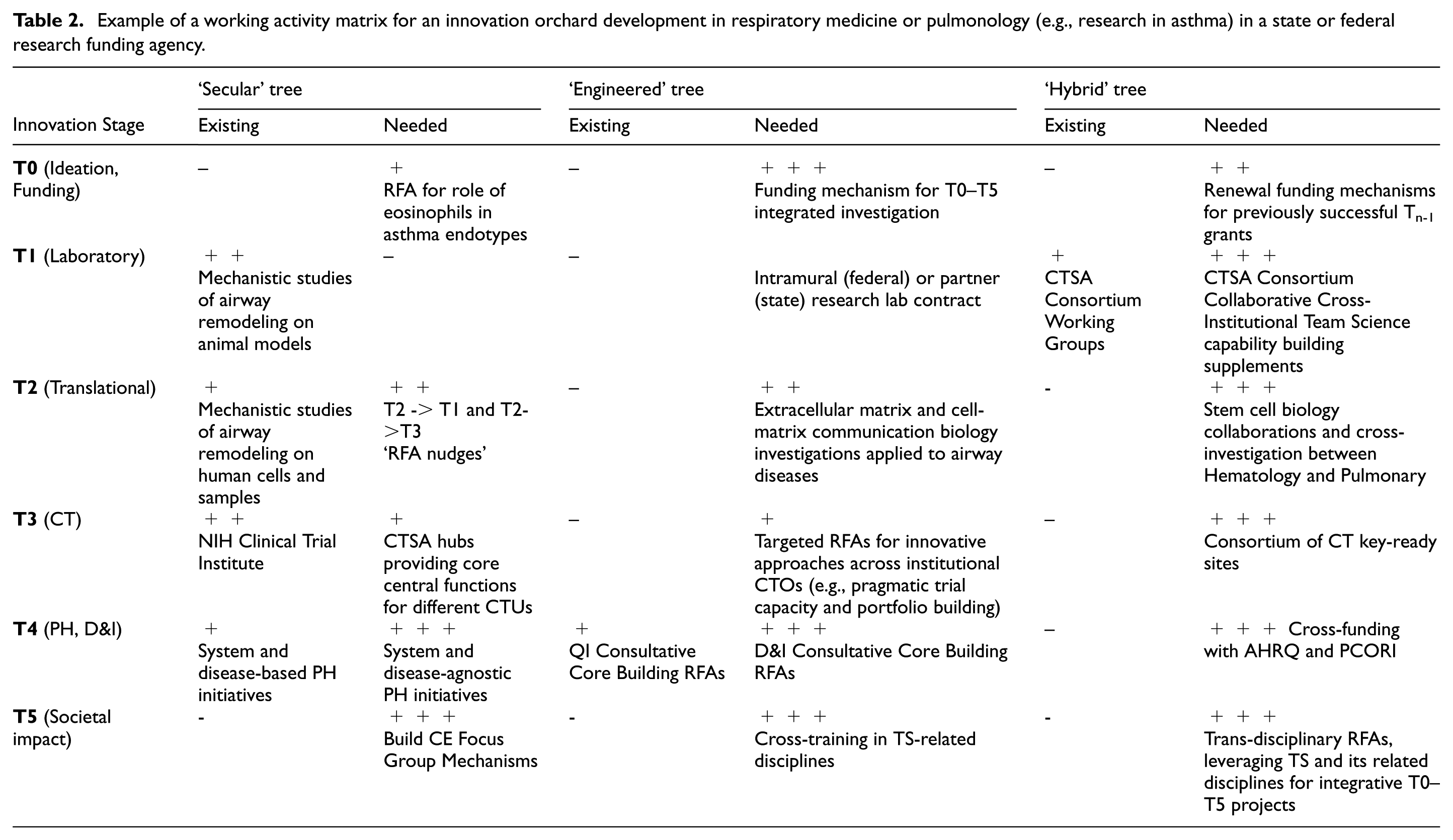
Example of a working activity matrix for an innovation orchard development in pulmonology (e.g., a procedure/device for airway thermoplasty in asthma) in a device company.
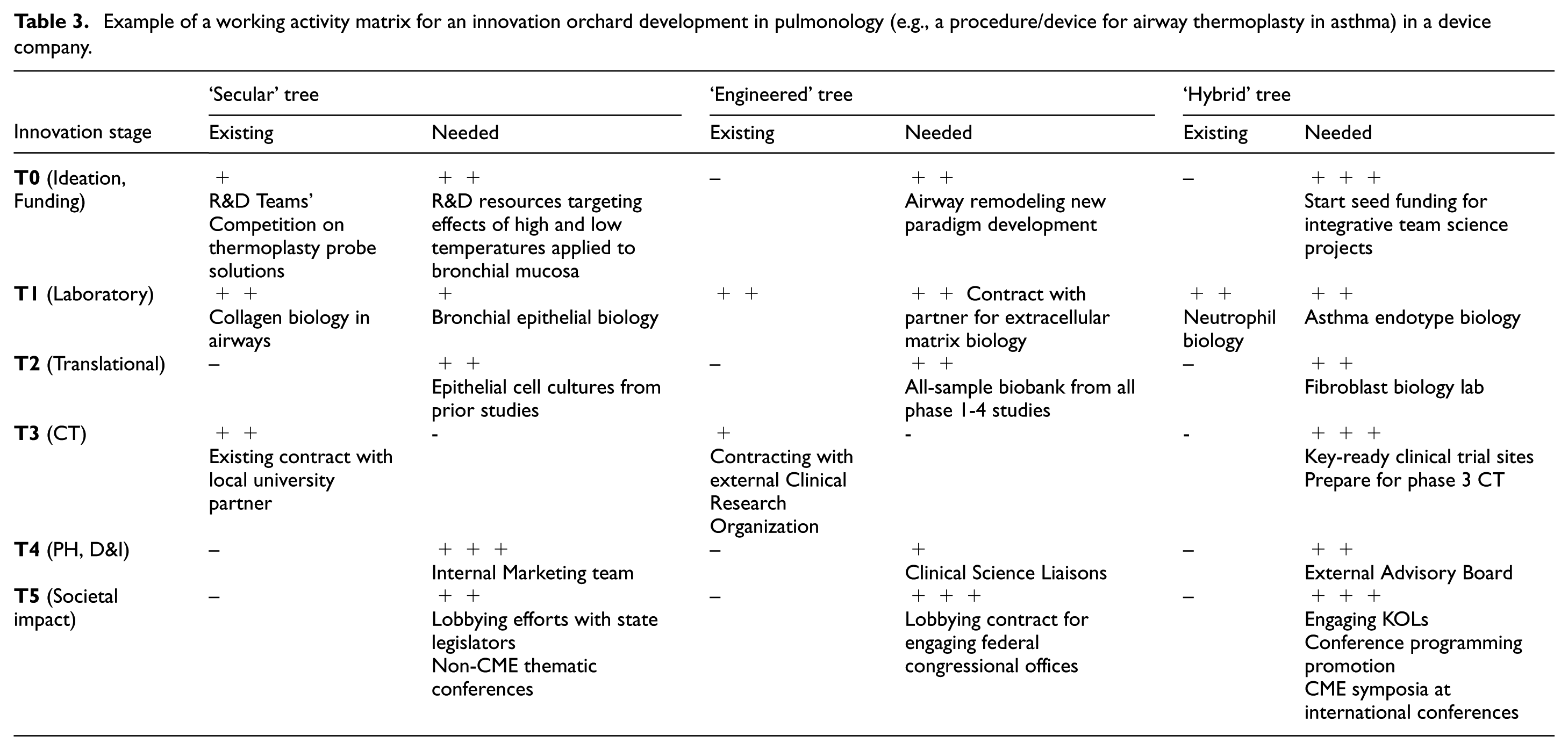
An example of a working priority matrix by existing and necessary resources, assets, and capabilities in a health sciences university (E: Existing; P1: Priority 1; P2: Priority 2; P3: Priority 3).
Fourth movement (Vivace): Summary
Using the proposed paradigm, one organization may find that its innovations flow easily down the branches of some ‘secular’ trees representing molecular biology capabilities, early and late clinical trials, but perhaps needs some ‘engineering’ for the branches required for implementation and dissemination capabilities. Other institutions may have competitive advantage at, for example, making very quickly AI-based 3D models and synthesizing various molecules based on the high-resolution scanned structure of patients’ receptors acting as personalized therapeutic targets in various diseases, but may lack the infrastructure to conduct the clinical trials on human subjects, so ‘engineering’ the branches required to perform this type of investigative work may be required. In another example, the throughput of discovery using cell biology departments in an organization may surpass significantly its ability to timely secure intellectual property rights for them or to conduct clinical trials involving these new products. Another institution may find its strengths in developing molecular targets for a certain disease, in data science capabilities, in securing intellectual property rights and in implementing its discoveries in the local hub, but lacks the reach to community stakeholders and potential target populations – this can be ‘synthesized’ the novo (organically or through acquisitions, alliances, innovation networks, etc.) for better efficiencies in the research ecosystem.
In summary, the current article proposes a conceptualization of what we call innovation orchards, applicable best to academic institutions, research and development departments, equipment and pharma industry, teaching hospitals, or research institutes. The model spells out the need for the organizations to identify the necessary resources, assets and capabilities to be able to design, build and maintain innovation orchards that provide the best care to existing translational ‘secular’ trees, synthesize adequate missing species of ‘engineered’ trees, and ultimately, provide the right grafting of ‘secular’ and ’engineered’ trees into necessary ‘hybrid’ innovation trees. The model may need further exploration and investigation, rigorous phenotypic and/or endotypic characterization, careful implementation and thorough outcome and impact evaluation, so that distinct models of successful translational science engines emerge and lead to the most agile innovation ecosystems of the 21st century.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
