Abstract
Advancements in cancer therapy have led to improved patient survival but have also introduced an increasing risk of cardiotoxicity, particularly with agents such as anthracyclines, trastuzumab, and various targeted treatments. Cardiotoxic effects may present as myocardial injury, systolic or diastolic dysfunction, or heart failure (HF), often developing insidiously before clinical symptoms become evident. Early detection is therefore crucial to preserve cardiovascular health in oncology patients. This narrative review examines the expanding role of cardiac biomarkers; including high-sensitivity troponins, natriuretic peptides, Galectin-3, soluble ST2, and novel molecular markers like microRNAs, in identifying and monitoring chemotherapy-induced cardiac injury. It also highlights how these biomarkers complement advanced imaging techniques such as global longitudinal strain, cardiac magnetic resonance, and myocardial deformation imaging to detect subclinical changes before ejection fraction declines. Furthermore, we explore the clinical relevance of biomarker-guided monitoring, the role of cardioprotective agents such as dexrazoxane, and the emerging utility of stress-based functional testing in evaluating cardiac reserve. Overall, the review emphasizes a tailored, multimodal approach that integrates biomarkers, imaging, and clinical risk profiling to enhance early intervention and sustain cancer treatment without compromising cardiac function.
Introduction
Modern cancer therapies have improved survival but increased cardiovascular complications. Treatments like chemotherapy, targeted agents, and immunotherapy can damage the myocardium, leading to HF, infarction, or dysfunction. 1 This growing risk has led to the rise of cardio-oncology, 2 which focuses on understanding and managing cardiovascular effects of cancer therapy while preserving oncologic treatment continuity.
The timely identification of cardiotoxicity is of paramount importance in order to avert irreversible myocardial damage, given that conventional imaging modalities may not consistently reveal subclinical cardiac impairment. In this regard, cardiac biomarkers have emerged as indispensable instruments for evaluating cardiovascular risk prior to the commencement of oncological treatment, monitoring cardiac health throughout the course of therapy, and assessing long-term sequelae in survivors. 3 These biomarkers, which can be quantified through serological analyses, yield critical information regarding myocardial stress and injury, frequently preceding the manifestation of clinical symptoms. 4
Key biomarkers, including cardiac troponins and natriuretic peptides, are extensively employed in the identification of cardiac dysfunction. Furthermore, recent investigations examine the significance of inflammatory markers, such as C-reactive protein, interleukin-6, myeloperoxidase (MPO), Galectin-3, and growth differentiation factor-15 (GDF-15), in forecasting cardiotoxicity. 5 Advances in genomics, proteomics, and microRNA profiling present promising novel methodologies for detecting patients at an elevated risk of cardiovascular complications and facilitating tailored cardioprotective interventions. 6
With the growing acknowledgment of the cardiovascular hazards linked to cancer treatment modalities, this literature review seeks to investigate the significance of cardiac biomarkers in the identification, surveillance, and management of cardiotoxicity. Through the incorporation of these biomarkers into clinical protocols, healthcare practitioners can facilitate the early detection, refine therapeutic strategies, and ultimately enhance both cardiovascular and oncological prognoses for patients receiving cancer interventions.
Definition of cardiotoxicity
In oncology, cardiotoxicity is increasingly recognized as more than a reduction in left ventricular ejection fraction (LVEF), although a decline of ≥10% to below 53% remains a commonly used threshold in imaging studies. 7 Breast cancer therapies, in particular, can impair cardiovascular health through diverse mechanisms, including arrhythmias, hypertension, ischemia, thromboembolism, and pericardial inflammation.
Traditionally, chemotherapy-induced cardiotoxicity has been classified into two types: type I, typically linked to anthracyclines and associated with dose-dependent, often irreversible myocardial injury; and type II, associated with agents like trastuzumab, characterized by non-dose-dependent and potentially reversible effects. However, this binary model is now viewed as overly simplistic. Combined use of these agents, along with radiotherapy or other cardiotoxic drugs, amplifies the risk of myocardial damage and underscores the limitations of this classification. 8
Early detection is critical, as many patients remain asymptomatic until advanced cardiac dysfunction or cardiomyopathy develops. Routine cardiovascular assessments, including echocardiography and clinical monitoring, are essential for identifying at-risk individuals. Preventive strategies and timely initiation of cardioprotective therapies can significantly reduce long-term complications. While some dysfunctions, particularly those linked to trastuzumab, may be reversible, others are complicated by comorbidities such as aging, hypertension, and diabetes, which further obscure the clinical picture. 9
Current perspectives in cardio-oncology
As cardio-oncology continues to evolve, professional organizations have worked to better define and categorize cardiotoxic effects associated with cancer therapies. The National Cancer Institute’s Common Terminology Criteria for Adverse Events (CTCAE) provides a framework for reporting cardiovascular complications, including both clinical symptoms and subclinical findings such as reduced ejection fraction or abnormal biomarkers. 1 However, CTCAE still lists outcomes like HF and systolic dysfunction separately, lacking a unified classification for cardiotoxicity. 10
In 2002, the Cardiac Review and Evaluation Committee introduced criteria for evaluating trastuzumab-related cardiotoxicity, incorporating clinical signs, physical exam findings, and imaging, including LVEF measurements. 11 In parallel, the American College of Cardiology/American Heart Association HF staging system offers a practical structure, ranging from at-risk but asymptomatic patients (Stage A) to those with advanced HF (Stages C and D). 12
Importantly, progression from Stage B (asymptomatic LV dysfunction) to Stage C (symptomatic HF) is linked to a sharp decline in 5-year survival, from 96% to 75%, and a fivefold increase in mortality. 13 This underscores the value of early recognition and intervention. Evidence shows that initiating standard HF therapies after detecting subclinical dysfunction, especially in anthracycline-related cases, can improve LV function and reduce adverse outcomes. 14
Cardio-oncology has evolved alongside new cancer therapies with cardiac risks. Despite growing awareness, predicting who will develop cardiotoxicity remains challenging due to nonspecific symptoms, variable timing, and unpredictable disease course. 15
Current methodologies for diagnosing cardiotoxicity
Cardiotoxicity, particularly characterized by left ventricular systolic dysfunction, represents a well-documented adverse consequence associated with various oncological therapies, notably anthracyclines and agents that target HER2. This phenomenon frequently initiates as a subclinical reduction in LVEF, which, if not identified, has the potential to advance to clinically evident HF. In order to counteract this progression, systematic cardiac surveillance prior to, during, and subsequent to chemotherapy is imperative for facilitating timely intervention.7,16
Transthoracic echocardiography remains the primary tool for baseline and ongoing cardiac assessment, with monitoring frequency tailored to individual risk factors such as age, comorbidities, prior radiation, and cumulative chemotherapy exposure. Advanced imaging techniques, including global longitudinal strain (GLS) and cardiac magnetic resonance (CMR), offer greater sensitivity for detecting early myocardial injury—often before LVEF declines.2,4 Complementary use of cardiac biomarkers such as troponins, brain natriuretic peptide (BNP), and galectin-3 provides additional insight into myocardial stress and fibrosis, supporting early detection and enhancing diagnostic accuracy. 1
Notwithstanding these advancements, significant variability in monitoring protocols remains evident across healthcare institutions (Table 1). Ongoing research is directed towards the development of risk-based surveillance strategies aimed at optimizing both patient outcomes and the utilization of resources.1,17
Summary of cardiotoxicity associated with breast cancer therapies.
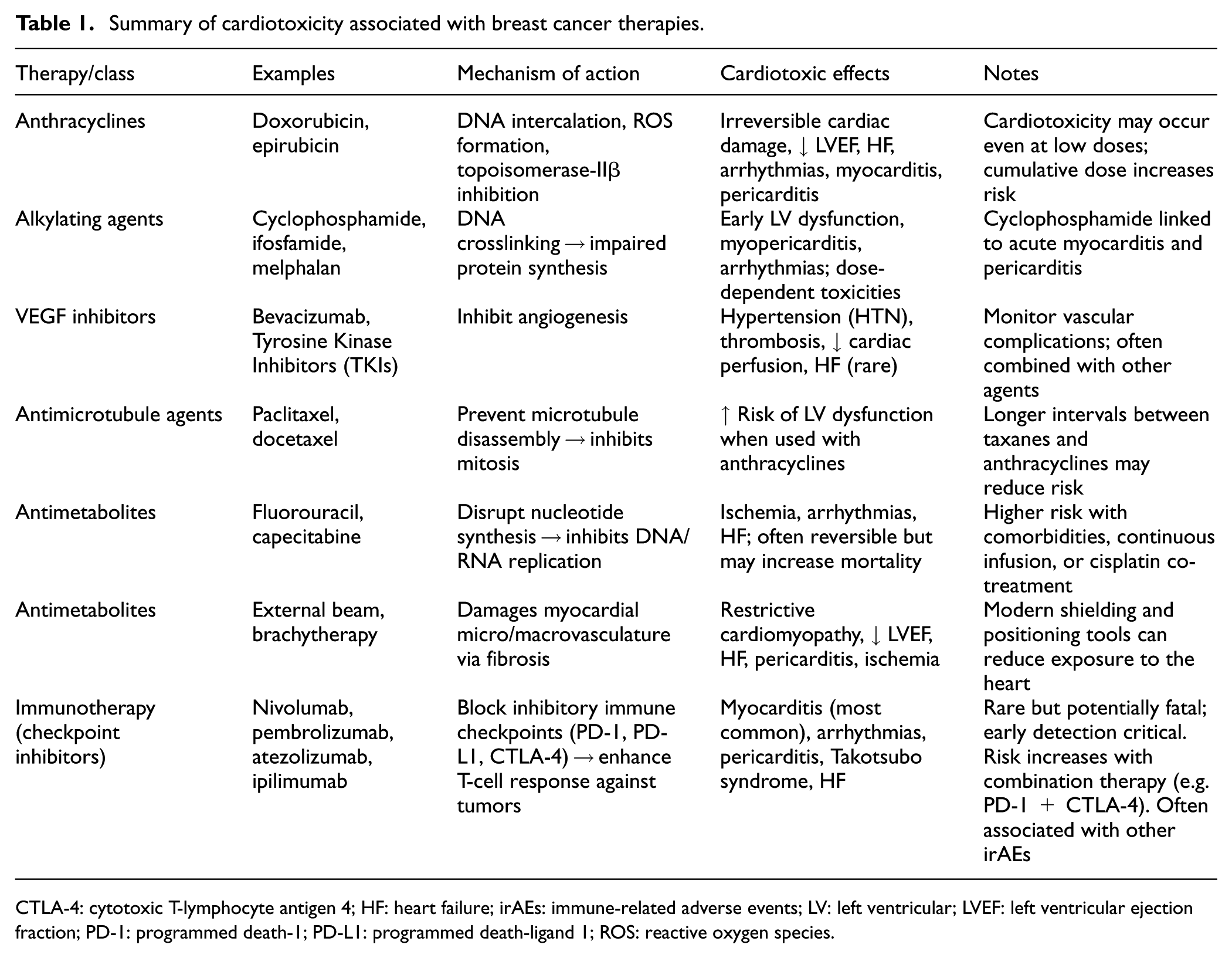
CTLA-4: cytotoxic T-lymphocyte antigen 4; HF: heart failure; irAEs: immune-related adverse events; LV: left ventricular; LVEF: left ventricular ejection fraction; PD-1: programmed death-1; PD-L1: programmed death-ligand 1; ROS: reactive oxygen species.
Types of LV dysfunction and monitoring strategies
Systolic dysfunction of the left ventricle
Echocardiographic assessment of LVEF remains the cornerstone for evaluating systolic function in patients receiving cancer therapy. A decline of more than 10 percentage points to below 53%, confirmed by follow-up imaging within 2–3 weeks, is considered diagnostic of systolic dysfunction according to current imaging society recommendations. 8 Advanced techniques such as three-dimensional echocardiography and contrast-enhanced imaging improve the detection of subtle myocardial changes and more accurately quantify LVEF decline. 6 Evaluation frequency should be individualized based on each patient’s risk profile and clinical status.
Although multiple-gated acquisition (MUGA) scans have traditionally been employed, their inherent limitations—particularly the deficiency in providing insights into cardiac morphology and diastolic function, as well as concerns regarding radiation exposure—significantly constrain their clinical applicability. 18 Moreover, research has indicated that MUGA may not consistently identify cardiotoxicity in patients treated with doxorubicin who subsequently progress to HF. 9
Cardiac MRI is considered the most accurate modality for evaluating structural and functional cardiac changes. However, its routine use in oncology is often limited by cost, availability, and patient tolerance. 9 Regardless of the imaging approach, early detection of cardiac dysfunction is essential to enable timely therapeutic adjustments and initiation of cardioprotective strategies. Given the absence of standardized guidelines for managing cardiotoxicity in breast cancer, a multidisciplinary approach is critical to ensure individualized patient care.19,20
Diastolic dysfunction of the left ventricle
The assessment of diastolic function in the context of oncological treatment has garnered heightened scholarly interest. Emerging evidence indicates that diastolic dysfunction may precede the onset of systolic dysfunction, thereby underscoring the necessity for a comprehensive evaluation of myocardial relaxation, filling pressures (e.g. early diastolic filling velocity of the left ventricle/early diastolic velocity of the mitral annulus (E/e′) ratio), and tissue Doppler imaging parameters during echocardiographic assessments. 21 The prompt identification of diastolic anomalies can create an essential opportunity for therapeutic intervention prior to the establishment of irreversible myocardial damage.
Myocardial deformation imaging in cardio-oncology
Advanced echocardiographic modalities have improved the early detection of subclinical myocardial injury, often preceding measurable declines in ejection fraction. Myocardial deformation indices; such as strain, strain rate, and torsion, provide sensitive markers of early dysfunction, particularly in patients undergoing cardiotoxic cancer treatments. These metrics are primarily evaluated using tissue Doppler imaging and speckle-tracking echocardiography (STE), with STE offering superior spatial resolution and angle independence. 22 STE has shown strong agreement with sonomicrometry and cardiac MRI, supporting its reliability as a non-invasive diagnostic tool. 2
GLS, a key deformation index, reflects myocardial shortening during systole and is assessable in longitudinal, radial, and circumferential planes. 23 GLS is more sensitive than LVEF in detecting early dysfunction and predicts cardiotoxicity in patients receiving trastuzumab or anthracyclines. Studies show that absolute GLS values <−19% or ≥11% relative declines forecast future LVEF reduction.24,25 A meta-analysis confirmed GLS as a stronger mortality predictor than LVEF in both cancer and non-cancer populations. 26
Altered torsion and rotational mechanics have been observed shortly after anthracycline therapy and in long-term survivors of pediatric cancer, even when LVEF is preserved. 27 Three-dimensional STE, which simultaneously assesses strain, torsion, and dyssynchrony, may offer enhanced sensitivity in detecting persistent myocardial injury. Preliminary data in pediatric survivors support its potential value over conventional metrics. 28
Despite these advantages, the clinical adoption of strain imaging faces several challenges, including variability across imaging platforms, dependency on high-quality image acquisition, and a lack of standardized thresholds and timing. 29 Nevertheless, longitudinal strain has shown strong reproducibility, 30 reinforcing its role in the routine surveillance of treatment-related cardiotoxicity.
Natriuretic peptides and early cardiotoxicity detection
A study of 149 breast cancer patients receiving anthracycline chemotherapy assessed BNP’s value in predicting cardiotoxicity. Those who developed cardiotoxicity had significantly higher BNP levels. A post-chemotherapy BNP cutoff of 107.9 pg/ml showed 53.8% sensitivity and 79.4% specificity. Elevated BNP also correlated with poorer cardiotoxicity-free survival, indicating its potential as a biomarker for early detection and risk assessment of anthracycline-induced cardiac damage. 8
Shared biomarkers between HF and cancer
Galectin-3, a β-galactoside-binding lectin, is linked to both HF and cancer due to its role in fibrosis and inflammation. Elevated galectin-3 levels are associated with myocardial remodeling and HF progression. In cancer, it promotes adhesion, angiogenesis, and metastasis. This dual role makes galectin-3 a promising biomarker for identifying individuals at risk of both cardiovascular and oncologic conditions. 31
Soluble ST2 (sST2), part of the IL-1 receptor family, is released during cardiac stress and inflammation and is a validated prognostic marker in HF. High sST2 levels are linked to fibrosis, remodeling, and increased mortality in HF. Beyond cardiology, ST2 affects tumor immunity, and dysregulated IL-33/ST2 signaling is tied to cancers like breast, lung, and colorectal. This dual role highlights ST2 as a biomarker for identifying patients at risk for both cardiac and oncologic disease. 32
CMR for early detection of cancer therapeutics
CMR provides superior evaluation of myocardial structure and function compared to other imaging modalities, with the ability to detect chemotherapy-related changes before declines in ejection fraction occur. 33 Techniques such as late gadolinium enhancement (LGE), T1/T2 mapping, and extracellular volume (ECV) assessment improve diagnostic sensitivity,34,35 allowing early identification of subclinical injury and real-time monitoring of cardioprotective interventions, particularly in patients receiving anthracyclines or HER2-targeted therapies.
T1-weighted imaging post-gadolinium can detect acute myocardial inflammation. In anthracycline-treated patients, increased myocardial signal intensity by day 3 predicted LVEF decline within a month. 36 LGE is reliable for identifying myocardial fibrosis in anthracycline cardiomyopathy, with some studies showing consistent subepicardial lateral wall enhancement in HER2-positive patients on combined therapy. Other studies, however, found minimal enhancement, reflecting clinical variability. These findings support CMR’s role in early detection of cardiotoxicity. 33
T1 mapping further enables quantification of ECV, often elevated after anthracycline treatment 33 and correlated with reduced LVEF and lower peak oxygen uptake (VO2 peak), suggesting early myocardial remodeling. Longitudinal studies also link decreases in LV mass, likely due to myocyte loss, to increased cardiovascular mortality and HF risk. Pulse wave velocity assessments have revealed post-treatment aortic stiffness, indicating vascular injury and heightened future cardiovascular risk. 37
Despite its strengths, CMR use in routine care is limited by costs, availability, and expertise requirements. Supporting data come mainly from small studies, stressing the need for larger, standardized trials. Still, current evidence supports integrating CMR into cardio-oncology protocols as a key tool for diagnosing, monitoring, and managing cardiotoxicity from cancer treatment.
Stress-based functional testing in cardio-oncology
Cancer therapies have the potential to diminish cardiovascular reserve, either through direct myocardial toxicity or indirectly via treatment-related deconditioning and alterations in lifestyle. 38 Functional stress testing—whether conducted through physical exercise or pharmacologic stimulation—has been extensively employed in cardiology to identify subclinical myocardial dysfunction and evaluate contractile reserve, which may serve as a prognostic indicator independent of LVEF. 39 Nevertheless, its utilization within the context of cardio-oncology has been comparatively constrained.
In a study of 57 asymptomatic breast cancer patients with preserved LVEF (≥50%), exercise echocardiography showed reduced stroke volume and cardiac index versus controls, suggesting impaired LV contractile reserve (LVCR). 38 McKillop et al. found that measuring LVEF during exertion raised doxorubicin cardiotoxicity detection from 58% to 100%. 39 Civelli et al. used low-dose dobutamine stress to evaluate LVCR during and after high-dose chemotherapy, finding that a ≥5% decline predicted subsequent LVEF drops below 50%. 40
Although certain studies utilizing exercise and pharmacological stress have exhibited potential in identifying cardiotoxicity among long-term survivors of pediatric malignancies treated with anthracyclines, the findings remain variable.41,42 These discrepancies underscore the necessity for additional validation of stress testing methodologies in oncology patient populations.
Cardiotoxicity may affect multiple systems. VO2 peak, reflecting integrated cardiovascular and pulmonary function, offers broader insights into systemic capacity. Low VO2 peak correlates with higher cardiovascular and all-cause mortality in patients with lung and breast cancer.43–45 Jones et al. found that breast cancer survivors had VO2 peak values 22% lower than sedentary, age-matched women without cancer, despite preserved resting LVEF—highlighting the value of functional testing beyond standard imaging. 45
Cardiac biomarkers for early detection of chemotherapy-induced cardiotoxicity
Cardiac troponins, particularly troponin I (TnI), have emerged as important blood-based biomarkers for the early detection of cardiotoxicity in patients undergoing cancer therapy. Several small studies have shown that transient elevations in TnI can predict both the onset and severity of LVEF decline, especially in patients receiving high-dose anthracyclines for hematologic or solid tumors.46,47 In women treated with anthracycline-trastuzumab regimens, TnI levels above 0.08 ng/mL were associated with a 23-fold increase in cardiotoxicity risk and a significantly higher likelihood of irreversible LVEF reduction, even after treatment cessation. 47
High-sensitivity (HS) and ultra-sensitive troponin assays offer improved prognostic value, particularly when combined with imaging modalities such as STE. However, predictive performance remains modest. For example, Sawaya et al. reported only 48% sensitivity and 73% specificity for ultra-sensitive TnI in identifying cardiotoxicity among early-stage breast cancer patients receiving anthracycline-trastuzumab therapy. 28 Moreover, the clinical utility of troponins across other chemotherapy protocols remains uncertain.
Natriuretic peptides—including BNP, NT-proBNP, and NT-proANP—have also been investigated but generally show lower reliability than troponins for detecting early cardiac dysfunction in oncology settings. 48 Despite their promise, both troponins and natriuretic peptides face limitations due to small, heterogeneous study cohorts, variations in chemotherapy regimens, and a lack of standardized protocols for assay timing, measurement techniques, and diagnostic thresholds. These issues currently hinder their broader integration into routine cardio-oncology practice. 49
Troponins as early indicators of cardiotoxicity in targeted cancer therapy
Emerging evidence supports TnI and troponin T (TnT) as early biomarkers of cardiotoxicity in targeted therapies. In a study of 251 breast cancer patients on trastuzumab, 14% had elevated TnI—62% of whom developed LV dysfunction (LVD) versus 5% without TnI elevation (p < 0.001). 50 Those with elevated TnI were less likely to recover cardiac function and had more cardiac events. This underscores TnI’s predictive value during trastuzumab treatment, especially following prior anthracycline exposure.
Remarkably, no elevations in troponin levels were detected in patients given trastuzumab without prior anthracycline exposure, suggesting the biomarker increase reflects anthracycline-induced damage, worsened by trastuzumab. 48 Similarly, Morris et al. noted TnI rises in patients receiving sequential anthracyclines, trastuzumab, and lapatinib, preceding LVEF decline. 49 Schmidinger et al. found 10% of metastatic renal cancer patients treated with sunitinib or sorafenib had elevated TnT, with many later showing LVEF impairment or regional wall motion abnormalities. 50
Troponins are valuable biomarkers for the early detection of cardiotoxicity in patients receiving both conventional and targeted cancer therapies, but their clinical utility is limited by inconsistent correlations with outcomes. These discrepancies stem from variations in treatment protocols, assay sensitivity, sample timing, and follow-up duration. 51 Older assays often fail to detect low-level troponin elevations post-chemotherapy, whereas HS assays may improve early identification of subclinical myocardial injury. 28
Overall, elevated troponin should not be interpreted as a reason to withhold cancer therapy. Instead, it serves as a valuable risk stratification tool, identifying patients who may benefit from closer cardiac monitoring and potential initiation of cardioprotective strategies. Incorporating troponin measurements into preclinical drug evaluations and cardio-oncology surveillance protocols may help to detect early subclinical cardiac injury and improve patient outcomes.28,50,51
HS troponins in cardio-oncology surveillance
Recent advancements in assay technologies have led to the development of HS troponin tests, which offer improved precision in detecting very low levels of cardiac troponin. This enhanced sensitivity is especially valuable in the oncology setting, where cardiac damage is often subclinical and associated with only slight elevations in biomarkers. 52
Sawaya et al. were among the first to investigate HS-TnI in patients undergoing chemotherapy with anthracyclines and taxanes, followed by trastuzumab. Their multicenter study revealed that absolute increases in HS-TnI measured at the end of anthracycline treatment were associated with a higher risk of developing LVD. 53 Additional markers, such as MPO, were also evaluated, although MPO demonstrated weaker statistical significance. In contrast, NT-proBNP and Galectin-3 did not correlate with later cardiotoxicity in that cohort.
Integration of HS-TnI with echocardiographic measures of myocardial deformation, such as longitudinal strain, has shown that early changes in these parameters, identified during anthracycline therapy, are more predictive of subsequent LVD than conventional markers like LVEF or diastolic function. 52 These findings highlight the value of combining biomarker surveillance with advanced imaging to detect myocardial injury before clinical symptoms emerge. In a study by Ky et al., 52 a panel of biomarkers, including HS troponins, was evaluated in breast cancer patients, reinforcing the utility of HS assays alongside imaging for identifying individuals at elevated risk. While results have been promising, larger, multicenter studies are needed to confirm the predictive accuracy of HS-TnI, particularly across diverse populations and with extended follow-up.
Immunotherapy and cardiotoxicity
The introduction of immune checkpoint inhibitors (ICIs) has transformed oncology, significantly improving outcomes across a broad range of malignancies. These agents enhance antitumor immunity by inhibiting regulatory pathways such as cytotoxic T-lymphocyte antigen 4 (CTLA-4), programmed death-1 (PD-1), and PD-ligand 1 (PD-L1). 53 The U.S. FDA has approved seven ICIs—including ipilimumab, three anti-PD-1 agents (nivolumab, pembrolizumab, cemiplimab), and three anti-PD-L1 agents (atezolizumab, avelumab, durvalumab)—for use in at least 12 cancer types. 54
Despite their efficacy, ICIs can provoke immune-related adverse events (irAEs) due to off-target immune activation. These typically affect the skin, liver, gastrointestinal tract, and endocrine glands, but may also involve the central nervous, pulmonary, cardiovascular, and hematologic systems. Among cardiovascular irAEs, myocarditis is particularly concerning due to its potential for severe morbidity and mortality. 55 Other cardiac toxicities include arrhythmias, cardiomyopathy, vasculitis, and acute coronary syndromes.
Mechanistically, ICI-related cardiotoxicity may result from immune cross-reactivity between neoplastic and cardiac antigens. Preclinical studies suggest that PD-1, PD-L1, and CTLA-4 pathways confer cardioprotection under stress, 56 and their inhibition may predispose to immune-mediated cardiac injury, including myocarditis.
ICI-induced myocarditis is a rare but potentially life-threatening complication that can closely resemble other cardiac conditions, such as acute coronary syndrome or viral myocarditis, making diagnosis particularly challenging. A high index of suspicion is essential when ICI-treated patients present with cardiac symptoms.53,56 Accurate diagnosis typically requires a multimodal approach, incorporating biomarkers, imaging, and occasionally angiography. The presence of concomitant conditions like myositis may support the diagnosis due to shared antigenic targets in cardiac and skeletal muscle. 55 Although infrequent, ICI-related cardiotoxicity significantly impacts clinical management, often necessitating immediate discontinuation of immunotherapy and initiation of high-dose corticosteroids to improve outcomes. 54
Discussion
Cardio-oncology has undergone a significant evolution with the integration of advanced cardiac biomarkers into routine oncologic care. While therapies such as anthracyclines, HER2-targeted agents, and ICIs have extended survival, they carry substantial cardiovascular risks. 1 There is growing consensus that early identification of cardiotoxicity—through a multimodal approach combining biomarkers and advanced imaging—is critical to preventing irreversible cardiac damage.1,57
HS cardiac troponins are now central to detecting early myocardial injury, often rising before overt LVD, particularly in patients receiving high-risk regimens like anthracyclines and trastuzumab. 57 However, their prognostic value depends on assay type, timing, and treatment context. Their utility is enhanced when combined with imaging modalities such as GLS, which is more sensitive than ejection fraction alone for identifying subclinical dysfunction. 58
With the rise of targeted and immune-based therapies, research into tailored cardioprotective strategies is accelerating. Early studies are evaluating IL-1 blockers, SGLT2 inhibitors, and prophylactic beta-blockers or ACE inhibitors to mitigate therapy-induced cardiac effects. Trials like SAFE-HEaRt and PROACT are investigating whether cardioprotective agents can be co-administered with HER2-targeted treatments without affecting cancer outcomes. These efforts reflect a shift toward proactive cardiotoxicity prevention in contemporary oncology.18,59,60
Natriuretic peptides and emerging biomarkers—such as Galectin-3, sST2, and GDF-15—offer further insights into myocardial stress, fibrosis, and inflammation. Some, including Galectin-3 and ST2, are implicated in both tumor progression and cardiac remodeling, highlighting a pathophysiologic overlap between cancer and HF.28,58
The introduction of ICIs has expanded the cardio-oncology landscape, with immune-mediated myocarditis emerging as a rare but potentially fatal complication. Despite its low incidence, its severity warrants high clinical vigilance.53–55 Timely diagnosis requires a comprehensive strategy involving biomarkers, cardiac MRI, and, when needed, histopathologic evaluation. Management typically involves immediate discontinuation of immunotherapy and initiation of high-dose corticosteroids, underscoring the need for clear protocols and multidisciplinary coordination. 56
Our investigation further emphasizes the pivotal importance of both preventive and protective measures. Pharmacological agents such as dexrazoxane have exhibited significant effectiveness in mitigating cardiotoxicity associated with anthracycline treatment, thereby necessitating their incorporation into therapeutic regimens for populations at elevated risk.58,61 Additionally, functional assessments (e.g. cardiopulmonary exercise testing) provide critical insights into cardiovascular reserve, particularly in instances where conventional imaging techniques yield ambiguous results. 61
Nonetheless, numerous deficiencies persist. Inconsistencies in monitoring methodologies, the absence of standardized biomarker thresholds, and insufficient data regarding specific cancer subpopulations hinder the establishment of cohesive clinical guidelines. Subsequent research endeavors should prioritize extensive, longitudinal investigations that substantiate multimodal surveillance methodologies, with a keen emphasis on tailoring care according to cumulative cardiovascular risk and the intensity of oncological treatment.
In conclusion, the amalgamation of biomarker information, functional imaging modalities, and individualized risk assessment constitutes a groundbreaking advancement in the field of cardio-oncology. This tailored strategy not only facilitates early identification but also improves risk stratification and promotes therapeutic interventions that safeguard cardiac integrity while maintaining oncological effectiveness.
Footnotes
Ethical considerations
Our institution does not require ethical approval for reporting individual cases.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
