Abstract
Inflammatory bowel disease (IBD), including Crohn’s disease and ulcerative colitis, is a chronic systemic immune dysregulated disorder affecting the gastrointestinal tract that often has extraintestinal manifestations. Limited data exist on the cardiovascular (CV) implications of IBD, but a higher prevalence of CV disease (CVD) has been observed compared to the general population, resulting in increased mortality risk. This review examines the pathophysiology and risk factors linking IBD to CVD, highlighting several key mechanisms: The “lipid paradox” in active IBD, arterial stiffness and endothelial dysfunction, proinflammatory cytokine activity, gut microbiota dysbiosis, and drug-induced myocardiopathy. The main findings include information about IBD patients exhibiting a higher incidence of CVD compared to the general population, independent of traditional risk factors. Chronic inflammation, altered lipid profiles, and gut microbiome imbalances play a significant role in heightened CV risk. Routine CV risk assessment should be considered during clinical assessment, to integrate into IBD management. Anti-inflammatory therapies may reduce CVD risk, while careful consideration is needed for drugs with potential CV side effects. Further research is required to understand the complex interplay between IBD and CVD, particularly regarding the impact of novel therapies and lifestyle interventions on long-term CV outcomes in IBD patients.
Keywords
Introduction
Cardiovascular disease (CVD) is the primary cause of mortality and morbidity in the world. 1 CVD is also the most significant cause of mortality and disability-adjusted life years. Inflammatory bowel disease (IBD) consists of Crohn’s disease (CD) and ulcerative colitis (UC). The fundamental causes of these autoimmune diseases include the interplay between genetic and environmental factors, excluding pathogenic infections.2–4 There have been findings of an increase in the incidence of IBD in children and adults worldwide5,6 with a global age-standardized prevalence rate of 59.25/100,000 people as reported in 2019 by the Global Burden of Disease Study. 7 Multiple studies demonstrated that the overall risk of serious cardiovascular (CV) events is related to IBD activity, with the highest incidence during flares and periods of persistent disease activity. 8 A recent population-based national cohort study involving more than 29 million patients showed that the prevalence of acute myocardial infarction (MI) was higher in both patients with CD and UC.9–12
One of the mechanisms suggested to account for the increased CV risk in patients with IBD is the underlying proinflammatory state acting as a strong stimulus for endothelial dysfunction and thrombogenesis.13,14 This has been demonstrated in studies that describe changes in CVD mortality in patients with chronic inflammatory diseases such as rheumatoid arthritis (RA) and IBD.7,14–17 It has been suggested that many inflammatory cytokines and interleukins (ILs) that are associated with worsening CVD and atherosclerosis are also involved prominently in IBD dysregulation, including tumor necrosis factor alpha (TNF-α).18,19 Treatment with anti-TNF-α agents has been shown to protect patients with IBD against the development of atherosclerosis and CVD, providing further evidence for the role of inflammatory processes in these conditions. 11
Additional findings in IBD patients show a link between dysbiosis (alteration of the microbiome) and increased plaque formation in blood vessels, along with loss of cell wall integrity, and ultimately increased risk of cardiac lesion development. This leads to a heightened, overall inflammatory burden via increased bacterial translocation. Subsequent metabolites released from the gut through microbial metabolism of choline, phosphatidylcholine, and L-carnitine directly contribute to CVD pathology. 20
Advanced IBD treatments, including small molecules and biologics, also impact CV risk. Recently, a novel class of drugs, the Janus kinase inhibitors, including Tofacitinib and Upadacitinib, have been labeled with black box warnings for major adverse cardiovascular events (MACE) and higher rates for venous thromboembolism (VTE) and cerebrovascular accidents. 21 Anti-TNF agents such as Infliximab and Adalimumab have cardioprotective effects by blocking the proinflammatory cytokine TNF-α, halting the progression of atherosclerosis, insulin resistance, and other inflammatory states; however, they are contraindicated in advanced decompensated heart failure.22–24 A newer therapeutic option recently approved for UC involves sphingosine-1-phosphate (S1P) receptor modulation with agents, such as Ozanimod and Etrasimod, which have CV implications for the heart rate, blood pressure, and cardiac autonomic regulation, also alternatively demonstrating cardioprotective mechanisms.25,26
There are significant gaps in the understanding of the complex relationship between IBD and CVD. The mechanisms underlying this association remain poorly understood, and the impact of IBD-specific medications on CV health is not fully clear. Furthermore, the relationship between traditional CV risk factors and IBD-specific inflammatory processes contributing to CVD risk has not been comprehensively explored.
This review aims to address these by (a) analyzing the pathophysiological mechanisms linking IBD and CVD, including the roles of chronic inflammation, endothelial dysfunction, and gut microbiota dysbiosis. (b) Examining the CV implications of current IBD treatments, from conventional therapies to advanced biologics and small molecules. (c) Exploring the potential for targeted interventions to mitigate CVD risk in IBD patients.
Understanding these aspects can lead to improved risk stratification and early intervention strategies for CVD in IBD patients. Second, it may inform the development of novel therapeutic approaches that address both intestinal and CV manifestations of IBD. It could also contribute to the optimization of current treatment regimens to balance IBD management with CV health.
By synthesizing the latest research on the IBD-CVD relationship, this review seeks to provide clinicians and researchers with a comprehensive understanding of this important area, ultimately aiming to improve the care of IBD patients.
Search strategy, inclusion/exclusion criteria, and data extraction methods
For this review manuscript, a formal systematic search strategy was not conducted. However, we conducted a targeted literature search using PubMed, focusing on articles categorized as clinical trials, meta-analyses, randomized controlled trials (RCTs), reviews, and systematic reviews. Our primary objective was to identify studies exploring the relationship between IBD and CVD. We emphasized our search on recent literature, prioritizing more recent publications to ensure the manuscript’s relevance. However, select earlier studies from the early 2000s were included based on their substantial contributions to the understanding of pathophysiological mechanisms and historical context. Articles were included based on their relevance to pathophysiology, risk factors, and therapeutic implications of CVD in the context of IBD. Data extraction was performed individually by the authors, with consensus-based selection of key findings deemed most pertinent to our review objectives.
Mechanisms of IBD and CVD interplay
IBD, CD, and UC cause immune disruption of the digestive system. Like many other autoimmune diseases, extraintestinal manifestations (EIM) may arise before or after gastrointestinal symptoms and diagnosis. In general, the incidence of EIMs ranges from 6% to 47% in IBD patients, 25% of which are present before the diagnosis is made and 75% starting afterward. There is evidence of a higher incidence of CVD in IBD patients compared to the general population. 27 This is consistent with other autoimmune conditions such as type 1 diabetes mellitus, RA, and psoriatic arthritis, among others. 28 These findings have been demonstrated to be independent of traditional CV risk factors include obesity, hypertension (HTN), and dyslipidemia.28,29
Several different mechanisms have been proposed to elicit the underlying pathophysiology including the lipid paradox, 30 arterial stiffness (AS), endothelial dysfunction,28,31–33 proinflammatory cytokines,27,28,34,35 gut microbiota dysbiosis,27,28,36 and drug-induced cardiomyopathy.27,34
The lipid paradox
The “lipid paradox” is described in the literature as a decrease in total cholesterol (high-density lipoprotein cholesterol (HDL-c) and low-density lipoprotein cholesterol (LDL-c)) in patients with active IBD vs patients in remission. 30 Lipid properties tend to be more pro-atherogenic, counteracting lower lipid levels. Some studies demonstrate a correlation demonstrating a lipid increase after initiation of Janus kinase inhibitors (Tofacitinib and Upadacitinib) and corticosteroids (CS). 30
Arterial stiffness and endothelial dysfunction
AS and endothelial dysfunction are a consequence of chronic inflammation. Levels are measured with pulse wave velocity (PWV).28,31 Aortic PWV (aPWV) has been shown to be a vascular biomarker and perhaps an early sign of a CV risk factor. ( 32 ) There is evidence showing that aPWV is increased in patients with active IBD. Once again, there has not been a positive correlation to an elevation of serum inflammatory biomarkers like C-reactive protein (CRP) and erythrocyte sedimentation rate. 32
Proinflammatory cytokines
The proinflammatory cytokine mechanism is based on early findings of atherosclerosis in IBD patients, with T helper (Th) 17 and Th1 as the key stimulus that increases the production of IL-1b, IL-6, IL-23, TNF, and interferon-gamma, CRP, and homocysteine levels. 35 Furthermore, IBD patients during a flare are up to three times more likely to develop a VTE compared to the general population. 22 This is suggestive of an increase in cytokine activation within the coagulation cascade, creating a pro-coagulant state.21,27 The combination of a chronic inflammatory state along with endothelial dysfunction creates the milieu for thrombosis. 22 It has also been revealed in vitro, with data demonstrating superoxide anion and reactive oxygen species stimulating nuclear factor kappa B (NF-kB), consequently activating inflammatory biomarkers (such as TNF-α and IL-1) that activate vascular cell adhesion protein-1, intracellular adhesion molecule 1, monocyte reactant protein 1, E-selectin, and cause macrophage infiltration, leading to, direct endothelial dysfunction via apoptosis, and inducing thrombosis (Figure 1). 28
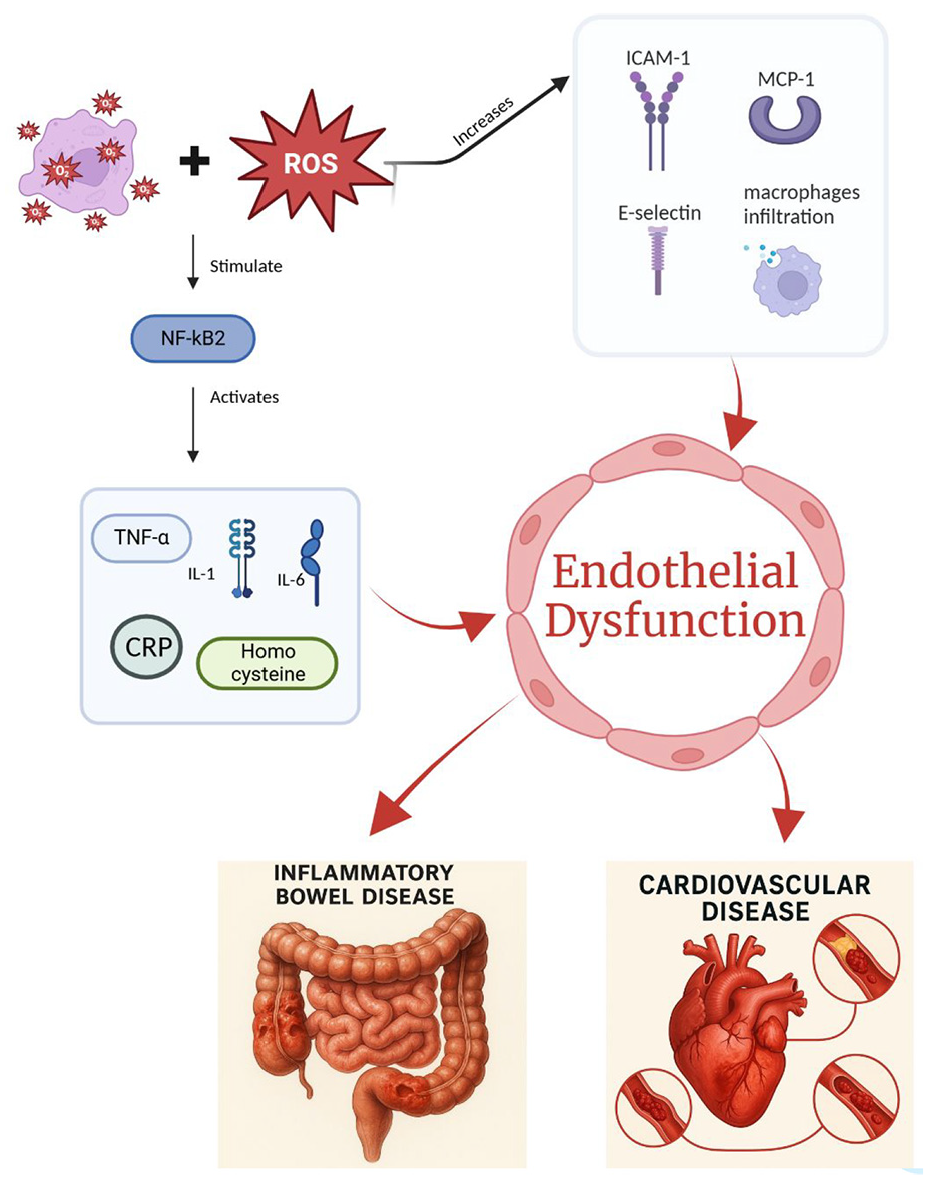
Demonstrates the two ROS pathways, one being the stimulation NF-kB, activating inflammatory biomarkers. The other pathway activates VCAM-1, ICAM-1, MCP-1, E-selectin, and macrophage infiltration. Both create a direct endothelial dysfunction via apoptosis and inducing thrombosis.
Gut microbiota dysbiosis
Intestinal flora and its role in inflammation
Intestinal flora is directly affected by inflammation within the gut. 37 Recent studies outline the relation between Firmicutes bacteria, particularly lower concentrations of Faecalibacterium prausnitzii,27,28 higher concentrations in Enterobacteriaceae and Streptococcus species (Streptococcus anginosus and Streptococcus dispar) impairing blood pressure regulatory mechanisms in another autoimmune disease, Systemic Lupus Erythematosus (SLE), contributing to increased CVD risk.28,38 One of the mechanisms described is from bacterial metabolites such as indole and phenyl derivatives of hippuric acid increasing accumulation of atherosclerosis and platelet activation, leading to a decrease in tri-methylamine N-oxide and toll-like receptor (TLR) 2 and 4 expression.27,28,39
Involvement of lipopolysaccharide and consequential TLR4 expression
IBD is also noted to disrupt the intestinal intracellular barrier, leading to increased endotoxin absorption. Lipopolysaccharide (LPS) is widely known to cause chronic systemic inflammation and vascular endothelitis. 40 Some studies demonstrate that LPS serotypes from different gram-negative species activate TLR4, causing downstream activation of mediators that culminate in the production of proinflammatory cytokines, such as TNF-α, NF-kB, IL-8, and IL-10, thereby inducing inflammation and intestinal epithelial permeability, enhancing the pathophysiology of IBD.28,41
LPS recognition on the cell surface involves multiple proteins. Initially, LPS-binding protein (LBP) recognizes the lipid A on the LPS, attaching to it and forming a complex that docks to the LPS receptor. Subsequently, LBP interacts with CD14, which transfers the LPS-LBP complex to TLR4, facilitated by the signaling molecule, MD-2. MD-2 is crucial for LPS binding to TLR4. Upon LPS binding to the TLR-CD14-MD-2 complex, TLR-4 activates, initiating downstream signaling pathways. Two primary pathways are triggered: the MyD88-dependent pathway, which is rapid and utilized by most TLRs, and the MyD88-independent pathway, which occurs later and involves endosomal trafficking and the recruitment of TRIF-related adaptor molecule (TRAM) and TIR domain-containing adaptor inducing interferon-β (TRIF). These pathways are pivotal in the immune responses triggered by LPS.42,43 Calprotectin, a stool biomarker, which has an excellent correlation to disease activity in IBD has also been described to increase TLR-4 expression and advanced glycation end products, the latter leading to cardiomyocyte dysfunction.27,28,42
N-oxide trimethylamine
In addition, N-oxide trimethylamine (TMAO), a metabolite found in meat and cheese (rich in choline and L-carnitine), has been associated with an increased risk of atherosclerotic CVD, thus proposing a link between diet-related inflammatory changes and the increased risk and progression of IBD (particularly in patients with UC) and CVD. The inflammatory process involves the metabolization of choline and L-carnitine into trimethylamine, which then gets converted into TMAO in the liver.28,36,44–46
Therapeutic alternatives
RCTs involving dietary interventions and alterations in gut microbiota, may provide alternative preventive measures to stop the atherogenic process in IBD patients. Other therapeutic alternatives, such as fecal microbiota transplantation, have been described since 1989 for patients with UC, as it has demonstrated a decrease in proinflammatory cytokines including IL-1β, IL-6, and TNF-α. 47
Drug-induced cardiomyopathy
Studies have shown an association between exposure to CS and an increase in risk of CVD in patients with IBD. CS also carries a side effect profile which includes increased risk of HTN, insulin resistance with elevated blood glucose, dyslipidemia, and obesity. 34
Shared risk factors between IBD and CVD
Chronic systemic inflammation is associated with early arteriosclerosis, 48 although other factors, such as obesity or metabolic syndrome, also contribute significantly to this risk in patients with IBD. 49
There are more IBD patients with both arterial and venous thrombotic events. In patients with IBD, the main CV events include VTE, deep vein thrombosis, and pulmonary embolism, occurring in a predominantly younger population, mostly women, during periods of disease exacerbation. This pattern contrasts with the typical CVD presentation observed in the elderly population. Therefore, it is vitally important to control inflammatory activity, which could reduce the likelihood of developing these complications.2,50,51
Some studies describe the risk of CVD to be higher in patients with CD compared with patients with UC.50,52
There is evidence that several pathophysiological mechanisms, including structural and functional vascular changes, as well as biochemical and genetic changes, may be involved in the risk of acute arterial events in patients with IBD. 53 Besides an increased carotid intima-media thickness and AS, microvascular endothelial dysfunction is observed in patients with IBD.16,54
Atrial fibrillation in inflammatory bowel disease: Pathophysiological links and clinical implications
Atrial fibrillation (AF) has recently been described as a CV comorbidity in patients with IBD. A meta-analysis of three cohort studies (2014–2019) showed that patients with UC had a higher risk of AF (OR 1.51, 95% CI 1.40–1.63). 7 AF is characterized by rapid, disorganized atrial electrical activity leading to compromised cardiac function, increased thromboembolic risk, and significant morbidity. In the past, it has been associated with HTN and structural heart disease, however, recently it is increasingly being linked to systemic inflammation, which is also a hallmark feature of IBD. This shared inflammatory foundation provides a correlation between the two conditions.55,56
Pathogenesis
The pathogenesis of AF in the context of IBD is multifactorial. Chronic inflammation contributes to atrial structural and electrical remodeling, fibrosis, and electrical instability, creating an ideal environment conducive to arrhythmogenesis. Elevated levels of proinflammatory cytokines, such as TNF-α, IL-6, and CRP, as observed in IBD flares, have been implicated in both atrial fibrosis and oxidative stress, which are well-established drivers of AF initiation and persistence.55,56
Outcomes in patients with AF and IBD
Recent systematic reviews and meta-analyses analyzed the clinical relevance of the higher prevalence of AF in IBD populations, particularly during flares, suggesting that active disease states may serve as a trigger for atrial arrhythmias. 2 These studies describe that IBD patients with concomitant AF had significantly worse clinical outcomes, including higher rates of hospitalization and increased mortality compared to IBD patients without AF. These findings highlight the importance of routine CV screening, especially among IBD patients during active disease phases.55,56
Risk factors
In addition to the inflammatory pathways, shared risk factors such as endothelial dysfunction, autonomic imbalance, and CS use may further enhance AF risk in IBD patients. Some studies provide additional context by outlining how inflammation-driven pathways not only predispose patients to AF but also accelerate coronary artery disease progression, suggesting the need of combined therapeutic strategies targeting systemic inflammation in chronic inflammatory diseases. 57
The importance of physical activity
Epidemiology
Sedentarism is a risk factor for CVD development, it also contributes to the development of several other chronic diseases. Around 30% of the adult population does not meet the recommended minimum of 150–300 min (about 5 h) weekly of moderate-intensity aerobic physical activity. 58 The CVD risk is increased by 1.5 times in people living a sedentary lifestyle. According to the American Heart Association, sedentary behavior has been identified as one of the leading preventable causes of death.59,60
Metabolic syndrome’s association with IBD
Obesity effects include increased insulin resistance, systemic inflammation, and a prothrombotic effect. There are further links to dyslipidemia, albuminuria, endothelial dysfunction, coronary artery disease, congestive heart failure, AF, stroke, and increased sympathetic activity. 61
Besides a high prevalence of obesity in patients with IBD, premorbid obesity has also been associated with a risk of developing CD, but not UC, though this relationship has not been consistently observed. 62 However, accumulation of intra-abdominal fat may contribute to mucosal inflammation, thereby affecting the clinical course in patients with established IBD.63,64
Dyslipidemia effects on IBD
The prevalence of overweight and obesity in patients with IBD is similar to or higher than that of the general population, as has been observed in some studies.59,65 This can generate a deleterious effect in the course of the disease since adipose tissue is a hormonally active organ and is capable of producing proinflammatory cytokines. 64 Other risk factors for CVD, such as diabetes mellitus, may also occur more frequently in patients with IBD. Dyslipidemia is one of the main CVD risk factors and chronic inflammation can generate important changes in the lipid profile. The detection of altered lipid levels in patients with IBD may also be due to the possible state of malnutrition, malabsorption or a history of intestinal resection. There is evidence that patients with IBD more frequently have decreased levels of HDL-c, and elevated levels of LDL, despite increased CVD risk. 50
Smoking
Smoking cigarettes is a dominant risk factor for CV and non-CV mortality and morbidity. It is estimated that by the year 2025, there will be 1.6 billion smokers in the world, and that 10 million people a year will die from smoking. 66 The 10-year risk of fatal CV events is doubled in smokers. Passive smoking also increases the risk of CVD. 67
Patients with CD who smoke have more frequent relapses and a greater requirement for surgery. This may be explained in part by potentiation of an occlusive microvascular injury. Cigarette smoking has been shown to induce morphological injury to endothelial cells: this injury is associated with the formation of microthrombi, impaired endothelial cell prostacyclin synthesis, and an increased aggregatory capacity for platelets. In addition, it has been reported that habitual smokers present decreased plasminogen and tissue plasminogen activators, along with increased plasma fibrinogen concentrations. These findings may explain, at least in part, the increased risk of thrombotic vascular disease in smokers, and possibly the tendency for smoking to aggravate CD. 68
Impact of IBD treatment on CVD risk
In recent years, there has been an increase in IBD therapeutic options. Presently, the therapeutic options for IBD management include 5-aminosalicylates (5-ASA), CS, immunomodulators, and advanced therapies, including small molecules and biologics. 69
Corticosteroids
Systemic CS effectively reduce inflammation and induce remission during IBD flares. However, prolonged use requires close monitoring due to risks such as edema, heart failure exacerbation, electrolyte imbalance, and increased blood glucose and pressure.70,71 In addition, metabolic effects of long-term steroid use, such as HTN, obesity, hyperlipidemia, and insulin resistance, have been associated with increased CV risk in the general population and chronic inflammatory disease.71–74 There have been reports that patients with UC have a higher incidence of ischemic heart disease (IHD) and MI with steroid use. 75
Overall, CS use might be beneficial in the short term, but it is not recommended for use in maintenance due to long-term side effects, including an increase in CVD and mortality.76,77
5-Aminosalicylates
Suggestive evidence showed that patients using 5-ASA 1 year after IBD diagnosis had less risk of IHD (incidence rate ratio (IRR) 1.16) than the patients with IBD not using 5-ASA (IRR 1.36). Even a lower risk (IRR 1.08) was shown in long-term users of 5-ASA. 8
On the other hand, the use of 5-ASA has been associated with unwanted side effects such as myocarditis and pericarditis, as well as bradycardia.78,79,85 A multicenter longitudinal study showed that salicylate use is associated with increased AS, as measured by carotid-femoral PWV. 80
Immunomodulators
Major adverse cardiovascular events
Debate surrounds whether immunotherapy increases or reduces the risk of MACE. A systematic review, encompassing 64 RCTs, on the impact of biologics and small molecules in patients with IBD, shows no increase in MACE association in patients with CD, for either induction or maintenance phases compared to placebo with the use of Infliximab (OR 0.63, 95% CI 0.07–6.14) and Ustekinumab (OR 0.50, 95% CI 0.03–8.04). Similarly, in patients with UC, there was no associated heightened risk of MACE with tofacitinib (OR 1.30, 95% CI 0.15–11.21) and Upadacitinib (OR 0.50, 95% CI 0.03–7.97). 81 However, no long-term data were evaluated in this meta-analysis. In contrast, a recent meta-analysis showed an increase in the risk of MACE and stroke with disease-modifying anti-rheumatic drugs such as methotrexate compared with anti-TNF therapy. 87 In addition, reports on S1P receptor modulators, such as ozanimod, suggest a dose-dependent increased risk of MACE with the potential to cause conduction abnormalities such as AV block and bradycardia, which is representative in patients with a predisposition and/or underlying intermediate to high CV risk. 82
Long-term effects
Long-term follow-up studies for patients treated with immunomodulators and small molecules are imperative to identify the CV effects, particularly in patients with IBD and a high-risk CV profile. 83 This condition inherently worsens the outcomes and contributes to additional consideration when choosing the type of biologics for patients with IBD. 84
Thiopurines and TNF-α antagonists
TNF-α levels are increased in Heart Failure ( HF) and MI patients. Reduction in TNF-α levels in mouse models demonstrates improvement in HF, leading to the exploration of anti-TNF as a treatment modality for HF and MI. However, some studies demonstrate increased risk of death and worsening HF in patients with NYHA class 3–4 with high doses of Infliximab, leading to contraindication of anti-TNF therapy in this subset of patients.85–87
Evidence shows that IBD patients have higher carotid-femoral PWV than healthy individuals. Anti-TNF-α therapy is linked to a lower risk of acute coronary syndrome, arterial events, and IHD, with benefits in AS and endothelial function. However, thiopurine exposure has not been associated with reduced arterial event risk.32,88,89,90
Janus kinase inhibitors
Tofacitinib is a first-generation JAK inhibitor. In 2019, the Food and Drug Administration (FDA) issued a post-marketing warning about the increased risk of VTE, MACE, and death with Tofacitinib, changing its indication for use in UC only as a second-line agent with a recommendation for early dose reduction. Data is described in patients older than 50 years, with CV risk factors.91–93
Studies also show that patients undergoing treatment with Tofacitinib have a reversible increase in total cholesterol, HDL, and LDL as compared to placebo. 94 Upadacitinib and Filgotinib, on the other hand, are more selective JAK-1 inhibitors and have been described as a safer option. 93
Tofacitinib black box warning
In a cohort of 476 patients with UC treated with Tofacitinib 10 mg, 1.05% patients developed a type of CV event during the induction phase compared to placebo, including acute coronary syndrome, coronary artery stenosis, extrasystoles, palpitations, and sinus tachycardia, which led to the discontinuation of treatment in 3.8% of patients in the tofacitinib group vs 1.6% from the placebo group and a black box warning. 84
IL-12/23 inhibitors
IL-12 is implicated in the pathogenesis of atherosclerosis, and inhibition of IL-12 in murine models has been implicated in its prevention.95,96 Ustekinumab has been used for the treatment of plaque psoriasis and psoriatic arthritis and is now available for the treatment of CD and UC. Studies with Briakinumab, another IL-12/23 inhibitor reported increased MACE events, which led to the discontinuation of the RCT in 2011. 97
Risankizumab and Mirikizumab are antibodies against IL-23 that have demonstrated efficacy and safety as an induction and maintenance therapy for moderate-to-severe CD and UC, respectively.98–100 Clinical trials have reported no MACE and a statistically significant histological and endoscopic remission on UC.101,102
Anti-integrins
Vedolizumab is a humanized selective α4β7 integrin monoclonal antibody that blocks the interaction of α4β7 with mucosal addressin cell adhesion molecule-1 and inhibits the migration of T-lymphocytes across the gastrointestinal endothelium. 95 Post-marketing data from the FDA’s adverse event database suggest a higher incidence of cerebrovascular events, including stroke and cerebral hemorrhage, in vedolizumab users compared to anti-TNF therapy. However, these findings may be influenced by self-reporting biases and missing data on CV risk factors and disease activity. 103
S1P agonist modulators
S1P receptor modulators are a class of oral small molecules approved as a treatment for UC and currently in randomized controlled trials (RTCs) for CD. These therapies target the interaction between S1P and S1P1 receptors, which regulate lymphocyte movement from the spleen and lymph nodes into the bloodstream, thereby reducing intestinal inflammation in IBD. In May 2021, the FDA approved Ozanimod as the first S1P receptor modulator for treating moderate-to-severe UC. Other S1P receptor modulators that have shown positive results in IBD include Etrasimod. 104
CV effects
Evaluating CV safety of S1P receptor modulators is warranted due to S1P receptor expression on cardiomyocytes and vascular endothelial cells. Studies describe the CV safety of Ozanimod, in patients with moderate-to-severe UC from the phase 3 True North and open-label extension; this analysis is consistent with previous results in patients with multiple sclerosis, demonstrating that Ozanimod has a well-tolerated CV safety profile; MACE or thromboembolic events were infrequent and unrelated or unlikely related to ozanimod. The proportion of patients with moderate-to-severe UC experiencing any CV effects was low and rarely symptomatic. Most heart rate effects occurred within the first 6 h of dosing. Ozanimod’s selectivity for S1P1,5 receptors, avoiding S1P3 activity, may contribute to its CV safety. 105
In addition, some studies show that Etrasimod (2–4 mg daily) has no clinically significant effect on QT/QTc intervals or PR intervals in healthy adults. The heart rate reduction was most pronounced after the first dose, mild, asymptomatic, transient, and diminished with continued use (Table 1). 106
Highlights of the impact of cardiovascular risk on inflammatory bowel disease therapeutic options.

5-ASA, 5-aminosalicylates; CVD, cardiovascular disease; DMARDs, disease-modifying anti-rheumatic drugs; HLD, hyperlipidemia; HTN, hypertension; IBD, inflammatory bowel disease; JAK, Janus kinase; IL, interleukin; MACE, major adverse cardiovascular events; S1P, sphingosine-1-phosphate;TNF-α, tumor necrosis factor alpha; VTE, venous thromboembolism.
Discussion
IBD has been linked to risks in CV health, where some of the known mechanisms proposed arise from a background of chronic systemic inflammation. This perpetual insult to the vessels promotes endothelial dysfunction, ultimately resulting in thrombogenesis. In addition to this, inflammatory cytokines and ILs, which are present in the context of chronic inflammation, also contribute to the increased formation of the atherosclerotic plaque, disrupting the cell wall integrity and leading to an increased risk of cardiac lesion development, ultimately suggesting a link be the increased prevalence of CVD in patients who suffer from IBD.
Among the recognized CV EIM of IBD, there are conditions known for their association with high-risk mortality such as pericarditis, endocarditis, MI, arrhythmias, and conduction disorders as reported in a cohort of 127 patients with IBD and electrocardiogram-proven increased QRS complex duration and interval, 107 venous and arterial thromboembolisms, and increased risk of atherosclerosis. In addition, studies suggest that AF should be considered a clinically relevant CV manifestation in IBD, with implications for both diagnosis and management.
Other well-documented factors that have been demonstrated to contribute to the presence of CVD in the general population, such as sedentary lifestyle, smoking, obesity, and Diabetes Mellitus (DM) may also exacerbate the prevalence of CVD in patients with IBD.
Conflicting evidence
While substantial evidence supports an increased CV risk in patients with IBD, conflicting data exist regarding specific CV outcomes and the CV safety profile of certain IBD therapies. For instance, evidence surrounding the CV impact of JAK inhibitors like Tofacitinib remains inconclusive, with some studies reporting increased risks of MACE and thromboembolic episodes, while others have found no significant difference compared to placebo. These discrepancies might arise from differences in patient selection, duration of follow-up, dosing regimens, and varying definitions or reporting of CV endpoints across studies. Moreover, heterogeneity in study populations, particularly concerning baseline CV risk, age distribution, and concurrent medication use, may further complicate direct comparisons and explain inconsistent findings.
Similarly, the relationship between biologic therapies such as anti-TNF agents and CV outcomes is complex, characterized by evidence of both cardioprotective effects and heightened risk in specific patient subsets. For example, anti-TNF agents have demonstrated benefits in reducing systemic inflammation and AS, thus potentially mitigating atherosclerotic risks. However, they are contraindicated in advanced HF due to the potential exacerbation risk, highlighting the need for patient stratification when selecting therapeutic options. 82
Clinical implications
Despite the pathophysiologic mechanisms of IBD explaining the rationale that advocates for the anti-inflammatory and immunomodulatory mechanisms to counteract both its intra- and extraintestinal deleterious effects, the reported CVD adverse events are latent. Thus, early recognition of CV risk factors and strict IBD drug monitoring are preventive strategies aimed at reducing the overall mortality related to CV adverse events and improving the clinical outcomes of IBD. More data on drug-specific therapies for IBD and their effect on the CV system are needed to support the current literature on IBD dosing and drug combination in relation to their safety on the CV effects.
Clinicians should maintain a high index of suspicion for CVD in patients with IBD, especially during active disease phases, and consider incorporating routine cardiac rhythm monitoring and risk stratification tools as part of comprehensive IBD care. As well as screening for traditional risk factors such as HTN, dyslipidemia, diabetes, as well as inflammatory biomarker testing like CRP and more importantly, aPWV, which has been associated with active IBD.
It is important that high-risk patients, especially those in active phases or flares, frailty, or polypharmacy, should be monitored closely. An interdisciplinary (including cardiology consultation) and comprehensive approach that focuses on effective mitigation of inflammation and addresses CVD symptoms and modifiable risk factors is essential pillars that aid in progression and relapse control.
Future research should prioritize prospective, controlled studies designed explicitly to evaluate CV outcomes in IBD populations. This approach could help clarify existing discrepancies, refine therapeutic guidelines, and ultimately enhance patient safety and clinical outcomes.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
