Abstract
The most important complication of familial Mediterranean fever (FMF) is secondary amyloidosis. The aim of this study is to investigate the risk of developing FMF-related amyloidosis with macrophage migration inhibitory factor (MIF), interleukin 4 (IL-4), and IL-1 receptor antagonist (IL-1RA) variants. This study included 62 FMF patients with amyloidosis, 110 FMF patients without amyloidosis, and 120 controls. The clinical information of the patient groups was compared. MIF-173G/C, IL-4 variant number tandem repeat (VNTR), and IL-1RA VNTR variants were analyzed for all participants. The use of colchicine, pleurisy, and appendectomy was more common in FMF patients with amyloidosis than in FMF patients without amyloidosis. MIF-173G/C C/C genotype and C allele were higher in both patient groups compared to controls. IL-1RA VNTR A1/A2 and A1/A4 genotypes and A1–A4 alleles were more common in both patient groups than controls. The IL-4 VNTR P1 allele was more common in FMF patients with amyloidosis compared to controls. The MIF-173G/C allele and the IL-1RA VNTR A1–A4 allele are associated with FMF in the Turkish population but not with amyloidosis risk in FMF patients. The IL-4 VNTR P1 allele is more common in FMF patients with amyloidosis than in healthy individuals.
Secondary amyloidosis is the most feared complication of FMF, an inherited auto-inflammatory disease.
MIF, IL-4, and IL-1RA gene activities and changes in their expression are significant in modulating immune and inflammatory activity.
MIF-173G/C C/C genotype and C allele and IL-1RA VNTR A1/A2, A1/A4 genotypes, and A1–A4 alleles are associated with FMF in the Turkish population, but not with amyloidosis risk in FMF patients.
The IL-4 VNTR P1 allele is more common in FMF patients with amyloidosis than in healthy individuals.
MIF-173 G/C, IL-4 VNTR, and IL-1RA VNTR variants can be evaluated in the clinical practice of FMF patients.
The risk of FMF-related amyloidosis can be assessed in individuals with the IL-4 VNTR P1 allele.
The results of this study can serve as the basis for further studies.
Introduction
Familial Mediterranean Fever (FMF), the most common inherited autoinflammatory disease in the world, is caused by mutations in the Mediterranean Fever (MEFV) gene, which encodes pyrin, an immune-regulating protein. FMF is characterized mainly by recurrent short-lived episodes of peritonitis, arthritis, pleuritis, and rash, usually with an accompanying fever. 1 In addition, the most severe complication of FMF is secondary amyloidosis, which is the result of the accumulation of extracellular amyloid protein in various tissues due to chronic inflammation. 2 Secondary amyloidosis, the most feared complication in up to 8.6% of FMF patients, typically reveals a nephrotic syndrome that rapidly progresses to end-stage renal disease. It may also involve the gastrointestinal tract, cardiovascular system, and central nervous system. 3
Macrophage migration inhibitory factor (MIF) is a cytokine that plays a critical role in innate and adaptive immune responses. 4 MIF is effective in macrophage function, regulation of the Th1/Th2 balance, lymphocyte immunity, and endocrine function. The human MIF gene is located on chromosome 22q11.2. 5 MIF, which can be released from macrophages and T lymphocytes stimulated by glucocorticoids, plays a role in the pathogenesis and progression of autoimmune diseases. A polymorphism in the MIF promoter region (rs755622) caused by the substitution of the G nucleotide by the C nucleotide affects promoter activity and expression. 4
Interleukin 4 (IL-4) is an immune-modulatory cytokine that plays a role in adaptive and humoral immune responses and is mapped to the q arm of the fifth chromosome. 6 Mainly, IL-4, produced by macrophages and T lymphocytes, controls the maturation of T helper-2 cells. 7 In addition, IL-4 induces inflammation in autoimmune diseases as an important regulator of immunoglobulin G. The third intron of the IL-4 gene has a variable number of tandem repeat (VNTR) (rs79071878) variants, and these variants have been associated with autoimmune diseases. 8
Interleukin-1 receptor antagonist (IL-1RA), which competes with IL-1α and IL-1β and thereby inhibits the activities of these cytokines, is an important anti-inflammatory molecule that modulates IL-1-related immune and inflammatory activity. 9 IL-1RA, which regulates various immune responses, has a polymorphic region containing 86 base pairs (bp) of VNTR (rs2234663). 10
Secondary amyloidosis is the most feared complication of FMF, the most common inherited autoinflammatory disease. On the other hand, changes detected in MIF, IL-4, and IL-1RA gene activities and expressions play an important role in modulating immunity and inflammatory activity. This study was designed to investigate the risk of developing FMF-related amyloidosis with MIF173G/C, IL-4 VNTR, and IL-1RA VNTR variants.
Materials and methods
Patients
A total of 172 FMF patients who applied to the Samsun Training and Research Hospital Medical Genetics Clinic were included in the study. Of these patients, 62 were FMF patients with amyloidosis, and 110 were FMF patients without amyloidosis. In addition, 120 healthy volunteers over the age of 18 without chronic disease were included in the study as a control group. All of the participants are of Turkish origin and live in the Northern Black Sea region. Clinical data were collected using a standard questionnaire. Written informed consent was obtained from all participants. The protocol of the study was approved by the local ethics committee (2016/8), and the study was conducted in accordance with the Declaration of Helsinki.
Genotyping
Five microliters of intravenous blood was taken from the participants into tubes with ethylenediaminetetraacetic acid. The genomic DNA was extracted using a commercial kit according to the manufacturer’s instructions. The MIF-173G/C, IL-4 VNTR, and IL-1RA VNTR variants were genotyped by performing the previously described polymerase chain reaction (PCR) and/or restriction fragment length polymorphism methods.11–13
For MIF-173G/C, a set of primers, (F) 5′-ACT-AAG-AAAGAC CCG-AGG-C-3′ and (R) 5′-GGG-GCA-CGT-TGGTGT-TTA-C-3′, was used to amplify the MIF-173G/C site. Genomic DNA was amplified in a 25-μL final reaction. PCR cycles were 95°C (5 min) for one cycle, followed by 95°C (45 s), 60°C (45 s), and 72°C (45 s) for 30 cycles. A final cycle of 72°C for 7 min completed the reaction. The amplified PCR product was digested in a 30-μL final reaction volume using 2 μL of reaction buffer and 1 μL of AluI restriction enzyme at 37°C for 12 h. Controls of known genotypes were included in every set of digestions carried out. The digested products were resolved on a 2% agarose gel stained with ethidium bromide and visualized using ultraviolet transillumination. Two fragments (268 and 97 bp) were seen when the G allele was present at position −173, while three fragments (206, 97, and 62 bp) were observed when the C allele was present.
For IL-4 VNTR, amplification was done using the (F) 5′AGG CTG AAA GGG GGA AAG C-3′ and (R) 5′-CTG TTC ACC TCA ACT GCT CC-3′ primers, with initial denaturation at 95°C for 5 min, 30 cycles of denaturation at 94°C for 30 s, annealing at 58°C for 45 s, extension at 72°C for 1 min and final extension at 72°C for 10 min. PCR was done in a 25 μL reaction mixture containing 50 ng of DNA, 0.8 μM of each primer, 200 μM of each dNTP, 2.5 mM MgCl, 1.5 units of Taq polymerase, and 2.5 μL of 10 × potassium chloride buffer. The resultant PCR products were examined by gel electrophoresis. The PCR products were 183 bp for the P1 allele and 253 bp for the P2 allele.
The identification of the IL-1RA VNTR variant was done by PCR using the (F) 5′-CTC AGC AAC ACT CCT AT-3′ and (R) 5′-TTC CAC CAC ATG GAA C-3′ primers following these conditions: first, an initial denaturalization at 94°C for 4 min; second, 30 cycles at 94°C, 51°C for 30 s, and 72°C for 45 s at each temperature; and third, a final extension at 72°C for 5 min. Four different alleles were identified as follows: allele 1, four repeats (410 bp); allele 2, two repeats (240 bp); allele 3, five repeats (500 bp); and allele 4, three repeats (325 bp). To check the results, 10% of the randomly selected samples were reworked, and a 100% match was found.
Statistical analysis
All data were analyzed using the Statistical Package for the Social Sciences (SPSS) for Windows (version 22.0; SPSS Inc, Chicago, IL, USA). The χ2 test was used for statistical analysis of genotype and allele frequencies in case and control groups. Continuous variables were expressed as mean ± standard deviation, and allele and genotype distributions were expressed as percent frequency. Statistical significance was accepted as p < 0.05.
Results
This study consisted of 292 individuals, including 62 FMF patients with amyloidosis, 110 FMF patients without amyloidosis, and 120 healthy controls. The mean age of FMF patients with amyloidosis was 31.10 ± 12.99 years, 29.61 ± 10.60 years in FMF patients without amyloidosis, and 32.90 ± 11.91 years in the control group. In all, 28 (45.16%) of FMF patients with amyloidosis, 64 (58.18%) FMF patients without amyloidosis, and 63 (52.50%) of the control group were women. There was no difference between the groups in terms of gender ratios or mean age. The demographic and clinical characteristics of the patient groups are presented in Table 1. In addition, the mutation distribution in the MEFV gene in the patient groups is presented in Table 2.
Baseline clinical and demographic features of the FMF patients (with/without amyloidosis).
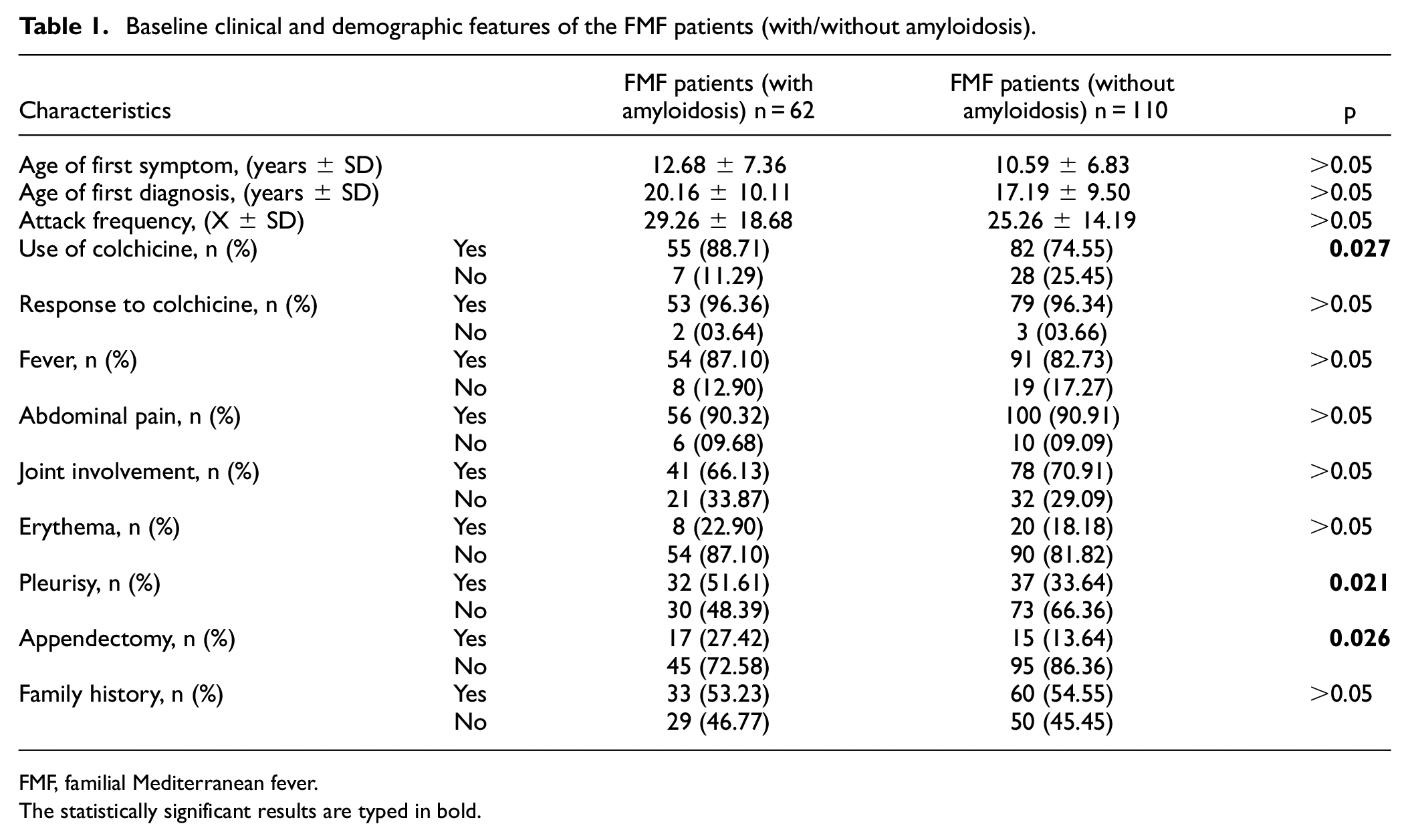
FMF, familial Mediterranean fever.
The statistically significant results are typed in bold.
Mutation distribution in the MEFV gene in patient groups.
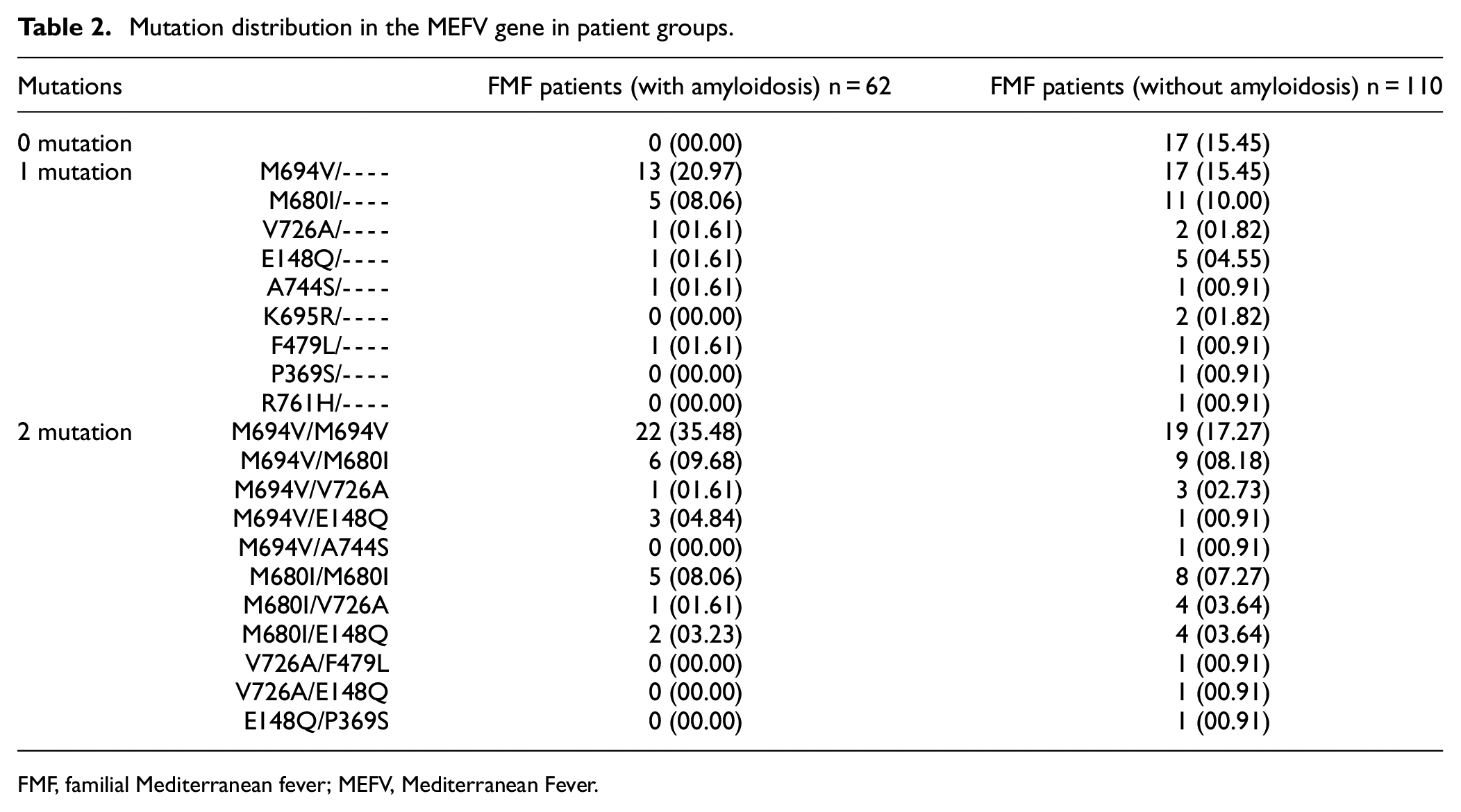
FMF, familial Mediterranean fever; MEFV, Mediterranean Fever.
There were statistical differences between FMF patients with amyloidosis and FMF patients without amyloidosis in terms of the use of colchicine, pleurisy, and appendicitis surgery. The use of colchicine, pleurisy, and appendectomy was more common in FMF patients with amyloidosis compared to FMF patients without amyloidosis (Table 1). There is no mutation in the MEFV gene in 15.45% of FMF patients without amyloidosis. However, at least one mutation in the MEFV gene was found in all FMF patients with amyloidosis. In the distribution of patients with a mutation in the MEFV gene in both patient groups with and without amyloidosis, the highest rate was observed in the M694V mutation. Similarly, when two mutations in the MEFV gene were found, the highest rate was found in the M694V/M694V mutation combination in both patient groups (Table 2).
The genotype and allele frequencies of MIF-173 G/C, IL-1RA VNTR, and IL-4 VNTR variants are presented in Table 3.
Genotype and allele frequencies of MIF-173G/C, IL-1RA VNTR, and IL-4 VNTR variants in groups.
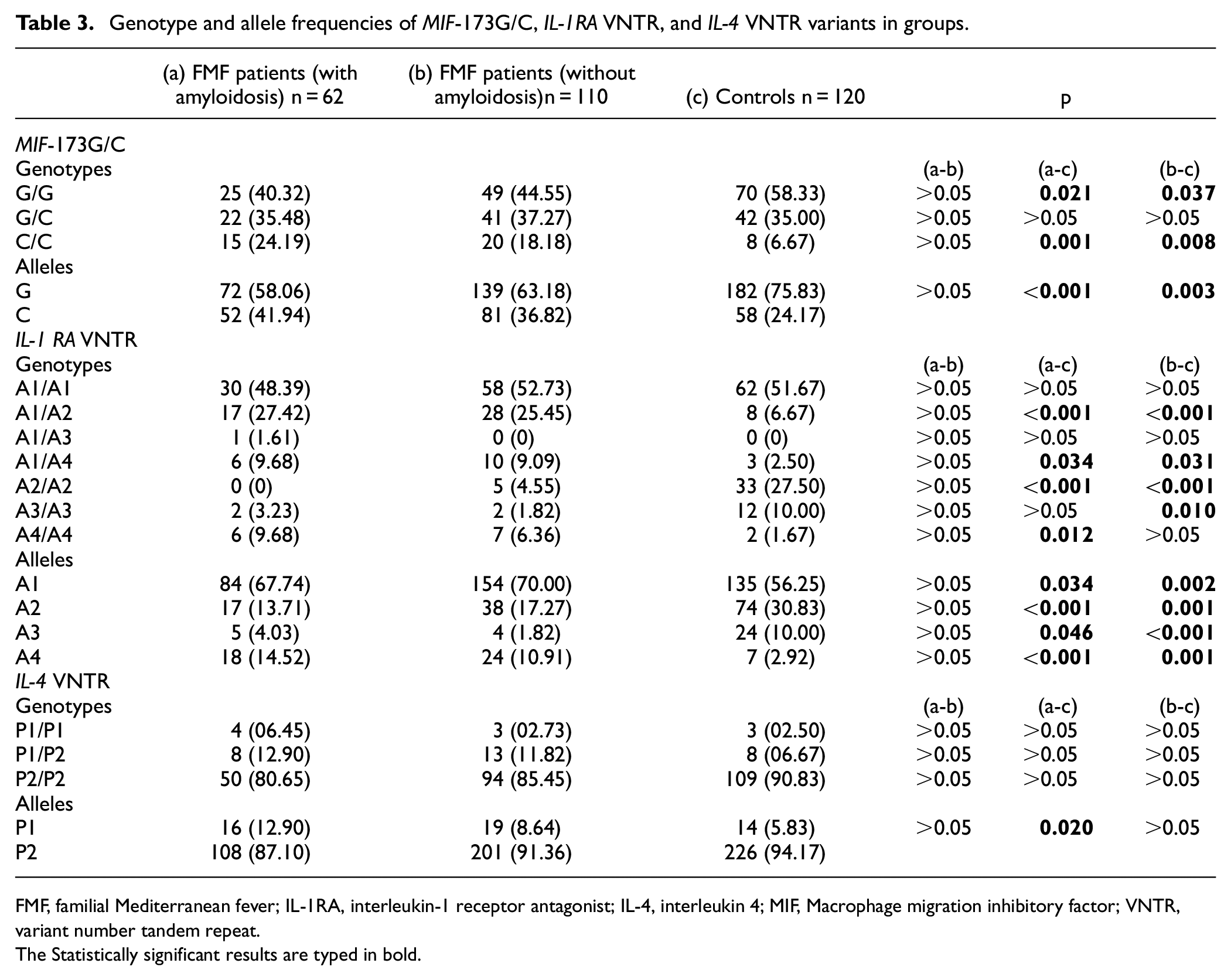
FMF, familial Mediterranean fever; IL-1RA, interleukin-1 receptor antagonist; IL-4, interleukin 4; MIF, Macrophage migration inhibitory factor; VNTR, variant number tandem repeat.
The Statistically significant results are typed in bold.
MIF-173G/C
The genotype and allele distribution of the MIF-173G/C variant were statistically different between the patient groups and the control group (Table 3). On the other hand, there was no statistical difference between the patient groups (Table 3). The C/C genotype frequency was higher in the patient groups compared to the control group (Table 3). The G/G genotype frequency was lower in the patient groups compared to the control group (Table 3). The C allele frequency was higher in the patient groups compared to the control group (Table 3).
IL-1RA VNTR
The genotype and allele distribution of the IL-1RA VNTR variant were statistically different between the patient groups and the control group (Table 3). On the other hand, there was no statistical difference between the patient groups (Table 3). A1/A2 and A1/A4 genotype frequencies were higher in the patient groups compared to the control group (Table 3). A2/A2 genotype frequencies were lower in the patient groups compared to the control group (Table 3). A3/A3 genotype frequency was lower in the FMF patient without amyloidosis group compared to the control group (Table 3). A4/A4 genotype frequency was higher in the FMF patient with amyloidosis group compared to the control group (Table 3). A1 and A4 allele frequencies were higher in the patient groups compared to the control group (Table 3). A2 and A3 allele frequencies were lower in the patient groups compared to the control group (Table 3).
IL-4 VNTR
There was no statistical difference between the groups in terms of genotype distribution of the IL-4 VNTR variant (Table 3). P2 allele frequency was lower in the FMF patient with amyloidosis group compared to the control group (Table 3).
We then evaluated the combined genotypes of the variants (Tables 4–6).
The combined genotypes distribution MIF-173G/C–IL-1RA VNTR variants in groups.
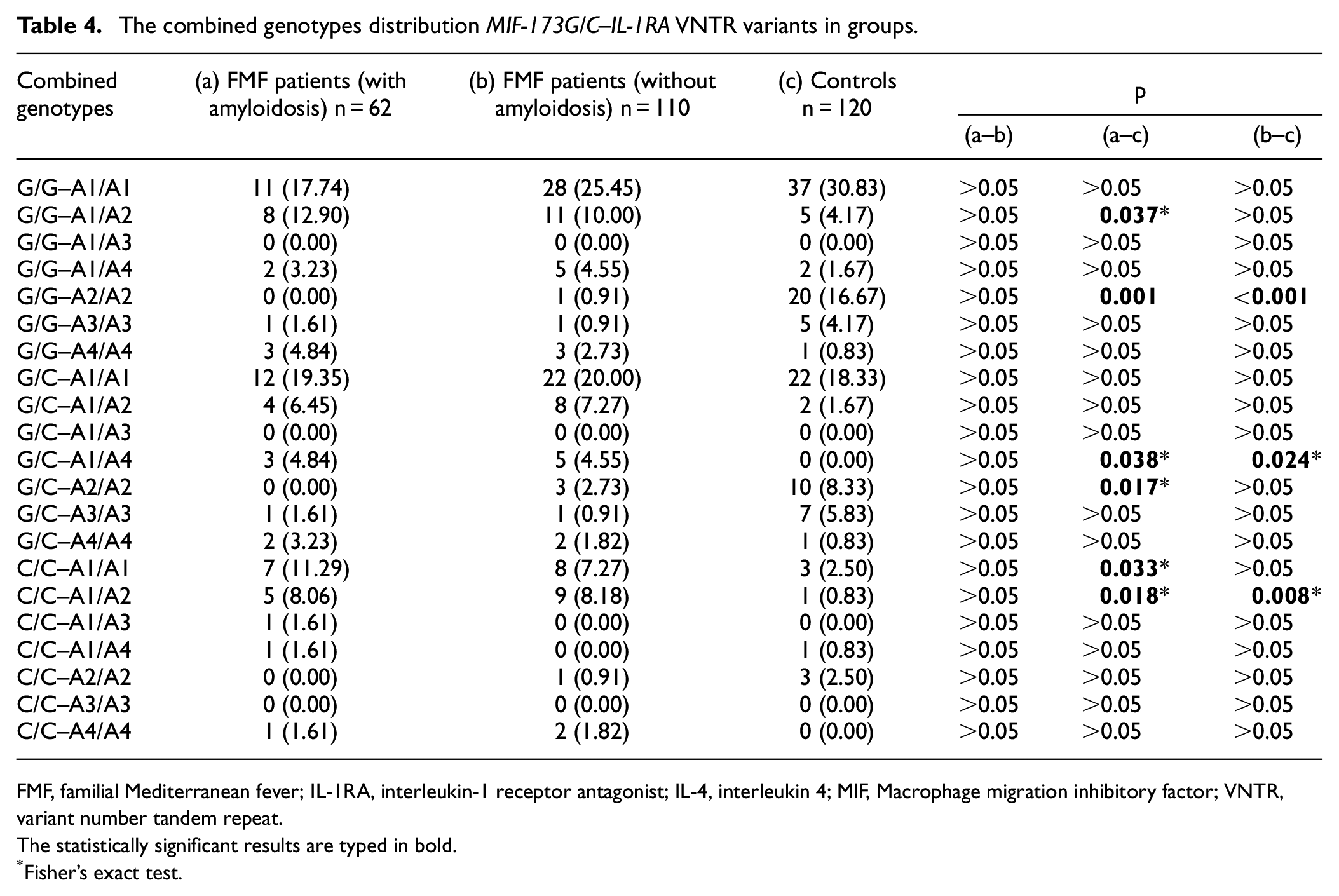
FMF, familial Mediterranean fever; IL-1RA, interleukin-1 receptor antagonist; IL-4, interleukin 4; MIF, Macrophage migration inhibitory factor; VNTR, variant number tandem repeat.
The statistically significant results are typed in bold.
Fisher’s exact test.
The combined genotypes distribution MIF-173G/C–IL-4 VNTR variants in groups.

FMF, familial Mediterranean fever; IL-1RA, interleukin-1 receptor antagonist; IL-4, interleukin 4; MIF, Macrophage migration inhibitory factor; VNTR, variant number tandem repeat.
The statistically significant results are typed in bold.
Fisher’s Exact Test.
The combined genotypes distribution IL-4 VNTR–IL-1RA VNTR variants in groups.
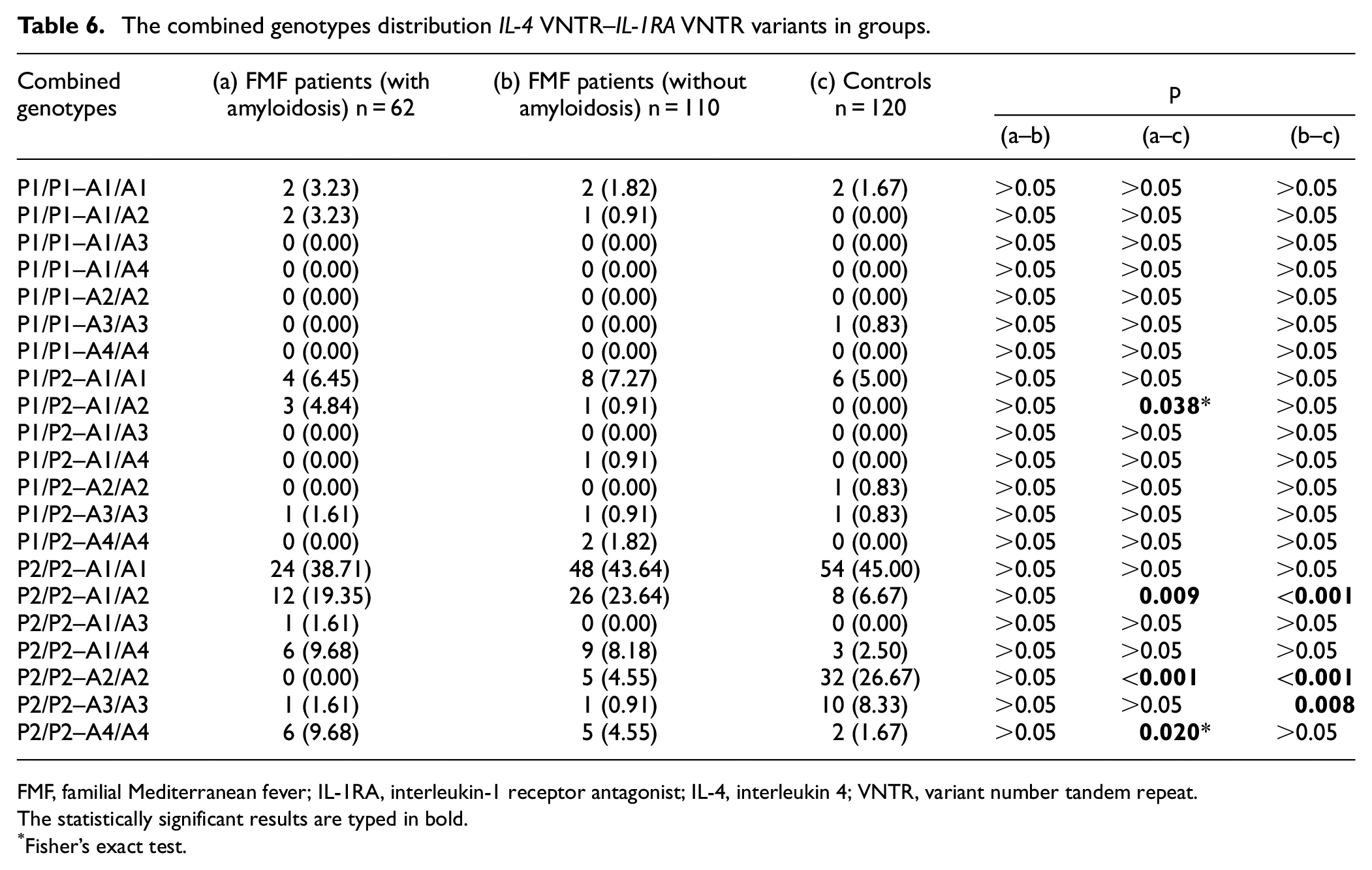
FMF, familial Mediterranean fever; IL-1RA, interleukin-1 receptor antagonist; IL-4, interleukin 4; VNTR, variant number tandem repeat.
The statistically significant results are typed in bold.
Fisher’s exact test.
MIF-173G/C–IL-1RA VNTR
The combined genotype distribution of the MIF-173G/C–IL-1RA VNTR variant was statistically different between the patient groups and the control group (Table 4). On the other hand, there was no statistical difference between the patient groups (Table 4). G/C-A1/A4 and C/C-A1/A2 combined genotype frequencies were higher in the patient groups compared to the control group (Table 4). G/G-A2/A2 combined genotype frequencies were lower in the patient groups compared to the control group (Table 4). G/G-A1/A2 and C/C-A1/A1 combined genotype frequencies were higher in the FMF patient with amyloidosis group compared to the control group (Table 4). G/C-A2/A2 combined genotype frequency was lower in the FMF patient with amyloidosis group compared to the control group (Table 4).
MIF-173G/C–IL-4 VNTR
The combined genotype distribution of the MIF-173G/C–IL-4 VNTR variant was statistically different between the patient groups and the control group (Table 5). On the other hand, there was no statistical difference between the patient groups (Table 5). The C/C-P2/P2 combined genotype frequency was higher in the patient groups compared to the control group (Table 5). G/G-P2/P2 combined genotype frequency was lower in the patient groups compared to the control group (Table 5). G/C-P1/P2 combined genotype frequency was higher in the FMF patient with amyloidosis group compared to the control group (Table 5).
IL-4 VNTR–IL-1RA VNTR
The combined genotype distribution of the IL-4 VNTR and IL-1RA VNTR variants was statistically different between the patient groups and the control group (Table 6). On the other hand, there was no statistical difference between the patient groups (Table 6). P2/P2–A1/A2 combined genotype frequency was higher in the patient groups compared to the control group (Table 6). P2/P2–A2/A2 combined genotype frequency was lower in the patient groups compared to the control group (Table 6). P1/P2–A1/A2 and P2/P2–A4/A4 combined genotype frequencies were higher in the FMF patient with amyloidosis group compared to the control group (Table 6). P2/P2–A3/A3 combined genotype frequencies were lower in the FMF patient without amyloidosis group compared to the control group (Table 6).
Discussion
Autoimmune diseases are a group of diseases characterized by the loss of immune tolerance of the immune system to one’s tissues. 14 The MEFV gene mutated in FMF, one of the most common autoimmune diseases, encodes the pyrin protein, which plays an important role in apoptosis and inflammatory signaling pathways. Pyrin induces inflammation with appropriate stimuli, followed by activation of caspase-1 and release of IL-1β and IL-18. 15 In addition, the MEFV gene mutation frequently seen in FMF patients is the M694V mutation. A relationship is also found between secondary amyloidosis, the most severe complication of FMF, and the M694V mutation. 16
In the distribution of patients with a mutation in the MEFV gene in both patient groups with and without amyloidosis, the highest rate was found in the M694V mutation. Similarly, in the distribution of patients with two mutations in the MEFV gene, the highest rate was found in the M694V/M694V mutation combination in both patient groups.
MIF, a cytokine with various immune-modulatory functions, modulates protective responses against intracellular pathogens. 17 MIF modulates the production and secretion of proinflammatory cytokines, including IL-1β, IL-6, tumor necrosis factor, and interferon-γ. 18 Cells from MIF−/− mice secrete less IL-1β and IL-18 than cells from wild-type control mice. 19 Savran et al. found increased MIF levels in FMF patients. 20 The MIF-173G/C variant C allele is associated with higher MIF levels. 21 The frequency of the MIF-173G/C C allele is higher in patients with hereditary periodic fever than in healthy individuals, and MIF levels increase significantly during the attack-free phases. 22 Cakan et al. reported that the MIF-173G/C C/C genotype was associated with FMF in children in Turkey. 23
In this study, the MIF-173G/C variant, C/C genotype, and C allele frequency were higher in the patient groups than in the control group. On the other hand, there was no statistical difference between the patient groups.
IL-1RA, a naturally occurring anti-inflammatory molecule, blocks the action of IL-1-induced proinflammatory activity. 24 The balance between IL-1RA and the proinflammatory cytokine IL-1β plays an important role in the development of FMF. 25 In addition, a study conducted in the Turkish population reported that the development of amyloid in FMF patients was associated with the acute phase response and the acute phase reactant serum amyloid A induced by IL-1β. On the other hand, it has been reported that there is no significant difference in allele and genotype frequencies of IL-1β and IL-1RA VNTR polymorphisms between FMF patients with and without amyloidosis and healthy individuals. 26
In this study, IL-1RA VNTR A1/A2 and A1/A4 genotype frequencies and A1 and A4 allele frequencies were higher in the patient groups than in the control group. In addition, the A4/A4 genotype frequency of FMF patients with amyloidosis was higher than that of the control group. On the other hand, there was no statistical difference between the patient groups.
IL-4, produced by activated CD4+ lymphocytes, mast cells, and basophils, is a potent anti-inflammatory cytokine. IL-4 enhances the anti-inflammatory effect and suppresses some proinflammatory cytokines. 27 Ibrahim et al. reported that during an attack, FMF patients had lower IL-4 levels than healthy individuals and FMF patients in remission. 28 On the other hand, Marzouk et al. reported that the IL-4 VNTR genotype distribution was not different between children with FMF and healthy children and was not associated with FMF severity or response to colchicine treatment. In addition, there was no significant difference in IL-4 levels between children with FMF attacks and children with FMF in remission. 29 A study with the Turkish population reported that the IL-4 VNTR P1 allele and P1/P1 genotype may be important molecular markers for FMF susceptibility. 30 Another study with the Turkish population reported that IL-4 polymorphism was associated with the risk of developing FMF. 31 In this study, there was no significant difference in IL-4 VNTR genotype distribution between the groups. The IL-4 VNTR P1 allele frequency of FMF patients with amyloidosis was found to be higher than that of healthy individuals. On the other hand, there was no significant difference between the frequency of the IL-4 VNTR P1 allele in FMF patients without amyloidosis and the frequency of the IL-4 VNTR P1 allele in healthy individuals.
In this study, which included FMF patients with amyloidosis, FMF patients without amyloidosis, and healthy controls, it was investigated whether MIF-173G/C, IL-4 VNTR, and IL-1RA VNTR variants play a role in the development of amyloidosis. This is the first study to evaluate these variants at risk for FMF-related amyloidosis in the Turkish population. In terms of clinical findings, the use of colchicine, pleurisy, and appendectomy was more common in FMF patients with amyloidosis than in FMF patients without amyloidosis. On the other hand, there was no significant difference in genotype and allele distribution of MIF-173G/C, IL-4 VNTR, and IL-1RA VNTR variants between FMF patients with amyloidosis and FMF patients without amyloidosis. In addition, there was no significant difference in the combined genotype distribution of variants between the two patient groups.
This study has several limitations. Our first limitation was the relatively small sample size. Another limitation is that MIF, IL-4, and IL-1RA levels were not evaluated. On the other hand, the absence of another study that comprehensively examined FMF-related amyloidosis with these variants can be considered the originality of the study. It can also form the basis for multicenter studies with larger sample sizes.
Conclusion
FMF is a common disease in our country. It is important to know the factors that can cause amyloidosis, which is an important complication of FMF. This is the first study in our country to evaluate the risk of FMF-related amyloidosis with these variants, to the best of our knowledge. In conclusion, MIF-173G/C C/C genotype and C allele and IL-1RA VNTR A1/A2, A1/A4 genotypes, and A1–A4 alleles are associated with FMF in the Turkish population, but not with amyloidosis risk in FMF patients. In addition, the IL-4 VNTR P1 allele is more common in FMF patients with amyloidosis than in healthy individuals. We believe that the results we have obtained can serve as a basis for further studies.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
