Abstract
Japanese mamushi (Gloydius blomhoffii) causes ∼3000 snakebites annually in Japan, but specific ultrasonographic findings have not been well characterized. Current severity assessment relies on visual grading systems based on local swelling and erythema, which may not provide sufficient information for timely and accurate treatment decisions. We present 3 pediatric patients (ages 7–9 y) with clinically diagnosed mamushi bites to the fingers, all presenting with Grade III envenomation (ie, swelling extending to the elbow). Point-of-care ultrasound examination revealed a remarkably consistent finding: a distinct hypoechoic band located in the deep subcutaneous layer immediately superficial to the muscle fascia. This characteristic band was most prominent at the bite site and gradually tapered off distally. Serial ultrasound examinations demonstrated progressive thinning and eventual disappearance of the hypoechoic band, correlating with clinical improvement following treatment with cepharanthine and anti-mamushi serum. Ultrasonography detected subcutaneous tissue changes beyond clinically apparent swelling in all cases, demonstrating superior sensitivity compared with visual assessment. All patients recovered without sequelae. These findings may differ from previously reported snakebite point-of-care ultrasound findings in terms of the anatomic location of the hypoechoic lesion. The hypoechoic bands observed in mamushi envenomation may serve as useful ultrasonographic markers for objective monitoring of treatment response and assisting with clinical management decisions, particularly regarding fasciotomy considerations and antivenom therapy guidance in the context of comprehensive clinical evaluation. These ultrasonographic features may not be species specific to mamushi but could represent common pathophysiologic responses to envenomation requiring validation across multiple species.
Introduction
Snakebite envenomation remains a significant global health concern, affecting millions of people worldwide annually. 1 The appropriate use of antivenoms is crucial for optimal patient outcomes because both underutilization and overutilization can lead to adverse consequences. In Japan, Gloydius blomhoffii (Japanese mamushi) is responsible for ∼3000 snakebite cases annually, with a mortality rate of 0.3%. 2 Systemic manifestations of mamushi envenomation may include coagulopathy, thrombocytopenia, and in severe cases acute kidney injury, although local tissue effects predominate in most cases.2,3 Current severity assessment relies on visual grading systems (Grades I–V) based on the extent of local swelling and erythema, but these approaches may not always provide sufficient information for timely and accurate treatment decisions. Importantly, on Honshu Island of Japan, the Japanese mamushi is the only medically important venomous species that causes local swelling. 3 Therefore, when patients present with progressive local swelling following snakebite in this geographic region, mamushi envenomation can be confidently diagnosed based on clinical presentation. However, mamushi bite diagnosis can be challenging because the fangs are ∼5 mm long and very thin, often leaving only small puncture wounds that may be mistaken for insect bites, particularly when bites occur at night. 3 The characteristic presentation of progressive local swelling provides the primary diagnostic criterion because laboratory changes may not appear for several hours after envenomation.
The utility of point-of-care ultrasound (POCUS) in evaluating local manifestations of snakebite has been increasingly recognized not only in Asian and African countries but also in North America, where similar findings have been reported in viper envenomations.4–6 Previous studies have reported that ultrasound can identify subcutaneous fat tissue swelling, hyperechoic changes, and cobblestone signs in snakebite cases.5,7 POCUS has demonstrated the ability to detect tissue damage that is not visible on clinical examination, predict severity progression, guide early antivenom administration decisions, and differentiate between muscular compartment syndrome and pseudo-compartment syndrome, thereby avoiding unnecessary fasciotomy.
However, detailed ultrasonographic findings in Japanese mamushi bites have not been comprehensively documented in the literature. We present a case series of 3 pediatric patients with clinically diagnosed Japanese mamushi bites to the fingers, all of whom demonstrated remarkably similar ultrasonographic findings characterized by distinct hypoechoic bands in the deep subcutaneous layer.
Methods
Ultrasonographic Examination
POCUS examinations were performed using different systems depending on case timing and availability: Case 1 was examined using a Canon Aplio 500 ultrasound system (Canon Medical Systems USA, Tustin, CA) with an 18-MHz linear transducer, whereas Cases 2 and 3 were examined using a GE LOGIQ E10 ultrasound system (GE HealthCare, Chicago, IL) with a 14-MHz linear transducer. All POCUS examinations were conducted by an experienced pediatric emergency physician (RH) with >10 years of ultrasound experience who has received specialized training in emergency ultrasound applications. No radiology specialists were directly involved in the bedside examinations.
Species Identification
In the absence of specimen-based identification by herpetologists or experts, we employed a retrospective syndromic approach for species diagnosis incorporating 3 key aspects:
Epidemiologic features: On Honshu Island, G blomhoffii (Japanese mamushi) is the only medically important venomous species that causes local swelling following snakebite.
3
The geographic location of all 3 cases (ie, Ibaraki Prefecture, Honshu) and outdoor exposure circumstances (ie, forest/schoolyard in warm months) are consistent with typical mamushi envenomation epidemiology. Clinical features: All patients presented with progressive local swelling extending proximally from the bite site, which is pathognomonic for mamushi envenomation in this geographic region. The characteristic fang mark pattern consisted of two small puncture wounds ∼1.0 cm apart, consistent with mamushi fang spacing. Serial laboratory monitoring: Monitoring showed no significant abnormalities in creatine kinase, coagulation parameters, or platelet counts in any case. Laboratory abnormalities do not necessarily correlate with local swelling severity in mamushi envenomation and were not required for diagnosis given the epidemiologic and clinical features.
This syndromic approach represents standard clinical practice in Japanese emergency medicine for mamushi bite diagnosis and has been validated by prior epidemiologic studies. 3
Case Reports
Case 1
A 7-y-old boy was bitten on his right third finger by a snake while catching insects in a nearby forest. He initially presented to a local clinic with finger swelling, which progressed proximally to the forearm, prompting referral to our hospital. On arrival (3 h after the snakebite), the boy was in good general condition with stable vital signs. Fang marks were observed on the right third finger (Figure 1A), and swelling had extended from the forearm to the lower part of the upper arm (Grade III). Neurologic examination was normal, and peripheral pulses were palpable. Based on the patient's clinical signs, characteristic bite pattern, and geographic location where mamushi is the only medically important venomous species causing local swelling, the bite was attributed to a mamushi (G blomhoffii). The patient was admitted and treated with cepharanthine and anti-mamushi serum (KM Biologics Co, Kumamoto, Japan), an equine-derived whole IgG preparation, 6000 units. Ultrasonography performed 3 h after the bite revealed marked swelling and increased echogenicity in the subcutaneous fat layer (Figure 1B). Notably, a hypoechoic band was observed parallel to the tissue layers in the deep subcutaneous fat, immediately superficial to the fascia, most prominent near the bite site and tapering off distally (Figure 1C–E). Ultrasonography detected subcutaneous thickening in the proximal upper arm region where no visible swelling was apparent on clinical examination. Despite antivenom administration, local swelling continued to progress for ∼4 h before stabilizing. The swelling gradually subsided with treatment, and the patient was discharged on hospital Day 4 without sequelae.
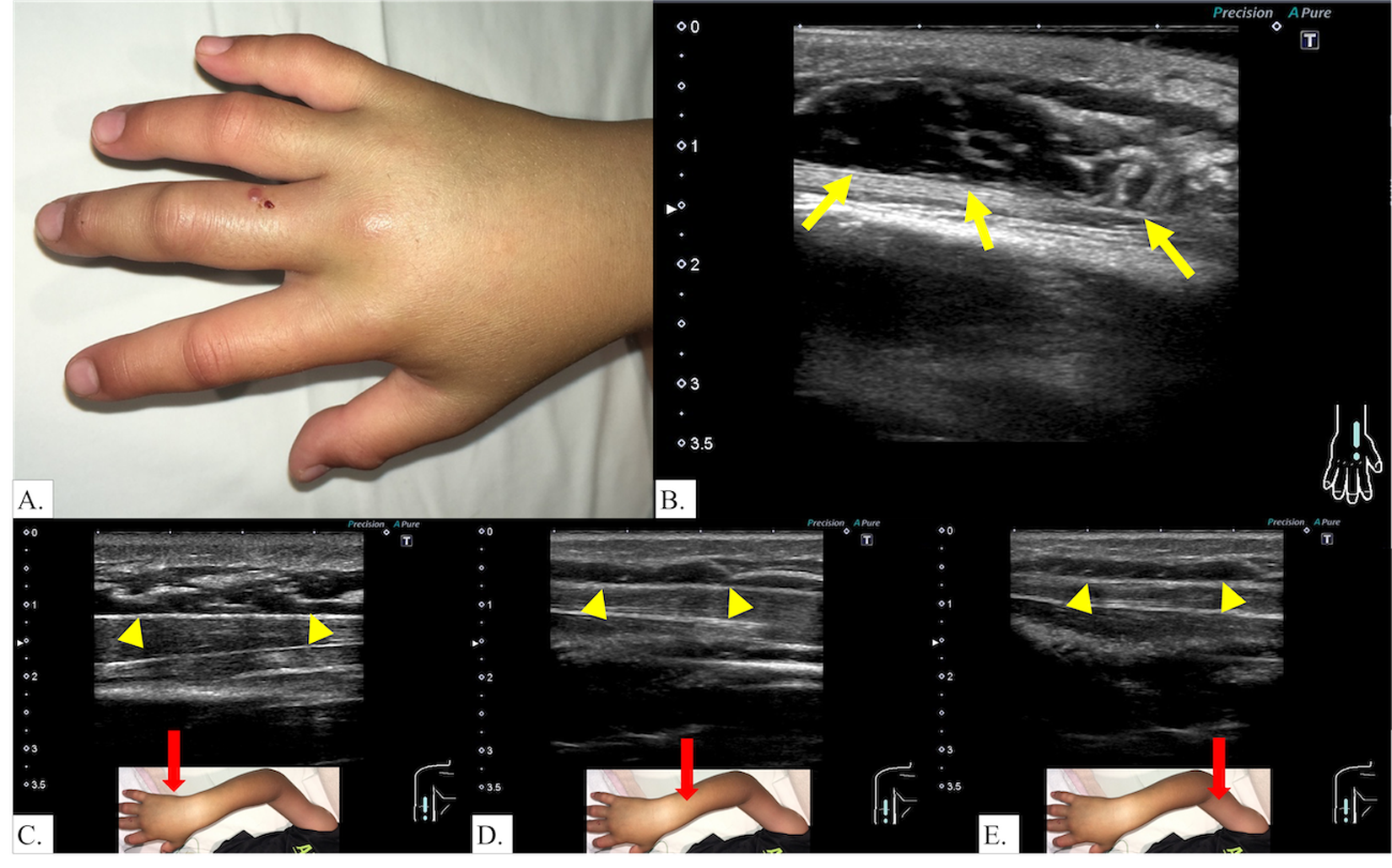
Case 1. Clinical presentation and ultrasonographic findings. A, Fang marks and local swelling on the right third finger 2 h after the bite. B, Ultrasound showing hypoechoic band (arrows) in deep subcutaneous fat above fascia, most prominent near the bite site. C–E, Hypoechoic bands (arrowheads) were most prominent at the bite site and gradually diminished with increasing distance from the bite site.
Case 2
An 8-y-old boy was bitten on his right second finger while handling a snake he had caught. Although the initial swelling was mild, the boy came to our emergency department at his parents’ request 2 h after the snakebite. He was hemodynamically stable and in good general condition. Fang marks were noted on the right second finger (Figure 2A), and swelling extended to the wrist at presentation. However, within 1 h of observation in the emergency department, the swelling progressed rapidly to the elbow. The radial pulse remained palpable. The snake responsible was identified as a mamushi (G blomhoffii) based on an actual photograph reviewed by experienced hospital staff familiar with local herpetofauna (Figure 2B). The envenomation was classified as Grade III, and treatment with cepharanthine and anti-mamushi serum (KM Biologics Co) 6000 units was initiated. Local swelling continued to progress for 8 h after antivenom administration before reaching peak severity. On hospital Day 3 (∼40 h after the bite), ultrasonography demonstrated edematous changes and increased echogenicity in the subcutaneous fat, along with a hypoechoic band located immediately superficial to the muscle fascia (Figure 2C). A characteristic hypoechoic band was observed running parallel to the fascial plane within the deep subcutaneous fat, demonstrating greatest prominence proximally at the bite site and progressive attenuation distally (Figure 2D, E). Similar to Case 1, subcutaneous tissue changes were detected ultrasonographically beyond the clinically apparent swelling boundary. The swelling gradually improved, and the patient was discharged on hospital Day 6 without sequelae.
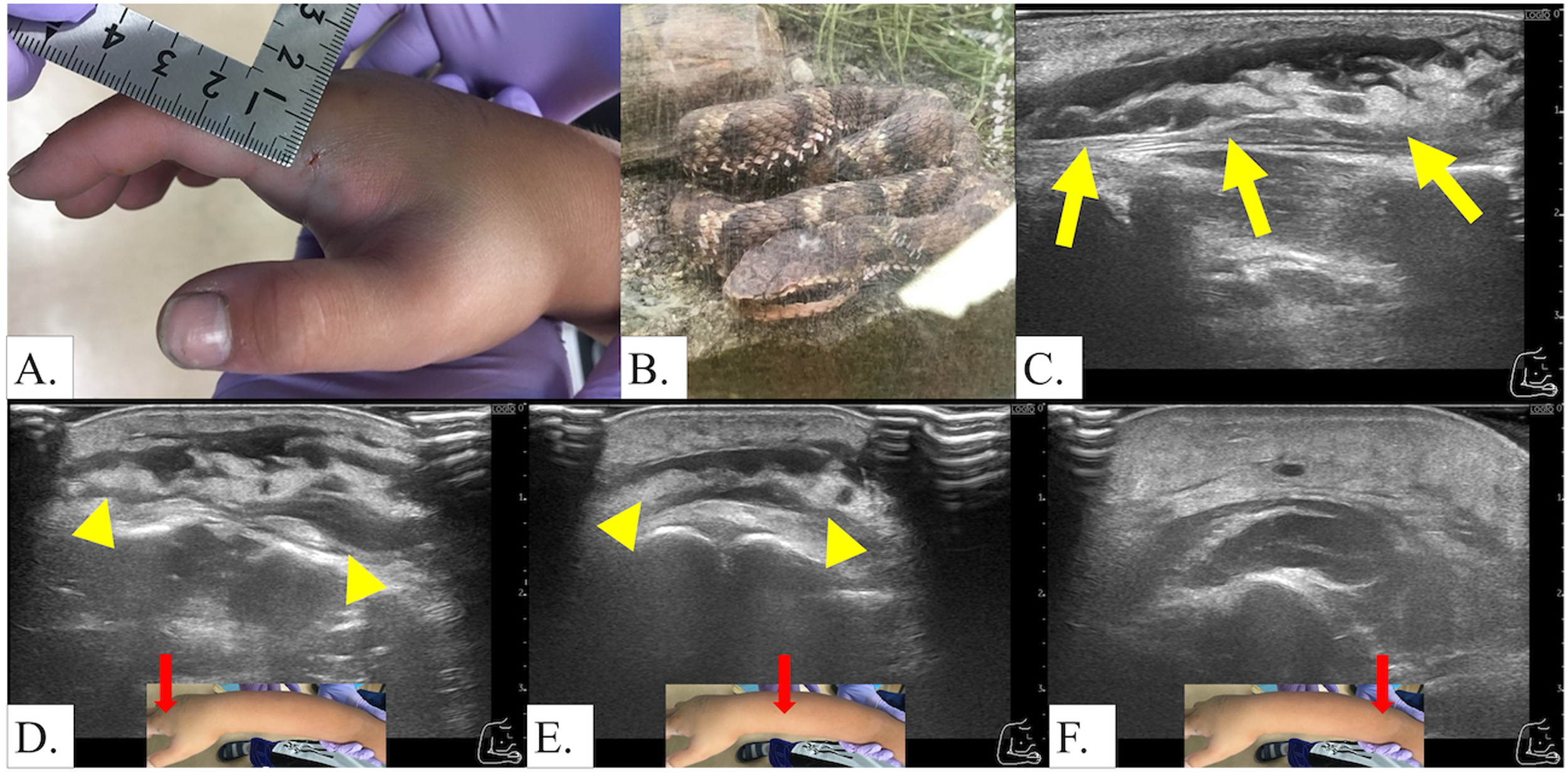
Case 2. Clinical presentation, snake identification, and ultrasonographic findings. A, Fang marks and local swelling on the right second finger with swelling extending to the wrist. B, Photograph of the snake (Gloydius blomhoffii) responsible for the bite, used for species identification. C, Ultrasound on hospital Day 3 showing hypoechoic band (arrows) immediately superficial to the muscle fascia. D, E, Ultrasound showing characteristic hypoechoic band (arrowheads) running parallel to the fascial plane within the deep subcutaneous fat, demonstrating greatest prominence at the bite site.
Case 3
A 9-y-old boy was bitten on the tip of his right second finger by a snake while approaching a group of children playing with the snake in a schoolyard. He was initially transferred to a local emergency facility and subsequently referred to our hospital due to progressive hand swelling. On presentation (2 h after the snakebite), he was in good general condition with stable vital signs. Fang marks were present on the right second finger (Figure 3A), and swelling extended to the wrist. Within a few hours, the swelling progressed to the elbow. Based on the clinical presentation, fang mark pattern, and geographic context, the envenomation was attributed to mamushi (G blomhoffii). The envenomation was classified as Grade III, and treatment with cepharanthine and anti-mamushi serum (KM Biologics Co) 6000 units was initiated. Despite treatment, local swelling progressed for ∼10 h before stabilizing. Ultrasonography performed 5 h after the bite revealed diffuse swelling and increased echogenicity in the subcutaneous tissue, along with a hypoechoic band parallel to the fascial layer (Figure 3B, C). Serial ultrasonographic examinations along the affected limb demonstrated the anatomic distribution of the hypoechoic band. The characteristic band was most pronounced at the bite site on the fingertip and gradually diminished in thickness as the examination extended proximally along the hand and forearm (Figure 3D–F). In the more proximal regions of the limb, only generalized subcutaneous thickening from edema was observed, without the distinct hypoechoic band seen at the envenomation site. Notably, ultrasonography detected subcutaneous tissue swelling in the proximal upper arm, where no visible swelling was apparent on clinical examination. This gradient pattern of ultrasonographic findings correlated with the clinical distribution of swelling and demonstrated the superior sensitivity of ultrasound in detecting tissue changes beyond those visible on physical examination.
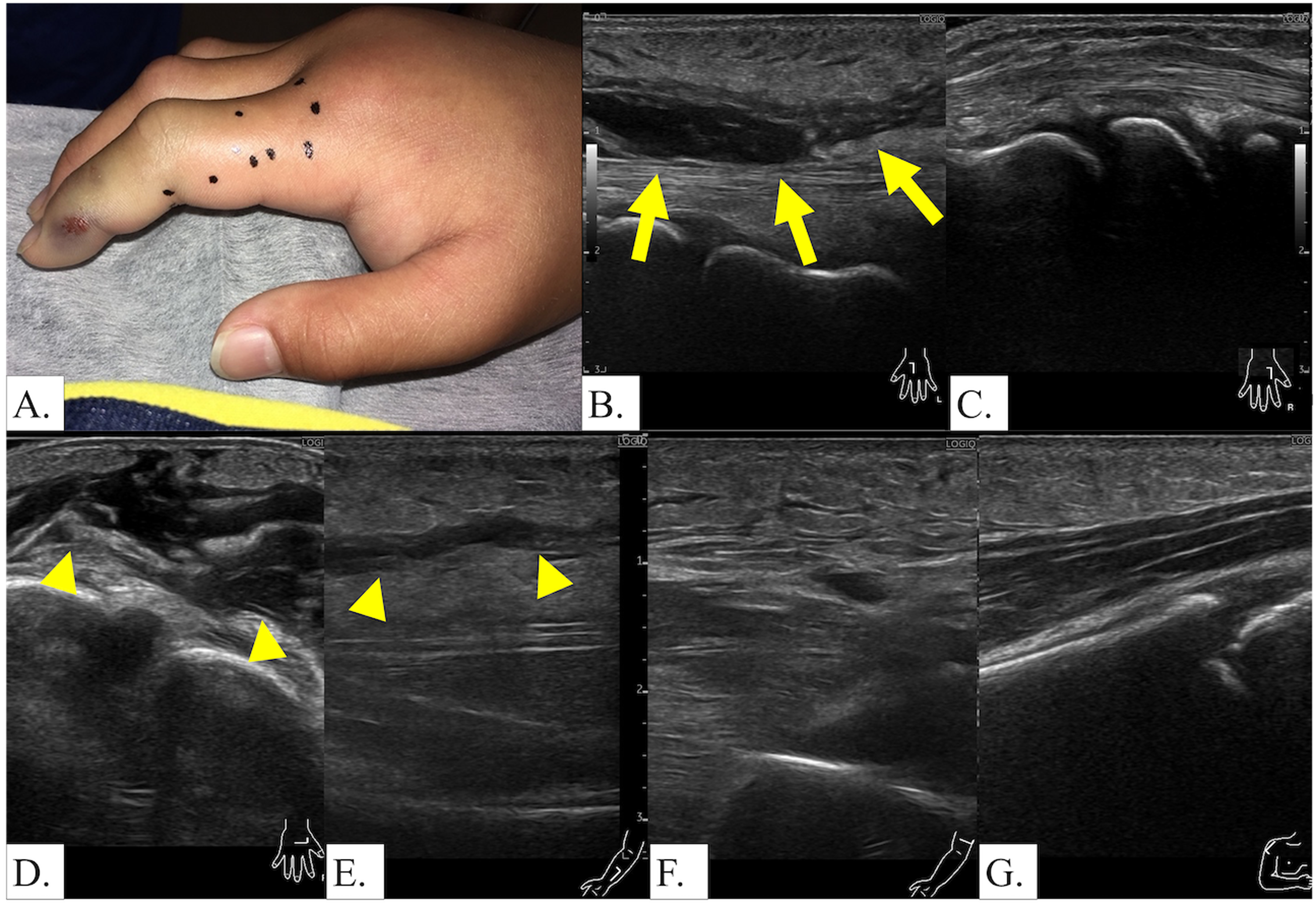
Case 3. Clinical presentation and anatomic distribution of ultrasonographic changes. A, Fang marks and local swelling on tip of right second finger with swelling extending to the wrist. B, Ultrasound 5 h after the bite showing hypoechoic band (arrows) parallel to the fascial layer. C, Ultrasound of the contralateral left second finger for comparison. D–F, Ultrasound showing characteristic hypoechoic band (arrowheads) running parallel to the fascial plane within the deep subcutaneous fat, demonstrating greatest prominence at the bite site. G, In the proximal regions, only subcutaneous thickening due to swelling was observed.
Follow-up ultrasound examinations were conducted daily, revealing progressive thinning of the hypoechoic band correlating with clinical improvement (Figure 4A–D). The band had disappeared by hospital Day 4, and the patient was discharged on Day 5 without sequelae.
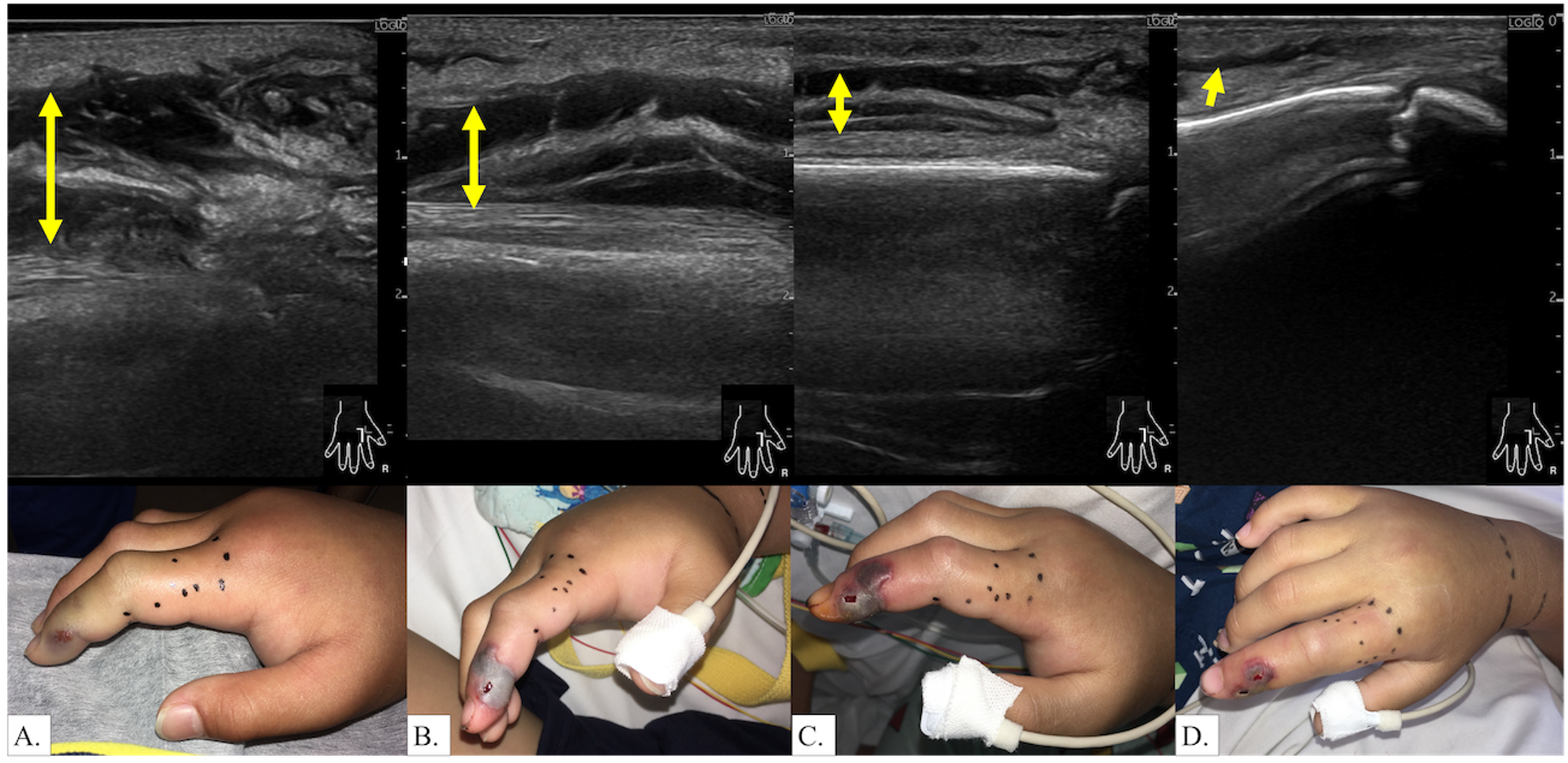
Case 3. Serial ultrasonographic follow-up demonstrating treatment response. A–D, Follow-up ultrasound demonstrating progressive thinning of the hypoechoic band with clinical improvement: (A) Day 1; (B) Day 2; (C) Day 3; (D) Day 4.
Discussion
Our findings suggest that the distribution pattern of edema differs significantly between snakebite envenomation and cellulitis. While cellulitis typically presents with diffuse edema throughout the subcutaneous tissue, 8 snake venom appears to preferentially affect the deep subcutaneous layer, creating the characteristic hypoechoic band observed in all 3 of our cases. This preferential deep layer involvement may be attributed to venom delivery by fangs that inject venom deep into tissues, whereas cellulitis typically originates from superficial skin injury. Mamushi venom contains multiple enzymatic components, including hyaluronidases, phospholipases A2, and metalloproteinases that increase vascular permeability, cause microvascular occlusion and lymphatic obstruction, and promote tissue edema. 2 The deep subcutaneous tissue, which mainly contains vascular structures within a loose connective tissue network, 9 may serve as a preferential site for edema spread. The exact pathophysiologic mechanisms underlying this preferential deep subcutaneous involvement require further investigation, as does the question of whether similar patterns occur with other snake species or are specific to mamushi envenomation. This anatomic distribution of edema has not been well emphasized in POCUS studies of snakebites to date. 5
In addition to the characteristic deep hypoechoic bands, all 3 patients demonstrated varying degrees of hyperechoic changes and swelling in the superficial subcutaneous tissues. However, the typical prominent cobblestone pattern commonly described in cellulitis 10 was not a dominant feature in our cases. The superficial tissue changes were generally milder, showing predominantly hyperechoic appearance with some heterogeneity, but lacking the pronounced cobblestone architecture typically emphasized in soft tissue infections. The most striking ultrasonographic finding in mamushi envenomation was the distinct bandlike hypoechoic appearance in the deep subcutaneous layer. Interestingly, the fluid-rich edematous changes in the superficial layer may have contributed to fluid accumulation preferentially along deeper anatomic planes, potentially explaining the characteristic deep subcutaneous band pattern observed in these patients.
Treatment in all cases included cepharanthine and equine-derived anti-mamushi serum. Cepharanthine, a natural bisbenzylisoquinoline alkaloid isolated from Stephania cephalantha Hayata, is the only bisbenzylisoquinoline alkaloid approved for human use and has been used clinically for >70 y with demonstrated safety. 11 It possesses multiple pharmacologic properties including immunomodulatory and anti-inflammatory activities. Experimental studies by Abe et al demonstrated dose-dependent inhibition of mamushi venom-induced lethality and circulatory disturbances in mice. 12 A large multicenter retrospective study of 234 mamushi bite cases in Japan demonstrated that in severe cases (Grades III–V), anti-mamushi serum was associated with significantly shorter hospital stays compared with cepharanthine alone (P=0.024), whereas no significant difference was observed in mild to moderate cases (Grades I and II). 13 Based on this evidence, both agents are routinely administered in many Japanese hospitals for moderate to severe envenomation, although the relative contribution of each agent to clinical outcomes remains a subject of ongoing investigation.
In our case series, local swelling continued to progress for several hours after antivenom administration in all 3 cases (∼4–10 h before stabilizing). This is a well-recognized phenomenon in snakebite management because antivenom does not necessarily prevent progression of local tissue damage and edema following envenomation.14,15 This observation underscores the importance of patient and family education regarding expected clinical course following antivenom administration because continued swelling does not necessarily indicate treatment failure.
Necrotizing fasciitis characteristically presents with deep fascial fluid accumulation on ultrasound examination. 16 The hypoechoic bands observed in our mamushi bite cases share similarities with this deep fascial fluid pattern, but several distinguishing features may aid in differential diagnosis. Although necrotizing fasciitis also may present with hyperechoic gas bubbles (reported in ∼62% of cases) and fascial thickening or irregularity (each found in about 10% of cases), these additional features were not observed in our mamushi envenomation cases.17,18 However, it is important to emphasize that no single ultrasonographic finding is universally present in necrotizing fasciitis, and the absence of these features should not be considered definitive for ruling out this serious complication. The diagnosis should be based primarily on clinical suspicion supported by comprehensive evaluation of symptoms, signs, and laboratory findings, with POCUS serving as a complementary diagnostic tool when interpreted alongside clinical findings such as fever, systemic toxicity, and laboratory markers.
Current severity grading relies on visual assessment, which may be less sensitive than POCUS findings. Our cases suggest that applying traditional grade classifications to ultrasound-detected changes might lead to overtreatment because ultrasound detected subcutaneous thickening even in areas without visible swelling. The thickness and extent of the hypoechoic bands, which appear to reflect more severe inflammation, potentially could serve as additional indicators for clinical assessment and treatment intervention decisions. This warrants further investigation. Given that the rate of proximal progression (extent of edema over time) can be monitored by ultrasound for decision making regarding antivenom indication or additional doses, 7 the determination of which sonographic findings should be used as indicators requires full validation in future studies.
Historically, compartment syndrome concerns led to aggressive fasciotomy approaches in snakebite management, but conservative treatment has become more prevalent.19,20 In our cases, POCUS clearly demonstrated that the hypoechoic bands were located above the muscle fascia, confirming that swelling was limited to subcutaneous tissue only. POCUS thus can help to identify which tissues are affected and contribute to more informed fasciotomy decision making.
Interestingly, similar horizontal hypoechoic bands have been reported in local allergic reactions to insect bites and stings, attributed to angioedema from increased vascular permeability. 21 While detailed descriptions of deep subcutaneous hypoechoic bands are limited in the snakebite literature, these findings suggest that vascular permeability changes may produce similar ultrasonographic patterns across different envenomation and allergic conditions.
Limitations to our study include the retrospective nature of this case series and the lack of definitive species identification with actual specimen examination. Although clinical diagnosis based on geographic distribution and characteristic presentation is standard practice in Japanese emergency medicine settings, this represents a limitation in scientific documentation. This study also has several other limitations. All 3 cases involved pediatric patients with finger injuries, representing a limited clinical scenario. The thickness and tissue composition of different anatomic sites may influence ultrasound findings. Additionally, this study focused exclusively on clinically diagnosed mamushi bites; different snake species with varying venom compositions and fang lengths may produce different ultrasonographic patterns. The examination was limited to two-dimensional ultrasound; future studies incorporating shear-wave elastography and color Doppler ultrasound may provide additional insights into tissue stiffness changes and vascular alterations following envenomation. Further accumulation of snakebite POCUS cases from various regions and with different species is needed to validate these findings.
Conclusions
We identified characteristic hypoechoic bands in the deep subcutaneous layer in all 3 pediatric patients with clinically diagnosed mamushi bites. Although these findings require validation in larger studies with multiple species, they may prove useful for monitoring treatment response and guiding clinical management decisions. This deep subcutaneous distribution pattern has not been well emphasized in previous POCUS studies of snakebite envenomation, although similar findings have been reported in allergic reactions to insect bites. Whether these represent species-specific features or common pathophysiologic responses that have been underreported remains to be determined. Future research should focus on accumulating ultrasonographic findings from various snake species to develop evidence-based guidelines for ultrasound-assisted clinical assessment, monitoring treatment response, and identification of situations where fasciotomy should be avoided as part of comprehensive clinical evaluation.
Previous Presentation
This study was presented in part at the 98th Annual Meeting of the Japanese Society of Ultrasonics in Medicine in Kyoto, Japan, May 30 to June 1, 2025.
Footnotes
Consent to Participate
Informed consent was obtained from the parents of all patients.
Author Contribution(s)
Financial/Material Support
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflicting Interests
The authors declare no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
