Abstract
This case series aimed to observe the impact of a single-stage ultramarathon (50–100 km) on gastrointestinal integrity, systemic immune responses, and exercise-associated gastrointestinal symptoms in 3 adolescent (aged 12–15 y) competitors. Body mass, venous and finger-prick blood samples and fecal samples were taken before and after the race for analysis of exercise-induced body mass loss, plasma osmolality, plasma intestinal fatty acid binding protein, soluble CD14, and C-reactive protein, total and differential leukocyte counts, and fecal bacterial alpha-diversity. Gastrointestinal symptom incidence were recorded before, during, immediately after, and 24 h after the race. Food and fluid intake were recorded for the 48 h before the race and each hour during the race. Body mass decreased in Youth 1 (−0.4%) and Youth 3 (−0.9%) and increased in Youth 2 (+0.5%) from before to after the race. No youth competitor met carbohydrate intake recommendations in the lead-up to (2.8–5.1 g·kg–1·d–1) or during the race (24–26 g·h–1). Pre- to post-race plasma intestinal fatty acid binding protein and C-reactive protein concentrations increased in the youth competitors, but was of no clinical significance (≤657 pg·mL–1 and ≤3.36 micrograms·mL–1, respectively). A pre- to postrace change in plasma soluble CD14 of clinical relevance was observed in Youth 3 (1.5 micrograms·mL–1), but not Youth 1 (−0.1 micrograms·mL–1). Total and differential leukocyte counts increased beyond the normal reference range in Youth 1 and Youth 2, but not Youth 3. The incidence and severity of gastrointestinal symptoms were low at all timepoints in the competitors. Large intra- and interindividual responses to ultramarathon events observed in this case series suggest that adolescent ultramarathon runners require individually tailored nutrition support.
Keywords
Introduction
Single-stage ultramarathon events (≥42.195 km) have gained popularity among athletes, attracting novice to elite competitors. 1 Events are characterized by challenging topographies and environmental conditions (ie, heat, wind, and/or altitude). 2 Such extreme exertional stress (ES) disrupts gastrointestinal (GI) homeostasis, leading to exercise-induced GI syndrome (EIGS). The pathophysiologic pathways of EIGS have been extensively discussed previously. 3 In short, the circulatory-gastrointestinal pathway of EIGS is linked to GI epithelial injury, luminal pathogenic translocation into the circulation, and systemic inflammatory response (SIR) outcomes due to redistribution of blood away from the GI tract to working muscle and peripheral circulation when exercising.3,4 The neuroendocrine-gastrointestinal pathway is linked to an exercise-associated increase in sympathetic drive that alters GI function (ie, motility, digestion, and/or absorption). A predominant outcome of these pathways is exercise-associated GI symptoms (Ex-GIS), which can have performance implications and/or signs of clinical implications for athletes.5,6 Age as an intrinsic factor exacerbating EIGS has garnered interest due to the increasing number of adolescents (≤18 y of age) participating in ultramarathons (eg, 7775 adolescent finishes between 1960 and 2017).7,8 Research investigating how these events impact the GI system at a time of important growth and development is limited. 8 Therefore, it is unknown whether adult recommendations established to safeguard athletes’ health and performance (ie, training, nutrition, and hydration)9,10 are applicable to adolescents.
Exercise gastroenterology studies on adolescent athletes have significant limitations, with insufficient ES protocols not reflective of real-life practices nor substantial enough to produce clinically relevant changes to EIGS biomarkers.11,12 Notably, a recent study applied best-practice methodologies to investigate EIGS in youth athletes within a laboratory setting and observed a similar presentation of GI perturbations compared with adults.11,13 However, it is challenging to replicate real-life ultramarathon conditions known to have significant physiological effects (eg, ambient conditions, altitude, and duration). In addition, although dietary control is an essential experimental practice in gastroenterology research, 11 it limits the understanding of before- and during-race nutrition habits and the impact on markers of EIGS and Ex-GIS. Considering the relationship between carbohydrate and fluid intake and the severity and incidence of EIGS,5,14–16 understanding the current nutrition practices of adolescents participating in ultramarathon events is significant and novel. This case series aimed to observe the impact of single-stage ultramarathon participation on GI integrity, SIRs, and Ex-GIS in 3 adolescent competitors (12–15 y of age), with consideration of competition nutrition and hydration practices. Observations may offer preliminary insights into whether adolescent age is an intrinsic factor impacting EIGS and if further research is warranted to determine the applicability of current prevention and management strategies of EIGS for youth athletes.
Methods
Presentation of Athletes
Following ethics approval (MUHREC 29429), 3 healthy recreational youth (≤18 y of age) endurance athletes (n=1 male and n=2 females) from 2 ultramarathon events (Surf Coast Century [SCC], December 4, 2021, and Margaret River Ultra Marathon [MRUM], May 7, 2022) volunteered to participate (Table 1). The 3 athletes were categorized as Youth 1, Youth 2, and Youth 3. Exclusion criteria were in accordance with Costa et al, 16 and a prerace questionnaire was completed to ensure that competitors were free of chronic and acute illness and injury.
Competitor and race characteristics.
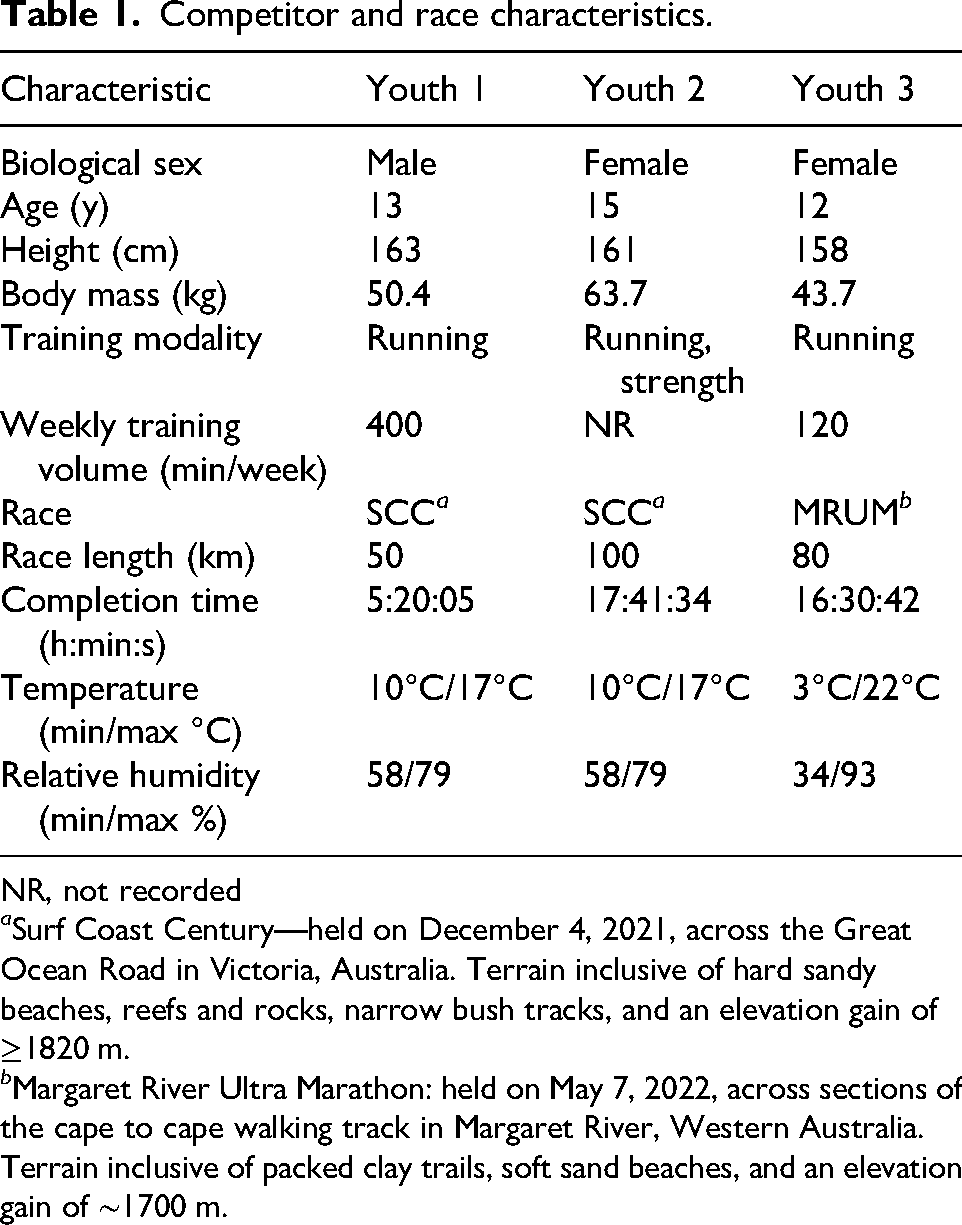
NR, not recorded
a Surf Coast Century—held on December 4, 2021, across the Great Ocean Road in Victoria, Australia. Terrain inclusive of hard sandy beaches, reefs and rocks, narrow bush tracks, and an elevation gain of ≥1820 m.
b Margaret River Ultra Marathon: held on May 7, 2022, across sections of the cape to cape walking track in Margaret River, Western Australia. Terrain inclusive of packed clay trails, soft sand beaches, and an elevation gain of ∼1700 m.
Study Design and Data Collection
Following recruitment and informed consent, competitors recorded, in real time, estimated amounts of all foods, fluids and supplements consumed for the 48 h leading up to the respective ultramarathon. In addition, competitors completed a paper-based race nutrition plan documenting planned food and fluid intake for each hour of the race. Immediately after the race, 3 trained sports dietetic researchers conducted a standardized interview (using a multiple-pass dietary recall interview technique) to capture total food and fluid ingestion during the race. Ex-GIS were determined by a modified visual analogue scale, 17 with participants completing the tool before the race, during the race, and immediately after and 24 h after the race, with during-race data obtained retrospectively.
To reduce competitor burden on race day, baseline characteristics and resting biomarkers were collected the afternoon before the race. Height (Seca 213 Portable Stadiometer, Seca GmbH, Hamburg, Germany) and nude body mass (Seca 813 Portable Scales, Seca GmbH) were measured on a solid surface. Multifrequency bioelectrical impedance analysis (Bodystat Quadscan 4000, Bodystat Ltd, Douglas, Isle of Man, UK) was used to determine pre-competition total body water and extracellular body water. Participants were required to lie supine for 10 min before conducting multifrequency bioelectrical impedance analysis. During this time, the conduction body points (right hand and foot) were cleaned and air dried prior to electrode attachment and analysis. Impedance calibration was performed prior to measurement in accordance with the manufacturer’s instructions using a device-specific calibrator (ie, impedance range between 496 and 503 Ω). Blood was collected via venipuncture from the antecubital vein into 2 vacutainers (6 mL lithium heparin and 4 mL K3EDTA; Becton Dickinson Pty Ltd, Macquarie Park, New South Wales, Australia) and immediately stored at −20°C before transferring to −80°C for longer-term laboratory storage until analysis. Finger prick blood sampling was conducted to determine leukocyte counts. Participants also were asked to provide an ∼30 g midflow fecal sample in a sterilized fecal collection container (Sarstedt Australia Pty Ltd, Mawson Lakes, South Australia, Australia), which was immediately stored at −20°C before transferring to −80°C for longer-term storage until processing and analysis. All measures were repeated within a 15 to 30 min timeframe after the race in the same order for consistency. Competitors were given a 24-h window to provide their first fecal sample following the race, as described previously. Youth 2 did not provide a postrace fecal sample.
Sample and Data Analysis
Dietary analysis was performed using FoodWorks.online V.2.0 Professional analysis software (Xyris Pty Ltd, Brisbane, Australia) by an accredited sports dietitian. Total and differential (ie, neutrophil, lymphocyte, and monocyte) leukocyte counts were determined by a HemoCue system (white blood cell differential; HemoCue AB, Ängelholm, Sweden); coefficient of variance [CV], 1.4%). Blood hemoglobin (CV, 1.2%) was determined by a HemoCue system (Hb201, HemoCue AB) in duplicate from heparinized whole blood samples. The capillary method determined hematocrit in triplicate from heparinized whole blood samples using a microhematocrit reader (CV, 1.1%; Thermo Fisher Scientific, Waltham, MA). Blood hemoglobin and hematocrit values were used to estimate changes in plasma volume relative to baseline to correct plasma variables. 18 Heparin and K3EDTA whole blood samples were centrifuged at 4000 rpm for 10 min. Plasma was aliquoted into Eppendorf tubes and frozen at −80°C until analysis, except for two 50-µL samples that were used to determine plasma osmolality in duplicate (CV, 0.3%) by freeze-point osmometry (Osmomat 030; Gonotec, Berlin, Germany). Circulating plasma concentrations of intestinal fatty acid binding protein (HK406, Hycult Biotech, Uden, Netherlands), soluble CD14 (HK320, Hycult Biotech, Uden, Netherlands), and C-reactive protein (KHA0031, Invitrogen, Thermo Fisher Scientific) were determined by enzyme-linked immunosorbent assay (CV, 3.2, 3.9, and 2.5%, respectively). Variables were analyzed in duplicate as per manufacturer's instructions, with standards and controls on each plate and each participant assayed on the same plate. Due to limited postrace sample collection, only plasma intestinal fatty acid binding protein was analyzed for Youth 2. Fecal bacterial profiling and sequencing for phylum, family, and genus amplicon sequence variants (AVS) was conducted, as described previously.19,20 For AVS, only bacterial groups with a relative abundance ≥0.5% were included to avoid the risk of artifact values in data resulting from sample-handling contamination. Bacterial calculations of n=5 phylum, n=18 family, and n=30 genus AVS were detected for relative abundance and alpha-diversity determination (ie, Shannon Equitability Index). Data in the figures are presented as full values for individual responses. Individual data in tables are presented as mean and/or mean ± SD. Individual raw values in text were compared against reference ranges previously established as suggestive of a change of clinical relevance. 12
Results
Diet and Fluid Practices
Dietary intake analysis for 48 h before the race and planned race and actual race intake is presented in Table 2. Youth competitors did not meet recommended carbohydrate intake for ultra-endurance sport in the lead-up to (2.8–5.1 vs recommended 10-12 g·kg–1·d–1) 21 and during the race (24–26 vs recommended 30–90 g·h–1). 9
Dietary intake analysis 48 h before the race and planned and actual race intake in preparation and during a single-stage ultramarathon (≥50 km).
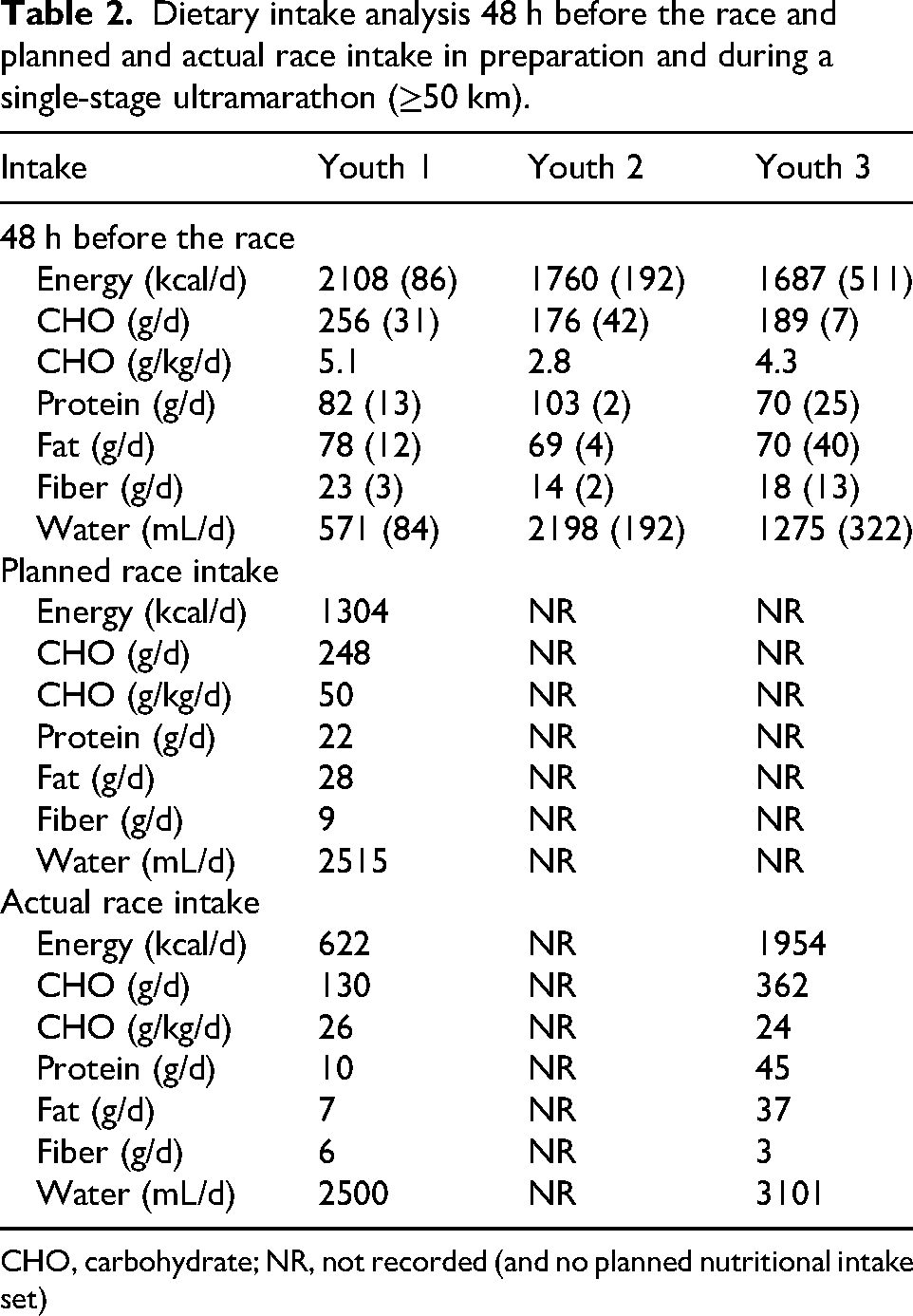
CHO, carbohydrate; NR, not recorded (and no planned nutritional intake set)
Hydration Markers
Body mass and fluid changes for competitors are presented in Table 3. In general, the change in body mass from before to after the race was low (mean: −0.3 kg). Based on plasma osmolality, youth competitors appeared adequately hydrated before the race. After the race, plasma osmolality was determined in only 1 competitor, who appeared to maintain euhydration.
Competitor body mass and fluid changes in response to a single-stage ultramarathon (≥50 km).
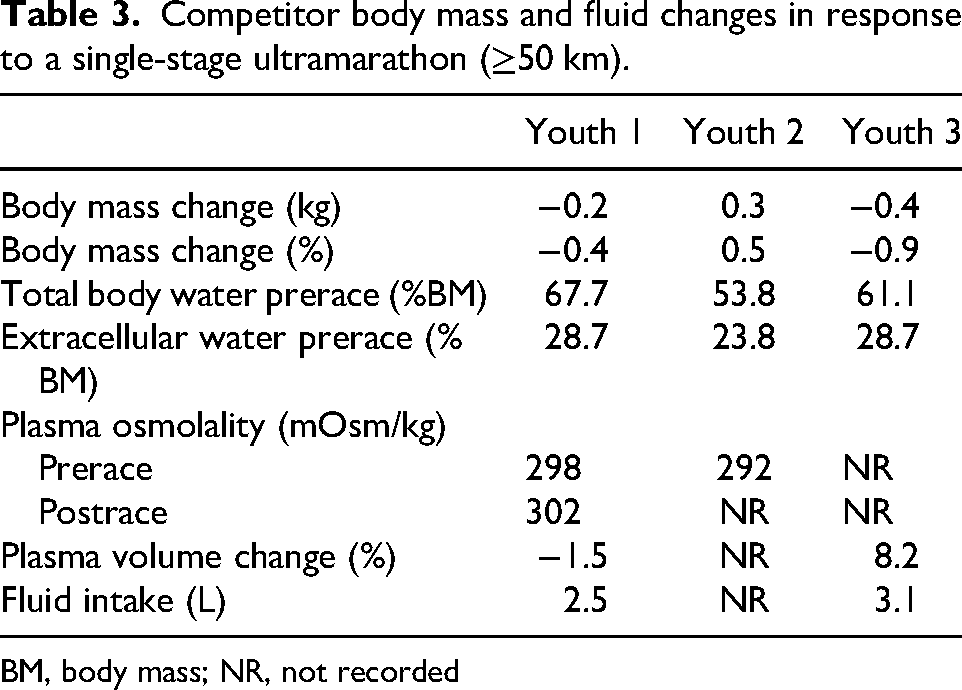
BM, body mass; NR, not recorded
GI Integrity
The change in pre- to postrace plasma intestinal fatty acid binding protein concentrations was minimal (≤660 pg·mL–1) in all competitors. Plasma soluble CD14 concentrations increased substantially from before to after the race in Youth 3 but not in Youth 1 (Figure 1B). Plasma soluble CD14 was not analyzed for Youth 2 due to limited postrace sample volume (Figure 2).
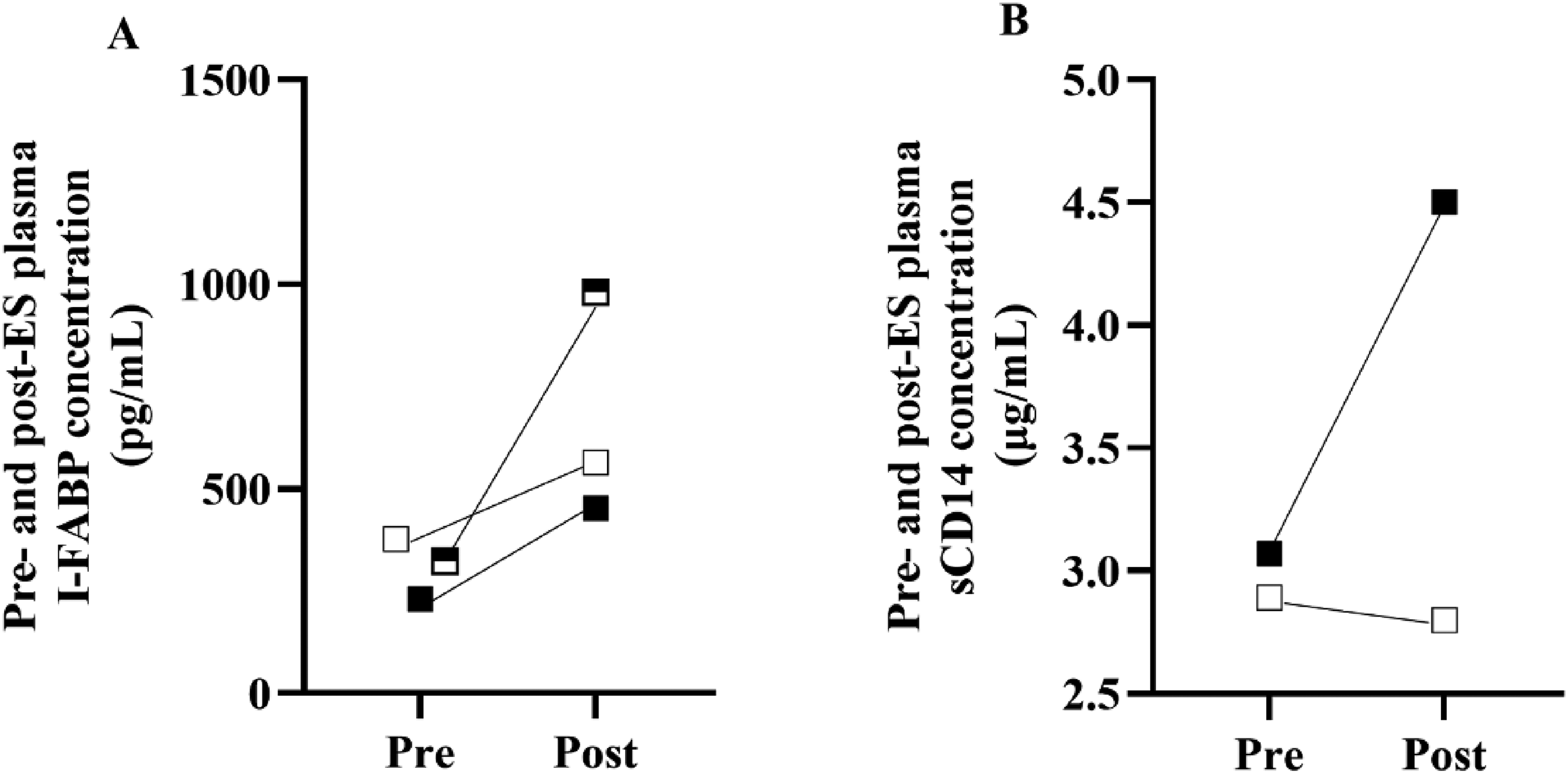
Individual pre- to postrace responses in plasma (A) intestinal fatty acid binding protein (n=3) and (B) soluble CD14 (n=2) concentrations in response to a single-stage ultramarathon (≥50 km) in Youth 1 (white square), Youth 2 (half square), and Youth 3 (black square).
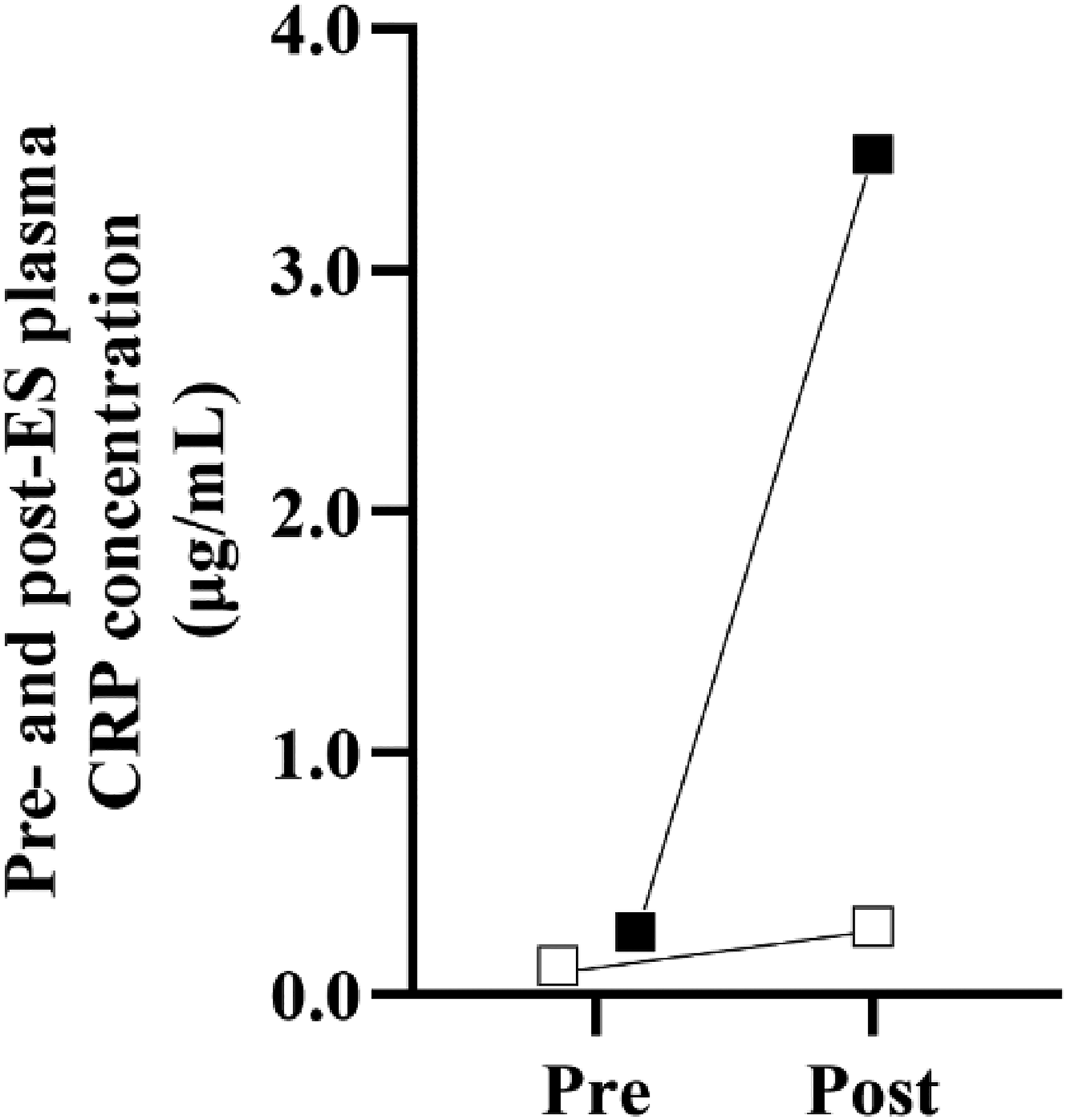
Individual pre- to postrace responses in plasma C-Reactive protein (CRP) concentrations (n=2) in response to a single-stage ultramarathon (≥50 km) in Youth 1 (white square) and Youth 3 (black square).
Immune Response
Leukocyte counts increased before and after the race in all competitors, with Youth 1 and Youth 2 counts increasing beyond the normative range (4.8–10.8 × 109/L, children) (Figure 3A). Neutrophilia occurring beyond the normative range (3.0–7.0 × 109/L, children) was observed in all competitors except Youth 3 (Figure 3B). Lymphopenia was observed in Youth 1 (Figure 3C), with an increase in lymphocytes observed in all other competitors, but not beyond the normative range (1.5–4.0 × 109/L). An increase in neutrophil-to-lymphocyte ratio was observed in all competitors from before to after the race (Figure 3D).
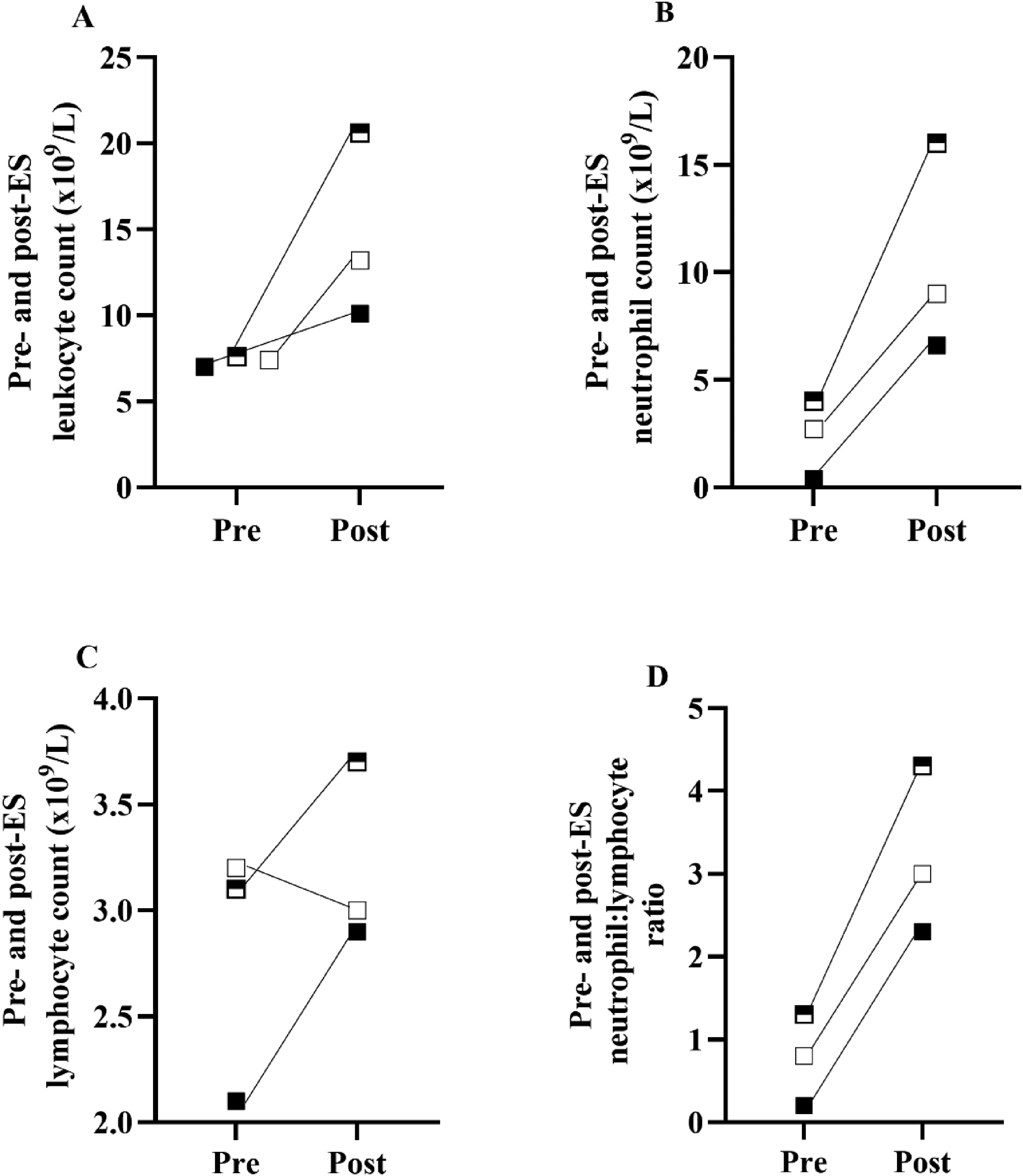
Individual pre- to postrace response in (A) leukocyte, (B) neutrophil, and (C) lymphocyte counts and (D) the neutrophil-lymphocyte ratio (n=3) in response to a single-stage ultramarathon (≥50 km) in Youth 1 (white square), Youth 2 (half square), and Youth 3 (black square).
Postrace plasma C-reactive protein concentrations increased slightly above the normative range (3.0 micrograms·mL–1) for Youth 3 (3.48 micrograms·mL–1) but not Youth 1 (0.28 micrograms·mL–1) (Figure 2). Plasma C-reative protein was not analyzed for Youth 2 due to limited postrace sample volume.
Fecal Microbial Taxa
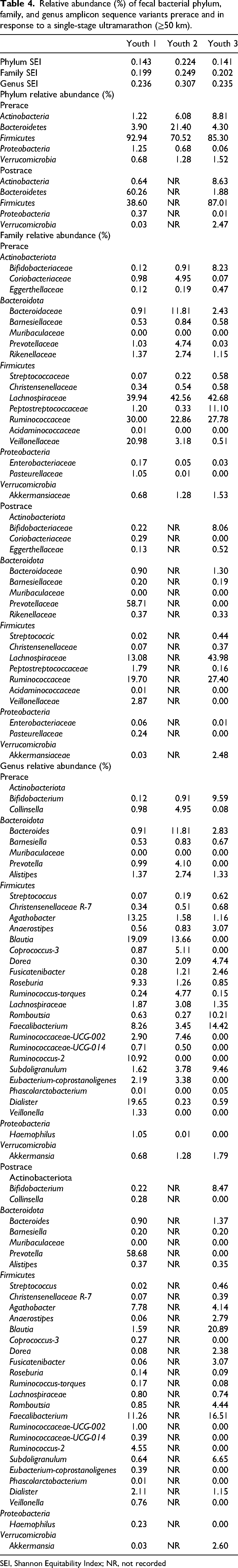
Bacterial taxa alpha-diversity (Shannon Equitability Index) and predominant fecal phylum, family, and genus bacterial groups are presented in Table 4.
Relative abundance (%) of fecal bacterial phylum, family, and genus amplicon sequence variants prerace and in response to a single-stage ultramarathon (≥50 km).
SEI, Shannon Equitability Index; NR, not recorded
GI Symptoms
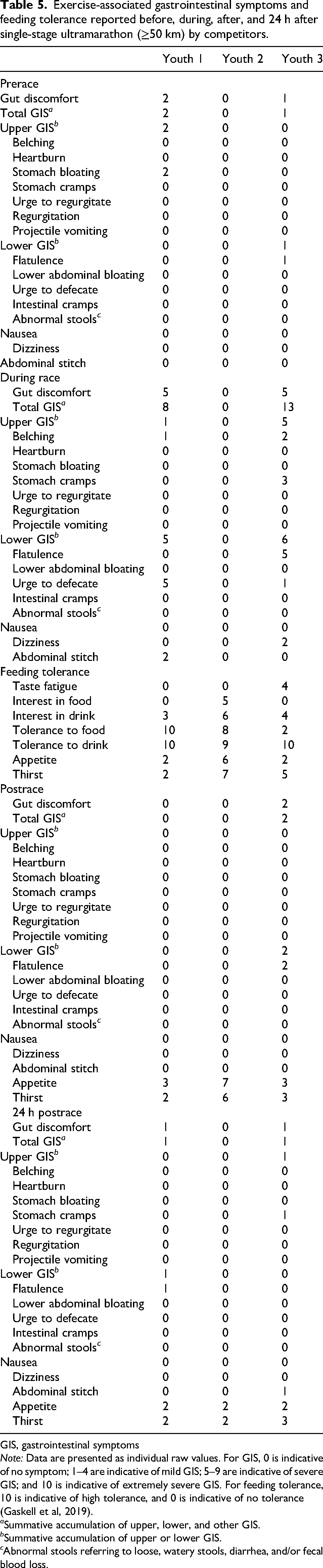
GIS before, during, and immediately after and 24 h after the race are presented in Table 5. GIS reported before and immediately after and 24 h after the race were mild (<5). The most predominant GIS reported by youth competitors during the race were belching and the urge to defecate. Youth competitors had a high tolerance for fluid throughout the race, with tolerance to food also high in Youth 1 and Youth 2 but not in Youth 3. Low appetite and thirst were reported by two youth competitors but not Youth 2.
Exercise-associated gastrointestinal symptoms and feeding tolerance reported before, during, after, and 24 h after single-stage ultramarathon (≥50 km) by competitors.
GIS, gastrointestinal symptoms
Note: Data are presented as individual raw values. For GIS, 0 is indicative of no symptom; 1–4 are indicative of mild GIS; 5–9 are indicative of severe GIS; and 10 is indicative of extremely severe GIS. For feeding tolerance, 10 is indicative of high tolerance, and 0 is indicative of no tolerance (Gaskell et al, 2019).
a Summative accumulation of upper, lower, and other GIS.
b Summative accumulation of upper or lower GIS.
c Abnormal stools referring to loose, watery stools, diarrhea, and/or fecal blood loss.
Discussion
This case series observed the impact of single-stage ultramarathon participation on markers of EIGS and Ex-GIS in 3 adolescents. Youth failed to meet carbohydrate intake guidelines in the lead-up to and during the race. Minimal disturbances to intestinal epithelial integrity (plasma intestinal fatty acid binding protein) and variable systemic endotoxemia (plasma soluble CD14) and inflammatory responses (plasma C-reative protein and leukocyte counts) were observed in response to the ultramarathon, which may be associated with differing diet and hydration practices. The youths experienced minimal GIS, indicative of tolerance to race nutrition and event characteristics. Observations suggest that individually tailored management of adolescent competitors may be required to attenuate the incidence and severity of EIGS.
Nutrition and Hydration
Ultramarathon nutrition guidelines emphasis adequate carbohydrate and fluid intake before and during competition, linked to performance and mitigation of EIGS-related GI disturbances. In this case series, youth competitors’ carbohydrate consumption ranged from 2.8 to 5.1 g·kg–1·d–1 48 h before the race, falling below the recommended 10 to 12 g·kg–1·d–1. 21 Observations are comparable to those of previous research, whereby a carbohydrate intake of ∼3.5 to 7.3 g·kg–1·d–1 has been reported in adult endurance athletes.22,23 Youth competitors failed to meet the lower end of carbohydrate recommendations during competition (30–90 g·h–1), 9 consuming 24 to 26 g·h–1. This is not dissimilar to amounts previously reported to be consumed by nonelite adult ultramarathon competitors, albeit toward the lower end (between 20 and 40 g·h–1).9,23,24 Past research has highlighted the notion that discrepancies in nutrition recommendations are linked to poor sports-specific nutrition knowledge among athletes and underreporting of 10 to 20% associated with self-reported food diaries and individual carbohydrate tolerability during exercise.9,23,25 From a practical translation perspective, it would be beneficial to explore the current sports-specific nutrition knowledge of youth ultra-endurance athletes.
Youth competitors presented adequately hydrated at baseline. Minimal before and after race body mass losses were observed in Youth 1 and Youth 3. Current adult recommendations stipulate to drink ab libitum to thirst during events and to avoid a fluid deficit of >2 to 3% of body mass.9,21 Low body mass loss in youth competitors (−0.4 to +0.5%) may be explained by appropriate fluid replacement during races. Another potential explanation may be lower sweat rates associated with slower race paces and/or a greater reliance on convective heat loss rather than evaporative cooling mechanisms related to pubertal age,26,27 with observations from a recent study revealing lower total sweat losses (1.5 L) in youth vs adult athletes (2.6 L) following 3-h treadmill running at 60% V˙O2max (ambient temperature 22.9°C) despite similar fluid volume consumption. 13 Youth 2 gained 0.3 kg from before to after the race, possibly related to slower completion time (ie, opportunities to drink and higher tolerability) and lower sweat rate. In addition, this was the first ultramarathon event for Youth 2, unlike the other competitors with previous experience, potentially contributing to greater knowledge of fluid guidelines in Youth 1 and Youth 3 (ie, hyperhydration risks). Although nutrition guidelines for ultra-endurance athletes are based on adult research, carbohydrate use and tolerability during prolonged running do not appear to be substantially different in adolescents. 13 Therefore, current carbohydrate recommendations should be appropriate to support adolescents in ultramarathons. Considering slower race times, age-related thermoregulation differences, and the likelihood of less experience in youth competitors, special considerations (ie, planned fluid intake) may be required to reduce the risk of fluid overconsumption.
GI Integrity and Immune Response
Despite modest disruptions to intestinal epithelial integrity (change in intestinal fatty acid binding protein of ≤1301 pg·mL–1) in all youth competitors, 12 changes in the systemic endotoxemia marker soluble CD14 were variable, with a clinically significant change (≥0.78 micrograms·mL–1) observed in Youth 3 but not Youth 1. 12 The mild disruptions to epithelial integrity, despite the duration of exercise being well established to perturb intestinal epithelial cells, may be explained by carbohydrate ingestion during the event, which is recognized to mitigate increases in intestinal fatty acid binding protein.14,15 Interestingly, although carbohydrate intake during the race was low (24–26 g·h–1) and lower than provided in previous studies (∼40 g·h–1) reporting attenuated disturbances,13,14 it may have been sufficient to mitigate GI integrity disruptions in the youth competitors. Further studies, adopting best-practice guidelines (ie, diet and hydration control), are required to confirm these findings. 11
Despite competitors being healthy adolescents and endurance trained, substantial individual variation in fecal bacterial profiles at phylum, family, and genus levels were observed, consistent with previous adult-focused exercise gastroenterology research.12,19,20 Fecal bacterial profiles of endurance-trained athletes appear dynamic and sensitive to various factors (eg, lifestyle, diet, training load, and seasonal variations). 19 Considering the numerous influential factors and the observational nature of this case series, lacking rigorous control of competitors, the impact of bacterial profiles on GI integrity markers is unclear.
All youth competitors, except Youth 3, exhibited clinically significant neutrophilia (≥9.0 × 109/L) and leukocytosis (≥13.2 × 109/L) after the race. 12 Youth 2 experienced a substantial increase in neutrophil counts (pre- to postrace change, 12.0 × 109/L) and associated leukocytosis (pre- to postrace change, 13.0 × 109/L), possibly related to ≥17-h race duration and a pre- to postrace body mass increase, which may reflect hyperhydration. Unfortunately, insights into race food and fluid consumption and systemic endotoxemia response (ie, soluble CD14), known to influence the inflammatory response, were unavailable for Youth 2, making it challenging to investigate potential associations. However, to attenuate SIRs, it may be beneficial to establish a fluid plan before the race to minimize body mass gain for inexperienced youth competitors, who are expected to race at lower intensities. 27 The absence of lymphopenia in 2 youth competitors likely represents sample timing variability after the race, with acute lymphopenia typically occurring >30 min after ES. 11 Despite efforts to standardize sample timing, it is challenging to control in field-based settings. Neutrophil-to-lymphocyte ratio changes in youth were comparable with changes observed in adults in previous studies.13,28
Plasma C-reactive protein concentrations increased from before to after the race in Youth 3 but not in Youth 1. Inconsistencies may be due to race distances (50 vs 80 km, respectively), with no to modest increases in plasma C-reactive protein previously observed in response to marathon distances. 29 Hepatic production of C-reactive protein also commences 6 to 8 h following inflammatory onset, peaking after ∼36 to –48 h. 30 The 50-km duration and immediate postrace sampling may explain minimal changes in Youth 1. Considering that Youth 3 completed the ultramarathon in 16.3 h, a more substantial plasma C-reactive protein increase is unsurprising. The low carbohydrate intake of Youth 3 (24 g·h–1) also may have contributed to the more substantial increase in C-reactive protein, with higher intake reported to attenuate increases in plasma C-reactive protein following ES. 31 Nevertheless, observed increases in plasma C-reactive protein were not dissimilar to previous ultramarathon research performed in adults and did not reach clinical significance (ie, severe trauma, bacterial infection, or surgery). 32 Inconsistencies in SIR markers in youth competitors is likely reflective of the use of nonspecific immune markers in addition to sample timing and limited study control (ie, diet and race duration and intensity). Further research analyzing SIR cytokines is required to determine youth athletes’ SIR to ultramarathon running.
GIS and Feeding Tolerance
Ex-GIS during the race were modest in youth competitors, with tolerance to food and drink generally high. The lower tolerance and higher taste fatigue in Youth 3 may be associated with a slightly higher total GIS and the consumption of mostly sweet-based foods and fluids (ie, lollies and sweetened beverages) compared with other competitors. Modest Ex-GIS reported by youth competitors are not comparable with the 60 to 96% prevalence of severe symptoms reported by adult ultramarathon competitors.3,9 In a previous laboratory-based study, adults also reported increased upper GIS compared with adolescent athletes during prolonged ES. 13 Disparities between age groups may be associated with 1) generally greater carbohydrate intake by adults, which is positively correlated with increased Ex-GIS,5,16 2) heightened sensitivity and/or awareness of Ex-GIS in adults due to experience in ultramarathon running, 13 and 3) minimal hypohydration in youth competitors, which is recognized to mitigate severity of Ex-GIS. 32 Observations demonstrate that GIS was not an issue for our youth competitors, but further field-based research is warranted exploring Ex-GIS in adolescents consuming carbohydrates at recommended levels (ie, ≥30 g·h–1). Youth 3 may benefit from following current nutrition guidelines recommending a variety of food choices of different forms (ie, fluids, gels, and bars) and taste profiles (ie, savory and sweet) to improve feeding tolerance. 9
Limitations
Although this case series provides novel preliminary insights into the impact of ultramarathons on the GI tract of adolescent athletes, limitations associated with the nature of field-based research, biomarker selection, and sample size impact the generalizability of observed results. For recruitment and adherence purposes, minimizing participant burden was necessary, resulting in adjustments to sample timing and data-collection techniques. For example, pre-ES measures collected the day before rather than the morning of the race do not capture overnight events that may impact resting biomarker levels (eg, sleep and diet), potentially affecting pre- to postrace observed changes. In addition, the reliance on retrospective data collection (eg, dietary intake and GIS) invites erroneous reporting resulting from recall bias. Although processes were followed to standardize sample timing and collection and dietary recall techniques between each competitor for comparison purposes, it is challenging to control for all potential confounders.
Participant burden and ease of transport also were considered in the selection of EIGS biomarkers. Leukocyte counts generally have low reliability in exercise gastroenterology research, 12 but immediately postrace sample collection was required to avoid interference of recovery nutrition restoring GI and immune homeostasis, deeming more reliable inflammatory measures that peak >1 h after ES (ie, cytokine interleukin-10) inappropriate for use. 11 To negate reliability issues, interpretation of leukocyte counts is in support of other inflammatory instigators (eg, soluble CD14) and markers (eg, C-reactive protein). Plasma C-reactive protein was selected as a broad-spectrum inflammatory marker, with use in previous field studies investigating immune response to single-, multistage and 24-h ultramarathons allowing for some comparative interpretation.29,32 However, a number of factors that can influence C-reactive protein response were unable to be controlled for (eg, race duration and intensity and diet); therefore, caution is again required when interpreting competitor results. Further research is needed using more reliable immune biomarkers to determine the response of youth ultramarathon competitors.
Lastly, considering the small sample size, additional research is warranted with a focus on ultramarathon events known to attract larger numbers of youth athletes (eg, European ultramarathons) 7 to establish whether the EIGS response to ultramarathon participation is comparable with that of adult competitors and the 3 youth competitors in this case series.
Conclusion
This case series offers preliminary insights into the impact of adolescent age on GI status investigated with respect to real-life ultramarathon conditions (eg, duration and environmental conditions) known to exacerbate EIGS. In addition, to our knowledge, this is the first study to report race nutrition and hydration practices of adolescent ultramarathon runners. Observations from the 3 youth competitors in this case series reveal large intra- and interindividual variability in GI integrity, systemic inflammation, and Ex-GIS responses. Observations also highlight an interplay of extrinsic factors (ie, duration, diet, and fluid intake) in the exacerbation of EIGS. Similar individual variations in EIGS perturbations are well established in adult endurance athletes, resulting in recommendations that hydration and nutrition strategies aimed at supporting both performance and GI status be individually tailored. Observations from this case series suggest that a similar approach may need to be adopted for youth ultramarathon athletes to help mitigate EIGS-associated disturbances and promote safe participation in events, although further studies using larger sample sizes are needed to confirm findings.
Footnotes
Acknowledgments
The authors thank the competitors for participating. The authors acknowledge and thank Peter Peeling and the University of Western Australia for allowing the processing and analysis of participant samples in the School of Human Sciences laboratory facilities.
Ethical Considerations
Ethical approval for this case series was provided by the Monash University Human Research Ethics Committee (Approval No. 29429) on June 14, 2021.
Consent to Participate
All participants provided written informed consent prior to participating.
Funding
This study was supported by the Ultra Sports Science Foundation (Research Service Agreement).
