Abstract
Objective
Neonatal necrotizing enterocolitis (NEC) is a serious intestinal inflammatory disease. We investigated intestinal fatty acid binding protein (I-FABP), I-FABP mRNA, and interleukin-6 (IL-6) as potential diagnostic biomarkers in NEC.
Methods
Forty mice were subjected to hypoxic-ischemic intestinal injury, and then serum I-FABP protein and mRNA levels were quantified. Ileal tissue pathological scores were determined by hematoxylin and eosin staining. I-FABP expression levels and translocation in these tissues were detected using western blotting and immunofluorescence, respectively. Samples from 30 human neonates with NEC and 30 healthy neonates had serum I-FABP protein/mRNA and IL-6 levels measured.
Results
The mouse ileal tissue pathological score and I-FABP levels, as well as serum I-FABP and I-FABP mRNA levels, were significantly higher in the model group than in the control group. Serum I-FABP, I-FABP mRNA, and IL-6 levels were significantly higher in human neonates with NEC than in the healthy group. Logistic regression and receiver operating curve analyses revealed that I-FABP protein/mRNA and IL-6 levels could be diagnostic biomarkers for NEC.
Conclusions
I-FABP protein/mRNA and IL-6 levels are useful biomarkers of intestinal ischemic injury in neonates with NEC. The combined detection of I-FABP protein/mRNA and IL-6 is recommended rather than using a single biomarker.
Keywords
Introduction
Neonatal necrotizing enterocolitis (NEC) is the main cause of neonatal deaths related to gastrointestinal diseases. According to the National Center for Health Statistics, the case fatality rate was 9.4 per 100,000 live births (0.094‰) in 2019, which was the tenth highest cause of neonatal deaths. 1 The incidence of NEC is higher in premature neonates, ranging from 2.53% to 17.08% in very low birth weight infants.2–4 However, NEC can also occur in full-term neonates (10% to 15%). 5 NEC can start slowly and implicitly, with some neonates exhibiting early symptoms such as decreased appetite and feeding intolerance. The early prediction of NEC is extremely important for neonatologists to avoid disease progression to requiring surgery. Therefore, identifying noninvasive and reliable biomarkers is necessary to predict neonatal NEC before the clinical signs appear.
Research on NEC biomarkers, such as interleukin-6 (IL-6), is ongoing. The routine examination of IL-6 levels is highly recommended by clinical guidelines for the diagnosis and treatment of neonatal necrotizing enterocolitis 6 because IL-6 upregulation usually suggests an early inflammatory response. As an inflammatory mediator, IL-6 is involved in multiple cytokine responses, has a wide range of biological effects, and plays vital roles in regulating inflammatory responses and the immune balance. However, other NEC biomarkers, such as fecal biomarkers, have demonstrated mixed results in neonates.6–9
As a specific indicator of intestinal injury, intestinal fatty acid binding protein (I-FABP) has potential clinical application as an objective basis for intestinal disease diagnosis. Several studies have shown a correlation between blood I-FABP levels and NEC,10,11 while other reports have examined FABP4 and FABP5 mRNA expression levels in NEC tissues. Egozi et al. 12 demonstrated differential expression of FABP4 and FABP5 between patients with NEC and neonates. However, no gene expression profiling studies in NEC tissues have indicated I-FABP as a differentially expressed gene.
To elucidate the relevant pathological mechanism, a mouse model of hypoxic-ischemic intestinal injury was generated following the animal model establishment method considered closest to neonatal patients with NEC. 13 We aimed to analyze the roles of I-FABP protein/mRNA, IL-6, and intestinal mucosal barrier disruption in the pathogenesis of NEC. Consistency between the NEC mouse model and human data was verified through macroscopic observation of specimens and microscopic pathological analysis and comparison of laboratory indicators. The predictive value of I-FABP, I-FABP mRNA, and IL-6 for diagnosing NEC was analyzed using receiver operating characteristic (ROC) curves to provide new ideas and perspectives for the early prediction of neonatal NEC to reduce its adverse outcomes.
Materials and methods
Study design and participants
This prospective cohort study enrolled neonates admitted to Fuzhou First General Hospital Affiliated with Fujian Medical University between April 2022 and July 2023. Neonates with NEC were included in the experimental group, while neonates with no history of hypoxia or cardiovascular disease were selected as the control group.
The study was approved by the Ethics Committee of the Faculty of Medicine, Fuzhou First General Hospital, Affiliated with Fujian Medical University, Fuzhou, China (no. 202104013; approval date: 12 April 2021) and the Experimental Animal Ethics Committee of Fujian Medical University, Fuzhou, China (no. IACUC FJMU2023-Y-0438; approval date: 3 March 2023). Verbal informed consent was obtained from the parents of all participants.
Patient selection criteria
The inclusion criteria for the experimental group were as follows: 1) neonatal patients with NEC Bell stage I 14 and a history of hypoxia at birth; 2) clinical symptoms, such as abdominal distension, vomiting, hematochezia, stool occult blood, unstable temperature, apnea, and declining heart rate; and 3) abnormal intestinal distension and significant intestinal wall thickening on abdominal X-ray imaging. Neonates with no history of hypoxia or cardiovascular disease were selected as the control group. Any patients with congenital organ malformations, congenital digestive tract malformations, severe infections, or severe complications were excluded.
Detection methods
In the experimental group, 2 mL of peripheral serum was collected within 24 hours of NEC onset using a high-speed freezing centrifuge (model H1850R; XiangYi, Hunan, China). I-FABP mRNA was quantified using quantitative real-time polymerase chain reaction (qPCR). Serum I-FABP, IL-6, and I-FABP levels were measured using enzyme-linked immunosorbent assays (ELISAs). To perform the ELISAs, different concentrations of standards were added to the microplate (DNM-9602; Beijing Pulang New Technology Co., Ltd., Beijing, China). After the reaction, the liquid in the microplate was washed away, and then the biotin anti-human I-FABP or IL-6 antibody working solution, avidin-peroxidase complex working solution, 3, 3′,5,5′-tetramethylbenzidine color development solution, and termination solution were added sequentially. During the experiment, the blue color turned to yellow, after which the optical density was measured at 450 nm (Nano-100; Allsheng, Hangzhou, China). The corresponding concentration of each sample was determined using a standard curve.
To perform qPCR analysis, total RNA was extracted from the serum samples and reverse transcribed to synthesize cDNA. Each cDNA reaction was diluted five-fold for qPCR. The qPCR reactions were performed using the following cycling conditions: 95°C pre-denaturation for 5 minutes, followed by 40 cycles of 95°C denaturation for 10 s, 60°C annealing for 30 s, and 72°C extension for 30 s (ABI 7500; Applied Biosystems, Foster City, CA, USA). The data were quantitatively analyzed using the ΔΔCt method with β-actin as a reference gene. All operations were performed by the same researcher in strict adherence to the manufacturer’s instructions.
Animal model establishment
One-week-old specific pathogen-free (SPF) BALB/c mice weighing 5.0 to 6.5 g were provided by SIPEIFU (Beijing) Biotechnology Co., Ltd. (Beijing, China; Certificate No. SCXK (Jing) 2019-0010). The animals were raised in the SPF animal room of SIPEIFU with the following conditions: a 12-hour light–dark cycle, 23°C ± 2°C temperature, and 50% ± 10% relative humidity. Six littermates (eight mice in each litter) were fed artificial milk for a week to adapt to the environment, after which the pups in the NEC groups were subjected to hypoxia for 3 days. The mice were randomly divided into a healthy control group and model group (20 mice in each group). Model mice were subjected to hypoxia twice daily, maintaining an oxygen concentration of 5% using an oxygen concentration monitor and nitrogen bottles. The timing began when the oxygen concentration in the hypoxic bin decreased to 5%. After 90 s, the nitrogen threshold was closed and the hypoxia box was opened, then the mice were removed and placed in a 4°C cold closet for 10 minutes of sustained cold stimulation. Hypoxia and cold stimulation were performed twice daily (at 8:00 AM and 8:00 PM). The above treatments were repeated for 3 days, after which the mice were sacrificed while fasting by cutting their head 24 hours after the last hypoxia and cold stimulation. Serum samples were collected from the mice and then their abdominal cavity was opened. The lower end of the duodenum to the upper rectum was removed and divided into two halves, the jejunum and ileum, and stored at −80°C and in 4% tissue cell fixative (P1110; Solarbio Technology Co., Ltd., Beijing, China) at 4°C, respectively. The distal ileum 4 cm proximal to the ileocecal valve was selected, leaving 3-cm-long specimens for ileum histological evaluation. The remaining tissue was flash frozen in liquid nitrogen for I-FABP detection.
All procedures and protocols for the use and care of animals were performed in accordance with the guidelines of the Ethics Committee of Fuzhou First General Hospital Affiliated with Fujian Medical University, Fuzhou, China.
Ileum histopathological score
A 3-cm-long section of the distal ileum was fixed in 70% ethanol and embedded in paraffin. Hematoxylin and eosin (H&E) was used to stain the nuclei blue and stain the cytoplasm red. The ileal histopathological score was evaluated within 1 to 5 minutes of staining and scored on a scale of 0 to 4 by two independent blinded evaluators according to the NEC scoring system.15–17 Histological scores of 2 or more were classified as NEC (Table 1).
Histopathological scores of necrotizing enterocolitis in the ileum of neonatal mice.

Western blot analysis
I-FABP protein expression levels in the ileal tissues of neonatal mice were detected by western blot analysis. Samples were added to protein lysate containing phenylmethylsulfonyl fluoride (Beyotime Biotech, Inc., Shanghai, China). The total protein concentration of the samples was determined using a BCA protein assay kit (Solarbio Technology Co., Ltd., Beijing, China). The absorbance was measured by a microplate reader (Pulang New Technology Co., Ltd., Beijing, China), and the protein concentration was calculated for each sample. Subsequently, sodium dodecyl sulfate-polyacrylamide gel electrophoresis was performed, and the samples were placed into an electrophoresis tank. The target band was cut after the electrophoresis. A polyvinylidene fluoride membrane (Millipore) was prepared and the protein was transprinted onto the membrane. The transferred membrane was placed in 5% bovine serum albumin blocking solution (Solarbio Technology Co., Ltd.) and blocked at room temperature for 2 hours. After blocking, the primary (β-actin, 1:1000; Abcam, Cambridge, United Kingdom and I-FABP, 1:2000; Proteintech Group, Inc., Wuhan, China) and secondary (mice antibody l: 5000, model HS201; TransGen Biotech, Inc., Beijing, China) antibodies were placed on the membrane and incubated at room temperature for 2 hours. The membrane was then placed flat on a transparent membrane, and enhanced chemiluminescence reagent (Thermo Fisher Scientific, Waltham, MA, USA) was added and the membrane was placed in the dark for 3 minutes. Band images were observed by luminescence detection using a gel imaging system (Peiqing Technology Co., Ltd., Shanghai, China). The relative target protein levels in the sample were determined using ImageJ software (version 1.4.3.67; National Institutes of Health, Bethesda, MD, USA) to analyze the band gray values.
Immunofluorescence
Sections were placed into an environmentally friendly dewaxing agent and anhydrous ethanol in turn for dewaxing. The sections were subsequently heated in EDTA antigen retrieval buffer (PH 9.0) for antigen repair. After cooling, the slices were shaken in a phosphate-buffered solution (PBS) and washed three times. After drying, the tissue was circled with a chemical pen, bovine serum albumin blocking solution was added, and the mixture was sealed for 30 minutes. The blocking solution was gently shaken off, and the primary antibody (27266-1-AP, 1:400/15073-1-AP, 1:100; Proteintech Group, Inc.) was added to the slices and incubated at 4°C overnight. The next day, the slices were shaken in PBS (pH 7.4) and washed three times. After drying the slices, the secondary antibody (ab6785, 1:1000/ab150076, 1:500; Abcam) was added and incubated at room temperature for 50 minutes. PBS (pH 7.4) was subsequently added and the mixture was washed three times. The mixture was then shaken and slightly dried, and 2-(4-amidinophenyl)-6-indolecarbamidine dihydrochloride (Beyotime Biotech, Inc.) dye solution was added. The mixture was then incubated at room temperature for 10 minutes, placed in PBS (PH 7.4) solution, and washed three times on a shaker (NuoMi; Naomi Medical Technology Co., Ltd., Taizhou, Zhejiang, China). Finally, the plate was sealed with an anti-fluorescence quenching agent (P0128M-2; Beyotime Biotech, Inc.). Sections were observed under a fluorescence microscope (MF53; Micro-shot Technology Co., Ltd., Guangzhou, China) and images were acquired using an imaging system (MF-BGU-LED; Micro-shot Technology Co., Ltd.). Nuclei counterstained with DAPI were colored blue upon excitation with ultraviolet light. Positive I-FABP protein staining with Texas Red was visualized as the corresponding fluorescein-labeled fraction. Images were analyzed using K-Viewer Installer (version 1.7.0.27; KFBIO Co., Ltd., Ningbo, China) to observe I-FABP translocation.
Neonatal mice serum I-FABP mRNA and I-FABP detection
The same ELISA and qPCR experimental procedures were used for neonatal mice serum I-FABP mRNA and I-FABP detection as described above for the human patient samples.
Statistical analysis
Data analysis was performed using IBM SPSS Statistics version 22.0 (IBM Corp., Armonk, NY, USA). For parametric data, the results are presented as numbers, percentages, means, and standard deviations. The Mann–Whitney U test and independent samples’ t-test were used to compare quantitative variables. Independent risk factors were determined using binary logistic regression analysis. ROC curve analysis was used to examine the diagnostic value of I-FABP, I-FABP mRNA, and IL-6 levels in neonatal NEC. A P value less than 0.05 was considered statistically significant.
Results
Mice ileal tissue H&E staining
Light microscopy revealed that the control group samples had a normal intestinal wall structure, intact villi and epithelium, and submucosa and lamina propria without edema or necrosis. The model group samples displayed an abnormal intestinal wall structure, loss of muscular layers, reduced and partial disappearance of villi, prominent edema of the lamina propria and submucosa, and necrosis in certain areas of the mucosa (Figure 1).
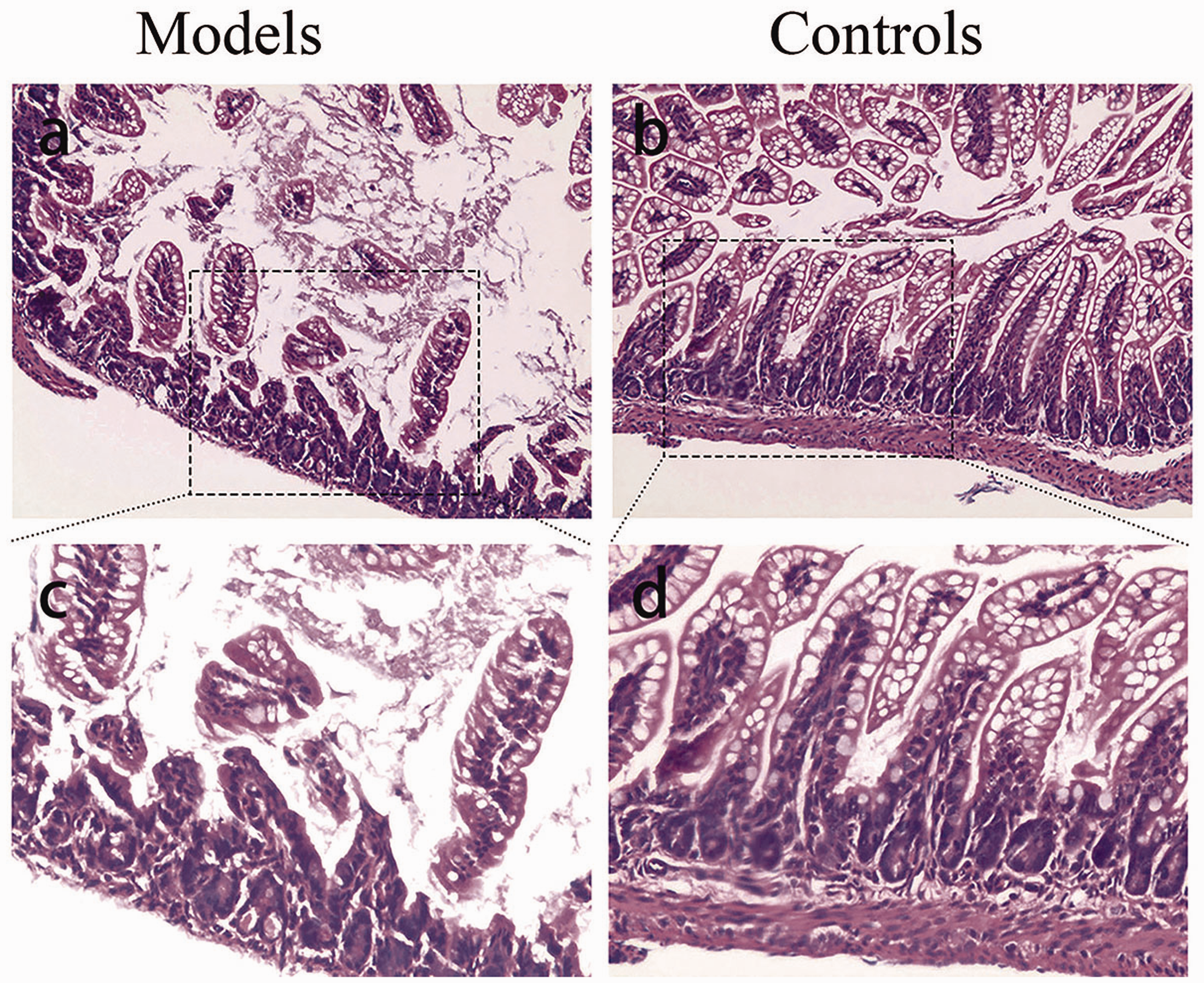
Comparison of neonatal mouse terminal ileum hematoxylin and eosin staining between the two groups. (a, c) Light microscopy revealed that the model group (pathological score: 3) tissues showed an abnormal intestinal wall structure, loss of muscular layers, reduced and partial disappearance of villi, obvious edema of the lamina propria and submucosa, and necrosis of some areas of the mucosa and (b, d) light microscopy revealed that the control group (pathological score: 0) tissues showed a normal intestinal wall structure, intact villi and epithelium, and submucosa and lamina propria without edema or necrosis. The images in panels a and b are 20× magnification; the images in panels c and d are 40× magnification.
Mice ileal tissue histopathology score and I-FABP expression comparisons
The pathological scores were determined using a double-blind scoring method. The pathological score was significantly higher (P < 0.05) in the model group (1.10 ± 1.16) than in the control group (0.30 ± 0.66). Additionally, model group tissues had significantly higher (P < 0.05) relative I-FABP expression levels (1.273 ± 0.376 pg/mL) than control group tissues (0.623 ± 0.266 pg/mL; Table 2 and Figure 2).
Comparisons of the pathological score, tissue I-FABP levels, serum I-FABP levels, and I-FABP mRNA levels between the two groups of neonatal mice.

*Comparisons between the two groups were performed using the independent samples t-test; P < 0.05 was statistically significant.
**Comparisons between the two groups were performed using the Mann–Whitney U test; P < 0.05 was statistically significant.
I-FABP, intestinal fatty acid binding protein.

Representative western blot bands of ileal tissue protein samples from the two neonatal mouse groups. Intestinal fatty acid binding protein (I-FABP) relative expression levels were significantly higher (P < 0.05) in tissues from the model group (1.273 ± 0.376 pg/mL) than in those from the control group (0.623 ± 0.266 pg/mL).
I-FABP translocation in mice ileal tissues
Translocation of I-FABP in the two groups of newborn mice was observed by immunofluorescence. I-FABP levels significantly increased in the cytoplasm and apical membrane of intestinal villus cells. However, they decreased in the nucleus and accumulated in the brush border membrane, central lacteal villi, basement membrane, and small intestine submucosa of model group tissues (Figure 3).
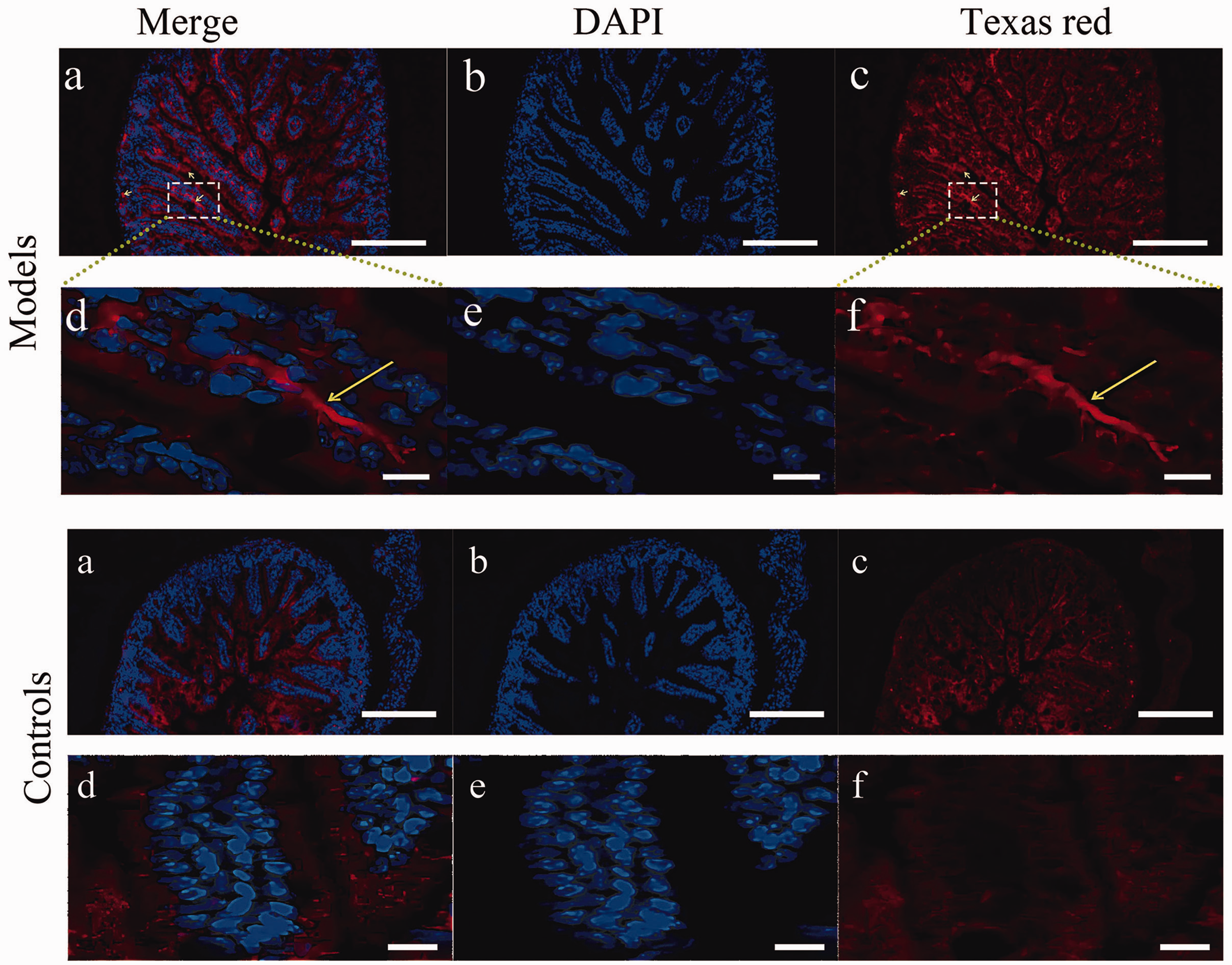
Comparison of intestinal fatty acid binding protein (I-FABP) translocation in ileal tissues from the two groups of neonatal mice. In the model group images, the yellow arrows point to I-FABP aggregation. Compared with the control group tissues, I-FABP expression was significantly increased in the model group tissues in the brush border membrane, central lacteal villi, basement membrane, and small intestine submucosa. The images in panels a, b, and c are 20× magnification, with the scale bar representing 100 μM; the images in panels d, e, and f are 80× magnification, with the scale bar representing 25 μM. DAPI, 2-(4-Amidinophenyl)-6-indolecarbamidine dihydrochloride.
Serum I-FABP and I-FABP mRNA comparisons in mice
Serum I-FABP (3083.332 ± 344.605 pg/mL) and I-FABP mRNA (4.911 ± 4.981) levels were significantly higher in the model group than in the control group (1535.666 ± 267.163 pg/mL and 1.219 ± 0.748, respectively) (P < 0.05; Table 2).
Clinical and demographic data of human patients
Sixty neonates were enrolled in this study, with 30 individuals included in the experimental and control groups. The neonates with NEC in the experimental group included 16 boys and 14 girls. Fifteen of these were delivered vaginally, while the other 15 were delivered by cesarean section. Their mean birth weight was 2861 ± 357 g, with a mean gestational age of 36.9 ± 2.8 weeks. The healthy neonates in the control group included 15 male and 15 female individuals. Thirteen of these were delivered vaginally, while the other 17 were delivered by cesarean section. Their mean birth weight was 3095 ± 683 g, with a mean gestational age of 38.1 ± 1.9 weeks. No significant differences in sex, delivery mode, birth weight, or gestational age were observed between the two groups.
Human patient serum I-FABP, I-FABP mRNA, and IL-6 comparisons
Significantly higher serum I-FABP (370.306 ± 264.621 pg/mL), I-FABP mRNA (5.104 ± 7.333), and IL-6 (319.277 ± 971.174 pg/mL) levels were observed in the experimental group than in the control group (232.083 ± 210.669 pg/mL, 3.556 ± 8.939, 15.327 ± 28.355 pg/mL, respectively; P < 0.05; Table 3, Figure 4).
Comparisons of serum I-FABP levels, I-FABP mRNA levels, and IL-6 levels between the two groups of neonates.

*Comparisons between the two groups were performed using the Mann–Whitney U test; P < 0.05 was statistically significant.
I-FABP, intestinal fatty acid binding protein; IL-6, interleukin-6.
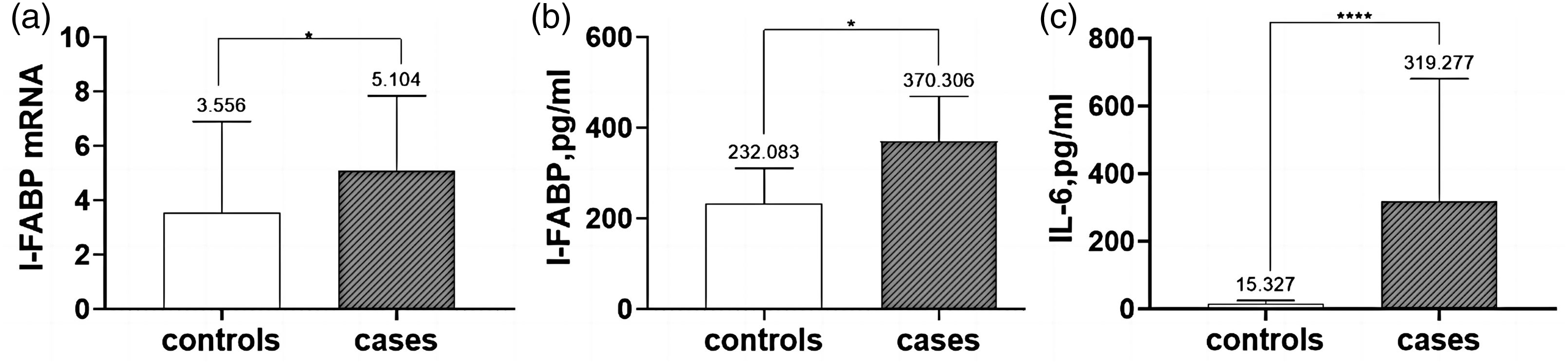
Serum intestinal fatty acid binding protein (I-FABP), I-FABP mRNA, and interleukin-6 (IL-6) levels in human newborn samples. Normality tests were performed first using GraphPad software. (a) The Shapiro–Wilk test results showed that neither data set was normally distributed. Nonparametric tests (Mann–Whitney U test) were therefore used for statistical analysis. The median value was 0.255 for the control group and 1.598 for the experimental group. We selected the mean value with the 95% confidence interval (CI) for plotting. I-FABP mRNA levels were significantly higher (P < 0.05) in the experimental group (5.104 ± 7.333) than in the control group (3.556 ± 8.939). (b) The Shapiro–Wilk test results showed that the control group data were normally distributed, while the experimental group data were not normally distributed. Nonparametric tests (Mann–Whitney U test) were used for statistical analysis. The median value was 208.3 for the control group and 297.7 for the experimental group. I-FABP levels were significantly higher (P < 0.05) in the experimental group (370.306 ± 264.621 pg/mL) than in the control group (232.083 ± 210.669 pg/mL) and (c) the Shapiro–Wilk test results showed that neither data set was normally distributed. Nonparametric tests (Mann–Whitney U test) were used for statistical analysis. The median value was 1.5 for the control group and 26.3 for the experimental group. We selected the mean value with the 95% CI for plotting. IL-6 levels were significantly higher (P < 0.05) in the experimental group (319.277 ± 971.174 pg/mL) than in the control group (15.327 ± 28.355 pg/mL).
Logistic regression analysis
Logistic binary regression analysis was then performed with NEC (no = 0, yes = 1) as the dependent variable and I-FABP, I-FABP mRNA, and IL-6 (continuous variables) as independent variables. This analysis revealed that the serum I-FABP level was an independent risk factor for patients with neonatal NEC (P = 0.043, Table 4). The overall prediction accuracy of this model was 70%. Hosmer’s test showed that the model and observed values were in good agreement (P = 0.422).
Logistic binary regression analysis of serum I-FABP levels, I-FABP mRNA levels, and IL-6 levels in NEC samples

NEC, necrotizing enterocolitis; B, regression coefficient; SE, standard error; Wald, Wals χ2-value; OR, odds ratio; CI, confidence interval; I-FABP, intestinal fatty acid binding protein; IL-6, interleukin-6.
Diagnostic value of I-FABP and I-FABP mRNA for neonatal NEC
ROC curve analysis was used to measure the diagnostic value of I-FABP, I-FABP mRNA, and IL-6 in patients with NEC. The cutoff value for I-FABP was 144.083 pg/mL, with an area under the ROC curve (AUC) value of 0.668 (95% confidence interval [CI]: 0.531–0.804), sensitivity of 86.7%, specificity of 46.7%, negative prediction rate of 77.8%, and positive prediction rate of 61.9%. The cutoff value for I-FABP mRNA was 0.655, with an AUC value of 0.658 (95% CI: 0.516–0.800), sensitivity of 70%, specificity of 66.7%, negative prediction rate of 69.0%, and positive prediction rate of 67.8%. The cutoff value for IL-6 was 4.95 pg/mL, with an AUC value of 0.786 (95% CI: 0.67–0.902), sensitivity of 86.7%, specificity of 66.7%, negative prediction rate of 83.4%, and positive prediction rate of 72.3%. The AUC value for the combined detection of I-FABP, I-FABP mRNA, and IL-6 was 0.768 (95% CI: 0.650–0.885), with a sensitivity of 56.7%, specificity of 90.0%, negative prediction rate of 67.5%, and positive prediction rate of 85.0% (Figure 5).

Receiver operating characteristic (ROC) curve analysis of intestinal fatty acid binding protein (I-FABP), I-FABP mRNA, and interleukin-6 (IL-6) levels in human neonatal necrotizing enterocolitis (NEC) samples. The area under the ROC curve (AUC) value for the combined detection of I-FABP, I-FABP mRNA, and IL-6 was 0.768 (95% confidence interval: 0.650–0.885), with a sensitivity of 56.7%, specificity of 90%, negative prediction rate of 67.5%, and positive prediction rate of 85.0%.
Discussion
Premature birth, intestinal dysplasia, enteral feeding, infection, hypoxia-ischemia, acidosis, and bacterial flora disturbance are essential factors that promote the occurrence and development of NEC.18,19 The main lesions involve ischemia and hypoxia in the mucosa and submucosa, leading to inflammation, bacterial displacement, and toxin invasion. These can result in serious consequences, such as intestinal wall necrosis or perforation.20–22
Most articles on NEC focus on preterm infants, but NEC has also been reported to occur in near-term infants. The NEC disease pattern is believed to differs between premature infants and full-term infants, mainly reflected by the onset time and inducement of onset. Regarding the onset time, premature infants generally develop symptoms late, from 2 to 3 weeks after birth, while symptoms can appear in full-term infants within the first week after birth. Regarding inducement of onset, premature NEC is associated with intestinal immaturity and low birth weight. In contrast, full-term NEC is associated with congenital malformations, perinatal hypoxia-ischemia, infection, glycemic dysfunction, or maternal hypertension. The precise pathophysiology of NEC between these two different populations remains to be studied. However, most experts believe that the pathophysiology of NEC is essentially related to the secondary inflammation caused by hypoxia or intestinal damage.23,24 In this study, late-stage neonates (around 37 weeks) were selected as the experimental group. Despite the incidence of NEC being lower in full-term infants than in preterm infants, it does have some research significance.
Although advanced NEC usually shows more obvious clinical symptoms, non-specific indications may appear at early stages. Therefore, identifying, predicting, and diagnosing early NEC using biomarkers will be crucial for timely intervention before the disease progresses.
Evaluating the intestinal barrier function is essential for the early diagnosis and treatment of NEC. This can be performed indirectly by collecting blood, urine, stool, intestinal tissue, and other specimens and measuring specific and non-specific indicators. There are several types of commonly used indicators: intestinal mucosal epithelium damage indicators, such as enterocyte metabolites (I-FABP), 10 intestinal permeability indicators, such as D-lactate and the lactulose to mannitol ratio, non-specific inflammatory markers, such as IL-6 and C-reactive protein, and small endogenous non-coding RNAs with regulatory functions, such as microRNAs. 25 However, certain NEC biomarkers, such as fecal calprotectin, fecal high mobility protein B1, and other fecal biomarkers, have controversial intestinal specificity. An example of this controversy is that calprotectin, a fecal biomarker of intestinal inflammation, has been found to have mixed results in neonates. Calprotectin levels are associated with gestational age and vary significantly during the first week of life, with the concentrations rapidly decreasing in subsequent measurements of preterm infants. Therefore, fecal calprotectin has limited value for early NEC diagnosis.6,7 Additionally, the role of high mobility group box-1 (HMGB1) in intestinal inflammation has been described in various studies, including with NEC and colitis, but its clinical significance in NEC remains unclear.8,9
IL-6 is a cytokine with a variety of biological activities and functions. This cytokine is widely involved in hematopoietic cells, acute phage responses, and immune responses. IL-6 expression levels typically increase earlier than C-reactive protein levels, inducing acute inflammatory reactions via acute hepatocyte stress and acute phase proteins, a good indicator of an early inflammatory response. IL-6 is a non-specific inflammatory indicator of NEC, with significantly higher levels in neonates with NEC stage III than in those with stage I or II. 6 Wisgrill et al. 26 showed that IL-6 levels are significantly higher in neonates with NEC requiring surgical intervention than in NEC stage I and II newborns that can be treated conservatively. Ma et al. 27 established an NEC mouse model, then treated them with an anti-IL-6 receptor (IL6R) antibody. These results suggested that IL-6 promotes the conversion of regulatory T (Treg) cells into IL-17-producing Treg cells. Additionally, blocking IL-6 signaling could improve NEC by increasing the proportion of Treg/IL17 cells. The authors concluded that blocking IL-6 is a promising new therapeutic strategy for NEC. Therefore, we selected IL-6 as the experimental index, which could provide further evidence for the early diagnosis of NEC patients.
Liu et al. 11 showed that measuring serum I-FABP levels could help identify patients with initially vague symptoms who eventually developed definitive NEC. This confirmed the value of I-FABP as a diagnostic and prognostic tool, even with non-specific gastrointestinal symptoms. Shaaban et al. 10 first showed that serum I-FABP levels were significantly higher in stage II compared with stage I, but also found that they were significantly higher in stage III compared with stages I and II. Furthermore, serum I-FABP levels and the NEC stage were significantly positively related in data from 40 preterm neonates with varying NEC severity (20 stage I, 12 stage II, and 8 stage III). In our study, we selected neonates with NEC Bell stage I, detected serum I-FABP and I-FABP mRNA levels, identified early NEC, made a timely intervention, and avoided progression. This workflow could mimic real-world clinical scenarios.
FABPs are small intracellular proteins generally composed of 126 to 134 amino acids that are expressed in different tissues and organs, actively participating in fat metabolism in the body. Their main function is to transfer long-chain fatty acids from the cell membrane to the phospholipid and triacylglycerol synthesis site. 28 Zhang et al. studied 18 species and identified 12 FABPs genes. However, none of these genes coexisted in a single species. 29 Nine genes encoding FABP have been identified in humans, which are expressed in the heart, brain, liver, testes, ileum, small intestine, epidermis, adipocytes, and myelin. The relevant gene in the small intestine encodes I-FABP and is located on the long arm of human chromosome 4. The protein consists of 132 amino acid residues. The excessive sloughing of epithelial cells associated with NEC damage in the lumen of the intestine is believed to cause I-FABP mRNA to be released into the serum.
I-FABP is an intracellular lipid chaperone with a low molecular weight (14 to 15 kDa). I-FABP is localized to the cytoplasm of mature enterocytes at the tip of the small intestinal villi and is expressed explicitly in the jejunum and ileum. I-FABP is a noninvasive marker for evaluating intestinal wall integrity and inflammation loss.30–32 Hydrophobic molecules, such as fatty acids, use soluble carrier proteins like I-FABP for transport into the hydrophilic cytoplasmic environment, where they are involved in various metabolic processes. 33 Normally, I-FABP is not present in the peripheral blood. However, when ischemia, hypoxia, or other injuries occur in the intestinal mucosa, enterocyte villi disruption leads to I-FABP being released from the cells into circulation. Therefore, serum I-FABP levels could theoretically be used as a specific marker for detecting NEC.34,35
Using I-FABP ablation, Lackey et al. 36 observed increased enterocyte mortality in the experimental mouse group with a shortened villus length, thinner myometrial layer, and decreased goblet cell density. Wang et al. 37 studied the importance of I-FABP by evaluating intestinal barrier dysfunction during the early stage of severe burns in 36 mice, which were randomly divided into healthy control and scalded groups. They showed that plasma I-FABP levels were significantly higher in the scalded group than in the healthy control group. Therefore, I-FABP may be increased by intestinal hypoxic-ischemic injury after scald burns. The neonatal model group results in our hypoxic-ischemic intestinal injury mouse experiments showed that the muscular layer of the ileum was absent, the villi were partially reduced, and the edema of the submucosa and lamina propria was evident, with some mucosal necrosis. A higher pathology score corresponded to more severe NEC damage. As the pathological score increased, the expression levels of I-FABP from the base to the top of the folded intestinal mucosa, especially in the cytoplasm of the brush border membrane of small intestinal villous epithelial cells, significantly increased. However, I-FABP expression decreased in the nucleus and accumulated in the villus central lacteal, basement membrane, and submucosa of the small intestine. Compared with the control mice, there were significantly increased serum levels of I-FABP and I-FABP mRNA, as well as ileal mucosa I-FABP levels, after ischemia and hypoxia. These data are consistent with the findings of previous studies.36,37 Similarly, in the neonatal clinical specimens, serum I-FABP and I-FABP mRNA levels were significantly higher in the experimental group than in the control group. After comparing laboratory indicators, the data generated with the animal model were consistent with the human data.
El-Abd et al. 38 compared blood and urinary I-FABP levels and blood lactate levels between 55 neonatal patients with NEC and 23 healthy neonates. They showed that these levels were significantly higher in the control group and increased with progression of the NEC stage. At a cutoff point of 3.24 ng/mL, I-FABP showed a negative predictive value of 94.7%, positive predictive value of 52.6%, sensitivity of 90%, and specificity of 72%. The AUC value for NEC diagnosis was 0.768. A retrospective study of 106 neonates with NEC by Hu et al. 28 showed that the AUC value of I-FABP for NEC diagnosis was 0.870, with a cutoff value of 12.10 pg/mL, 72.6% specificity, and 92.5% sensitivity. Serum I-FABP levels were significantly higher in deceased patients than in surviving patients, and the logistic multiple regression analysis showed that IL-6, IL-1β, and I-FABP were independent risk factors for mortality in neonatal NEC. Our study showed the following diagnostic value of I-FABP, I-FABP mRNA, and IL-6 levels for NE: an accuracy of 0.668, 0.658, 0.786, and 0.768; sensitivity of 86.7%, 70%, 86.7%, and 56.7%; specificity of 46.7%, 66.7%, 66.7%, and 90%; negative prediction rate of 77.8%, 69.0%, 83.4%, and 67.5%; and positive prediction rate of 61.9%, 67.8%, 72.3%, and 85.0%, respectively. Because I-FABP, I-FABP mRNA, and IL-6 were detected with high sensitivity, they can be used as important biomarkers for the early prediction of NEC. The combined detection of I-FABP, I-FABP mRNA, and IL-6 showed high accuracy for NEC prediction and had higher positive prediction rate and specificity values than with using any of these indicators alone. Therefore, combined detection is recommended to improve the accuracy of NEC diagnosis to reduce the misdiagnosis rate. When these indicators were detected separately, the negative prediction rate was higher than the positive prediction rate, indicating that the possibility of NEC was low if the detection result was negative. If the results with these indicators separately are positive, then other clinical indicators should be used to aid with the NEC diagnosis. However, when these indicators were detected in combination, the positive prediction rate was higher than the negative prediction rate, indicating that the possibility of NEC was high if the detection result was positive.
Study limitations
This study has several limitations. First, the small sample size reduced the accuracy of the results. However, the data are still acceptable, considering the sample type, number, detection method, and high heterogeneity between the different cohorts. Further research with a larger sample size is needed to increase the accuracy of the predictive value of these markers. Second, the interactions between I-FABP and other regulatory proteins, such as PPAR, nuclear localization processes, and signaling pathways, were not examined in depth. Additional studies are needed to determine the intracellular interactions of I-FABP.
Conclusions
In this study, we found that I-FABP genes may be widely involved in regulating FABP expression in small intestinal tissues, providing a fundamental theoretical basis for fatty acid metabolism regulation. Furthermore, serum I-FABP, I-FABP mRNA, and IL-6 levels have high sensitivity values as biomarkers in NEC, suggesting their potential use as specific indicators for early disease assessment. Examining serum I-FABP levels in NEC showed lower specificity than measuring the levels of its mRNA. The early combined detection of I-FABP, I-FABP mRNA, and IL-6 is recommended to improve detection accuracy, facilitate diagnosis, and provide an objective basis for assessing neonatal NEC progression and outcomes.
Footnotes
Author contributions
JC designed the study, performed the primary research, and wrote the manuscript. ZY supported manuscript revision. ZL performed the sample collection and detection. YF supported the specimen collection and detection for all animal experiments. XB, XC, and AZ supported the data collection and analysis. All authors read and approved the final manuscript.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This study was supported by grants from the Fujian Natural Science Fund (grant number: 2021J011300) and the Municipal Key Clinical Specialty Construction Project of Fuzhou City in 2019 (grant number: 20191204). The funding bodies played no role in the study design, data collection, analysis, interpretation, or manuscript writing.
