Abstract
Objective
Hypothermia increases mortality among trauma patients. It is difficult to identify hypothermia in conscious patients due to the limited availability of diagnostic equipment yielding accurate measurements. Shivering is a symptom associated with hypothermia, and it serves as a parameter for clinical staging of hypothermia. Because shivering is part of the body's response to counteract hypothermia, the resulting thermogenic effect and expected cooling rates in shivering individuals warrant further investigation. This study aimed to describe core temperature trajectories in a setting of cold air, mild wind, and wet clothing, simulating a clinically relevant prehospital scenario.
Methods
Fifteen healthy volunteers were dressed in wet clothing and placed in a windy climate chamber set at 5.0°C for 3 h. Core temperature was measured using an esophageal probe (Teso) and an ingestible capsule (Tcap). Shivering was estimated based on oxygen consumption (VO2) and by objective observations of the participants.
Results
The temperature reductions from baseline were 0.25°C for Teso (95% CI, 0.06–0.44°C, P=0.014) and 0.11°C for Tcap (95% CI, −0.13–0.34°C, P=0.36). Initially, participants exhibited a significant increase in temperature. The cooling rate in the climate chamber was 0.17°C·h–1 (Teso). VO2 corresponded with objective observations of shivering and was inversely related to Teso.
Conclusions
Healthy individuals exposed to the described windy, wet, and cold conditions with only light clothing retained a normal core temperature for 3 h. The heat-producing mechanism of shivering is considered to be a significant contributing factor; therefore, a lack of shivering in patients in cold environments should be considered a warning sign for hypothermia.
Introduction
Accidental hypothermia is an independent risk factor for increased mortality among patients with traumatic injuries.1,2 Diagnosis, prevention, and treatment of hypothermia are highly prioritized by prehospital personnel. In prehospital settings, the demand for rescue services sometimes exceeds the available resources, and healthcare personnel must decide whether to prioritize hypothermia treatment above other medical emergencies or rapid transport to definitive care.
Identifying hypothermia in a prehospital environment is complex and technically challenging. Esophageal temperature probes yield precise measurements and are considered the “gold standard” measurement for obtaining a correct core temperature. 3 However, in our experience, most patients are conscious and unlikely to tolerate an esophageal probe. Tympanic and other noninvasive measurements tend to be inaccurate,4,5 and even though thermistor-based tympanic measurements are considered more accurate, they do not seem to provide accurate core temperatures for initial diagnostics. 6 Rectal temperature measurements could require further exposure of the patient to the cold environment, and measurements lag in cases of rapid temperature change.7–9 Overall, there are limited accurate and feasible tools available for measuring the core temperature of conscious patients in a prehospital setting.
In the absence of objective measures, hypothermia is sometimes staged based on clinical symptoms. The human body employs a range of homeostatic mechanisms to counteract deviations in core temperature and preserve thermal equilibrium, such as vasoconstriction of the peripheral capillaries. Shivering is one of the most efficient and apparent heat-generating mechanisms and is frequently associated with the onset of hypothermia. The Swiss staging system categorizes hypothermia as mild (35–32°C), moderate (32–28°C), or severe (<28°C) based on the presence of shivering and the level of consciousness alone. 10 This scale does not include the cold-stressed patient group (35–37°C). Notably, it is not necessarily correct to use shivering as a sign of reduced core temperature because the shivering threshold is usually higher than 35°C, which is the definition of hypothermia. 11 Shivering is initiated in response to increased heat loss and occurs well before any measurable change in core temperature. Recent research has shown that the Swiss staging system is imprecise due to the high degree of overlap between categories and provides a correct staging of hypothermia in only 50 to 60% of cases. 12
If neither measurement devices nor symptoms can provide an accurate core temperature in conscious patients, data regarding the expected cooling rates under certain conditions can guide clinical decision making. This approach to core temperature estimation has been examined under conditions of immersion in snow and cold water.13–18 The effect of cold ambient air on cooling rates, however, remains insufficiently described. In our experience, patients typically experience heat loss through exposure to cold ambient temperatures combined with windy or wet conditions. Accordingly, a Polish study of the prevalence of accidental hypothermia reported that cold ambient air was the predominant mechanism for heat loss. 19 To design this study to examine the most prevalent conditions, we reviewed meteorologic data from the last 3 decades. The average year-round temperature in Oslo, Norway, is 7.0°C. 20 In Switzerland—a similar country with mountain ranges—the average temperature is 5.8°C. 21 Notably, the prehospital environment is also frequently characterized by precipitation and wind.
In this study, we aimed to describe the core temperature trajectory in healthy volunteers exposed to 5°C ambient air in a wet and windy environment. The primary outcome measures were esophageal and gastrointestinal temperature, and the secondary outcome was estimation of shivering.
Methods
Study Design
The data used in this study were obtained from a clinical trial involving experimental hypothermia that was completed at Haukeland University Hospital, Bergen, Norway, in November 2023. 22 The participants in the drug trial received shiver-inhibiting drugs or placebo in a randomized, blinded, crossover design with a 1-wk washout period. The present analysis uses data from the placebo arm of the study. Note that certain data were employed in both the current paper and in the previously published effect paper. These papers answer different research questions and apply the data using different methodologic approaches as well as differing with respect to the substantive focus.
Selection of Participants
Fifteen healthy volunteers 18 to 40 years of age with a body mass index of <30 kg/m2 were recruited. The participants were asked to refrain from caffeine, alcohol, and strenuous exercise for 24 h prior to the experiment. They ate a light meal on the day of the experiment, and no nicotine products were used at the study site.
Protocol
One hour before entering the climate chamber, the participants ingested a temperature capsule. Subsequently, an esophageal probe was inserted, and the participants were connected to a multimonitor (Corpuls3; GS Elektromedizinische Geräte G. Stemple GmbH, Kaufering, Germany). Participants were fitted with a face mask to enable measurement of oxygen consumption (VO₂), which served as a proxy for shivering. After instrumentation, the participants rested in a supine position on stretchers in thermoneutral conditions for 10 min before baseline measurements were recorded. Before entering the climate chamber, the participants dressed in single-layer cotton pants and sweaters with an estimated thermal insulation value of 1 clo, according to ISO 9920. The garments had been soaked in cold water and subsequently compressed by hand to remove excess water such that they were not dripping. To minimize discomfort, participants wore dry hats, mittens, socks, and shoe covers. The participants walked into the climate chamber (20 m) and laid in the supine position on sleeping pads (Z-Lite [R-value 2.0]; Therm-a-Rest, Seattle, WA). The climate chamber was a (2.3×5.5×2.3m) refrigerated steel container set to 5°C. For 3 h, the participants laid still with air fans (Ryobi 18V R18F5-0; Ryobi Tools, Marlow, UK) next to their feet, set to maximum wind speed. The resulting median ambient temperature [min, max] was 5.9°C [5.5, 6.8], and wind speed was 1.8 m·s–1 [0.9, 2.8]. After the experiment, the subjects were actively warmed in an infrared sauna until their esophageal temperature reached at least 36°C. For an overview of the timeline, see Figure 1.
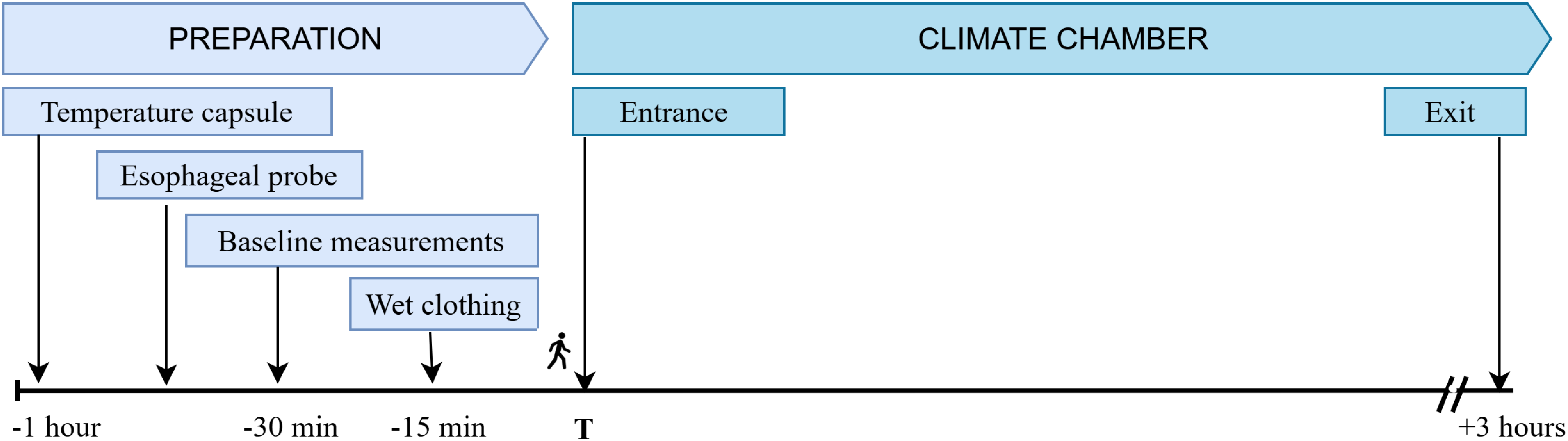
Timeline of the study protocol. All time points are in reference to time T, which was the entrance to the climate chamber.
Outcomes/Measurements
The primary outcome was the change in temperature from baseline to the end of the experiment. An esophageal probe was inserted through the nose to the level of the heart and connected to the multimonitor. Measurements were performed at all time points, revealing the trajectory of temperature change throughout the experiment. Temperature also was measured by an ingestible thermal telemetry capsule (e-Celsius; BodyCAP, Caen, France).
The secondary outcome was how core temperature corresponded to shivering. Shivering was estimated based on an increase of oxygen consumption (VO2) per milliliter per kilogram per minute. VO2 was logged breath by breath while participants were in the climate chamber using the portable metabolic system COSMED K5 (COSMED, Rome, Italy) with OMNIA Software v.2.2 (COSMED) calibrated according to the manufacturer's specifications. The participants wore K5 face masks for the duration of the trial, fixed in position by compatible headgear (Hans Rudolph, Inc., Kansas City, MO). Shivering also was evaluated as none, mild, moderate, or severe on a scale from 0 to 3 by a research assistant every 20 min using the Bedside Shivering Assessment Scale (BSAS). 23 Ambient temperature and humidity were measured continuously using a logger placed on the floor of the climate chamber (OM-CP-MICRORHTEMP; DwyerOmega Engineering, Irvine, CA). Wind speed was measured at the thoracic level every 20 min using handheld equipment (Testo, Titisee-Neustadt, Germany).
To ensure that they remained within predefined safe levels, the participants’ vital signs (ie, heart rate, respiratory rate, oxygen saturation, blood pressure, electrocardiogram, and end-tidal carbon dioxide) were measured using the Corpuls3 monitor (GS Elektromedizinische Geräte G. Stemple GmbH). Skin temperature was monitored using iButtons (Maxim Integrated Products, Beaverton, OR) placed on forehead, chest, upper arm, dorsal surface of the hand, thigh, calf, and foot. A lower safety limit for skin temperature was predefined at 8°C. Confirmation that skin temperatures remained above this threshold was obtained post hoc through visual inspection of the recorded temperature data.
Analysis
We characterized the sample using descriptive methods. Temperature differences were assessed using 2-sided paired t tests, applying a significance level of P<0.05. Mean cooling rates and corresponding 95% CIs were calculated using the Student t distribution. The boxplot of temperatures at relevant time points illustrates the median (line), the interquartile range (box), the minimum and maximum value (whiskers), and the outliers (defined as values outside ±2 SD, shown as circles). VO2 data were smoothed using the moving average of 5 measurements graphically illustrated and numerically described.
Data handling and computations were performed using R 4.4.0 (R Foundation, Vienna, Austria), and graphics were generated with MATLAB (MathWorks Inc, Natick, MA). 24 Analyses were performed using all collected data. Missing data were not replaced using imputation methods, with the exception of esophageal temperature, for which single missing values and obviously erratic measurements (temperature <34°C or a change of >0.3°C per minute) were replaced by the average of the 5 previous and 5 subsequent measurements.
Results
Fourteen participants completed the 3-h-long cooling protocol (Table 1). A technical measurement error led to early experiment termination for 1 participant, and data from this participant were excluded from analysis.
Participant characteristics.
From baseline to exit from the climate chamber, the esophageal temperature (Teso) decreased by 0.25°C (95% CI, 0.06–0.44°C, P=0.014; Figure 2). Measurements by ingestible capsule (Tcap) showed a nonsignificant decrease of 0.11°C (95% CI, −0.13–0.34°C, P=0.36). Individual temperature curves are available as Supplemental Material in the online version of the journal.
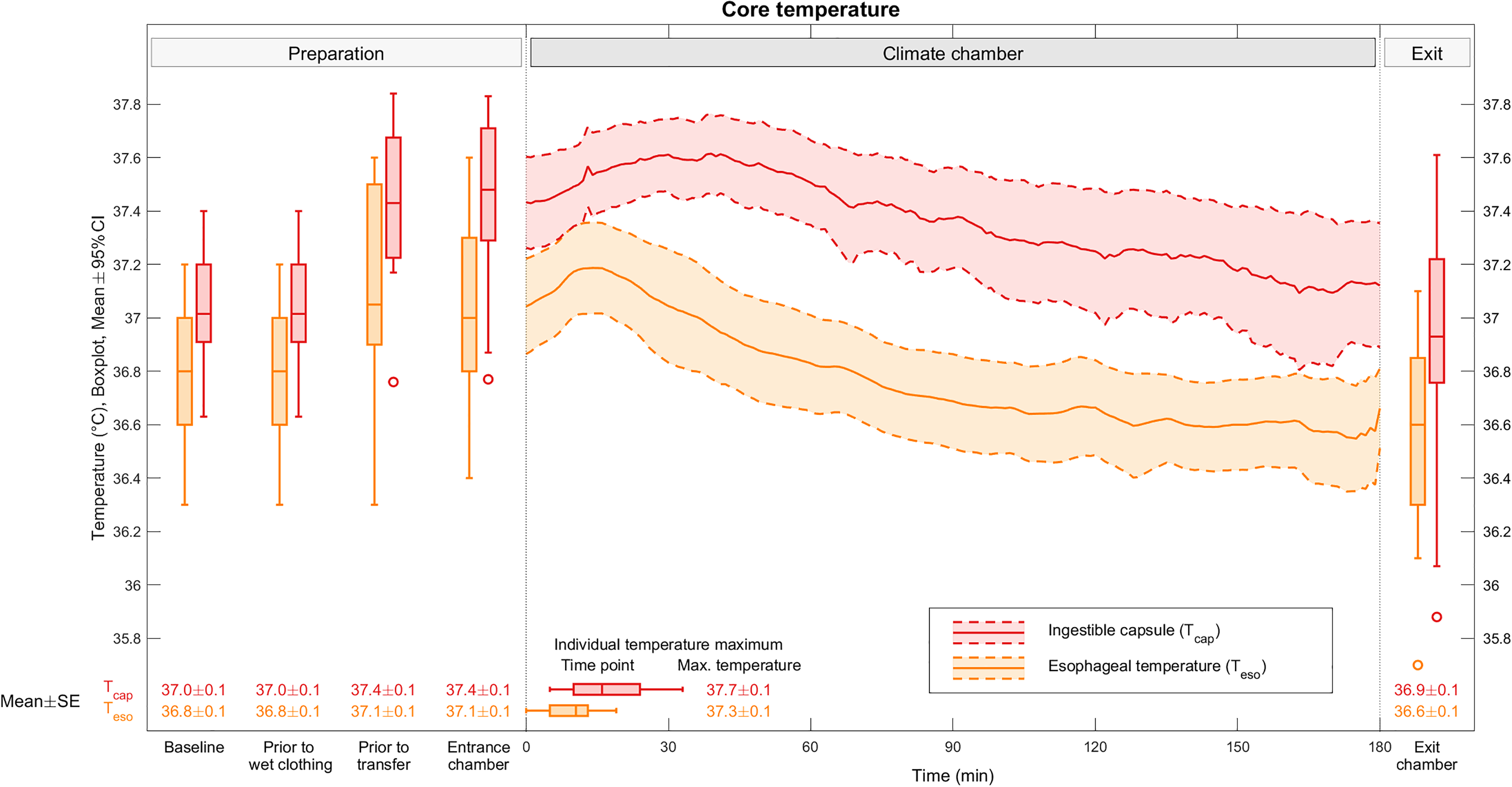
Participants’ core temperature trajectories measured by esophageal probe (Teso) and ingestible capsule (Tcap). Temperature was documented at baseline and continuously logged during the preparation phase and during exposure inside the climate chamber.
After being dressed in wet clothing, participants exhibited an increase in core temperature, according to both esophageal and gastrointestinal measurements. Teso increased by 0.29°C (95% CI, 0.18–0.39°C, P<0.001). Core temperature also increased after participants entered the cold climate chamber, with Teso increasing by 0.20°C (95% CI, 0.10–0.31°C, P<0.001) during the first minutes. For the remainder of the trial, core temperature decreased at a slightly gradual slope, resulting in a mean Teso of 36.6±0.4°C after 3 h. Based on the 3 h spent in the climate chamber, the cooling rate was −0.17°C·h–1 (95% CI, −0.23 to −0.11°C·h–1) for Teso and −0.18°C·h–1 (95% CI −0.25 to −0.11°C·h–1) for Tcap. When accounting for the cold stimulus induced by application of wet clothing and defining T–15 min as the starting point—at which the core temperature remained equivalent to baseline—the resulting cooling rates were Teso 0.08°C·h–1 (95% CI, −0.13 to −0.02°C·h–1) and Tcap 0.04°C·h–1 (95% CI, −0.12–0.03°C·h–1). The core capsule displayed consistently higher temperature values than the esophageal probe.
VO2 was 4.1±0.9 mL·kg–1·min–1 at baseline and increased to 4.6±2.1 mL·kg–1·min–1 on entrance to the climate chamber. The peak core temperature at ∼15 min after entering the climate chamber was accompanied by a reduction of oxygen consumption to near baseline values (Figure 3). Thereafter, oxygen consumption increased gradually during the experiment, reaching 5.8±2.0 mL·kg–1·min–1 after 3 h. Bystander observations documented shivering at 89% of all time points registered (BSAS>0). Most of the negative observations (shivering not observed, BSAS = 0) occurred at 20 min after entrance, in accordance with decreasing VO2 at the time at which temperatures increased (Figure 3).
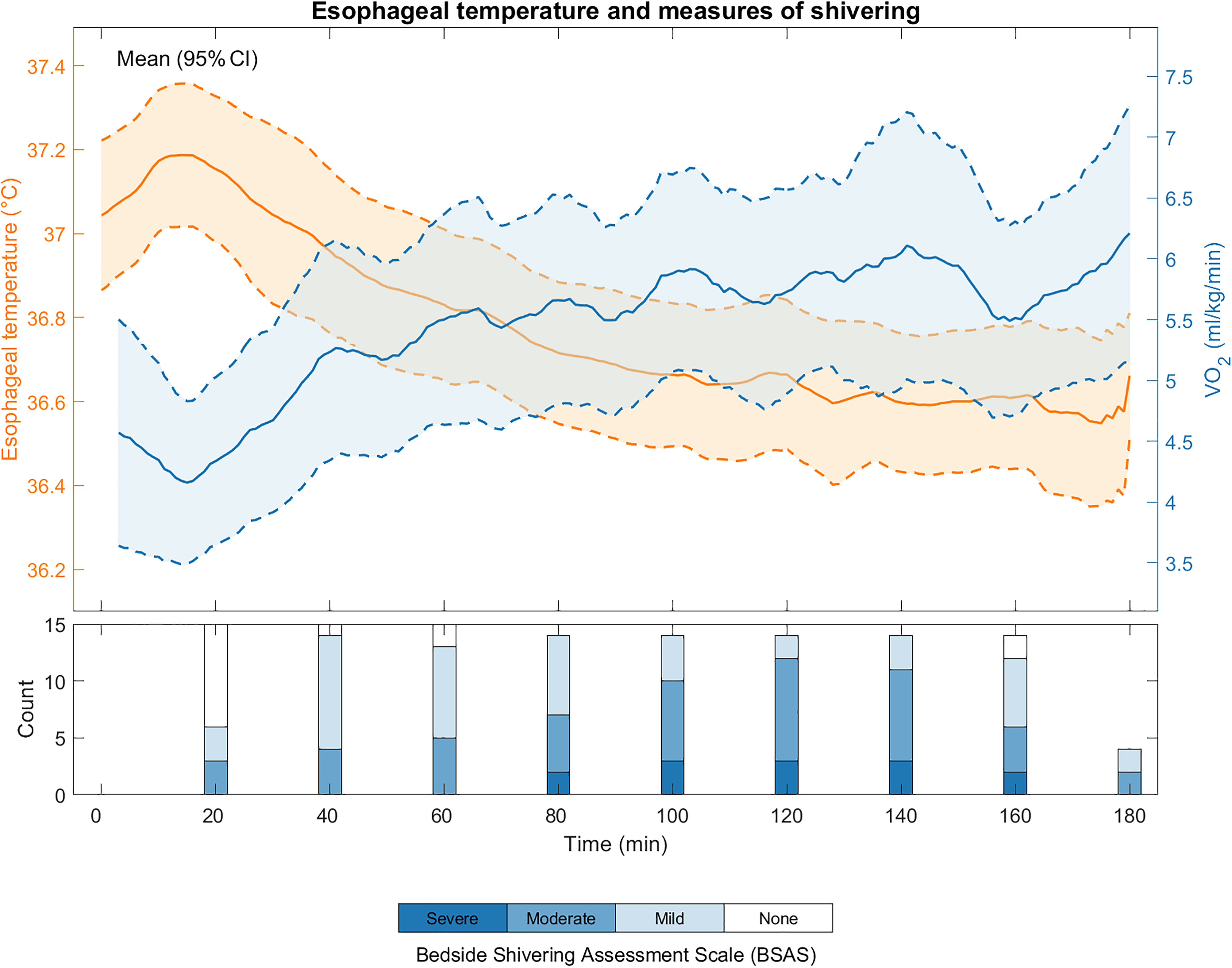
Esophageal temperature and VO2. Shivering, as estimated by objective observations (Bedside Shivering Assessment Scale), and oxygen consumption (VO2, in mL·kg–1·min–1) relative to mean esophageal temperature. Mean VO2 is shown along with the 95% CI. Participants entered the climate chamber at time=0. The absence of observations for the final Bedside Shivering Assessment Scale scoring at 180 min was due to a timing overlap between the scheduled shivering assessment and the conclusion of the experiment.
There was no need to discontinue study interventions based on safety parameters. Skin temperatures are available as Supplemental Material in the online version of the journal.
Discussion
In this study, healthy volunteers retained a normal core temperature throughout 3 h of exposure to windy, wet, 5.9°C conditions. Our results illustrate the remarkable impact of endogenous mechanisms in maintaining core temperature in harsh environments and demonstrate the human body's inherent capacity for heat conservation and production.
In accordance with our findings, a small study of healthy male participants published in 1993 documented rectal temperatures of 36.4 to 36.5°C after 3 h in a 5°C environment. 25 In that study, the participants were not wearing wet clothing, and the wind speed was lower than in this study (1 vs 1.8 m·s–1). These conditions of dry clothing and minimal wind speed are similar to those of another study from 1985, in which 4 male volunteers spent 24 h at an ambient temperature of 7.5°C and exhibited a core temperature reduction of only ∼0.8°C, demonstrating that core temperature could be maintained over prolonged periods. 26 The preliminary study procedure used an ambient temperature of 5°C, but the 2 participants undergoing these conditions could not complete a 24-h cooling phase due to thermal discomfort and were removed from analysis. Moreover, 1 of the remaining participants exhibited a core temperature approaching 35°C and had to be given warm wrappings. These results reveal wide interindividual variations in cold-stress endurance. Although the generalizability of these findings is limited by the small sample sizes, there being no female participants, and the site for temperature measurements being rectal, the general temperature trajectory aligns with our findings. In another study, volunteers wore wet clothing and were exposed to −18.5°C for 20 min, followed by 30 min wearing insulation; the participants exhibited no change in esophageal temperature. 27 In that study, the ambient temperature was low, but the time window was short, and there was no wind. The results may indicate the importance of wind speed when predicting cooling rates, perhaps due to the large convective heat loss. All the abovementioned studies have included healthy volunteers. Steeper cooling rates likely will be observed among children and elderly, fatigued, intoxicated, or injured patients with reduced capacity for heat conservation and production.
When core temperature increases, the stimulus to maintain shivering is reduced. Accordingly, our data showed decreasing VO2 in association with the initial rise of core temperature immediately following entrance to the climate chamber (Figure 4). The absence of shivering at the time point of peak temperature was supported by the bystander's observation of shivering behavior. The subsequent gradual, slight decrease in core temperature during the remaining hours in the climate chamber was accompanied by a gradual increase of shivering, estimated by both increased VO2 and bystander observations. This gradual increase is in accordance with a previous report of heat production during a 24-h experiment. 26
There is a long-reigning perception that shivering is a sign of hypothermia. A New England Journal of Medicine review of accidental hypothermia from 2012 claimed that hypothermia can be staged clinically using the Swiss system, 28 and this claim was repeated in the 2016 update. 29 According to the Swiss clinical staging system, a patient exhibiting both clear consciousness and shivering would be placed in the 32 to 35°C category. 10 Using this scale, all our participants would have been categorized as hypothermic throughout the entire trial; however, none of them were hypothermic (<35°C) at any point. A clinical decision-making tool for differentiating hypothermic from normothermic patients could help rescuers prioritize resources as long as it provides an accurate diagnosis. A 2022 update on accidental hypothermia noted that shivering is not a consistent symptom of hypothermia. 30 We wish to expand on this observation by pointing out that in trials of experimental hypothermia, the deliberate suppression of shivering enables a reduction of core temperature. 22 We therefore wish to emphasize that the absence of shivering may indicate an increased risk of hypothermia.
The healthy individuals in our study were able to withstand hypothermia under the tested conditions, but it remains unclear how long a person can endure such conditions. The increasing VO2 illustrates how the heat produced by shivering nearly compensated for the heat lost to the surroundings. The mean VO2 increased by ∼50% from baseline to exit, which is far below the maximum increase of nearly 500% from baseline that has been shown in other studies exploring peak heat production from shivering. 31 If we extrapolate data from our experiment, using the cooling rate of 0.17°C·h–1, healthy individuals would be able to resist hypothermia for >24 h under similar conditions. However, instead, there may be a point of fatigue at which individuals can no longer conserve or produce heat, resulting in an increased cooling rate. Further studies are needed to investigate longer time periods, fatigued participants, lower temperatures, and other wind strengths to increase our understanding of how these conditions affect our patients.
The initial temperature increase observed immediately following exposure to a cold stimulus was unexpected. Participants’ core temperature after entrance to the climate chamber was higher than at baseline. The application of wet clothing at T–15 min appeared to activate the endogenous thermoregulatory response, inducing visible shivering and increasing core temperature. The most plausible explanations include heat generated by shivering, along with nonshivering thermogenesis and reduced heat loss due to vasoconstriction and perhaps increased vasomotor tone. 32 The endogenous thermoregulating mechanisms not only maintained the core temperature at baseline value but actually increased the core temperature. Regrettably, data on shivering were not documented during this time interval. These endogenous thermoregulating mechanisms likely also caused the increase in temperature after entering the climate chamber, possibly with a contribution from muscular work due to the short walk. The core temperature at T–15 min was equal to the baseline value (Teso and Tcap), hence representing core temperature ahead of any cold stimulus. When the cooling rates were calculated by incorporating the 15 min prior to entrance, the temperature decreased by <0.1°C·h–1, with these values reflecting the complete trajectory of core temperature increase and decline. This finding adds to our understanding of the body's response to cold stimuli. The initial response of increased core temperature when exposed to a cold stimulus may suggest that rescuers approaching shivering patients who have been exposed to similar conditions for similar time periods could modify the predicted level of urgency and criticality with respect to hypothermia, which may allow more time to consider danger on approach or other critical tasks.
Notably, this does not mean that shivering patients exposed to a similar cold environment for <3 h would not benefit from early hypothermia treatment. Although the thermoregulating mechanisms are highly effective, they do require oxygen. Injured, bleeding patients and medically ill patients in oxygen deficit should avoid expending resources on shivering when possible. Trauma patients exhibit increased mortality even with a core temperature of 36°C. 33 Thus, shivering patients should receive isolation and active warming as early as possible. Ideally, all patients would receive prophylactic hypothermia treatment in prehospital settings; however, the innate characteristics of prehospital emergency medicine include having limited time and resources.
Our results are reliable because we measured core temperature using an esophageal probe, which yields accurate values in cases of swift temperature changes. 7 The temperature trajectory documented by esophageal measurements was similar to that revealed by core capsule measurements, and the measurement sites showed nearly identical cooling rates in the climate chamber. In general, compared with the esophageal measurements, the temperatures measured by core capsule were a little higher, and the response to cold stimuli lagged behind. This likely represents an accurate measurement of the temperature in the gastrointestinal tract and indicates how the heat is distributed throughout the body. In a state of cold stress, sympathetic vasoconstriction could reduce circulation to the bowel. Additionally, the gastrointestinal measuring site is more isolated from the cold environment, which may explain the variation between measuring sites.
Another strength of this study is the choice to cool patients by mimicking a common clinical setting, which improves the transferability of our results to a clinical setting. However, our results are limited to the given study setting of 5.9°C ambient air, slight wind, and wet clothing.
Our findings are applicable solely to healthy adult individuals in the conditions described. Cooling rates also may depend on clothing worn, wetness, nutrition status, and body composition, and changes in these variables could impact the resulting cooling rates. Another limitation of our study is that the participants’ hands and feet were covered in dry clothing for comfort. This differs from a plausible clinical scenario, and the effect on the cooling rate is unknown. It is possible that the core temperature reduction would have been larger if this clothing also had been soaked in cold water, but it is also possible that the stronger cold stimulus provided would have induced a larger magnitude of shivering. Additionally, the wind measurements are associated with uncertainty because the handheld anemometers showed large variations with small adjustments during the trial. The timing of ingestion of the temperature capsule (1 h prior to entering the climate chamber) also could have affected the results due to a short transit time in the gastrointestinal tract.
In summary, our results indicate that healthy individuals exposed to the harsh conditions described are able to effectively preserve normal core temperature for 3 h. The heat-producing endogenous mechanism of shivering is considered a significant contributing factor. A lack of shivering among patients in cold environments should be considered a warning sign by clinicians. The minor reduction of core temperature is a relevant reference point that can be useful when evaluating the risk of hypothermia among shivering individuals in similar prehospital settings.
Supplemental Material
sj-docx-1-wem-10.1177_10806032251378099 - Supplemental material for Minor Decrease of Core Temperature in Shivering Volunteers Over a 3-Hour Exposure to Cold, Wet, and Windy Conditions
Supplemental material, sj-docx-1-wem-10.1177_10806032251378099 for Minor Decrease of Core Temperature in Shivering Volunteers Over a 3-Hour Exposure to Cold, Wet, and Windy Conditions by Ane M. Helland, Sigurd Mydske, Jörg Assmus, Guttorm Brattebø, Øystein Wiggen, Haakon K. Kvidaland and Øyvind Thomassen in Wilderness & Environmental Medicine
Footnotes
Previous Presentation
The results of this study were presented at the Congress of the International Commission for Alpine Rescue (ICAR) in Thessaloniki, Greece, on October 19, 2024, and at the Fall Meeting of the Norwegian Society of Anesthesiology in Oslo, Norway, on October 23, 2024.
Acknowledgments
We thank our volunteer participants and our research assistants for their altruistic contributions. We also express our gratitude to Sivert Helland Veseth for his outstanding assistance with the K5 apparatus during the trial and to Safeguard Medical and Algeco for providing us with necessary equipment.
Author Contribution(s)
Design (AMH, SM, JA, GB, OW, OT); protocol (AMH); trial conduct and data collection (AMH, SM, OT); VO2 measurements and management of the K5 apparatus (HKK); statistical analysis (JA, AMH); manuscript draft (AMH). All authors substantially contributed to and approved the final version of the manuscript.
Consent to Participate
All participants gave their signed informed consent prior to participation.
Data Availability
The complete study protocol and the deidentified data subset for this investigation are available on reasonable request.
Ethical Considerations
The protocol was approved by the Norwegian Ethics Committee for Clinical Trials on Medicinal Products and Medicinal Devices and the Reginal Committee for Medicinal and Health Research Ethics (REC) Southeast Norway and was registered in the EudraCT database (ID 2023-506020-81-00).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The trial was funded by the Norwegian Air Ambulance Foundation (NAAF).
Declaration of Conflicting Interests
The authors declare no conflicts of interest. AMH, SM, JA, and OT are employed by the Norwegian Air Ambulance Foundation (NAAF). NAAF had no role in the design of the study, does not have access to the data, and was not involved in the process of analyzing, interpreting, or publishing the results.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
