Abstract
Introduction—
Mount Aconcagua (6961 m) in Argentina is the highest peak in the Americas, and more than 3000 climbers attempt to summit annually. High altitude pulmonary edema (HAPE) is a leading cause of mortality and evacuation on Aconcagua.
Objective—
This study sought to describe the characteristics of climbers who developed HAPE on Aconcagua to aid in future prevention efforts.
Methods—
Climbers diagnosed with HAPE in January 2024 were surveyed regarding demographics, preparation, acclimatization, and illness. Healthy climbers on Aconcagua also were surveyed for the same period, providing a control population.
Results—
Seventeen climbers were diagnosed with HAPE. These climbers were similar to climbers without HAPE in age, sex, origin, home elevation, hypoxic tent use, staged ascent, recent virus exposure, fitness metrics, and ascent-rate plans. Climbers with HAPE spent fewer nights at the 4300-m base camp (3.6 vs 5.0 nights). Prior to developing HAPE, 71% of patients reported unresolved acute mountain sickness symptoms. HAPE onset median elevation was 5500 m, with a median of 8.0 days over 3000 m and a median lowest SpO2 of 60% at diagnosis. There was a nonsignificant trend between acetazolamide use and increased HAPE. All surveyed HAPE patients descended, required helicopter evacuation, and survived.
Conclusion—
This study examined climbers who developed HAPE on Mount Aconcagua, yielding implications for high altitude illness prevention efforts and further study. HAPE cases were associated with insufficient nights at the 4300-m base camp and unresolved acute mountain sickness symptoms. The relationship between acetazolamide and HAPE warrants further study on Aconcagua.
Introduction
Mount Aconcagua (6961 m) in Argentina is the highest peak in the Americas, and more than 3000 climbers attempt to summit annually. The climb's popularity is likely related to its nontechnical route, which makes it accessible to more climbers than many peaks of similar elevation. Of the climbers who ascend Aconcagua, most ascend via the “normal” route via the Horcones Valley with variable rates of ascent. 1 Aconcagua is managed by Aconcagua Provincial Park (APP) and includes medical services that provide medical screenings in the park. The incidence of acute mountain sickness (AMS) has been reported to be 39% on Aconcagua, 1 but more concerning is high altitude pulmonary edema (HAPE), which is a frequent reason for medical evacuation. In 2013, HAPE was reported as the second leading cause of mortality on Aconcagua after trauma, with 21.2% of deaths attributed to HAPE. 2
Acclimatization schedules on Aconcagua vary widely among private climbers and guide services operating on the mountain. Many climbers fly into Mendoza (746 m) and spend a night or two as they organize for the trek. Most guide services will then travel to Penitentes (2579 m) for a night, which puts them geographically close to the entrance to APP for an easy start the next day. Figure 1 Most climbers then spend 2 nights at Confluencia Camp (3400 m), with an acclimatization hike to Plaza Francia (4200 m) on the second day. The next camp is Plaza de Mulas (4300 m). There is no typical camp between Confluencia Camp and Plaza de Mulas. Guide services vary by how long (often 4–6 nights) climbers will sleep at 4300 m before ascending to higher sleeping elevations. Many of these itineraries are available online at publicly accessible guide service websites.
The unique aspect of hiking Aconcagua is that on Day 3, climbers hike ∼8 h through a dry, dusty valley from 3400 to 4300 m. This is a 900-m jump in sleeping elevation, which is nearly double the typically recommended increase for a single day. A rise in sleeping elevation of >500 m falls within the published acceptable guideline as long as the average does not exceed 500 m per night and there is a rest day every 2 to 3 d prior to summit day. 4
HAPE classically presents as exertional dyspnea, fatigue, and cough. As the edema progresses, the dyspnea will occur at rest, and the cough may become productive with pink, frothy sputum. 5 The primary pathophysiologic alteration in HAPE is an exaggerated hypoxic pulmonary vascular response leading to increased capillary and epithelial permeability. 5 Although much research has been directed at the pathophysiology, prevention, and treatment of HAPE, one of the gaps in knowledge remains in determining what factors may place climbers at higher risk of developing this deadly altitude illness.6–9
Aconcagua is a unique mountain to study high altitude illness because of the large number of climbers and the required climber screening system, which allows for contact with climbers. APP policy is to medically screen all climbers as they progress along the climbing route. Climbers are screened at Confluencia (3400 m) and Plaza de Mulas (4300 m) before being permitted to ascend to a higher sleeping elevation. To our knowledge, Aconcagua is the only large mountain that requires a similar medical screening evaluation. This presents a safety valve for the climbers, a benefit to the park professionals responsible for their rescue on the mountain should they get sick, and an ideal circumstance for high altitude research. The park medical service is also charged with caring for all ill or injured climbers on Aconcagua, including all those who develop HAPE.
This study sought to better understand the characteristics of climbers who developed HAPE on Aconcagua and determine whether differences exist between healthy climbers on the mountain to aid in global understanding and future prevention efforts.

Aconcagua, spatial view of the normal route through the Horcones Valley. 3
Methods
All climbers diagnosed with HAPE from January 5 through 24, 2024, were surveyed at Plaza de Mulas (4300 m) regarding their demographics, preparation, acclimatization, and illness. Aconcagua medical service physicians made the diagnosis of HAPE based on the standard diagnostic criteria of pulmonary crackles, dyspnea, and hypoxemia below expected for that altitude. Informed consent was obtained, and participation was voluntary and anonymous. Surveys were in English or Spanish. Surveys were performed after their initial medical evaluation, while patients were receiving treatment, and during the process of evacuation. As per study design, no climber was impeded in their treatment or evacuation in order to complete a survey. Climbers were permitted to complete only partial surveys if desired or necessary to facilitate treatment or evacuation.
Information gathered by the survey included demographics; altitude of residence; pre-acclimatization such as staged ascent and hypoxic tent use; medication use; recent virus exposure or symptoms such as rhinorrhea, pharyngitis, cough, or fever; training and preparation; concurrent symptoms of other altitude illness such as those of AMS; and HAPE symptoms and treatment. Climbers self-reported heart rate, oxygen saturation (SpO2) from pulse oximetry, and blood pressure if recalled from their own pulse oximetry devices or park medical service exams. Healthy climbers on Aconcagua also were surveyed for the same time period and provided a control population.
This survey-based study was determined to be exempt from institutional review board review at the University of Vermont. The APP medical service granted permission to conduct the study.
Statistics
Categorical variables were analyzed using Fisher's exact tests due to small sample sizes in some categories. Means were compared with 2-sample t tests. If variables were not normally distributed, they were compared using nonparametric Wilcoxon rank-sum tests. Analyses were conducted using SAS 9.4 (SAS Institute Inc, Cary, NC).
Results
Table 1 displays the characteristics of climbers who developed HAPE on Aconcagua. Over the study period, 17 climbers were diagnosed with HAPE by the park medical service. One climber was initially surveyed as a healthy control but later developed HAPE and was moved to the climber group that developed HAPE for this study.
Characteristics of climbers who developed high altitude pulmonary edema on Mount Aconcagua.

HAPE, high altitude pulmonary edema; AMS, acute mountain sickness.
Climbers who developed HAPE had an average age of 38 (9) y and were predominantly male (n=15; 88%). Climbers with HAPE were mainly from Europe (n=10; 59%) or North America (n=5; 29%). Of climbers who developed HAPE, 12 (71%) completed the survey question on whether AMS symptoms resolved before developing HAPE, and all 12 (100%) reported that their symptoms of AMS had not resolved before developing HAPE. Eleven (92%) of these 12 HAPE patients with concurrent unresolved AMS symptoms reported headache plus other general symptoms, and 1 (8%) reported only antecedent fatigue. The onset of HAPE occurred at a median elevation of 5500 (interquartile range [IQR] 4300–5500) m, with a median of 8.0 d ascending >3000 (IQR 5–13) m before HAPE development. At the onset of HAPE, the median lowest SpO2 was 60% (IQR 54–69%). A minority of climbers received oxygen (n=3; 18%). All surveyed HAPE patients (n=17; 100%) descended, were evacuated by helicopter, and survived. These data are displayed in Table 1.
Table 2 compares the 17 climbers who developed HAPE with a population of 42 healthy climbers as a control group. Climbers who developed HAPE were similar to healthy climbers in age, sex, altitude of residence, pre-acclimatization including staged ascent and hypoxic tent use, recent virus symptoms and exposure, fitness and training metrics, and overall ascent-rate plans. Climbers with HAPE spent fewer nights at 4300 m base camp (mean 3.6 vs 5.0 nights; P=.01). Climbers who developed HAPE had a nonsignificant trend toward more commonly taking acetazolamide (n=8 of 17; 47% vs n=8 of 42; 19%; P=.05).
Comparison of climbers with high altitude pulmonary edema with healthy climbers on Mount Aconcagua.
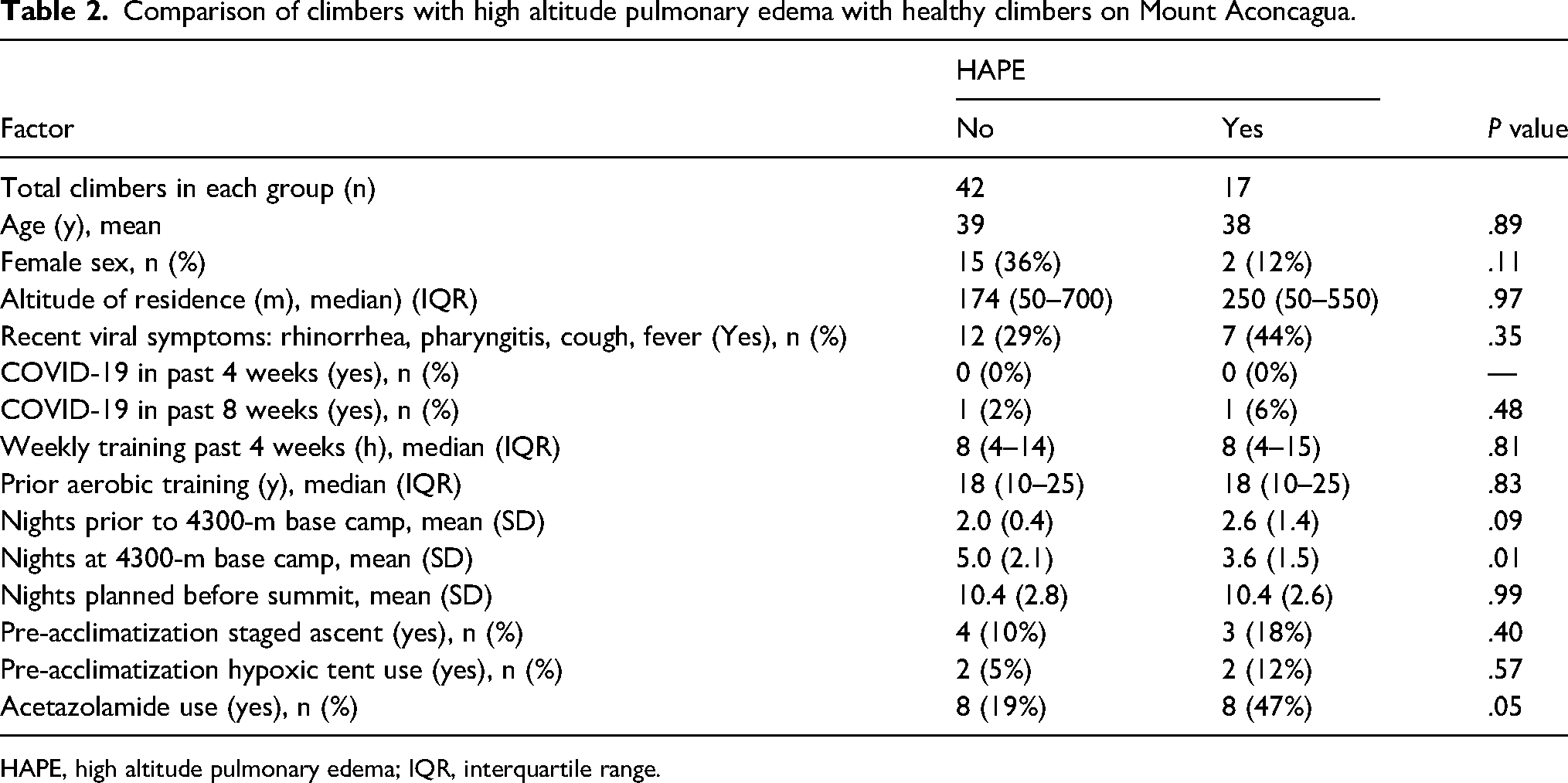
HAPE, high altitude pulmonary edema; IQR, interquartile range.
Discussion
This study examined 17 climbers who developed HAPE while on Mount Aconcagua over 3 weeks in January 2024 and yields implications for high altitude illness prevention efforts and further areas of study. Significant findings include the number of nights spent at 4300 m as being protective against developing HAPE and not ascending with AMS symptoms. Conditioning metrics did not have a significant protective effect on the development of HAPE.
Rate of Ascent
In this study, climbers who spent more nights at the base-camp elevation (Plaza de Mulas, 4300 m) were less likely to develop HAPE (Table 2). Spending 5 nights at 4300 m vs 3.6 nights was found to be protective against developing HAPE. The increased time at an elevation of 4300 m would allow for a greater time for acclimatization and an increased likelihood of resolving any AMS symptoms before ascending to higher sleeping elevations. The physiologic processes that occur with acclimatization take time. Studies show that a slower ascent and more time sleeping at an altitude allows optimal acclimatization.10,11 This is a specific and logistically feasible recommendation that could be useful for future APP preventive efforts and educational initiatives.
AMS Symptoms Preceding HAPE
It was found that 71% of HAPE patients did not have resolution of their AMS symptoms before developing HAPE (Table 1). This finding is significant and consistent with guidelines that indicate the need for the resolution of AMS symptoms prior to further ascent in the mountains. 4 The Wilderness Medical Society Clinical Practice Guidelines suggest that a climber experiencing AMS should be monitored for up to 3 d and should descend if they show no improvement. Ascent to a higher elevation is contraindicated if AMS symptoms have not resolved. 4 Recommending that climbers with AMS do not ascend may be a typical recommendation, but this study provides evidence to substantiate this rationale for preventive efforts and climber education from APP.
HAPE Presenting Symptoms
This study found that climbers with HAPE presented most commonly with the initial symptoms of hypoxia (71%), dyspnea (65%), cough (53%), and, less frequently, chest pain and hemoptysis. Mean SpO2 was 60%, indicating a potential red flag value, with the caveat that this represents a mean and not an absolute or cutoff value. These symptoms and signs provide educational opportunities for the APP medical service regarding attributes of climbers developing HAPE on Aconcagua.
Elevation of HAPE Onset
Climbers developed HAPE at an average elevation of 5260 m and 8.6 d at >3000 m. These findings substantiate the need for medical service and physician presence at higher camps on Aconcagua, not just base camps, when feasible. According to a 2013 study, 2 HAPE has caused significant mortality on Aconcagua in past years. The presence of medical care at a location near the onset of the majority of HAPE cases, such as at the Nido de Condores Camp at ∼5500 m and at base camps, appears crucial for APP.
Helicopter Evacuation
Despite being a nontechnical climb, all 17 (100%) of the HAPE cohort (Table 1) received helicopter evacuation. This is a significant difference when looking at other major climbing peaks across the world. On Kilimanjaro, 4 of 30 (13%) HAPE patients used helicopter evacuation, 12 and on Everest, 35 of 56 (62%) HAPE patients used helicopter evacuation. 13 The descent from Aconcagua's 4300 m base camp is a 24-km dry and rocky hike to the entrance of the park. This is an arduous hike for healthy climbers. When climbers develop HAPE, the mainstay of treatment is decent. The universally recommended priority and treatment are to descend in altitude. 4 On Aconcagua, this is extremely difficult to do by walking out; therefore, a majority of climbers with HAPE require helicopter evacuation. Sympathetic stimulation through further activity or hiking can lead to further exaggerated pulmonary vasoconstriction, exacerbating symptoms.14–16 Excess exertion with HAPE may contribute to deterioration through increased pulmonary artery pressure, worsening pulmonary edema, and lower oxygen saturations. 17 Therefore, helicopter evacuation from the 4300-m base camp appears to be necessary and lifesaving for HAPE patients on Aconcagua.
Acetazolamide Use
Acetazolamide has been shown to prevent AMS.10,18,19 Our data showed a borderline association between climbers taking acetazolamide and HAPE (P=.05). Although the intent of our surveys was to inquire about prophylactic acetazolamide use, some climbers may have reported taking a self-treatment dose of acetazolamide because many in the HAPE cohort also had unresolved symptoms of AMS. This differentiation presents an area of future possible research. Acetazolamide has not been shown to prevent HAPE in climbers, and it is not recommended for HAPE prophylaxis. 4 However, we are not aware of studies finding that acetazolamide may predispose a climber to HAPE. This finding was not statistically significant in this study but warrants further targeted evaluation while controlling for potential confounding variables. A larger study or more specific survey question could better ascertain how acetazolamide is associated with HAPE on Aconcagua.
Upper Respiratory Illness
Climbers with recent upper respiratory tract illness symptoms (44%) showed no increased likelihood of developing HAPE in this study. A larger sample size or study specifically targeting this variable could be beneficial to assess this risk factor more adequately. The underlying inflammatory process during a respiratory infection may contribute to the overall proinflammatory state and a lower threshold for capillary leakage that also occurs in HAPE. In turn, the physiologic processes could be additive. 8
Limitations
Primary study limitations include the brief time period allotted for the survey while the research group was present on the mountain. Despite this limitation, due to large climber numbers on Aconcagua, 17 climbers with HAPE were able to be enrolled. An increased sample size in a future study could improve the power to detect small differences. Additionally, questions regarding acetazolamide could have been more specific to the indications for use, including dosage and timing. Finally, the study relied on climber reporting of information, which could introduce inaccuracies and potential bias because the climbers were discussing with doctors who they could have perceived as potential barriers to proceeding on their climb if they didn’t answer questions a certain way, although precautions were taken to keep surveys independent of the APP medical screening process.
Conclusion
This study examined climbers who developed HAPE while on Mount Aconcagua over a 3-wk period in January 2024 and yields specific information for high altitude illness prevention efforts and areas needing further study. This study reaffirms the importance of an appropriate acclimatization plan in preventing high altitude illness. Five nights at the 4300-m camp were found to be protective against HAPE, although gradual ascent to and above this elevation is also prudent. Unresolved AMS is a red flag for progression to HAPE, and our findings are consistent with the current recommendations to caution against ascent when symptoms of AMS have not resolved completely. Acetazolamide use was not significantly associated with HAPE but appears to warrant further study on Aconcagua. Certain variables such as the benefit of staged ascent and upper respiratory tract illness symptoms could be explored further with a larger sample size. Hospitalization of patients was not tracked but could be informative for outcomes in a future study.
Footnotes
Acknowledgments
The authors thank Aconcagua Provincial Park and the park medical service for their assistance. Specifically, we thank Pablo Perello, chief ranger of the high mountain zone, Aconcagua Provincial Park, for his assistance with background information. Roz King and Miles Lambertson at the University of Vermont assisted with study design and preparation. Peter Callas at the University of Vermont assisted with statistical analysis.
Author Contribution(s)
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the University of Vermont.
Correction (February 2025):
This article has been updated with minor style corrections since its original publication.
