Abstract
Introduction
The sharpened Romberg test (SRT) is a physical maneuver that has been used to identify ataxia in individuals in resource-limited settings. Previous research has suggested that performance on balance testing may be affected by hypocapnia. In this study, we sought to determine whether acute hyperventilation-induced hypocapnia affects performance on the SRT at 501 meters above sea level.
Methods
We recruited 22 healthy subjects. Each subject performed a baseline SRT. Subjects were then asked to hyperventilate to the point of hypocapnia, confirmed by measurement with a capnometer. Subjects were then asked to re-perform SRT. The primary endpoint was time to loss of balance, measured as time-to-stepout.
Results
Time-to-stepout (TTS) on SRT at baseline had a mean ± standard deviation of 101 ± 117 s. In the hypocapnic condition, TTS was reduced to 48 ± 68 s. TTS normalized to 121 ± 132 s after recovery to normal capnic levels. Time-to-stepout was found to be significantly shorter in the hypocapnic measurement compared to the baseline measurement (P = .0128). Statistical analysis was conducted using one-tailed, paired sample T-tests using a P-value of < .05.
Conclusions
Our study found a statistically and clinically significant reduction in performance on a balance test (SRT) when exposed to acute hyperventilation-induced hypocapnia compared to a eucapnic control. Our results suggest that acute hypocapnia may contribute to neurological dysfunction independently of hypobaric hypoxia.
Introduction
High-altitude illnesses include neurologic and pulmonary syndromes that arise due to maladaptation in the setting of rapid ascent to altitude. The neurological impacts of high-altitude illness may first manifest as benign and self-limiting conditions, such as high-altitude headaches. However, if an individual does not rapidly descend, they may find themselves suffering from conditions such as acute mountain sickness (AMS) or high-altitude cerebral edema (HACE). 1 The rapidly progressive, incapacitating, and potentially life-threatening nature of neurological high-altitude illnesses presents unique diagnostic and treatment challenges.
The 2018 Lake Louise acute mountain sickness score provides an easily administered screening test to diagnose AMS in the field. This score synthesizes self-reported symptoms of headache, gastrointestinal dysfunction, fatigue, dizziness, and functional impairment into a single numerical score, thereby facilitating the diagnosis of AMS and stratifying individuals into mild, moderate, or severe categories. 2 Ataxia is not a sensitive or specific sign of AMS, but it is an important and commonly present feature of HACE. 3 Early identification of HACE is critical in the field, as expeditious descent and evacuation are vital to improving outcomes. Encephalopathy and ataxia are both signs that may be difficult to measure objectively in austere environments, making accurate diagnosis of HACE challenging, especially in its early stages.
Previous research has investigated the SRT as a potential diagnostic tool to identify ataxia in the field. During the SRT, an individual is asked to stand in place, feet placed inline heel-to-toe, arms folded across the chest, knees slightly bent, and eyes closed. He or she is asked to hold this position for as long as feasible, with a test considered “passing” if the individual does not open their eyes, move a hand from the opposite shoulder, or break the heel-to-toe stance for the duration of the test. 4 One study demonstrated the superiority of SRT over conventional heel-to-toe testing to identify ataxia at high altitudes. 5
Hypocapnia is a well-documented consequence of compensatory hyperventilation during acclimatization to high altitude. 6 It has been shown that even a brief duration of hypocapnia can have multiple neurologic effects, including impaired balance, slowed reaction time, and impaired verbal learning.7–10 The mechanism of hypocapnia-related ataxia is likely multifactorial. When an individual experiences hypobaric hypoxia, the cerebral arteries acutely vasodilate, which may cause cerebral edema. 11 In contrast, compensatory hyperventilation in hypobaric hypoxic conditions causes hypocapnia, which causes significant cerebral vasoconstriction.12–14 Cerebral vasoconstriction reduces cerebral blood flow, thereby reducing the risk of cerebral edema. However, if severe hypocapnia causes prolonged cerebral vasoconstriction, then decreased cerebral oxygen delivery can affect brain function and may lead to cerebral ischemia. 15 In summary, hypoxia and hypocapnia have opposite effects on cerebral blood flow, which in most circumstances maintains homeostasis between cerebral vascular tone and cerebral edema. In addition to perfusion-mediated effects on cerebral function, hypocapnia has also been shown to directly affect corticospinal excitability independently of cerebral blood flow. 16
It is possible that mountaineers with varying levels of hypocapnia may experience significantly variable performance on a test of balance, thereby confounding the measurement of underlying ataxia. In our study, we sought to further elucidate this potential association by investigating the effects of acute hypocapnia on the performance of the SRT.
Methods
The study protocol was approved by the Mayo Clinic Institutional Review Board and was carried out at a testing site at 501 meters elevation.
Equipment and Procedures
Subjects were instructed to perform an SRT, wherein an individual stands with feet placed inline heel-to-toe, crosses their arms over their chest with each hand resting on the opposite shoulder, slightly bends their knees, and then closes their eyes (see Figure 1). Subjects were asked to hold this position without opening their eyes, removing either hand from the opposite shoulder, or stepping out of the heel-to-toe foot position for as long as they could. All subjects were afforded practice to become familiar with the test procedure and its proper performance. A stepout was recorded if a subject opened their eyes, removed at least one hand from their opposite shoulder, or stepped out of the heel-to-toe foot position. The elapsed time between assuming the SRT position and stepout was recorded as “time-to-stepout.”
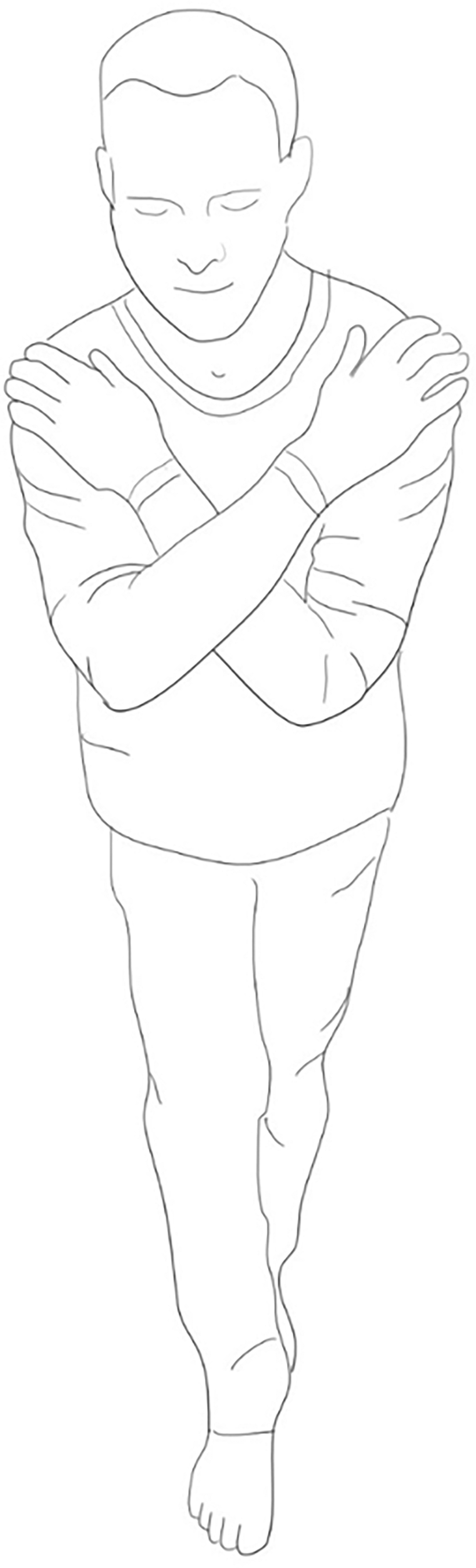
Drawing of the SRT.
Each subject performed an SRT at baseline. Time-to-stepout was recorded for each participant. At the conclusion of the baseline SRT, end-tidal CO2 (ETCO2) was measured over 5 breaths using the Emma Capnograph (Irvine, California). All baseline data were recorded after quiet breathing and no talking for 2 minutes.
To induce acute hypocapnia, subjects were asked to breathe at the maximum possible rate and depth for 2 minutes, an exercise known as maximum voluntary ventilation (MVV). At the conclusion of MVV, end-tidal CO2 was again measured over 5 breaths to quantify the level of hypocapnia. Immediately thereafter, subjects were asked to repeat the SRT, and time-to-stepout was recorded.
Subjects were then allowed to recover to normal capnic status. To aid in rapid recovery, subjects were instructed to place an effervescent electrolyte tablet on their tongue and inhale by mouth to enrich their inhaled air with carbon dioxide. After subjective recovery, the researchers again measured end-tidal CO2 in order to confirm that CO2 levels had returned to normal. The participant then performed a final SRT post-recovery, and ETCO2 was measured at the conclusion of the test.

Statistical Analysis
A one-tailed, paired samples t-test was used to compare time-to-stepout between baseline measurements and post-hyperventilation measurements. Data were reported as mean ± 2*SD.
This study used a P-value of <.05 to determine statistical significance and a power of 0.90. The effect size on performance on the SRT was estimated to be a 10% decrease in SRT time, as informed by previous studies on athletes’ centers of pressure and postural sway. These previous studies were used as an allegory for time-to-stepout on the SRT.
Results
A sample of 22 subjects (7 men, 15 women) was enrolled. Subject demographics were age for both sexes (33 ± 7 years, range 21–55), male age (36 ± 12 years), female age (31 ± 6 years), height (172 ± 10 cm), weight (80.4 ± 23.7 kg) and BMI (26.9 ± 6.8 kg/m2). There was no significant difference in age, BMI range, or mean values by sex. The subjects were all nonsmokers.
Time-to-stepout was 101 ± 117 s in the baseline measurement, 48 ± 68 s in the hypocapnic condition, and 121 ± 132 s in the post-recovery measurement.
Time-to-stepout was found to be significantly shorter in the hypocapnic measurement compared to the baseline measurement (P = .0128; Figures 2–4). Time-to-stepout was shown to be significantly longer in the post-recovery measurement compared to the hypocapnic measurement (P = .0002; Figures 2–4).
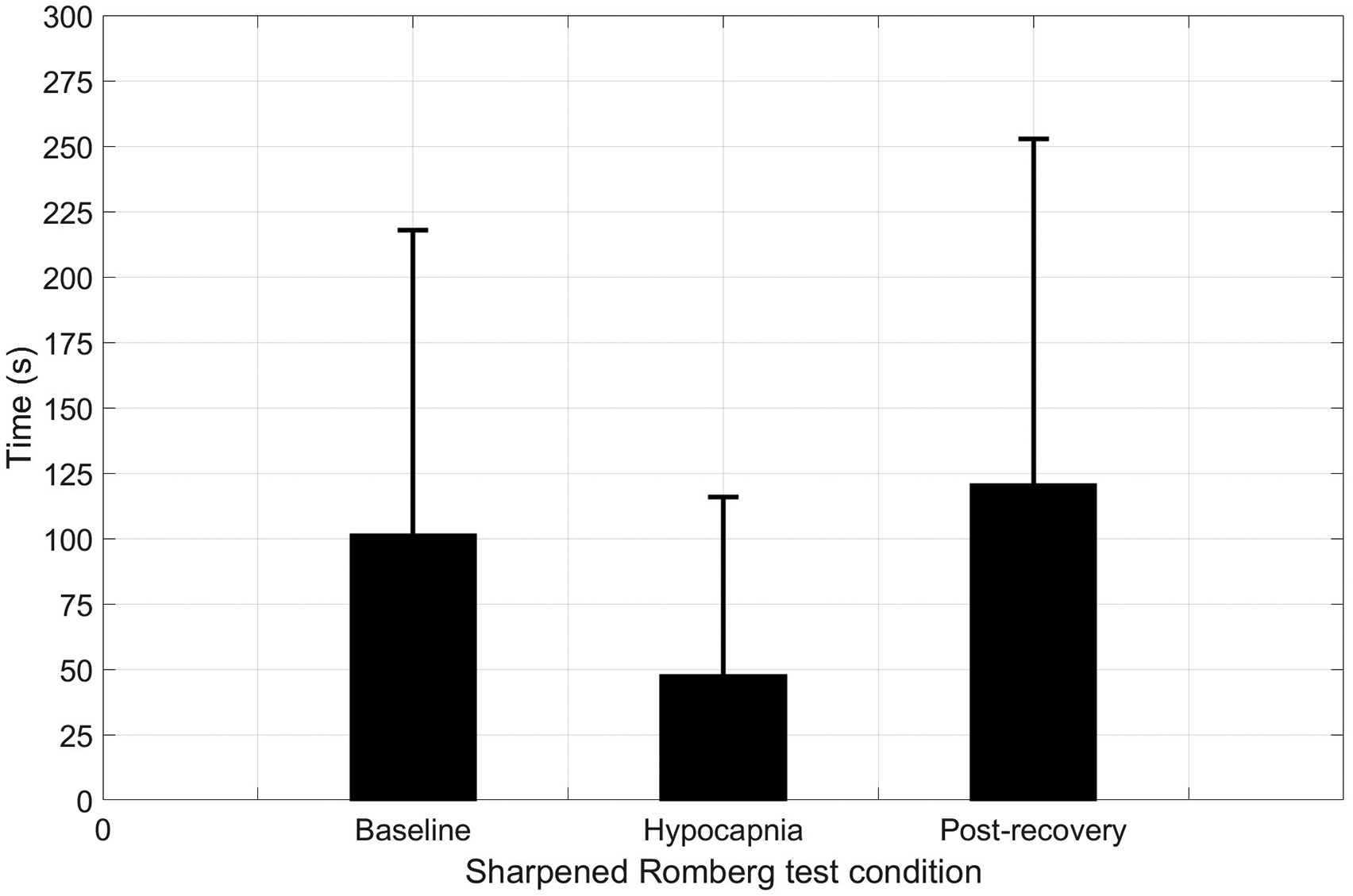
Subjects experienced a statistically significant decline in time-to-stepout (mean ± SD) compared to baseline after hyperventilation, as well as a statistically significant return to baseline after subjective return to normocapnia.
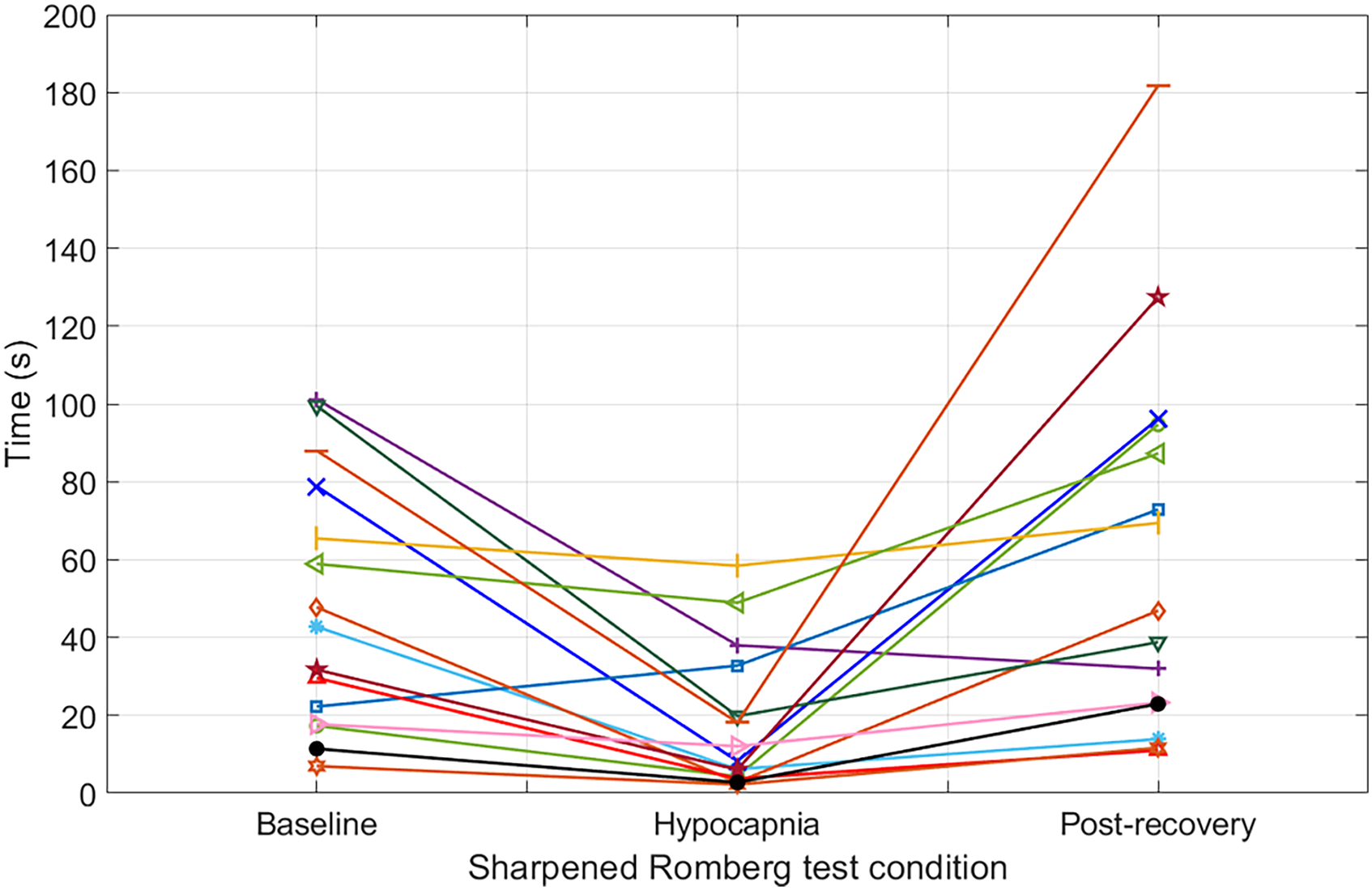
Spaghetti plot detailing the results of the SRT of all tested individuals with baseline time ≤100 s.
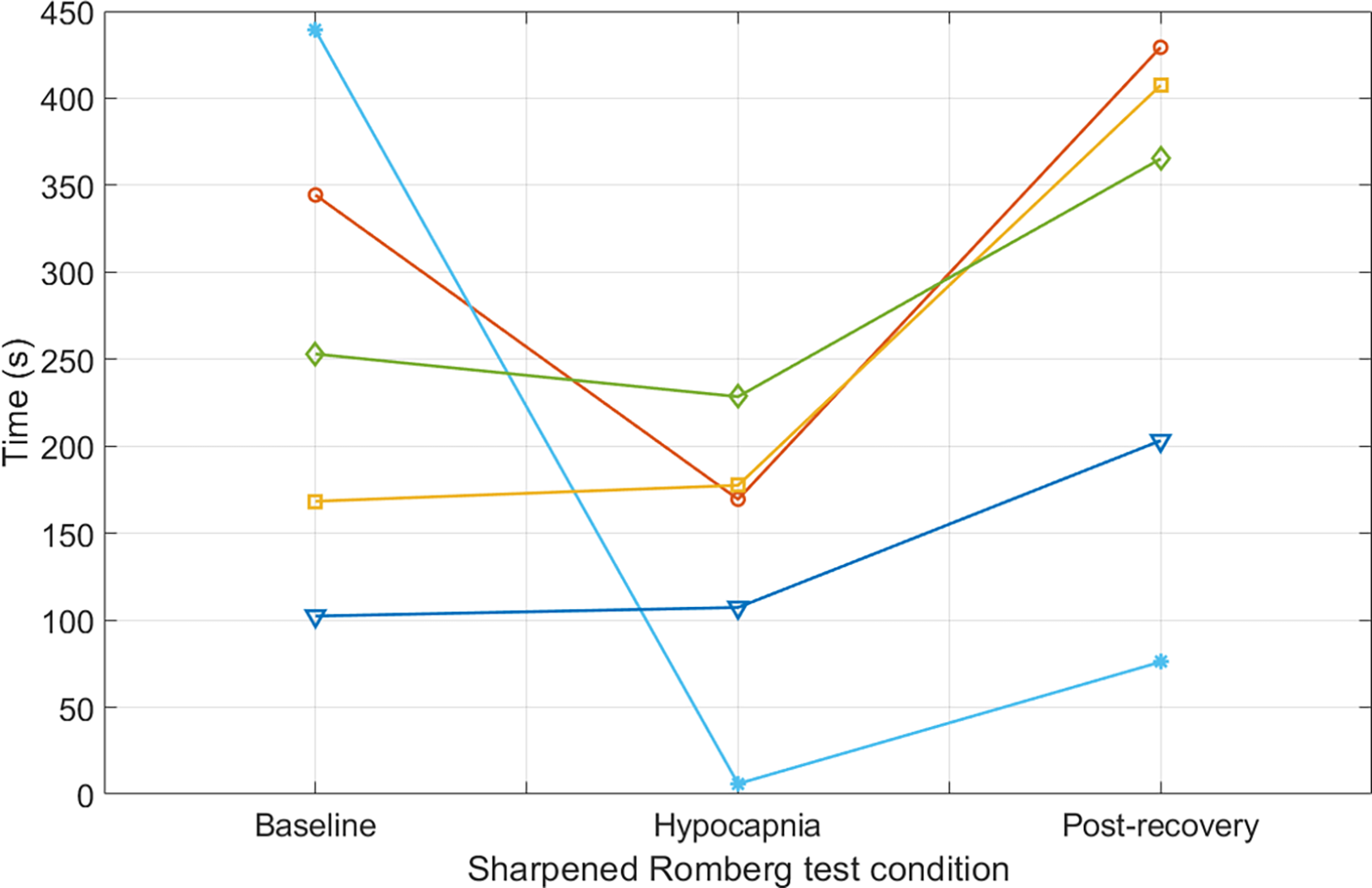
Spaghetti plot detailing the results of the SRT of all tested individuals with baseline time ≥100 s.
Discussion
In this study, we tested the effects of hypocapnia on standing balance in 22 healthy volunteers. Subjects were asked to complete an SRT at baseline, then again after inducing acute hypocapnia through hyperventilation, and a third time after restoring eucapnia. We demonstrated that acute hypocapnia significantly worsened performance on the SRT, suggesting that individuals may experience hypocapnia-related disruption in standing balance in the absence of altitude illness or hypoxia. Notably, performance on the SRT returned to baseline rapidly after restoring eucapnia through the inhalation of supplemental carbon dioxide.
Individuals at high altitudes may experience varying levels of hypocapnia based on myriad physiologic factors such as base level of fitness, rate of ascent, prior history of altitude illness, and compensatory increase in ventilation in response to ambient reduction of barometric pressure. 17 Therefore, individuals at altitude are likely to score variably on both end-tidal capnography and the SRT, regardless of whether they are suffering from altitude-related illness. These confounding factors illustrate the difficulty of diagnosis and treatment of altitude-related neurological disorders in the field.
The clinician caring for mountaineers at elevation should be keenly aware that the mechanisms affecting neurocognitive functioning are an interplay between environmental conditions, such as reduced partial pressure of oxygen at altitude causing hypobaric hypoxia, and the physiologic response of the body, such as compensatory hyperventilation resulting in hypocapnia. Pre-ascent administration of carbonic anhydrase inhibitors, such as acetazolamide, should be considered in individuals at high risk of altitude-related illness to maintain capnic balance during acclimatization. Individuals with clinically significant hypocapnia may be administered supplemental carbon dioxide in an effort to restore capnic homeostasis. We utilize CO2 emitted from a dissolving effervescent tablet administered orally to provide a safe, lightweight, and readily available source of carbon dioxide in the backcountry.
Limitations
We recognize the limitations in translating our study directly to austere environments. Our study was conducted at 501 meters above sea level, which does not reflect the conditions of hypobaric hypoxia that would be encountered at high altitudes. Secondly, our subjects were exposed to acute hypocapnia, while individuals at altitude commonly experience hypocapnia due to subacute or chronic hyperventilation. Additionally, athletes at high altitudes commonly experience significant sleep deprivation and fatigue, which may further impede their ability to perform tasks and/or testing. We did not account for these potential confounding variables in our study.
One additional potential limitation of our study was bias due to learning effect. As subjects completed sequential SRT, they could have gained additional practice with each sequential test, potentially becoming more skilled at performing the test with each subsequent repetition. We attempted to control for this variable by standardizing practice time for all subjects prior to testing. Another limitation was the wide standard deviation in all three conditions, indicating notable variation in performance on SRT. This may be due to natural differences between participants in their balance and ability to effectively learn SRT within the time limit of the experiment.
Conclusions
Our study demonstrates that healthy volunteers performed significantly worse on the SRT when exposed to acute hyperventilation-induced hypocapnia compared to a eucapnic control. Our results suggest that acute hypocapnia may contribute to neurological dysfunction independently of hypobaric hypoxia. Physicians caring for individuals with altitude-related neurologic syndromes should consider hypocapnia in their differential diagnosis as an important contributing factor in diagnostic and management decisions.
Footnotes
Acknowledgments
We gratefully acknowledge the assistance and thoughtful review of the paper by Michael Cevette, PhD, neurovestibular and balance specialist and co-director of the Aerospace Medicine & Vestibular Research Laboratory (AMVRL) at Mayo Clinic in Arizona.
Author Contribution(s)
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Drs. Stepanek and Pradhan are inventors on USPTO patent number 11529492 (methods and materials for treating hypocapnia).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded entirely by Mayo Clinic institutional funding made possible by the Burns Family Philanthropic Fund.
