Abstract
Okinawa prefecture is at risk for emerging infectious diseases due to its subtropical climate and its location within the Indo-Pacific region. Understanding the existing vectors and infectious agents contextualizes current threats, guides treatment, and informs prevention, and may be of unique concern in the setting of complex emergencies.
Introduction
Okinawa prefecture is the southernmost province of Japan. The Okinawa Archipelago consists of 113 islands over approximately 550 square miles. The prefecture is located near the southernmost island of mainland Japan (Kyushu), Taiwan, Vietnam, the Philippines, and China. Its humid subtropical climate interspersed with tropical rainforest has not impeded development with the bulk of its 1.4 million population residing on the Okinawa main island.
Its climate and location within the western Pacific Ocean make Okinawa ripe for an array of infectious tropical diseases. The relative absence of below-freezing temperatures allows vectors to live year-round, and intermediate reservoirs of disease may exist throughout the year. The prefecture's location makes it particularly susceptible to typhoons and post-typhoon flooding with standing water. To combat this, the government of Japan implemented a parasite elimination program in the late 19th and early 20th century resulting in a dramatic reduction in environmental infectious diseases throughout the prefecture. 1 These control measures, in conjunction with vigilant import and customs practices, effectively eliminated malaria, filariasis, and a host of other parasitic diseases. 2 Despite these efforts, a few key infectious agents, such as leptospirosis,3,4 Strongyloides, 5 and Japanese encephalitis virus (JEV), remain endemic. 6
Recent post-pandemic increases in international tourism 7 may introduce infectious agents into the prefecture. A previously described climate risk factor (crf) index was high in Japan and at particular risk for climate change. 8 The CRF is a validated, predictive model based on temperature, humidity, and precipitation. It is specifically designed for dengue transmission, though it can be used as a proxy estimate of diseases with similar vectors (mosquitoes).
Similarly, geopolitical challenges in the region may worsen infectious disease threats. Natural and human-made disasters may enhance these risks. These disasters could range from drought 9 to typhoons (tropical cyclones), tsunamis, and armed conflict. Understanding the existing vectors and infectious agents contextualizes current threats, guides treatment, and informs prevention, and may be of unique concern in the setting of complex emergencies.
Current Threats
Leptospirosis
Leptospirosis is a disease caused by spirochete bacteria found in freshwater throughout Okinawa. Leptospirosis generally persists in an animal host (often rodents) within the proximal renal tubules. 10 Animals excrete the bacteria through their urine, which can contaminate soil and water sources, exposing humans. Specifically, exposure to freshwater sources, including swimming and kayaking, can expose individuals to leptospirosis. Leptospira grow better in warm, humid, and rainy environments, and thus Okinawa is an ideal environment for the organism. Between 2006–2022, 267 cases were reported in Okinawa, 4–43 cases per year. 11 Recreational exposure to freshwater accounted for 44% of the cases, and agriculture/labor within rivers accounted for 27% of the cases. 3 Among US military personnel, several outbreaks have occurred. Two notable military outbreaks occurred as part of the Jungle Warfare Training Center in the northern part of Okinawa main island.12,13 In these cases, patients reported swallowing water while in freshwater sources. During these outbreaks, servicemembers who were exposed to the contaminated water but did not ingest it appeared to not be at risk for subsequent infection. Within Japan (including Okinawa), there appears to be a correlation of infections following typhoons. 14 Increased typhoon activity with climate change may worsen leptospirosis throughout the prefecture. Typhoons increase this risk by the relative increases in standing water, wading in water, and contamination of freshwater sources.
Leptospirosis is an acute febrile illness with a variety of symptoms ranging from a mild form with only common coldlike symptoms to a severe form (Weil's disease) with jaundice, bleeding, and kidney damage. Early symptoms can often be confused with a variety of other tropical diseases. Two severe and notable complications include acute renal failure and pulmonary hemorrhage. 15 Preferred laboratory diagnosis can be made with PCR assays and serologic testing with microscopic agglutination test (MAT). Serologies are based on serum IgG and may have false negative results early in infection. Strong consideration should occur for retesting 7–14 days after symptom onset due to delayed IgG production. Treatment with penicillins, cephalosporins, and tetracyclines is preferred, as well as supportive care for severe cases.
Strongyloides
Strongyloides is a genus of parasitic nematode worms that is common in the tropics and is endemic to Okinawa. Strongyloides have a unique lifecycle, where the filariform larval stage exists freely in the soil until it penetrates the skin of a host organism. The larvae then travel through the blood, into the lungs, and cross the alveoli. They travel up the trachea and the larynx, where they are swallowed by the host. They are able to produce eggs once in the GI tract that are then excreted with feces in the soil and drinking water. 16 Autoinfection can occur when the rhabditiform larvae mature into filariform larvae within the host. This autoinfection can make eradication difficult.
Up to 64% of infected patients are asymptomatic. 17 Okinawa has a high human T-cell lymphotropic virus type 1 (HTLV-1) infection rate. HTLV-1 in Okinawa has a strong association with Strongyloides, and coinfection is associated with an increased rate of accelerated autoinfection. Prevalence throughout Japan remains fairly low, with a reported 15.5 case incidence per year.5,18 Common symptoms include rash of the exposed skin, respiratory symptoms (a Loeffler's-like syndrome), fever, diarrhea, vomiting, and epigastric pain. Diagnosis can be made with microscopic analysis of the patient's stool and serology for filariform larvae antigen IgG. 19 Ivermectin is generally used for the treatment of Strongyloides infection. Fortunately, this is generally a problem for immunocompromised patients and elderly patients who were infected in earlier periods with poor sanitation.
Japanese Encephalitis Virus
Japanese encephalitis is a mosquito-borne flavivirus. It is a single-stranded RNA virus, similar to other flaviviruses such as West Nile virus, Zika virus, dengue virus, and Yellow fever. It is transmitted primarily by Culex mosquitoes, with alternative zoonotic hosts of domesticated pigs, 20 though other vertebrates can be hosts, including wading birds. Culex mosquitos are endemic to the region, and Okinawa has a large population of pig farms that act as a reservoir of JEV. Wild boars similarly appear to be large hosts of the virus. 21 Annual cases in Japan remain low, though widespread vaccination campaigns have been helpful in reducing the rate of new infections. 22 Worldwide, there are approximately 68 000 cases annually. 23
Neurologic sequelae from the virus are perhaps the most notable features of JEV, including altered consciousness, headache, hyporeflexia, seizures, flaccid weakness, and abnormal brainstem reflexes. JEV should be considered in patients with acute encephalitis in the endemic regions. Diagnosis of JEV classically involves the presence of JEV-specific IgM in the CSF by ELISA. Serum analysis is unreliable but can be considered in patients where CSF is unable to be obtained.20,24 No specific treatment for JEV exists, and thus care is supportive in nature. Prevention with widespread vaccination appears to be effective as well. The JEV vaccine appears to be extremely effective after at least 2 doses. 25 The aging population within Japan was historically vaccinated with inactivated JE vaccine, and there appears to be waning immunity. Vaccine rates dropped in the late 2000s following reports of post-vaccination events. 26 Increased vaccination rates in nearby Asian countries recently may have decreased the global burden of disease. 27
Widespread vaccination programs in Japan, including Okinawa prefecture, have resulted in low rates of infection within the country, especially when compared with nearby countries. 28 The last publicly available published data had 3 cases throughout Japan in 2021 and no cases in Okinawa prefecture. 28 Recent mosquito sampling within the prefecture resulted in no JEV identified within the sample, suggesting a very low prevalence. 29 Environmental change within the prefecture may contribute to the decreases in JEV—specifically, changes in farmland from rice paddies to sugar cane, as well as the increased overall urban development of the island. 29
Interestingly, JEV strains in Okinawa prefecture have seen a recent change in antigenicity and genotype. 30 This shift demonstrates the possibility of the introduction of new and emerging infectious diseases into Okinawa from nearby countries. While JEV has persisted endemic in the region, new strains may have been transmitted by wind-blown mosquitoes, birds, or even traveling humans. This suggests other infectious diseases may similarly move through the region as a conduit from Southeast Asia to Japan and beyond.
Recent Infectious Disease
Okinawa prefecture may be at risk of emerging infectious diseases due to its subtropical climate and its location within the Indo-Pacific region. Several infectious diseases have emerged over the last 2 decades in Okinawa, including COVID-19 (SARS-CoV-2), Japanese spotted fever, and scrub typhus. COVID-19 spread worldwide and provided interesting modeling for Okinawa prefecture, which required the introduction of the virus via either ferry or air travel. An interesting additional component was the influence of the US military on the predictive models of COVID-19 infection within the prefecture. 31 The proximity of the prefecture to other areas of East Asia makes the rapid spread of emerging infectious diseases possible. Understanding some of the recent infectious disease additions may help predict future risks.
Japanese Spotted Fever
Japanese spotted fever (JSF) is a rickettsial (Rickettsia japonica) tick-borne disease endemic to Japan and Okinawa prefecture. JSF was first identified in 1984, and the first case in Okinawa prefecture was in 2001.32,33 Along with scrub typhus, JSF infections have been suggested to be increasing due to climate change and increasing temperatures within Japan. 34 Nationwide, JSF generally had <10 cases/year in the early 1990s, with a steady increase to over 250 cases/year recently. 35 Typhoons or tropical cyclones worsened by climate change can contribute to epidemics of rickettsial disease, JSF, and dengue within the region. 36 Because JSF is a tick-borne disease, exposure to tick-laden areas makes patients susceptible. This includes agricultural areas and the many forested areas within Japan. 37 Increased recreational activity in forested areas, especially among elderly populations, may have led to increased exposure.
JSF is caused by patients being bitten by an infected tick. Symptoms generally develop 2–8 d after infection. Similar to other rickettsial diseases like Rocky Mountain spotted fever (Rickettsia rickettsii) and epidemic typhus (Rickettsia prowazekii), JSF symptoms include fever, erythema, and eschar from the tick bite. 33 Severe complications can include disseminated intravascular coagulation (DIC), organ failure, meningoencephalitis, respiratory distress, and death. The definitive diagnosis is made based on R. japonica PCR or from R. japonica serology. Other markers may include leukocytosis or leukopenia, thrombocytopenia, elevated C-reactive protein, and elevated liver enzymes. 33 Tetracyclines are the preferred treatment.
Scrub Typhus/Tsutsugamushi Disease (Orientia tsutsugamushi)
Scrub typhus (or tsutsugamushi disease) is a bacterial infection caused by Orientia tsutsugamushi, an obligate intracellular gram-negative bacteria. It is transmitted by the chigger mite, Leptotrombidium deliense (Figure 1). The chigger mite and larvae generally live in moist grassy or thick vegetation environments, which is why O. tsutsugamushi infection is known as scrub typhus. Scrub typhus has been endemic to the main Japanese island of Honshu since at least the 1800s.
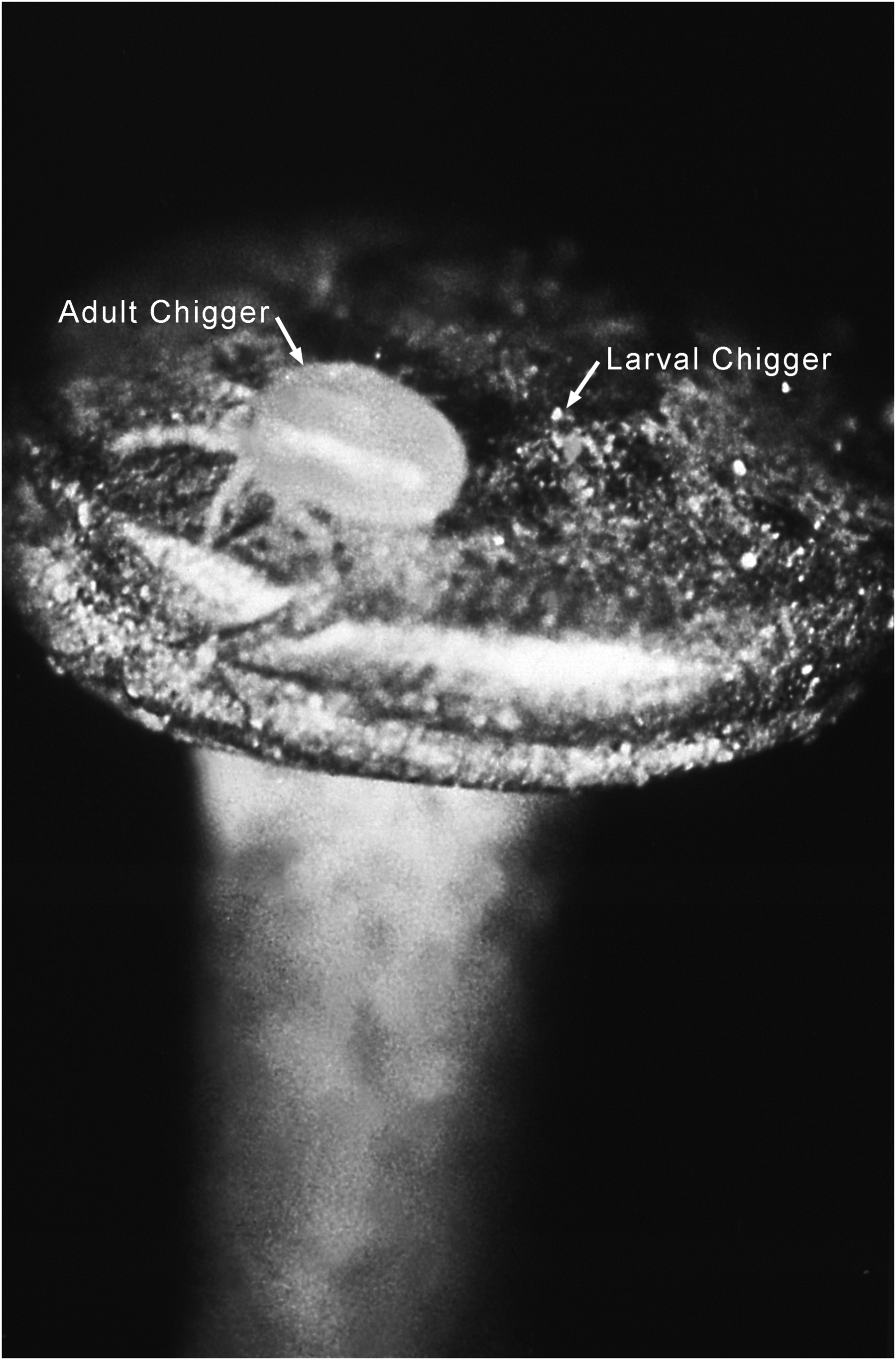
Adult and larval chigger. Image courtesy of Cornelius B. Philip, PhD, of the National Institute of Allergy and Infectious Disease (NIAID) [Leptotrombidium deliense]; https://phil.cdc.gov/.
Historically, scrub typhus was not present in Okinawa. Scrub typhus was present in nearby Taiwan, at least prior to World War 2. Japan saw decreases in annual cases in the mid-to-late 20th century (appr. 10 cases per year), but this increased to an average of 400 cases annually over the last 30 y. 38 The low case numbers in the mid-century have been attributed in part to high rates of tetracycline and chloramphenicol use for febrile disease, 38 while the increases in case numbers recently have alternatively been attributed to increased outdoor recreation among the elderly population. 38
Okinawa notably saw its first case in 2009. Genetic analysis of these cases (mostly from Miyakojima in Okinawa) revealed 3 O. tsutsugamushi strains present more typically in Taiwan (Gilliam, Saitama, and Karp variants). 39 It is unknown when the vector was introduced to the region, but after the case in 2009, field research identified the presence of the chigger in Ikema island (part of Miyakojima). This is concerning—not only because new diseases are present but because continued infections suggest that the vector persists and may now be endemic.
Climate change and other meteorologic features appear to play a role in the rates of scrub typhus, primarily through changes in vector abundance. The major determinants appear to be temperature and rainfall, though humidity, sunshine, pressure, and wind speed all contribute. 40 From 2009 until 2022, 44 cases were reported, with 0–10 per year. Among 44 cases, 28 were males (64%), 16 were females (36%), and 40 cases(91%) were above the age of 50. 41
Symptoms of scrub typhus include fever, rash, and eschar formation. Severe symptoms, including organ failure, can occur. Prior to antibiotic use, case fatality rate was greater than 30%. Diagnosis historically involved serology (including the Weil-Felix agglutination reaction) but now may involve DNA amplification with loop isothermal amplification (LAMP) or PCR. 42 In Japan, minocycline is the preferred treatment.
Angiostrongylus cantonensis (Rat Lungworm)
The parasitic nematode A. cantonensis, or rat lungworm, can be pathogenic in humans. This can cause eosinophilic meningitis, with potentially fatal outcomes. Initially identified in China in 1935, the nematode's definitive host is the rat, where larvae are spread via feces. The larvae are ingested by snails, where they further develop, and are eaten by rats, where they develop into adults. Humans may be infected following consumption of the intermediate host (the snails or other possibly infected hosts). 43 The giant African land snail has been implicated in spreading A. cantonensis throughout the world. 44 This snail was introduced to Okinawa and was implicated in a 2000 epidemic of rat lungworm infection in the prefecture. 45 During this epidemic, it appeared to be spread from contaminated vegetables, as opposed to consumption of the snails themselves.
Patients with signs of meningitis who traveled to an endemic area should be evaluated for A. cantonensis infection. Diagnosis can be assisted with a lumbar puncture and cerebrospinal fluid analysis. Presence of larvae is diagnostic, though PCR can be used if no larvae are identified. MRI of the brain can be helpful to aid diagnosis as well, though it has low sensitivity, especially early in infection. 46 Treatment involves corticosteroids and anti-helminthic medications.
Future Infectious Disease Threat
Okinawa prefecture is at risk for emerging infectious diseases. Its location and environment allow for potential regional spread. This risk may increase with climate change, as seen recently with scrub typhus and JSF. Another potential risk may include complex disasters that have disrupted public health efforts over the last 70 years.
If viewed as a major health and humanitarian disaster, the Battle of Okinawa serves as a valuable case study in public health. This period—marked by limited sanitation and public services, displaced populations, and malnutrition—saw tremendous numbers of disease-related illnesses among US and Japanese troops as well as Okinawan locals. The humid subtropical climate, as well as endemic vectors and diseases, only compounded the man-made destruction wrought on the islands and their inhabitants.
Of the total 548 000 US military forces over the 81-d campaign, there were an estimated 26 000 nonbattle casualties. 47 The Allied Pacific campaign battled parasitic infections, scrub typhus, dysentery, malaria, and other tropical diseases. An account of Okinawan civilians forced to evacuate in the Yaeyama region during June 1945 estimates that over 16 000 people contracted malaria, with over 3600 deaths attributed to the disease. 48 Malaria and other tropical diseases are particularly damaging during humanitarian crises when populations are displaced. People sharing limited shelter in crisis-riddled lands increase the transmissibility of such mosquito-borne illnesses. This is to say nothing of this vector's proliferation in the standing water made abundant from the destruction of civic infrastructure. Malaria was later eradicated from the region in 1962 after combined US and local efforts; however, the prefecture is still home to several known vectors of Plasmodium, filarial disease, dengue, and JEV.
The experience during the Battle of Okinawa demonstrates how robust malaria control in peacetime does not necessarily equate to adequate control following war or disasters. At the start of the battle, there were almost no cases of malaria in Okinawa, with only 8 positive samples in over 2200 tested patients. 49 Several months into the battle, approximately 10% of the civilian population tested positive for malaria. Following the battle, DDT use was widespread on the island, and the number of malaria decreased significantly. With the anopheles mosquito (Figure 2) currently endemic in the prefecture, malaria outbreaks remain possible.

Anophelese mosquito. Image courtesy of Walter Reed Biosystematics Unit, 2023 [Anopheles gambiae]; https://wrbu.si.edu/.
There was a similar experience with dengue fever during the battle of Okinawa, with a similar rise in cases, followed by a fall after mosquito control was implemented. 50 Tokyo (almost 1000 miles to the north of Okinawa) experienced a dengue outbreak in 2014. The outbreak was centered around Yoyogi Park, a large urban park within the city. 51 This was unusual in Japan, with the last dengue outbreak in 1945 during the war. Several causes have been suggested, including climate change. The relative vectorial capacity (rVc) is an indicator of dengue epidemic potential, 52 and the summer of 2014 had a particularly high rVc, correlating well with the Tokyo outbreak. Temperature, precipitation, and rVc (including the presence of Aedes mosquitos) may continue to place Japan at risk of epidemics. 53 Increased temperature and humidity with climate change may worsen over the next several decades, leaving more areas of Japan susceptible to dengue outbreaks.
Okinawa remains at risk for outbreaks and even a full transmission cycle, with A. albopictus (Figure 3) mosquitoes able to survive throughout the year in the Okinawan climate. 54 As is the case with malaria, disaster-related changes in the environment and mosquito control may dramatically impact disease transmission within the prefecture. Potentially, a variety of other diseases could spread with changing climate and humanitarian disasters. Flaviviruses, including Zika, West Nile, and yellow fever, could reasonably be transmitted within the prefecture via the Culex mosquito (Figure 4). Chikungunya virus, filariasis, influenza, and novel viruses could all potentially spread during disasters.

Aedes mosquito. Image courtesy of Walter Reed Biosystematics Unit, 2023 [Aedes albopictus]; https://wrbu.si.edu/.
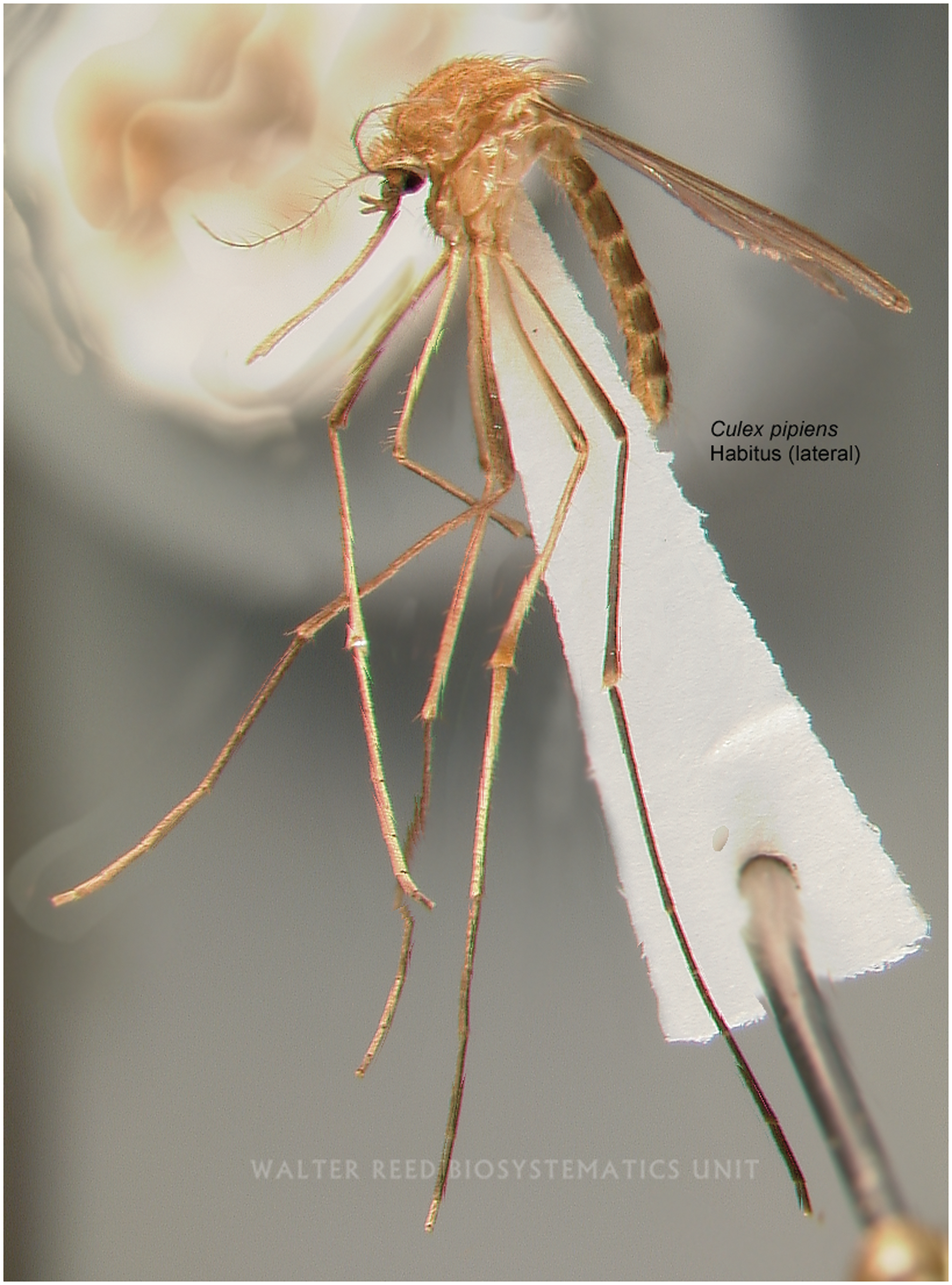
Culex mosquito. Image courtesy of Walter Reed Biosystematics Unit, 2023 [Culex pipiens]; https://wrbu.si.edu/.
Conclusions
Okinawa prefecture has several endemic infectious diseases important to individuals traveling to the area, especially with plans to spend significant time outside modern structures. Several emerging infectious diseases have been introduced recently to the prefecture, including Japanese spotted fever and scrub typhus. Ongoing climate change has been suggested as a possible cause for this and may potentially worsen with time. Additionally, wartime experiences have demonstrated the potential for worsening tropical diseases like dengue and malaria during humanitarian crises. The presence of compatible mosquito vectors within the prefecture makes this a distinct possibility.
Strong consideration should be made for future public health countermeasures against these potential threats, including during times of humanitarian disasters. These can include the eradication or reduction of vectors for vector borne disease, increased coverage of vaccination (and vaccine development), and education and avoidance of behaviors that increase the risk of disease transmission. Understanding the potential future threats for tropical diseases within the prefecture may help prepare for and mitigate the risk of future epidemics.
Footnotes
Author Contribution(s)
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
