Abstract
The Wilderness Medical Society (WMS) convened an expert panel in 2011 to develop a set of evidence-based guidelines for the recognition, prevention, and treatment of heat illness. The current panel retained 5 original members and welcomed 2 new members, all of whom collaborated remotely to provide an updated review of the classifications, pathophysiology, evidence-based guidelines for planning and preventive measures, and recommendations for field- and hospital-based therapeutic management of heat illness. These recommendations are graded based on the quality of supporting evidence and the balance between the benefits and risks or burdens for each modality. This is an updated version of the WMS clinical practice guidelines for the prevention and treatment of heat illness published in Wilderness & Environmental Medicine. 2019;30(4):S33–S46.
Introduction
Heat illness is a common occurrence worldwide. The European heat wave of 2003 resulted in at least 70,000 fatalities. 1 From 2008–2014, the United States averaged over 700 deaths annually associated with excessive heat exposure. 2 Acute heat surges caused 700 excess deaths over 10 days in Chicago in 1995 3 and 600 deaths in the states of Washington and Oregon during a single week in 2021. 4 Heat illness is the leading cause of morbidity and mortality among US high school athletes, 5 while also causing excess cardiovascular deaths in those over 65 years of age. 3 Large-scale studies of heat stroke outcomes show findings that are variable but grave, with an overall case fatality rate of 5% while survivors have a 16% chance of poor neurologic outcomes, defined as a modified Rankin score ≥4.6,7 For patients presenting with hypotension, mortality increases to 33%. 8 Outcomes worsen with increasing magnitude and duration of hyperthermia,9–11 making early recognition and treatment priorities.
The Wilderness Medical Society (WMS) assembled an expert panel in 2011 to develop a set of practice guidelines for the recognition, prevention, and treatment of heat illness. This is an updated version of the WMS Practice Guidelines for the Treatment and Prevention of Heat-Related Illness originally published in Wilderness and Environmental Medicine in 2013 12 and most recently updated in 2019. 13 We present a review of the classifications, pathophysiology, and evidence-based guidelines for planning and preventive measures, as well as best practice recommendations for both field and hospital-based therapeutic management of heat illness. Though we discuss the full range of heat-related illnesses, we focused mostly on exertional heat stroke (EHS). Throughout these guidelines, discussion of heat stroke will refer to EHS unless otherwise specified. This update provides new details for ice-water immersion treatment in field and hospital environments.
Methods
The WMS chose the original panel members with research expertise in human physiology and clinical expertise in emergency medicine, primary care, and critical care; the panel membership expanded in 2022 to broaden its research foundation. The panel used the 2019 update as its foundation and identified new articles in the PubMed database using the following keywords: hyperthermia, heat stroke, heat illness, heat syncope, and heat exhaustion. The panel then performed a hand search from references in the initial PubMed search. We included case reports, observational studies, and controlled trials; we excluded abstract-only reports. The panel used a consensus approach to develop recommendations for the recognition and management of heat illness, with levels of evidence and strength of recommendation graded according to the classification scheme of the American College of Chest Physicians (ACCP). 14 The panel considered the balance between the burden of evidence and the risks and benefits of treatment in accordance with the ACCP classification scheme (Supplementary Table 1).
Definitions
Heat illness manifests as a spectrum of diseases from minor to severe, including heat cramps, heat syncope, heat exhaustion, and heat stroke. The body and the environment exchange heat through four fundamental mechanisms: conduction (heat transfer from the body to the surrounding environment along a temperature gradient by direct contact), evaporation (heat transfer from the body to moisture resulting in liquid water in sweat and respiratory secretions transitioning to vapor phase), thermal radiation (infrared spectrum radiation between 2 objects of differing temperatures such as sun onto skin), or convection (transfer of heat from the body to free fluids or gas moving across the skin surface). Given that heat is being generated metabolically, the environmental gradients must result in a net loss of heat in order to avoid hyperthermia. Hyperthermia is an increase in core temperature above the normal physiologic setpoint. The exact value of a normal temperature is generally accepted to lie between 36 °C and 38 °C.15,16 To accommodate diurnal variation and individuals’ variability in set point temperature, studies often use the clearly abnormal 40 °C as a threshold for hyperthermia. Even this level of exertional hyperthermia may be well tolerated, rendering no symptoms. One warm-weather study that observed core temperatures of 18 soldiers who ran 21 kilometers in a wet bulb globe temperature (WBGT) of 26.5 °C found all of the subjects were asymptomatic despite having core temperatures >39 °C; 10 subjects were >40 °C. 17 A cool-weather study that observed core temperatures of 227 civilian subjects who ran 15 km in WBGT of 11 °C found that even in these cooler conditions, 15% of participants had core temperatures >40 °C, with none reporting heat-related complaints. 18 Therefore, absolute temperature thresholds may suggest increased risk but are not diagnostic of heat illness in asymptomatic individuals.
The terms used for various types of heat illness may not precisely reflect the underlying pathophysiologic processes, yet they are useful to describe the clinical findings, aid in treatment, predict outcomes, and for epidemiologic purposes.19,20 The term heat cramps describes the clinical phenomenon of involuntary diffuse large muscle contractions associated with exertion in hot environments. It was originally described in 1933 and attributed to electrolyte imbalance. 21 Some research shows that dehydration and electrolyte disturbances are associated with heat cramps, but causation is not clearly established. 22 It is also possible that the phenomenon originally described as cramps has more in common with exercise-associated muscle cramps (EAMC). Given that EAMCs are potentiated in heat but not clearly caused by associated electrolyte disturbances, there is now a unifying hypothesis that altered neuromuscular control is the true underlying mechanism. 23 Heat edema is a benign self-limiting condition that occurs when interstitial fluid accumulates in dependent extremities because of hydrostatic pressure, vascular leak, and cutaneous vasodilation. Heat syncope refers to a transient loss of consciousness with a relatively rapid return to normal neurologic baseline associated with exposure to heat. Contributing factors may include peripheral vasodilation, orthostatic pooling of blood, prolonged standing, advanced age, dehydration, medications, dietary supplements, and coexisting medical conditions that reduce cardiac output, such as ischemic heart disease. Although syncope can accompany more severe forms of heat illness, the term heat syncope generally refers to a relatively mild clinical condition that should resolve with rest and rehydration at comfortable ambient temperatures.24,25 A differential consideration for loss of postural tone during exercise includes exercise-associated collapse, which describes a loss of postural tone with or without loss of consciousness because of impaired baroreceptor reflexes and lower-extremity venous pooling in the absence of hyperthermia or dehydration. 24
Heat exhaustion results from exposure to high ambient temperature or from strenuous exertion. It manifests as a constellation of symptoms that range from uncomfortable to debilitating9,26 and may limit the continuation of exercise in the heat. 9 Symptoms are variable and may include weakness, fatigue, thirst, headache, nausea, dizziness, and muscle aches. 26 Heat exhaustion is a mild-to-moderate heat illness that may progress to heat stroke if untreated in a hot environment, although heat stroke may occur suddenly without a preceding phase of identifiable heat exhaustion. Heat stroke is classically defined as a core temperature above 40 °C (104°F) combined with central nervous system dysfunction (eg, encephalopathy, seizures, or coma). Heat stroke is generally divided into two categories: classic or nonexertional heat stroke (NEHS) resulting from passive exposure to high environmental temperatures, and exertional heat stroke (EHS) resulting from pathologic (nonphysiologic) hyperthermia during strenuous exercise (Table 1).
Categories of heat illness.

Heat Dissipation and Pathophysiology
Heat loss is controlled by 2 sets of mechanisms: peripheral mechanisms in the skin and other organs and, to a greater degree, by the central nervous system via the hypothalamus. A rise in blood temperature by less than 1 °C triggers hypothalamic thermoregulation to increase blood flow to the skin by up to 8 L·min−1 via sympathetic cutaneous vasodilation. The forearm and the glabrous (hairless) skin areas of the palms, soles, and face show up to 6-fold blood flow increases, thus utilizing their unique vascular arrangements that provide countercurrent heat exchange. 27 As blood is shunted to the periphery to facilitate heat loss, renal and splanchnic perfusion is reduced by 30%, which can cause gastrointestinal symptoms such as cramping, nausea, and diarrhea. 28
When the core temperature increases during exercise, the skin is a critical site for heat dissipation. If the skin is kept at an elevated temperature by ambient environmental conditions or clothing, then it cannot dissipate heat normally. This results in impaired cooling at first, then as skin temperatures rise further, the skin may begin to heat, not cool, the body. When metabolic heat production outpaces heat dissipation, core temperature rises and heat illness can occur. Heat stroke occurs when core temperature rises above a critical level, leading to a cascade of cellular and systemic responses. These responses include thermoregulatory dysfunction, an acute phase response, and a heat shock protein response. The acute phase response to heat stress involves an inflammatory reaction of interleukins, cytokines, and proteins that progresses in a similar sequence as that seen in sepsis. Prognostic scores first developed for inflammatory response and sepsis have correlated with fatality risk in heat stroke. 7 Exaggerated acute phase reaction and inflammatory responses mark the progression from heat stress to heat stroke, possibly incited by hypoperfusion of the gastrointestinal tract. Increased mucosal permeability from inflammatory mediators may allow endotoxins from the gut to enter the systemic circulation. The resulting release of lipopolysaccharides and similar compounds can be mitigated by prior heat acclimatization. Contributing more than just metabolic heat to this pathophysiologic cascade, skeletal muscle strained by heat and exercise releases increased amounts of myokines and other inflammatory mediators. 29 The resulting endotoxemia and cascade of inflammatory cytokines lead to alterations in the microcirculation, further endothelial and tissue injury, and impaired thermoregulation, precipitating heat stroke and hypotension. 30 An overlapping hypothesis presupposes that at a similar threshold temperature, the expression of protective heat-shock proteins is altered, decreasing their ability to prevent thermal denaturation of structural proteins and enzymes that start to fail at a cellular level, with ensuing end-organ dysfunction.31,32 At critical levels of hyperthermia, heat causes direct tissue injury and death via apoptosis or necrosis. The severity of the injury is determined by the level and duration of thermal stress.33,34 This complex constellation of overlapping events leads to thermoregulatory failure, heat stroke, and circulatory shock.
Prevention and Planning
Heat illness is a potentially fatal condition, yet the risk can be greatly reduced through low-tech, low-cost preparation and planning. Risk reduction begins with risk assessment. Structured risk assessments can be developed and validated for population-level use35,36 or for a specific situation considering the individual's physiology, the environment, and the planned activity. 37 Because some risk factors are fixed in any given scenario, planners must identify and focus on the most modifiable factors available.
Individual Factors
The most powerful strategy for the prevention of heat-related illness is acclimatization. As few as three exposures of 90 min duration over the course of one week can reduce physiologic strain by approximately 20%, although individual responses vary. 38 In general, one can expect that the benefits of acclimatization are 75–80% complete after 7 days of moderate exertion in the heat. 39 Additional heat-exposed exertion for 1–2 h per day for 10–14 d results in adaptations that further reduce heat-related physiologic strain.40–42 These adaptations may persist for up to a month, even if the person is removed from the hot environment.43,44 The key adaptation to repeated heat exposures appears to be plasma volume expansion and the resulting increased sweat capacity. 38 Heat acclimatization may be achieved by gradual exposure after arriving in a hot environment, 45 introduction of necessary personal protective equipment, periodic heat exposure prior to traveling to a hot environment, 46 and overdressing in a temperate environment. 47 A bout of heat stroke may acutely reset these thermoregulatory adaptations, causing elevated risk for subsequent heat injury for months after the initial event. 48 However, case reports indicate that heat tolerance can be recovered fully.49,50 Individuals with high levels of cardiopulmonary fitness tolerate more activity in conditions of high heat strain (whether due to hot temperatures or heat exchange limitations from clothing and gear) and acclimatize to heat more rapidly because of increased sweat capacity achieved through normal training.
The most readily modifiable physiologic risk factor is hydration status. While endurance athletes may comfortably tolerate weight losses of 3–4% during events,51,52 fluid losses that result in a 2–3% decrease in body weight correlate with greater core temperatures at a given workload in the heat.41,53,54 Dehydration increases physiologic strain, decreases sweat rates, increases perceived exertion, and increases core temperatures.55–57 However, adequate hydration cannot prevent heat accumulation in all circumstances, especially when challenged by a high metabolic rate, high ambient temperature, and factors that reduce the effectiveness of evaporative cooling, such as high humidity and impermeable clothing. 58
Maintaining euhydration during activity is typically done through drinking, either through a simple “drink-to-thirst” approach or through a scheduled drinking regimen with target volumes. A scheduled regimen carries the risk of accidental over or underconsumption, while drink-to-thirst is guided by the body's drive to maintain appropriate osmolality. An increase in plasma osmolarity by 2% triggers both the sensation of thirst and renal water retention through the arginine vasopressin pathway. 59 If one is experiencing thirst during activity, then the kidneys are avidly retaining free water, a key step toward exercise-associated hyponatremia. The technique of hyperhydration uses osmotic agents such as glycerin to augment water absorption, allowing one to load extra water into the plasma volume. Although some methods of hyperhydration have been shown to decrease physiologic strain from heat, 60 these methods have not reliably been shown to be protective from heat illness. Precooling may offer performance benefits for short periods of exertion in hot environments but is unlikely to be of benefit during longer periods of activity. 61 Female sex is associated with less heat loss, and regardless of sex, heat loss declines as age advances. 62 Studies of military recruits found a significantly increased risk of heat illness among those with high body mass indices.20,40 The physiology of pediatric and elderly patients leads to vulnerabilities that are outside the scope of these guidelines.63–65 High-risk populations warrant heightened efforts for prevention.
Any condition that limits heat loss through the skin may lead to heat retention. Examples include hypohydrosis, extensive scars, and the diminished cardiopulmonary reserve seen in the extremes of age. Small studies have linked acute sunburn with impaired sweating for 7 d, considerably longer than the associated pain and erythema. 66 Impaired sweating is a risk factor for heat accumulation and heat illness.
Drugs can predispose individuals to heat illness by increasing heat production or by compromising the function of thermoregulatory mechanisms (Table 2).67,68 In addition to prescription medications, stimulants such as nicotine and the constantly evolving array of compounds in preworkout supplements may increase the risk for heat-related illness. Moderate caffeine intake appears to have no detrimental effect. 22
Medications and drugs that may contribute to heat illness.

Environmental Considerations
As the environmental temperature increases, the body will eventually incur a net heat gain through conductive, convective, and radiative processes, leaving evaporation as the only potentially effective cooling mechanism. The vaporization of 1.7 mL of sweat consumes 1 kcal of heat. 69 Evaporative cooling is less effective in highly humid environments as humidity lowers the water vapor pressure difference between the sweat on the skin and the water in the surrounding air. Recreational clothing and occupational personal protective equipment can significantly reduce evaporative cooling. 30 The wet-bulb globe temperature index (WBGT) is a composite index of temperature, humidity, and solar radiation that expresses the total thermal strain on an individual. A predetermined set of WBGT values can be set as thresholds to activate guidelines for rehydration, active cooling, and limitation or cancellation of physical activity. 66 An alternative to the WBGT that is more readily available is the heat index, a measure of the contribution that high temperature and high humidity make in reducing the body's ability to cool itself. The WBGT is the preferred metric for environmental heat strain for military, 70 occupational, 71 and civilian groups,63,72 but it is rarely available when making decisions due to its localized measurement with specialized equipment. Heat index is more readily available. Guidelines that correlate the heat index with the risk of heat injury and outline parameters for limiting physical activity are also available. 73
Activity Considerations
The metabolic thermal output of exertion is the product of its intensity and duration. The increased metabolic heat from increased intensity may be offset in some activities in which intensity also enhances heat loss to the environment, such as convection in water by a swimmer or cool air by a cyclist. During breaks, metabolic heat production is expected to fall. Heat dissipation can be simultaneously increased by entering shade or a water mist and by removing clothing or protective gear that insulates or creates a vapor barrier. Occupational,58,71,74 military, 70 medical, 72 and sport guidelines recommend breaks in proportion to metabolic demand and ambient conditions, but there are few studies examining how to optimize the dosing of breaks.
Clothing and Equipment
Clothing, personal protective equipment, or other gear worn during an activity may limit or enhance the body's thermoregulatory efficiency.58,75 Of particular importance is equipment that occludes regions of skin, resulting in compromise of evaporative, convective, radiative, or conductive heat transfer. For example, the American football uniform prevents full heat exchange across much of the torso and head and can, therefore, contribute to heat accumulation. 76 A similar effect has been found for other occupational undergarments, 30 helmets, 77 and body armor. 78 Unfortunately, the necessary safety protection provided by some occupational gear results in an elevated risk of heat accumulation—a safety compromise that may be optimized but not eliminated. Sports medicine guidelines as well as military occupational guidelines provide example protocols to reduce clothing and equipment based on WBGT thresholds.72,79
Field Treatment
Optimal field management of heat illness may be challenging because of limited resources and extreme environmental demands. Moreover, most of the available data to guide therapy are based on case series or extrapolation of results from exercise-associated hyperthermia models because randomized controlled trials for treatments of EHS are ethically not feasible. The methods and aggressiveness of cooling in the field depend on the type of heat illness to be treated (Table 3). Regardless of the underlying cause, removal from the heat and rapid cooling are critical as the extent of morbidity is directly related to both to the degree and duration of hyperthermia.11,40,80–83 Initial treatment in the field is first directed to stabilization of the patient's airway, breathing, and circulation before proceeding to more specific cooling therapy. If no life-threatening complications exist, on-scene cooling is potentially life-saving and should be immediately implemented prior to evacuation if logistically possible (Figure 1). In transitioning patient care to EMS providers, any cooling techniques undertaken in the field should be described in the hand-off report, and cooling of the patient by the best available means should be continued during transport. All these priorities and steps are consistent with the current US prehospital care consensus statement and thus are considered best practice across wilderness and nonwilderness first responder settings. 84
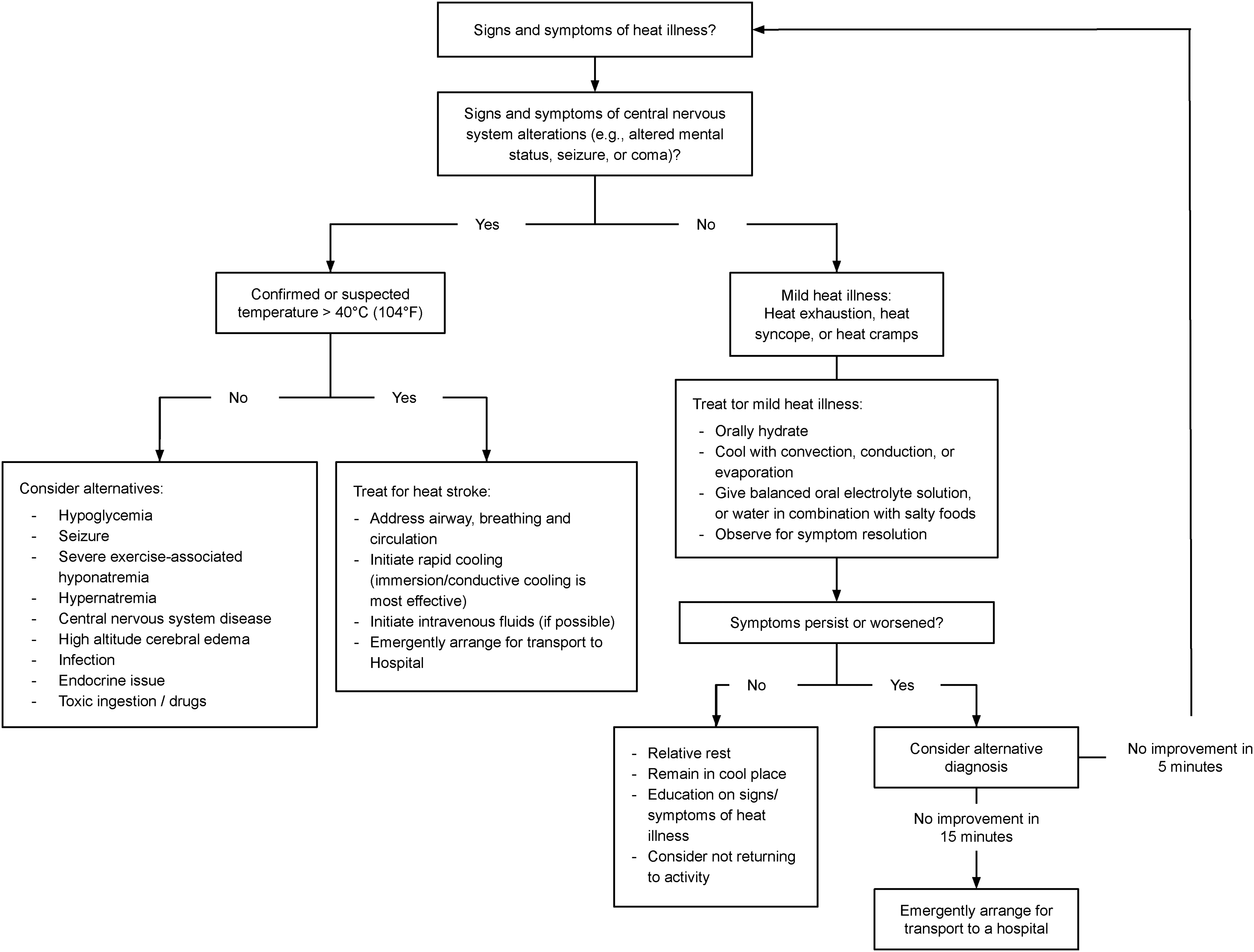
Heat illness treatment algorithm.
Heat illness treatments.

a Intravenous hydration with isotonic fluids (0.9% saline or lactated Ringer's), with 3% saline indicated if concern for exercise-associated hyponatremia as cause of encephalopathy.
b Initiate EMS if unable to rapidly cool patient, prolonged encephalopathy, or concern of multiorgan dysfunction.
Minor Heat Illness Treatment
There is scant evidence supporting treatments for minor and moderate heat illness. Most treatments have not been studied in trials but persist in clinical practice (Table 3). Heat cramps, historically described as generalized, 21 seem to differ from the focal exercise-associated muscle cramps seen in endurance athletes. More recent research23,85 has sought a common mechanism to explain both clinical presentations, yet definitive evidence is lacking to clearly unite or separate these clinical patterns of cramping and adequately explain their differing response to treatment. Generalized heat cramps can be relieved with oral salt solutions or electrolyte replacement that may be isotonic or hypertonic.21,86 In contrast, exercise-associated muscle cramps that occur with repetitive movement and neuromuscular fatigue seem to be better relieved with passive stretching. 85 Heat edema can be relieved by extremity elevation or wearing of compression stockings. Diuretics are ineffective for treating heat edema and may worsen volume depletion. 87
Heat syncope is self-limiting. After consideration of other medical causes of syncope and managing any resultant trauma from a fall, treatment consists of rest in a cool environment, while ensuring euhydration with isotonic oral fluids may enhance recovery. 24 Individuals at risk for heat syncope should move often and flex their larger leg muscles to prevent peripheral pooling of blood caused by cutaneous vasodilation. An individual with heat syncope may benefit from further acclimatization, though no studies provide clear guidance for return to activity.
Heat exhaustion, like heat stroke, results from a combination of cardiovascular and thermal strain. Various forms of rest and whole-body cooling are dictated by the severity of symptoms. Mild cases are generally resolved by removing the patient from the hot environment, cessation of physical activity, and rehydration with oral isotonic fluids. Severe heat exhaustion typically has more pronounced volume depletion that may require intravenous replacement of fluids as well as conductive and convective cooling. Heat exhaustion can lead to cessation of physical activity and collapse, rendering self-care ineffective. It is important to intervene to reverse the process of heat exhaustion, as the patient can progress to heat stroke without proper cooling techniques. Heat exhaustion or acute heat stress can precipitate EKG changes, symptomatic arrhythmias, and cardiac arrest with features of underlying Brugada syndrome.88,89
Temperature Measurement
Accurate measurement of core temperature is an important step in differentiating heat stroke from less severe heat illness, as altered mental status with a core temperature below 40 °C should not be attributed to heat stroke. Rectal temperature is widely considered the standard field measurement because it is a reliable and practical measure of core temperature that is more accurate than temporal, axillary, oral, or tympanic thermometry.27,81,90,91 Esophageal and ingestible thermistors have been validated and provide an accurate measurement of core temperature but are impractical in most wilderness settings. Rectal temperature measurement carries implicit challenges in maintaining patient privacy and hygiene. Initial assessment and aggressive cooling should be implemented based on clinical suspicion, regardless of the degree of hyperthermia or mode of measurement.
Passive Cooling
Simple measures can reduce the patient's exposure to heat. Moving the victim into the shade can decrease the radiative heat gains and reduce the ambient temperature—this is most effective in temperatures < 20 °C (68°F). 81 Conduction of heat from the ground can be decreased by placing the victim on cool ground, if available, or an insulating barrier such as a sleeping pad or sleeping bag. Loosening or removing any tight-fitting clothing to optimize air circulation aids in convective heat exchange. 92
Hydration
Euhydration is an important factor in reducing hyperthermia.92,93 Dehydration predisposes patients to hyperthermia by decreasing sweat rates and increases thermal strain at a given temperature.57,93–95 Oral and intravenous hydration have been shown to be equally effective in replenishing water deficiencies related to heat stress.93,96
Optimizing hydration in heat stroke decreases both cardiovascular and thermal strains. In a heat stroke victim with altered mental status and risk of seizure, the intravenous route minimizes the risk of aspiration and airway compromise. Few data exist on the optimal type and amount of intravenous fluid to use for treating heat illness. Heat stroke can occur in both euhydration and dehydration, so care should be taken not to overhydrate patients, especially those with cardiac comorbidities, as this may increase the risk of pulmonary edema. 97 Any effort to provide hydration in suspected heat stroke should not delay rapid whole-body cooling. 98 If intravenous hydration is provided, field monitoring of blood pressure, heart rate, lightening of urine color, and increase in urine output can help guide patient response and fluid status.
Symptomatic exercise-associated hyponatremia (EAH) may present similarly to heat exhaustion,26,99 but severe EAH may present more like heat stroke with altered mental status (eg, obtundation, coma, or seizures) without another explanation such as hypoglycemia, trauma, or hyperthermia >40 °C. These two life-threatening diagnoses share risk factors and clinical manifestations yet need different treatments—a troubling situation in wilderness settings where diagnostic testing is unavailable. While graded recommendations about the treatment of EAH are outside the scope of this guideline, we recommend that the assessment of heat illnesses should include consideration for EAH using available clinical guidelines.100,101 In the absence of point-of-care testing, if clinical suspicion is high for severe EAH, then treatment should be started with up to 3 boluses of 3% saline, 100 mL each, given every 10 min or until resolution of altered mental status, with rapid transfer to a medical facility.100,101
Cold Water Immersion Therapy
Cold water immersion (CWI) therapy is the optimal field treatment to achieve rapid temperature reduction below critical levels in heat stroke. Immersion is a convective method of cooling that takes advantage of water's high thermal conductivity, which is 24 times greater than air, 102 and the high thermal gradient that exists between cold water and skin, 103 which translates into a greater capacity for heat transfer. While a wide range of temperatures can be effective, the colder the water, the faster the rate of cooling. 104 The theoretical concern that cold water immersion causes peripheral vasoconstriction and shivering that slow down cooling or may even increase the core temperature is a prevalent misconception. 105 While shivering has been observed in immersions lasting longer than 10 min in healthy volunteers,106,107 such shivering may be less problematic in actual heat-stroke patients with failing thermoregulation.108,109 Cold water immersion is achieved by removing insulating clothing and equipment and submersing the patient's trunk and extremities in a bath of ice water or the coldest water available. Alternatively, the patient can be placed onto a plastic sheet or tarp, then covered in ice and water added to form a slurry. The sheet edges should be elevated to form walls that keep the slurry in place (“tarp taco”). 110 A body bag may also be used to contain the patient, ice, and water. 111 Ice-water cooling lowers core temperature twice as rapidly as covering the body in soaked towels to enhance evaporative cooling (0.20 °C·min−1 vs 0.11 °C·min−1). 48
In the field, using a natural body of water such as a stream, pond, river, or lake may be another treatment option. Protect the patient against currents and ensure the head does not go underwater. The victim should never be left alone because of the risk of aspiration and drowning. If immersion is not being used, the patient may be repeatedly doused with cold water or snow. Multiple military studies on immersion cooling of young, healthy heat stroke victims report no fatalities. 112
Evaporative Cooling
If immersion or conductive cooling is unavailable, evaporative cooling should be initiated by loosening or removing clothing and dousing the victim with cold water with the goal of ensuring all the skin surface area is wet and able to contribute to evaporative cooling. 113 Convection is then facilitated with air movement by fanning. Cooling by evaporation plus convection has been studied predominantly in patients with nonexertional heat stroke, 114 with mean cooling times of 40–68 min.108,115 Limited studies on evaporative cooling have been done with EHS or heat exhaustion, with reported cooling rates half as fast as immersion cooling. 116
Chemical Cold Packs and Ice Packs
There is traditional advocacy for cooling by ice packs or chemical cold packs strategically applied to the skin covering the neck, axillae, and groin to cool blood flowing in major vessels as an adjunctive cooling measure. 117 Limited studies show no benefit in heat reduction with this technique.118,119 However, ice packs have greater cooling capacity than chemical cold packs 120 and, if used, are most efficacious when wet and covering the entire body to optimize conductive cooling. 118 In a small translational study, chemical cold packs applied to the glabrous skin of the palms, soles, and cheeks showed twice the cooling rate compared to traditional placement on major vascular locations because of the high-capacity blood flow of the subcutaneous arteriovenous anastomoses. 121
Antipyretics
Antipyretic medications such as ibuprofen and aspirin work by inhibiting prostaglandin formation, and acetaminophen lowers the thermoregulatory setpoint. 122 While this setpoint may be elevated in infectious causes of hyperthermia, it is not disturbed in exercise-induced hyperthermia. In these cases, antipyretic drugs are ineffectual.123,124
Hospital Treatment
The traditional teaching has been that all heat stroke patients require hospital-based evaluation and treatment, given the severity of the disease and high risk of morbidity and mortality. 125 There is ample precedent, however, for definitive treatment in field medical stations without the need for hospital transfer. In a large civilian case series, hundreds of athletes with EHS at an annual road race have been treated on-site in medical tents and discharged home.126,127 Similarly, hundreds of military recruits have been treated for EHS at base infirmaries. 112 In both settings, treatment has involved immediate cooling in prepared tubs with an ice-water slurry and a period of observation before discharge. These studies have not reported a standard duration of observation. No fatalities or lingering morbidity were reported.
In the absence of such medically supervised, pre-established field-cooling protocols, patients with heat stroke should be transported to a medical facility capable of managing critical illness with the potential for multiple organ injury or failure. Patients at athletic events or military bases who are treated according to standing protocols but whose mental status or physical examination has not returned to baseline should also be transferred to a higher level of care. Cooling measures should continue during transportation for patients whose temperature remains elevated.
In the hospital setting, the primary treatment goals for heat stroke are rapidly lowering core temperature and supporting organ system function. Heat stroke patients may develop multiple organ failure with shock, respiratory failure, acute kidney and liver injury, rhabdomyolysis, disseminated intravascular coagulation, and intestinal ischemia with loss of barrier integrity, leading to bacterial translocation into the bloodstream with resultant sepsis.31,125,128 Depending on the patient's clinical status, supportive treatment may include administering supplemental oxygen, performing intubation and mechanical ventilation, establishing intravascular or intraosseous access, restoring intravascular volume with intravenous isotonic crystalloid solution, placing a bladder catheter to monitor urine output and temperature, and initiating vasopressors to support blood pressure if needed after adequate volume resuscitation. Such interventions should be undertaken simultaneously with any ongoing cooling measures to help prevent further clinical deterioration.
Although large, randomized controlled trials on treating heat stroke patients are lacking, the best available evidence supports ice-water immersion as the most rapid, effective treatment for young, athletic patients with EHS.31,129 A cooling protocol used for over 15 y involving an ice-water slurry has been applied effectively with no fatalities or adverse effects in hundreds of civilian and military individuals, with cooling rates of 0.15–0.22 °C·min−1 and mean cooling times of 12–18 min.112,116
In an emergency department without a tub, a waterproof body bag may be filled with an ice-water slurry and arranged on a stretcher as a makeshift basin (Table 4). 111 In the absence of a tub or basin, the entire body may be covered with cubed ice or ice packs before water is poured over the patient, increasing the surface area for conductive cooling. 118 If ice packs are used, these should cover the entire body. If a shortage requires strategic placement of ice packs, use them on the glabrous skin of the palms, soles, and cheeks. Ice-filled packs remain colder longer and may have more cooling capacity compared with instant chemical cold packs. 120
Steps for hospital-based ice-water immersion.

Strategic application of ice packs to the axillae, neck, and groin contributes minimally to cooling under experimental conditions and would not be expected to be effective in treating heat stroke. 118 This last point should be emphasized, as published articles continue to promote such treatment despite a lack of proven effectiveness as a primary cooling method. 130 The application of icy, wet towels as a primary cooling method is also ineffective. 119
If ice is unavailable, then copious water should be poured over the unclothed patient and strong air currents directed at the patient for evaporation and convection. Large electric fans and commercially available air compressors have been used to create air currents. Techniques using evaporation plus convection cool only half as fast as cold water immersion, with mean cooling times of 40–68 min.108,115,131 Therefore, CWI should be used in EHS whenever possible.
Historically, cooling by evaporation plus convection has been used predominantly in older, more infirm patients who present more often with NEHS, with mortality rates ranging from a low of 0–6% to a high of 11–15%.113–115,132 Whether the higher mortality associated with evaporation plus convection versus CWI is attributable to the older age and associated medical conditions (such as diabetes, high blood pressure, and heart disease) versus the cooling technique remains uncertain. Evaporation plus convection has an accepted role in the hospital-based treatment of NEHS, although recent reviews have suggested that CWI may be preferable, even in patients with NEHS, because of its shorter cooling times.31,129 The benefits of immersion cooling should be balanced against the theoretical concerns of impaired access to an immersed patient who may require advanced cardiac monitoring or resuscitation.65,109,113,133
Shivering, agitation, or combativeness may occur during cooling of encephalopathic heat stroke patients. The conditions can often be pharmacologically controlled with benzodiazepines or opiates. The theoretical concern that shivering and the accompanying reflex peripheral vasoconstriction might slow immersion cooling has not been observed in clinical practice because the modest amount of total heat generated by shivering is overwhelmed by the rapid heat loss to ice-water bath. Furthermore, pronounced shivering is observed more often during CWI lasting longer than 10 min in conscious, healthy volunteers and less often in obtunded heat-stroke patients with failing thermoregulation.104,107,109
Adjunctive, Combined, or Alternative Cooling Methods
Applying crushed ice or ice packs over patients cooled via evaporation plus convection11,134 may increase cooling, but the extent of any incremental benefit on cooling rates or outcomes has not been established.
Cold intravenous fluids may supplement cooling but have not been shown to adequately serve as a primary treatment for heat stroke. 98 In healthy volunteers, an intravenous 30 mL/kg bolus of chilled isotonic fluids at 4 °C over 30 min decreased core temperature by 1 °C compared to 0.5 °C with room temperature isotonic fluids. 135
Invasive techniques of body-cavity lavage with cold isotonic fluid have been reported but not adequately studied.136,137 Noninvasive external cooling systems, such as the Arctic Sun™, have been used successfully in individual case reports but require further study. 138 Intravascular cooling devices have been used in pilot studies on both EHS and NEHS patients but require both time and special equipment to set up, potentially leading to delays in cooling.139–141
The use of cooling blankets in heat stroke has not been well described or studied. 11
Target Cooling Temperatures
Cooling to a target temperature of 38.3–38.8 °C by ice-water immersion has been well tolerated in multiple studies with no fatalities, adverse outcomes, or core temperature after drops resulting in hypothermia.22,142–144 Once patients are cooled to this range, endogenous thermoregulation should resume and help return the body to normal temperature.
Pharmacologic Treatment
No pharmacologic agent has been approved to treat heat stroke. A clinical trial using dantrolene in NEHS showed no difference in cooling rates or outcome. 132 Recently, a new drug application has been filed in the United States to use dantrolene in EHS, and a clinical trial is in progress. Antipyretic agents such as aspirin, ibuprofen, and acetaminophen are not recommended in heat stroke because fever and heat stroke raise core temperature through different physiological mechanisms. In addition, nonsteroidal anti-inflammatory agents theoretically risk exacerbating coagulopathy and renal injury. Acetaminophen (paracetamol) similarly risks exacerbating hepatic injury.
Investigations of agents that block inflammatory pathways may lead to future pharmacologic therapies for heat stroke. Potential agents include activated protein C, thrombomodulin alpha, type III antithrombin concentrate, serine proteases, and Rheum palmatum or Chinese rhubarb. 125
Emerging Trends
Research is ongoing on serum biomarkers that may predict severity of disease or mortality in heat stroke. These include serum procalcitonin, C-reactive protein, HMGB1 protein, plasma diamine oxidase, and intestinal fatty acid binding protein. Levels of plasma diamine oxidase and intestinal fatty acid binding protein may also correlate with intestinal injury and increased bacterial translocation into the bloodstream, although such findings have been inconsistent. No conclusive recommendations can be made yet on the role of biomarkers in predicting clinical outcomes or guiding the management of heat stroke, though this remains an active area of exploration.7,128,145,146 The practical roles of serum biomarkers are not yet established for predicting outcomes or guiding clinical management of heat stroke.
Conclusions
We recommend that patients with heat stroke be cooled by whole-body ice-water or cold water immersion as immediately as can be safely accomplished. Evaporative and convective cooling may be augmented with the addition of ice packs over the entire body to promote conductive cooling. Future areas of research should include direct comparisons of available cooling modalities in controlled models, as well as further evaluation of endovascular catheters and hospital-based systems for cooling. Implementation efforts in a hospital setting should focus on eliminating cultural barriers among personnel to ease the adoption of ice-water immersion in available containment devices such as body bags or tarpaulins. This method provides the most rapid cooling in a fashion that is readily available, low cost, and most effective for the patient.
Supplemental Material
sj-pdf-2-wem-10.1177_10806032241227924 - Supplemental material for Wilderness Medical Society Clinical Practice Guidelines for the Prevention and Treatment of Heat Illness: 2024 Update
Supplemental material, sj-pdf-2-wem-10.1177_10806032241227924 for Wilderness Medical Society Clinical Practice Guidelines for the Prevention and Treatment of Heat Illness: 2024 Update by Kurt P. Eifling, Flavio G. Gaudio, Charles Dumke, Grant S. Lipman, Edward M. Otten, August D. Martin and Colin K. Grissom in Wilderness & Environmental Medicine
Footnotes
Author Contribution(s)
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplementary material associated with this article can be found in the online version at https://doi.org/10.1177/10806032241227924.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
