Abstract
The Wilderness Medical Society convened an expert panel to develop a set of evidence-based guidelines for the prevention and treatment of frostbite. We present a review of pertinent pathophysiology. We then discuss primary and secondary prevention measures and therapeutic management. Recommendations are made regarding each treatment and its role in management. These recommendations are graded on the basis of the quality of supporting evidence and balance between the benefits and risks or burdens for each modality according to methodology stipulated by the American College of Chest Physicians. This is an updated version of the guidelines published in 2019.
Introduction
The Wilderness Medical Society (WMS) convened an expert panel to develop a set of evidence-based guidelines for the prevention and treatment of frostbite to guide clinicians and first responders and disseminate knowledge about best practices in this area of clinical care. We present the main prophylactic and therapeutic modalities and make recommendations about their role in injury management. Recommendations are graded on the basis of the quality of supporting evidence and balance between the benefits and risks or burdens for each modality. We then provide suggested approaches for prevention and management that incorporate these recommendations.
The original expert panel was convened at the 2010 Annual Winter Meeting of the WMS in Park City, UT. Members were selected on the basis of their clinical or research experience. Relevant articles were identified through the MEDLINE database using the search terms frostbite, frostbite management, prehospital frostbite treatment, prehospital frostbite management, frostbite prevention, first aid frostbite treatment, and first aid frostbite and were restricted to the English language. Studies in these categories were reviewed, and the level of evidence was assessed. The panel used a consensus approach to develop recommendations regarding each modality and graded each recommendation according to criteria stipulated by the American College of Chest Physicians statement on grading recommendations and strength of evidence in clinical guidelines (Online Supplementary Table). 1 This is an updated version of the guidelines published in 2019. 2
Pathophysiology of Frostbite
Frostbite is a freezing injury that involves 4 overlapping pathologic phases: prefreeze, freeze-thaw, vascular stasis, and late ischemic. The prefreeze phase consists of tissue cooling with accompanying vasoconstriction and ischemia and without actual ice crystal formation. Neuronal cooling and ischemia produce hyperesthesia or paresthesia. If cooling stops at the prefreeze stage, permanent damage does not occur (see later discussion). In the freeze-thaw phase, ice crystals form intracellularly (during a more rapid-onset freezing injury) or extracellularly (during a slower freeze), causing protein and lipid derangement, cellular electrolyte shifts, cellular dehydration, cell membrane lysis, and cell death. 3 The thawing process may initiate ischemia, reperfusion injury, and an inflammatory response. In the vascular stasis phase, vessels fluctuate between constriction and dilation; blood may leak from vessels or coagulate within them.4–6 The late ischemic phase results from progressive tissue ischemia and infarction from a cascade of events, including inflammation mediated by thromboxane A2, prostaglandin F2alpha, bradykinin, and histamine; intermittent vasoconstriction of arterioles and venules; continued reperfusion injury; showers of emboli coursing through the microvessels7,8; and thrombus formation in larger vessels. 9 Destruction of the microcirculation is the main factor leading to cell death. 10 The initial cellular damage caused by ice crystals and the subsequent post-thawing processes are made worse if refreezing follows the thawing of injured tissues.11,12
Classification of Frostbite
Frostnip is a superficial nonfreezing cold injury associated with intense vasoconstriction on exposed skin, usually cheeks, ears, or nose. Ice crystals, appearing as frost, form on the skin’s surface. Frostnip is distinct from and may precede frostbite. With frostnip, ice crystals do not form within the tissue, and tissue loss does not occur. Numbness and pallor resolve quickly after warming the skin with appropriate clothing, direct contact, breathing with cupped hands over the nose, or gaining shelter. No long-term damage occurs. Frostnip signals conditions favorable for frostbite; appropriate action should be undertaken immediately to prevent injury.
Frostbite has historically been divided into 4 tiers or “degrees” of injury following the classification scheme of severity for thermal burn injury. These classifications are based on acute physical findings and advanced imaging after rewarming.
13
The classifications can be difficult to assess in the field before rewarming because the still-frozen tissue is hard, pale, and anesthetic. An alternate 2-tiered classification more appropriate for field use (after rewarming but before imaging) is suggested after the following 4-tier classification:
First-degree frostbite causes numbness and erythema. A white or yellow, firm, and slightly raised plaque develops in the area of injury. No gross tissue infarction occurs; there may be slight epidermal sloughing. Mild edema is common. Second-degree frostbite injury causes superficial skin vesiculation; a clear or milky fluid is present in the blisters, surrounded by erythema and edema. Third-degree frostbite causes deeper hemorrhagic blisters, indicating that the injury has extended into the reticular dermis and beneath the dermal vascular plexus. Fourth-degree frostbite extends completely through the dermis and involves the comparatively avascular subcutaneous tissues, with necrosis extending into muscle and bone. Superficial—no or minimal anticipated tissue loss, corresponding to first- and second-degree injury. Deep—anticipated tissue loss corresponding to third- and fourth-degree injury.
For field classification, after spontaneous or formal rewarming but before imaging, we favor the following 2-tier severity classification scheme:
The severity of frostbite may vary within a single extremity. Once thawing occurs and a patient reaches a field clinic or hospital, one can further classify or characterize the frostbite injury via additional methods. The Cauchy classification method
13
(Table 1) measures extent of frostbite anatomically using the following grades: 0, no discoloration; 1, discoloration on the distal phalanx; 2, discoloration on the middle phalanx or proximal phalanx for the thumb/big toe; 3, discoloration on the proximal phalanx except for the thumb/big toe; 4, discoloration on the metacarpal/metatarsal; 5, discoloration on the carpal/tarsal. The Cauchy classification method may assist caregivers in predicting amputation risk, which helps to inform evacuation and other care decisions. For example, a necrotic fingertip (labeled grade 4 by the 4-tiered system but unlikely to involve significant amputation) would be designated a Grade 1 on the Cauchy classification method, designating lower severity. Higher grades in the Cauchy classification method designate more proximal injuries with greater risk for functionally important amputation. We note that Grade 2 “fingernail sequelae” is more accurately defined as “nailbed sequelae” and can include the toes as well.
Cauchy classification scheme for severity of frostbite injuries
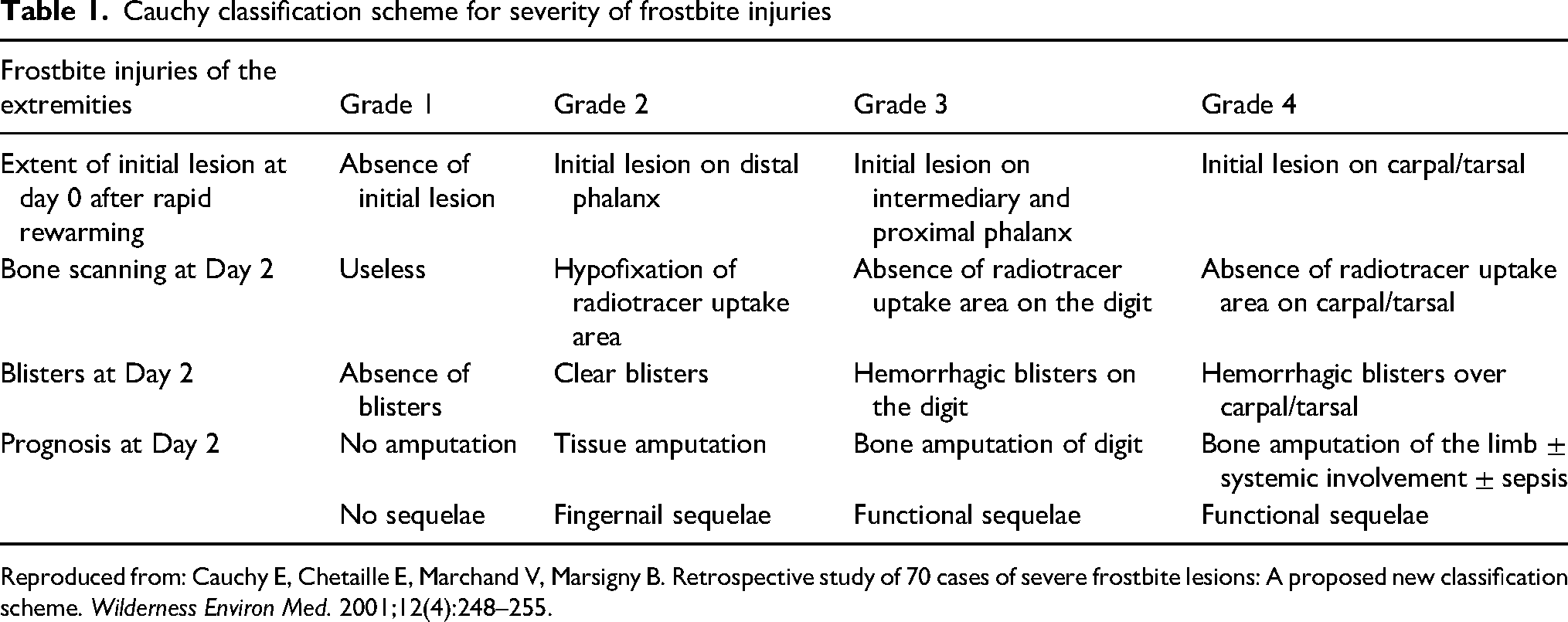
Reproduced from: Cauchy E, Chetaille E, Marchand V, Marsigny B. Retrospective study of 70 cases of severe frostbite lesions: A proposed new classification scheme. Wilderness Environ Med. 2001;12(4):248–255.
Summary of field treatment of frostbite (>2 h from definitive care)

Prevention
The adage that “prevention is better than treatment” is especially true for frostbite, which is typically preventable and often not improved by treatment. Underlying medical problems such as peripheral vascular disease, malnutrition, Raynaud's disease, diabetes mellitus, and tobacco use may increase the risk of frostbite; prevention must address both environmental and health-related aspects. Frostbite injury occurs when tissue heat loss exceeds the ability of local tissue perfusion to prevent freezing of soft tissues (blood flow delivers heat). One must both ensure adequate perfusion and minimize heat loss to prevent frostbite. The individual should recognize cold-induced “numbness” as a warning that frostbite injury may be imminent if protective or avoidance measures are not taken to decrease tissue cooling. Subsequent loss of sensation does not mean the situation has improved; rather, receptors and nerves are not conducting pain/cold signals because they are nearing the freezing point.
Maintaining Peripheral Perfusion
Preventive measures to ensure local tissue perfusion include: 1) maintaining adequate core temperature and body hydration; 2) minimizing the effects of known diseases, medications, and substances that might decrease perfusion (including awareness and symptoms of alcohol and drug use); 3) covering all skin and the scalp to insulate from the cold; 4) minimizing blood flow restriction, such as occurs with constrictive clothing, footwear, or immobility; 5) ensuring adequate nutrition; and 6) using supplemental oxygen in severely hypoxic conditions (eg, >7500 m). 14 Recommendation: Strong recommendation, low-quality evidence.
Exercise
Exercise is a specific method to maintain peripheral perfusion. Exercise enhances the level and frequency of cold-induced peripheral vasodilation (CIVD). In one study, cold-induced peripheral vasodilation occurred in the toes of 58% of subjects who were exercising versus only 28% in those subjects not exercising. 15 Another study found increased skin temperature in the hands during exercise. 16 An additional study demonstrated the CIVD response may be negatively affected by the introduction of hypoxia, whereas metabolic heat production via exercise may counteract adverse effects of hypoxia and improve CIVD responses. 17 However, using exercise to increase warmth can lead to exhaustion, with subsequent profound systemic heat loss should exhaustion occur. Recognizing this caveat, exercise and its associated elevation in core and peripheral temperatures may be protective in preventing frostbite. Recommendation: Strong recommendation, moderate-quality evidence.
Protection from Cold
Measures should be taken to minimize exposure of tissue to cold. These measures include the following: 1) avoiding environmental conditions that predispose to frostbite, specifically below 0 °C (some risk 0 to −15 °C and increasing risk below −15 °C, even with low wind speeds) 18 ; 2) protecting skin from moisture, wind, and cold; 3) avoiding perspiration or wet extremities; 4) increasing insulation and skin protection (eg, by adding clothing layers, changing from gloves to mitts); 5) ensuring beneficial behavioral responses to changing environmental conditions (eg, not being under the influence of illicit drugs, alcohol, or extreme hypoxemia) 19 ; 6) using chemical hand and foot warmers and electric foot warmers to maintain peripheral warmth (note: warmers should be close to body temperature before being activated and must not be placed directly against skin or constrict flow if used within a boot); 7) regularly checking oneself and the group for extremity numbness or pain and warming the digits and/or extremities as soon as possible if there is concern that frostbite may be developing; 8) recognizing frostnip or superficial frostbite before it becomes more serious; and 9) minimizing duration of cold exposure. 14 Emollients do not protect against—and might even increase—the risk of frostbite. 20 The time that a digit or extremity can remain numb before developing frostbite is unknown; thus, digits or extremities with paresthesia should be warmed as soon as possible. An extremity at risk for frostbite (eg, numbness, poor dexterity, pale color) should be warmed with adjacent body heat from the patient or a companion, using the axilla or abdomen. Recommendation: Strong recommendation, low-quality evidence.
Field Treatment and Secondary Prevention
If a body part is frozen in the field, the frozen tissue should be protected from further damage. Remove jewelry or other constrictive extraneous material from the body part. Do not rub or apply ice or snow to the affected area. 21
Refreezing Injury
A decision must be made on whether to thaw the tissue. If environmental conditions are such that thawed tissue could refreeze, it is safer to keep the affected part frozen until a thawed state can be maintained. Prostaglandin and thromboxane release associated with the freeze-thaw cycle21–23 causes vasoconstriction, platelet aggregation, thrombosis, and, ultimately, cellular injury. Refreezing thawed tissue further increases release of these mediators, and significant morbidity may result. One must absolutely avoid refreezing if field thawing occurs. Recommendation: Strong recommendation, moderate-quality evidence.
Spontaneous or Passive Thawing
Most frostbite thaws spontaneously and should be allowed to do so if rapid rewarming (described in the following) cannot be readily achieved. Warming with available methods, such as against warm body parts (eg, axilla), should be employed if rapid rewarming is not possible. Do not purposefully keep tissue below freezing temperatures because this will increase the duration that the tissue is frozen and might result in more proximal freezing and greater morbidity. If environmental and situational conditions allow for spontaneous or slow thawing, tissue should be allowed to thaw. Recommendation: Strong recommendation, low-quality evidence.
Strategies for 2 scenarios are presented:
Therapeutic Options for Both Scenarios
Many of these guidelines parallel the state of Alaska’s cold injuries guidelines. 24 Therapeutic options are as follows.
Treatment of Hypothermia
No studies examine concurrent hypothermia and frostbite. Hypothermia frequently accompanies frostbite and causes peripheral vasoconstriction that impairs blood flow to the extremities. Mild hypothermia may be treated concurrently with frostbite injury. Moderate and severe hypothermia should be treated effectively before treating frostbite injury following recommendations in the WMS Hypothermia Clinical Practice Guidelines. 25 Recommendation: Strong recommendation, low-quality evidence.
Hydration
Vascular stasis can result from frostbite injury. No studies have specifically examined the effect of hydration status on frostbite outcomes, but appropriate hydration and avoidance of hypovolemia are important for frostbite prevention and recovery. Oral fluids may be given if the patient is alert, capable of purposeful swallowing, and not vomiting. If the patient is nauseated or vomiting or has an altered mental status, IV fluids should be given to maintain normal urine output. Intravenous fluids should optimally be warmed (minimally to 37 °C but preferably to 40 °C to 42 °C with a method that has been proven to be effective in the present environmental conditions) before infusion and be infused in small (eg, 250 mL), rapid boluses because slow infusion will result in fluid cooling and even freezing as it passes through the tubing. Fluid administration should be optimized to prevent clinical dehydration. Recommendation: Strong recommendation, low-quality evidence.
Low Molecular Weight Dextran
Intravenous low molecular weight dextran (LMWD) decreases blood viscosity by preventing red blood cell aggregation and formation of microthrombi and can be given in the field once it has been warmed. In some animal studies, the extent of tissue necrosis was found to be significantly less than in control subjects when LMWD was used26–29 and was more beneficial if given early. 30 In one animal trial, 30 tissue in the LMWD group thawed slightly more rapidly, but overall tissue loss was no different from that of control animals. A test dose before administration is recommended because of the low risk of anaphylaxis. This low risk of anaphylaxis should not deter administration. The slight risk of bleeding is minimal, and benefits seem to outweigh this risk; however, availability is limited in the United States and the United Kingdom. The use of LMWD has not been evaluated in combination with other treatments, such as thrombolytics or iloprost. If systemic treatment such as thrombolytic or iloprost therapy is not available or considered, LMWD should be administered if locally available. Recommendation: Weak recommendation, low-quality evidence.
Ibuprofen
Nonsteroidal anti-inflammatory drugs (NSAIDs) block the arachidonic acid pathway and decrease production of prostaglandins and thromboxanes. 31 These mediators can lead to vasoconstriction, dermal ischemia, and further tissue damage. No studies have demonstrated that any particular anti-inflammatory agent or dosing is clearly related to outcome. Aspirin has been proposed as an option and is used in many parts of the world for anti-inflammatory and inhibition of platelet aggregation. One rabbit ear model study showed 23% tissue survival with aspirin versus 0% in the control group. 32 However, aspirin theoretically blocks the production of certain prostaglandins that are beneficial to wound healing, 33 and the authors of the rabbit ear model study recommend ibuprofen in their treatment algorithm. No studies specifically compare aspirin with ibuprofen in frostbite. Ibuprofen should be started in the field at a dose of 12 mg/kg per day divided twice daily (minimum dose to inhibit harmful prostaglandins 31 ) to a maximum of 2400 mg/d divided 4 times daily. Recommendation: Weak recommendation, low-quality evidence.
Specific Recommendations—Scenario 1
Therapeutic options for frostbite in Scenario 1 (no active thawing) are as follows.
Dressings
No evidence supports applying a dressing to a frostbitten part intended to remain frozen until rewarming can safely be achieved. If this is considered, it should only be done if practical and will not interfere with mobility. Bulky, clean, and dry gauze or sterile cotton dressings should be applied to the frozen part and between the toes and fingers. Recommendation: Weak recommendation, very low-quality evidence.
Ambulation and Protection
If at all possible, a frozen extremity should not be used for walking, climbing, or other maneuvers until definitive care is reached. If use of the frozen extremity for mobility is considered, a risk-benefit analysis must consider the potential for further trauma and possible poorer outcome. Although it is reasonable to walk on a foot with frostbitten toes for evacuation purposes, it is inadvisable to walk on an entirely frostbitten foot because of the potential for resulting morbidity. This risk is theoretical and based on the panel's opinion. Mills described frostbite patients who ambulated on frozen extremities for days and sustained no or limited amputation. 34 If using a frozen extremity for locomotion or evacuation is unavoidable, the extremity should be padded, splinted, and kept as immobile as possible to minimize additional trauma. Recommendation: Weak recommendation, low-quality evidence.
Specific Recommendations—Scenario 2
Therapeutic options for frostbite in Scenario 2 (thawing and continued warming) are as follows.
Rapid Field Rewarming of Frostbite
Field rewarming should only be undertaken if the frozen part can be kept thawed and warm until the victim arrives at definitive care. Field rewarming by warm water bath immersion can and should be performed if the proper resources are available and definitive care is more than 2 h distant. Other heat sources (eg, fire, space heater, oven, heated rocks) should be avoided because of the risk of thermal burn injury. Rapid rewarming by water bath has been found to result in better outcomes than slow rewarming.21,29,34 Water should be heated to 37 °C to 39 °C (98.6 to 102.2°F) using a thermometer to maintain this range. 35 If a thermometer is not available, a safe water temperature can be determined by placing a caregiver's uninjured hand in the water for at least 30 s to confirm that the water temperature is tolerable and will not cause burn injury. Circulation of water around the frozen tissue will help maintain correct temperature.36,37 Because the water will cool once the frostbitten tissue is immersed, the water should be regularly rewarmed and replaced while taking care not to burn the immersed skin. For example, immersion could alternate between two containers: one for thawing while the other is being reheated to the target temperature. If a thermometer is not available to measure water temperature, a healthy hand should be able to be fully immersed long enough to experience a very warm but tolerable sensation. Thawing tissue in a container that is actively being heated increases the risk of burn injury. If this is the only option available, immersed tissue should not touch the container itself. The water should be stirred, and its temperature must be continually monitored with a thermometer or healthy hand. Rewarming is complete when the involved part takes on a red or purple appearance and becomes soft and pliable to the touch. This is usually accomplished in approximately 30 min, but the time is variable depending on the extent and depth of injury. The affected tissues should be allowed to air dry or be gently dried with a blotting technique (not rubbing) to minimize further damage. Under appropriate circumstances, this method of field rewarming is the first definitive step in frostbite treatment. Recommendation: Strong recommendation, moderate-quality evidence.
Antiseptic Solution
Most injuries do not become infected, but adding an antiseptic solution (eg, povidone-iodine, chlorhexidine) to the rewarming water has theoretical benefits of reducing skin bacteria. Evidence for this practice does not exist for frostbite care, however. Adding an antiseptic solution to the water while rewarming is unlikely to be harmful and might reduce the risk of cellulitis if severe edema is present in the affected extremity. Recommendation: Weak recommendation, very low-quality evidence.
Pain Control
During rewarming, pain medication (eg, NSAIDs or an opiate analgesic) should be given to control symptoms as dictated by individual patient situation. Recommendation: Strong recommendation, low-quality evidence.
Spontaneous or Passive Thawing
According to the foregoing guidelines, rapid rewarming is strongly recommended. If field rewarming is not possible, spontaneous or slow thawing should be allowed. Slow rewarming is accomplished by moving to a warmer location (eg, tent or hut) and warming with adjacent body heat from the patient (eg, frozen hands/fingers in the axillae) or a caregiver, as previously described. The expert panel agrees that slow thawing is a reasonable course of action to initiate the rewarming process if it is the only means available. Recommendation: Strong recommendation, very low-quality evidence.
Debridement of Blisters
Debridement of blisters should not be routinely performed in the field. If a clear, fluid-filled blister is tense and at high risk for rupture during evacuation, blister aspiration and application of a dry gauze dressing should be performed in the field to minimize infection risk. Hemorrhagic bullae should not be aspirated nor debrided in the field. These recommendations are common practice but lack evidence beyond case series. Recommendation: Weak recommendation, low-quality evidence.
Topical Aloe Vera
Aloe vera ointment has been shown in an observational study 38 and an animal model 32 to improve frostbite outcome by reducing prostaglandin and thromboxane formation. Topical agents do not penetrate far into tissues, however, so aloe vera is theoretically only beneficial for superficially injured areas. The study supporting the benefit of aloe vera examined its application on unroofed blebs where it would be able to penetrate underlying tissue. Topical aloe vera should be applied to thawed tissue before application of dressings. Recommendation: Weak recommendation, low-quality evidence.
Dressings
Bulky, dry gauze dressings should be applied to the thawed parts for protection and wound care. Substantial edema should be anticipated, so circumferential dressings should be wrapped loosely to allow for swelling without placing pressure on the underlying tissue. Recommendation: Strong recommendation, low-quality evidence.
Ambulation and Protection
A risk-benefit analysis must consider the potential for further trauma and, ultimately, potentially higher morbidity if a thawed part is used for ambulation. For example, it might be reasonable to walk on a foot with thawed toes for evacuation purposes, but it is inadvisable to walk on a recently thawed frostbitten foot because of the potential resulting morbidity. Very little evidence is available to guide recommendations. In one study, mobilization within 72 h after thawing did not affect tissue loss, complications, or hospital length of stay. 39 After the rewarming process, swelling should be anticipated. If passive thawing has occurred, boots (or inner boots) may need to be worn continuously to compress swelling. Boots that were removed for active rewarming may not be able to be redonned if tissue swelling has occurred during the warming process. The panel's clinical experience supports the concept that a recently thawed extremity should ideally not be used for walking, climbing, or other maneuvers and should be protected to prevent further trauma.38,40 Recommendation: Weak recommendation, low-quality evidence.
Elevation of Extremity
If possible, the thawed extremity should be elevated above the level of the heart, which might decrease the formation of dependent edema. Recommendation: Strong recommendation, low-quality evidence.
Oxygen
Recovery of thawed tissue partly depends on the level of tissue oxygenation in the postfreezing period. One small study that measured hand temperature at normobaric hypoxia found decreased skin temperatures with decreasing FIO2. 14 However, hyperoxia has been found to cause vasoconstriction in the extremities 41 and may increase mortality in patients with accidental hypothermia. 42 While the exact effects of hypoxia and hyperoxia are not completely described, oxygen should not be applied routinely to patients who are not hypoxic. Although evidence is lacking to support use of supplemental oxygen for frostbite, oxygen may be delivered by face mask or nasal cannula if the patient is hypoxic (oxygen saturation <88%) or at high altitude above 4000 m. Recommendation: Weak recommendation, low-quality evidence.
For a summary of the suggested approach to the field treatment of frostbite, see Table 2.
Summary of initial hospital management of frostbite

Immediate Medical Therapy—Hospital (or High-Level Field Clinic)
Once the patient reaches the hospital or field clinic, a number of treatments should be initiated. After reaching the hospital or field clinic, potential therapeutic options for frostbite are as follows.
Treatment of Hypothermia
Similar recommendations apply to hospital or field clinic treatment of hypothermia before frostbite treatment (see previous). Recommendation: Strong recommendation, low-quality evidence.
Hydration
Similar recommendations apply in the hospital or field clinic regarding hydration (see previous). Recommendation: Strong recommendation, low-quality evidence.
Low Molecular Weight Dextran
Similar recommendations apply in the hospital or field clinic regarding LMWD (see previous). Recommendation: Weak recommendation, low-quality evidence.
Rapid Rewarming of Frozen Tissues
Frozen tissue should be assessed to determine whether spontaneous thawing has occurred. If tissue is completely thawed, further rewarming will not be beneficial. Rapid rewarming should be undertaken according to the field protocol described previously if the tissue remains partially or completely frozen. Recommendation: Strong recommendation, moderate-quality evidence.
Management of Blisters
Clear or cloudy blisters contain prostaglandins and thromboxanes that may damage underlying tissue. Hemorrhagic blisters are thought to signify deeper tissue damage extending into the dermal vascular plexus. Common practice is to drain clear blisters (eg, by needle aspiration) while leaving hemorrhagic blisters intact.36,38,40,41,43,44 Although this approach to frostbite blister management is recommended by many authorities, comparative studies have not been performed and data are insufficient to make absolute recommendations. Some authors argue that unroofing blisters might lead to the desiccation of exposed tissue and that blisters should only be removed if they are tense, likely to break or be infected, or interfere with the patient's range of motion. 45 Others recommend debridement or aspiration because the advantages of minimizing the desiccation of the epidermis as the fluid collects outweigh the infection risk. 46 In a remote field situation, draining or unroofing blisters may not be under the control of the provider. Blisters most often will have been broken by the patient's boots. In this case, the most important treatment is applying aloe vera and a sterile dressing to the unroofed blister. Debridement or aspiration of clear, cloudy, or tense blisters should be performed after the patient arrives at the hospital. Recommendation: Weak recommendation, low-quality evidence.
Topical Aloe Vera
Topical aloe vera cream or gel should be applied to the thawed tissue before application of dressings. Aloe vera is reapplied at each dressing change or every 6 h. 38 Recommendation: Weak recommendation, low-quality evidence.
Systemic Antibiotics
Frostbite is not an inherently infection-prone injury. Therefore, antibiotic administration specifically for preventing infection during or after frostbite injury is not supported by evidence. Some authorities reserve antibiotics for situations when edema occurs after thawing because of the notion that edema increases skin susceptibility to infection by gram-positive bacteria. 40 However, this practice is not based on evidence. Systemic antibiotics, either oral or parenteral, should be administered to patients with significant trauma, other potential infectious sources, or signs and symptoms of cellulitis or sepsis. Recommendation: Strong recommendation, low-quality evidence.
Tetanus Prophylaxis
Tetanus prophylaxis should be administered according to standard guidelines. Recommendation: Strong recommendation, low-quality evidence.
Ibuprofen
If NSAIDs have not been initiated in the field, ibuprofen should be administered at a dose of 12 mg/kg divided twice daily (to inhibit harmful prostaglandins but remain less injurious to the gastrointestinal system 31 ) until the frostbite wound is healed or surgical management occurs (typically for 4 to 6 wk). Recommendation: Weak recommendation, low-quality evidence.
Thrombolytic Therapy
The goal of thrombolytic therapy in frostbite injury is to lyse and clear microvascular thromboses. For deep frostbite injury with potential amputations, the use of either IV or intra-arterial tissue plasminogen activator (tPA) within 24 h of injury may salvage some or all tissue at risk. While there are no randomized or controlled studies in human frostbite injury with thrombolytics, many case reports and series support its use. A retrospective, single-center review by Bruen et al 47 demonstrated a reduction in digital amputation rates from 41% in those patients who did not receive tPA to 10% in patients receiving intra-arterial tPA within 24 h of injury. The 20-y series presented by the Regions Hospital group found that two-thirds of patients who received intra-arterial tPA responded well and that amputation rate correlated closely with angiographic findings. 48 The Massachusetts General Hospital group has proposed a screening and treatment tool for thrombolytic management of frostbite based on a case report and their evaluation of the Utah and Minneapolis experiences. 49 Twomey et al 50 from Hennepin County Medical Center have developed a specific protocol based on a small group of good outcomes with intravenous tPA. Further study is needed to compare intra-arterial versus IV tPA for tissue salvage and functional outcome. Animal studies demonstrate the benefit of thrombolytics. 51 When considering using a thrombolytic, a risk-benefit analysis is necessary. tPA is contraindicated in trauma, recent surgery, recent stroke, and many other conditions that might pose a bleeding risk. Only deep injuries with likely amputations (eg, extending into the proximal interphalangeal joints of digits, Grades 3–4) should be considered for thrombolytic therapy. Potential risks of tPA include systemic and catheter site bleeding, compartment syndrome, and failure to salvage tissue. The long-term, functional consequences of digit salvage using tPA have not been evaluated. Thrombolytic treatment should ideally be done in a facility familiar with the technique and with intensive care monitoring capabilities in case of complications. If a frostbite patient is being cared for in a remote area, transfer to a facility with tPA capabilities should be considered if tPA can be started within 24 h of tissue thawing. Time to thrombolysis appears to be critical, with best outcomes when administered within 12 h and ideally as soon as possible. Recent work from Hennepin County has found that each hour of delay of thrombolytic therapy results in a 28% decrease in digit salvage. 52 Field use of tPA has shown variable results 53 and should only be undertaken with extreme caution because bleeding complications may be impossible to detect and treat. Evidence of significant trauma in the field setting is an absolute contraindication to tPA. However, tPA initiated remotely in medical centers prior to transfer to a regional treatment center has shown improved results with lower odds of amputation compared to patients with tPA initiated at the tertiary care center or the group receiving no tPA, with only one patient in the remote tPA group having significant bleeding requiring blood transfusion. 54
Method of Administration
IV dosing of tPA is typically 0.15 mg/kg bolus followed by 0.15 mg/kg/h for 6 h plus heparin for 3–5 d. IA dosing varies; a typical regimen is a 2-4 mg bolus followed by 1 mg/h for 24 to 48 h, plus heparin or LMWH. 46 Intra-arterial angiography or IV pyrophosphate scanning should be considered to evaluate the initial injury and monitor progress after tPA administration as directed by local protocol and resources. The various reports on the use of thrombolytics in frostbite through 2022 55 include the following: 1 randomized controlled prospective trial in which tPA was combined with iloprost, 16 patients), 56 1 retrospective observational study (125 patients), 57 3 retrospective cohort studies (59 patients),47,52,58 11 retrospective case series (∼200 patients),48,50,59–67 and 3 case reports.49,68,69 Further work is needed to determine the absolute efficacy of tPA for frostbite injury and to compare intra-arterial and IV tPA to IV prostacyclin.
We suggest IV or intra-arterial tPA for the treatment of severe frostbite (Cauchy Grade 3 and 4) within 24 h of injury, especially if iloprost is not available. tPA initiated early and remotely following a standardized protocol at community hospitals prior to transfer to a tertiary care center may safely decrease the amputation rate. 54 Recommendation: Strong recommendation, low-quality evidence.
Iloprost
Iloprost, a prostacyclin (PGI2) analogue, is a potent vasodilator that also reduces inflammation, inhibits platelet aggregation, down-regulates lymphocyte adhesion to endothelial cells, 70 and may have fibrinolytic activity. 71 Intravenous iloprost was first used for treatment of frostbite by Groechenig in 1994 in 5 patients with second- and third-degree frostbite. He infused iloprost daily, starting at 0.5 ng/kg and increasing to 2.0 ng/kg total dose over 3 d, and then continued for between 14 and 42 d. 72 Recovery without amputation was achieved in all patients. Subsequent reports are limited to case series with one exception.
An open-label randomized trial by Cauchy et al assessed the efficacy of: 1) IV buflomedil 400 mg/d, an alpha-blocker vasodilator; 2) iloprost, 0.5 ng to 2 ng/kg/min IV for 6 hours per day; or 3) IV iloprost 2 ng/kg/min for 6 hours per day plus fibrinolysis with 100 mg recombinant tissue plasminogen activator (tPA) for the first day only. 56 All patients received 250 mg aspirin PO and 400 mg IV buflomedil prior to randomization. Forty-seven patients with severe frostbite (Stages 3–4), with 407 digits at risk, were randomly assigned matched for clinical severity to 8 d of treatment with the 3 different regimens. Iloprost alone (0% amputation rate) was found superior to tPA plus iloprost (19%) and buflomedil (60%) groups. Although these differences were significant, the confidence intervals were wide, and the data overall were considered low quality by a Cochrane Review. 73 A further limitation of this study was that ischemia was not documented with angiography or technetium scanning before treatment.
The only other studies of iloprost for frostbite are case series. A Canadian study documented full recovery of Grade 3 frostbite when iloprost was started within 48 h of injury in 2 long-distance runners. 74 In a Finnish study, iloprost was partially beneficial with a digit salvage rate of 78% in 4 persons: 2 with contraindication for tPA, 1 with failed tPA therapy, and 1 with vasospasm without thrombosis on angiography. 59 One patient with minimal response to tPA had complete reperfusion with iloprost. Extending the treatment window, Pandey et al 75 reported good results with iloprost therapy up to 72 h after injury. In 5 Himalayan climbers with 34 digits at risk, 5 d of daily iloprost infusion produced excellent outcomes in 4 of 5 patients. Treatment delayed beyond 72 h has not been beneficial except in 1 patient.76,77
Recent studies from Canada in rural 73 and urban centers 78 have shown improved outcomes with iloprost. In urban Calgary, in a retrospective study with 90 patients with Grades 2–4 frostbite (26 treated with iloprost and 64 with standard care), there was no amputation in Grade 2 frostbite for either group. Grades 3 and 4 injuries had lower amputation rates with iloprost compared to usual care (18% vs 44% amputation rate in Grade 3 injuries, and 46% vs 95% in Grade 4 frostbite). 78 Among 22 patients in Yukon territory with frostbite of 142 digits, all were treated with iloprost, and some received alteplase. There was 100% digit salvage rate for Grades 2 and 3 injuries treated with iloprost (15 patients). Grade 4 frostbite (5 patients) treated with iloprost + alteplase (tPA) + heparin and iloprost alone (2 patients) showed digit salvage rate of 50% with 4/7 patients requiring amputation. Two out of 5 patients in the iloprost + alteplase + heparin group had bleeding complications requiring blood transfusion while 2/17 in the iloprost group had complications attributable to anticoagulant/ibuprofen use and not to iloprost. 10 Iloprost was well tolerated without major side effects in all these studies. Despite the limitations of these case studies, iloprost has shown consistently favorable effects and appears safe.
In the absence of large-scale randomized trials and in light of available evidence, intravenous iloprost should be considered a first-line therapy for Grades 3 and 4 frostbite <48 h after thawing, and possibly for up to 72 hours. It should be given as soon as possible after rewarming. In countries in which the IV form of iloprost is not yet approved, thrombolysis with IV or IA tPA is the available treatment for Grade 4 frostbite, and possibly Grade 3 frostbite. It is unclear if iloprost is indicated in Grade 2 frostbite. Field use of both iloprost and IV tPA has been advocated to reduce delay in treatment for mountaineers who will invariably take >48 h for evacuation to a hospital. 53 In these situations, iloprost may be the safer alternative since it eliminates the bleeding risks of tPA. The optimal duration for iloprost therapy has not been established. Clinical reports vary in duration from one day to 48 days, with most reports using 5 to 8 days. Considering the lack of robust data, a reasonable approach is to use iloprost for 5 to 8 days, perhaps guiding termination or extension of therapy based on evidence of adequate reperfusion, such as with fluorescein microangiography, technetium bone scanning, or other imaging.
We suggest iloprost for deep frostbite extending to the distal interphalangeal joint or more proximal (Grades 2–4), up to 72 hours after rewarming but ideally as soon as possible. Recommendation: Strong recommendation, low-quality evidence.
Heparin
No evidence supports the use of low molecular weight heparin or unfractionated heparin for initial management of frostbite in the field or hospital, although climbers and practitioners in many regions use these medications. Evidence supports use of heparin as adjunctive therapy in tPA protocols, as described previously. Heparin has been used in conjunction with iloprost as well; the 5 patients in the 1994 Groechenig iloprost study, 72 the 1 Israeli traveler with excellent outcome in the Kathmandu study, 75 and 4 patients in the Finnish study 59 were treated with low molecular weight heparin (enoxaparin) in addition to iloprost. Whether low molecular weight heparin offers additional benefit when combined with iloprost requires further investigation; currently data are insufficient for a recommendation on this combination. Recommendation: We do not suggest the use of heparin as monotherapy: weak recommendation, low-quality evidence.
Other Vasodilator Therapy
Vasodilators, such as prostaglandin E1, 79 nitroglycerin, 49 pentoxifylline,80,81 phenoxybenzamine, nifedipine, reserpine,82,83 and buflomedil,56,84,85 have been used as primary and adjunctive therapies for treatment of frostbite. In addition to vasodilation, some of these agents might also prevent platelet aggregation and microvascular occlusion. Sheridan et al 49 recommend intra-arterial infusion of nitroglycerin during angiography before tPA infusion. A study in rabbits that did not undergo rapid rewarming found benefit from intra-arterial administration of prostaglandin E1. 79 Buflomedil is an alpha-adrenolytic agent that was used widely in Europe with preliminary and anecdotal evidence of good results56,85; however, animal models have not replicated these findings. 84 Buflomedil is not approved by the US Food and Drug Administration and has been withdrawn in Europe because of reports of severe adverse neurological and cardiac events after its administration. 73 Intra-arterial reserpine studied in a controlled trial was found not to be effective. 82
Pentoxifylline, a methylxanthine-derived phosphodiesterase inhibitor, has been widely used for the treatment of peripheral vascular disease and yielded promising results in animal81,86,87 and human frostbite. 80 Hayes et al 80 recommend pentoxifylline in the controlled-release form of one 400 mg tablet 3 times a day with meals, continued for 2 to 6 wk. Controlled studies of pentoxifylline in management of frostbite have not been performed.
Certain vasodilators have the potential to improve outcomes and can be used with minimal risk. However, as discussed earlier, data demonstrating benefit are limited. Iloprost is the only vasodilator with reasonable scientific evidence supporting its use.
For a summary of the suggested approach to hospital or advanced field clinic treatment of frostbite, see Table 3.
Other Post-thaw Medical Therapy
Once the patient has received initial frostbite therapy, long-term management is initiated to reduce long-term sequelae. Therapeutic options for frostbite after thawing are as follows.
Hydrotherapy
Daily or twice-daily hydrotherapy at 37 °C to 39 °C (98.6 to 102.2°F) has been recommended in the post-thaw period.34,36–38,88 Hydrotherapy theoretically increases circulation, removes superficial bacteria, and debrides devitalized tissue. 40 No trials support improved outcomes, but the practice has few negative consequences and has the potential to benefit recovery. Data are insufficient to recommend specific temperature, timing, or duration of therapy. Recommendation: Strong recommendation, low-quality evidence.
Hyperbaric Oxygen Therapy
The use of hyperbaric oxygen therapy (HBOT) is well-studied in wound care, but there is a paucity of data for use in the treatment of frostbite. HBOT has a direct action on tissue ischemia by improving oxygen in the tissue, decreasing blood viscosity, reducing the inflammatory cascade, and decreasing bacterial load, helping to prevent infection. 89 HBOT can decrease edema and compartment syndrome and has a positive effect on ischemia–reperfusion. 90 With repeated HBOT sessions, vascular endothelium growth factor is activated, as well as fibroblast and collagen production, therefore lessening tissue damage. 91
HBOT was first reported in frostbite injuries in 1963 by Ledingham. 92 Despite anecdotal success in limited case series,93–99 randomized controlled studies on HBOT and frostbite may not be performed due to the time and cost involved in HBOT. However, the investigations in retrospective studies 13 and a recent prospective case-controlled study by Magnan et al 100 showed improvement in wounds when HBOT was used.
Additionally, there are an increasing number, albeit still few, case studies showing clinical improvement in frostbite injury after HBOT, even in delayed presentations. HBOT may improve severe frostbite outcomes, thus decreasing significant morbidity. The time, expense, and availability of HBOT limits its use, however. If HBOT is available and feasible, incorporating HBOT as an adjunct into the treatment plan may be of benefit. Exact treatment protocols need to be studied. Recommendation: No recommendation owing to insufficient data.
Imaging
In patients with delayed presentation (4–24 h from the time of the frostbite thawing), noninvasive imaging with technetium pyrophosphate 13 or magnetic resonance angiography 101 can be used at an early stage to predict the likely levels of tissue viability for amputation. Cauchy et al 13 described the combination of a clinical scoring system and technetium scanning to successfully predict subsequent level of amputation on Day 2 after frostbite rewarming. Single photon emission computed tomography (SPECT/CT) combines the anatomic precision of CT with the functional vascular information obtained from multiphase bone scintigraphy. Kraft et al used single photon emission CT/CT for 7 patients with frostbite and found it improved surgical planning for deep frostbite injuries by enabling early and precise anatomic localization of nonviable tissues.102,103 The techniques are evolving, and multiphase [Tc99m]-MDP bone scintigraphy with SPECT/CT after a severe frostbite has been described. 104
If available, appropriate imaging should be used to assess tissue viability and guide timing and extent of amputation. Recommendation: Weak recommendation, low-quality evidence.
Other potentially useful imaging techniques include Doppler ultrasound 59 ; triple phase technitium59,105; indocyanine green microangiography 98 ; and thermal imaging. 14 Although some of these techniques show potential, further studies are required to determine their exact role.
Sympathectomy
Because blood flow is partly determined by sympathetic tone, chemical or surgical sympathectomy has been proposed in the immediate postexposure phase to reduce tissue loss. In a rat lower limb model, early surgical denervation (within 24 h of exposure) reduced tissue loss but had no effect if performed after 24 h. 106 In a rabbit ear model, procaine-induced sympathectomy had no demonstrable beneficial effect. 107 Frostbite patients often experience long-term delayed symptoms, such as pain, paresthesia, and numbness. Chemical or surgical sympathectomy to treat these symptoms has been performed with variable results. In some studies, surgical sympathectomy has been found to reduce the duration of pain and expedite demarcation of tissue necrosis. However, it has not been found to reduce the ultimate extent of tissue loss.43,108 Acute treatment success with IV guanethidine has been reported 109 but was not beneficial in another case report. 110 Sympathectomy may have a role in preventing certain long-term sequelae of frostbite, such as pain (putatively caused by vasospasm), paresthesia, and hyperhidrosis.111,112 Despite many years of study, the data on surgical sympathectomy are limited and conflicting and have been delegated to a historical treatment with no demonstrable benefit with significant risk and potential long-term sequelae. Recommendation: We suggest that sympathectomy not be considered. Weak recommendation, low-quality evidence.
Hospitalization
Hospital admission and discharge are determined on an individual basis. Factors should include severity of the injury, coexisting injuries, comorbidities, and need for hospital-based interventions (tPA, vasodilators, surgery) or supportive therapy, as well as ease of access to appropriate community medical and nursing support. Significant swelling should prompt evaluation for compartment syndrome and admission for observation. Patients with superficial frostbite can usually be managed as outpatients or with brief inpatient stays followed by wound care instructions. Initially, deep frostbite should be managed in an inpatient setting. Recommendation: Weak recommendation, low-quality evidence.
Fasciotomy
Thawing results in reperfusion of ischemic tissue and, in turn, sometimes results in elevated pressures within closed soft-tissue compartments. Compartment syndrome clinically manifests as tense, painful distention with reduced movement and sensation. Urgent attention is necessary to evaluate compartment pressures. If elevated compartment pressures are present, prompt surgical decompression is indicated for limb salvage. 21 Recommendation: Strong recommendation, low-quality evidence.
Surgical Treatment or Amputation
After frostbite occurs, complete demarcation of tissue necrosis may take 1 to 3 mo. Angiography, technetium-99 bone scan, or magnetic resonance imaging may be used to assist determination of surgical margins44,101,113 in conjunction with clinical findings. If the patient exhibits signs and symptoms of sepsis attributed to infected frostbitten tissue, amputation should be performed expeditiously. 88 Otherwise, amputation should be delayed until definitive demarcation occurs. The affected limb is often insensate. Therefore, an approach that addresses footwear and orthotics is essential to provide optimal function. Our experience has found that early involvement of a multidisciplinary rehabilitation team produces better long-term functional results. Telemedicine or electronic consultation with a surgical frostbite expert to guide local surgeons should be considered when no local expert is available. Because significant morbidity may result from unnecessary or premature surgical intervention, a surgeon with experience evaluating and treating frostbite should assess the need for and the timing of any amputation. Recommendation: Strong recommendation, low-quality evidence.
Conclusions
This summary provides evidence-based guidelines for prevention and treatment of frostbite. Many important questions remain and should serve as a focus for future research. This includes elucidation of pathophysiology, medications to assist in the prevention of frostbite, peri-thawing procedures to reduce injury and decrease morbidity, and post-thaw therapies that might improve long-term outcomes.
Supplemental Material
sj-pdf-1-wem-10.1177_10806032231222359 - Supplemental material for Wilderness Medical Society Clinical Practice Guidelines for the Prevention and Treatment of Frostbite: 2024 Update
Supplemental material, sj-pdf-1-wem-10.1177_10806032231222359 for Wilderness Medical Society Clinical Practice Guidelines for the Prevention and Treatment of Frostbite: 2024 Update by Scott E. McIntosh, MD, MPH, Luanne Freer, MD, Colin K. Grissom, MD, George W. Rodway, PhD, APRN, Gordon G. Giesbrecht, PhD, Marion McDevitt, DO, MPH, Christopher H. Imray, MD, Eric L. Johnson, MD, Prativa Pandey, MD, Jennifer Dow, MD, and Peter H. Hackett, MD in Wilderness & Environmental Medicine
Footnotes
Author Contribution(s)
Drafting of the manuscript; critical revision of the manuscript; and approval of final manuscript (SEM, LF, CKG, PSA, GWR, AC, GGG, MM, CHI, ELJ, PP, JD, PHH).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material(s)
Supplementary material associated with this article can be found in the online version at https://doi.org/10.1177/10806032231222359.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
