Abstract
Introduction
Bothriechis schlegelii is a Crotaline viperid species of Central America and Northern South America. The characteristics of its envenomation have not been well established. We present clinical characteristics of human cases evaluated and treated in a hospital in southwestern Colombia.
Methods
We evaluated data from patients who suffered Bothriechis schlegelii envenomation and were seen at Fundación Valle del Lili Hospital, Cali, Colombia between 2011 and 2022.
Results
Eight patients were included, with a median age of 24 years. Snakebites occurred in rural areas. Six (75%) patients were bitten on the upper extremities in relation to the arboreal habits of this animal. The most common symptoms were pain and edema (N = 8, 100%), ecchymoses (N = 2, 25%), and paresthesia (N = 2, 25%). The most common systemic findings were hypofibrinogenemia (N = 8, 100%) and prolonged prothrombin time in five patients (N = 5, 62.5%). All were treated with polyvalent antivenom for Colombian snakes, with a good response and outcome.
Conclusions
Most bite sites from B. schlegelii were on the upper limbs. All patients had both local manifestations, including edema, pain, and systemic effects with hypofibrinogenemia, but none had systemic bleeding. Every patient received antivenom and had favorable outcomes.
Introduction
Bothriechis schlegelii (eyelash viper or eyelash pit viper) is a venomous snake species inhabiting regions from southern Mexico to northern Peru and belonging to the family Viperidae and subfamily Crotalinae. 1 It is a relatively small species, rarely exceeding 75 cm in length and has a triangular head, vertical pupils, a loreal pit on each side between the eye and the snout, prominent supraciliary scales (from which its popular name is derived), and prehensile tail (Figure 1A). The color is variable (Figure 1B).1–3 It inhabits tropical and humid forests, humid subtropical forests (cloud forests), and very humid mountain forests. 1 It feeds on small rodents, frogs, snakes, lizards, and small birds. It is ovoviviparous and has arboreal habits; hence, it bites humans on the upper limbs and head more frequently than on the lower limbs.4,5

A, Classical characteristics of B. schlegelii according to the illustration in the book Los ofidios venenosos del Cauca by Evaristo García (1896): Arboreal snake with a prehensile tail and supraorbital scales in the form of eyelashes (Ref. 2) (With permission for its publication given by the “Evaristo Garcia Foundation” publishers). B, Two individuals from two geographical areas of southwestern Colombia. (Photographs by the author FCH).
The venom of the eyelash viper, similar to that of all snakes of the Viperidae family, is comprised of a cocktail of substances with biological effects that function to immobilize and kill prey and possibly initiate the digestive process. 6 Bites in humans occur infrequently, usually as the result of a defensive reflex. Therefore, envenomation has been rarely described.7–10 The biological effects of the components of the venom and the derived clinical manifestations are of various types. The most important components are phospholipase A2 related to the production of edema, tissue damage and myotoxicity, metalloproteases with dermonecrosis, L-amino acid oxidases with tissue damage, serine proteases with tissue damage and hemorrhagic diathesis, lectin type C with tissue damage and hemorrhagic diathesis, disintegrins with detachment of cells from their extracellular matrix leading to blisters and platelet function impairment, bradykinin-potentiating peptides with hypotension, enzymes that degrade fibrinogen, plasminogen activators, prothrombin activators, factor V activators, factor X activators, and anticoagulant activities (including inhibitors of prothrombinase complex formation, inhibitors of thrombin, phospholipases, and protein C activators).7,11
This paper presents the clinical characteristics and outcome of eight cases of envenomation by B. schlegelii treated in a tertiary health center hospital in Cali, Colombia, where patients from southwestern Colombia are treated.
Methods
We evaluated the electronic medical records of patients with envenomation by B. schlegelii treated between January 2015 and June 2022 at the Fundación Valle del Lili (FVL) Hospital, a private quaternary-level university hospital that serves as a referral center for cases of envenomation by vipers in southwestern Colombia. It is in the city of Cali, has 668 beds, and attends around 90,000 emergencies per year. Since 2015, the clinical and laboratory records of patients treated for snake envenomation have been retained. Patient characteristics, laboratory data, and the classification of the degree of envenomation and treatment, including antivenom administration and doses required to control the disease, were obtained.
The snake species that caused the envenomation were identified either by photographic registries or by direct evaluation of the living or dead specimens. The authors FCH and SCV identified the snakes. Both of them are herpetologists with extensive field experience in Colombia; FCH, a professor at the Universidad del Valle in Cali, Colombia, has been recording snakes from southwestern Colombia for more than 40 years and is very familiar with this species. As our hospital is a private teaching hospital, by protocol, any snakebite patients are usually required to be hospitalized for at least 24 h to determine envenomation severity.
No specific antivenom is available for B. schlegelii. Based on the knowledge that polyvalent antivenoms created from Bothrops atrox, B. asper, Crotalus durissus, and Lachesis muta venoms have a broad spectrum of neutralizing efficacy against venoms of snakes of the other genera, such as Agkistrodon, Atropoides, Bothriechis, Bothrops, Cerrophidion, and Porthidium,8–12 we used a polyvalent antivenom approved for Colombian viper envenomation (produced by Laboratorios Probiol, Carrera 8 # 187A-53, Bogotá D.C., Colombia, http://www.probiol.com); each vial contains 10 mL of equine polyvalent antivenom that the manufacturer reports can respectively neutralize at least 25, 10, and 5 mg of the venom of B. atrox/asper, C. durissus, and L. muta. The antivenom is obtained from horses immunized with whole venom.
The number of vials of the polyvalent antivenom was calculated according to the initial classification of the envenomation grade. These recommendations are given by national guidelines for “bothropic type” envenomation:
12
Grade 0: Patient free of local or systemic symptoms or signs 6 h after the bite. Do not administer antivenom serum. Grade I: Local edema of less than 20 cm. Moderate local pain; no systemic symptoms or signs. No changes were observed in the coagulation test results. Administer two vials. Grade II: Edema greater than 20 cm. Local pain and bleeding and mild systemic symptoms. Fibrinogen levels are between 150 and 200 mg/dL. Administer between 3 and 5 vials. Grade III: Marked local and systemic symptoms, emesis, diarrhea, systemic bleeding, and abnormal renal function (creatinine levels greater than 1.3 mg/dL) and coagulation test results. Fibrinogen levels are between 50 and 150 mg/dL. Administer between 6 and 9 vials. Grade IV: Edema with rapid proximal progression reaching the trunk, shock, oliguria, anuria, no detection of plasma fibrinogen, and prothrombin time (PT) and activated partial thromboplastin time (aPTT) >150 and 340 s, respectively. Administer between 10 and 15 vials.
Subsequent doses (1–4) were added every 6–8 h until the results of the coagulation tests (PT, aPTT, and fibrinogen levels) normalized. Disturbance in coagulation has been considered a good indicator of venom activity. Studies about redosing are lacking in this regard.
This study complied with the Declaration of Helsinki and was approved by the Ethics Committee of FVL (protocol number 599). Patients 1 and 2 (Table 1) signed informed consent forms and declared their authorization to publish their clinical images (Figure 2B and Figure 2C). Informed consent and authorization to publish the image (Figure 2A) of Patient 7 (Table 1) were provided by her mother.
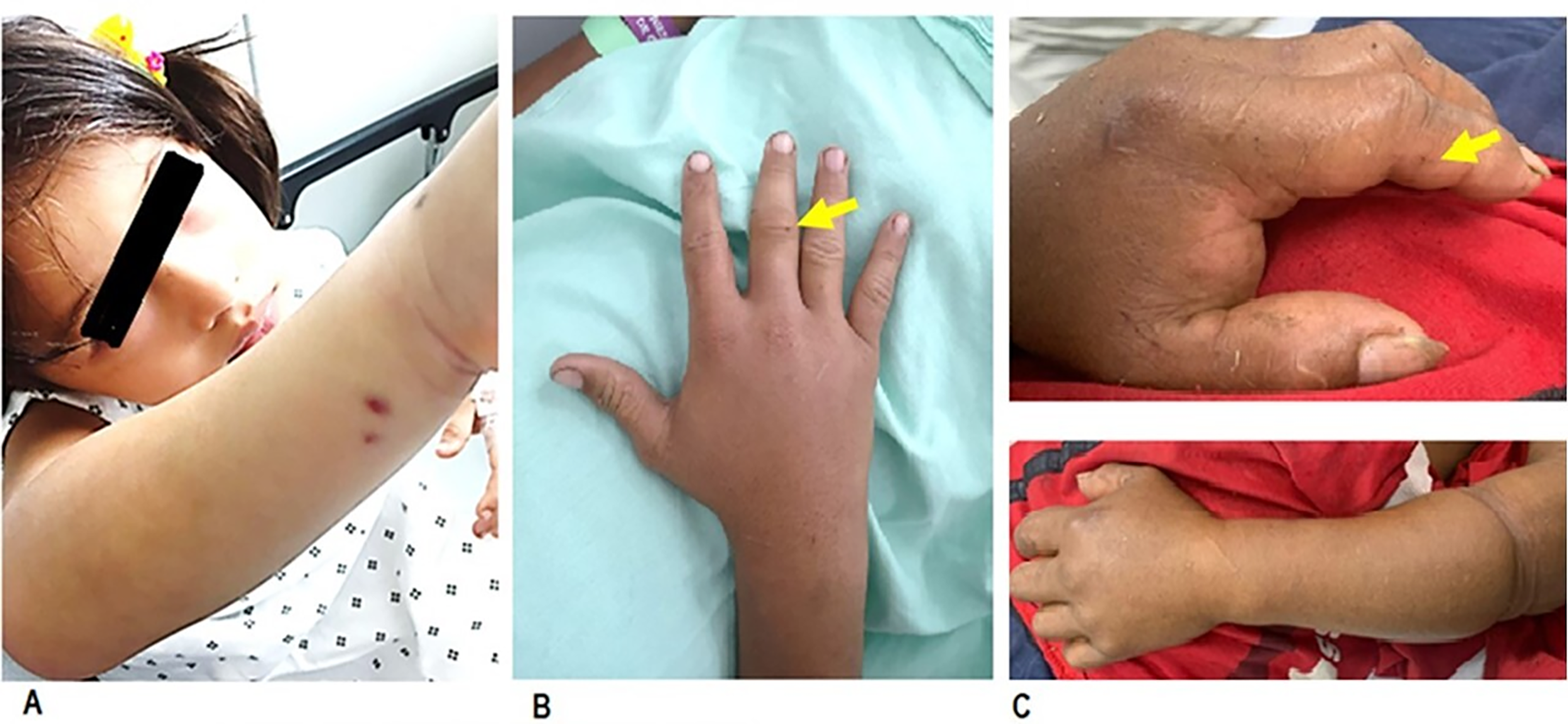
A, A 4-year-old patient was bitten on the posterior aspect of her right forearm, where her skin is thin. The punctate wounds related to the entry of both fangs are very well appreciated. The wounds showed slight bleeding, and minor edema was observed. The degree of envenomation was classified as Grade II. B, A 23-year-old patient with a single fang bite on the third finger of the right hand. He presented moderate edema in his hand. Envenomation was classified as Grade III. C, A 42-year-old patient. The entrance of the fangs is barely noticeable on the second finger of the left hand. The skin of his fingers is calloused due to his farming profession. However, he presented with Grade IV envenomation with significant edema in the left upper extremity. The misused tourniquet effect is appreciated. (Photographs by the author CAC).
Clinical, laboratory, and antivenom treatment characteristics of 8 patients with B. schlegelii envenomation.
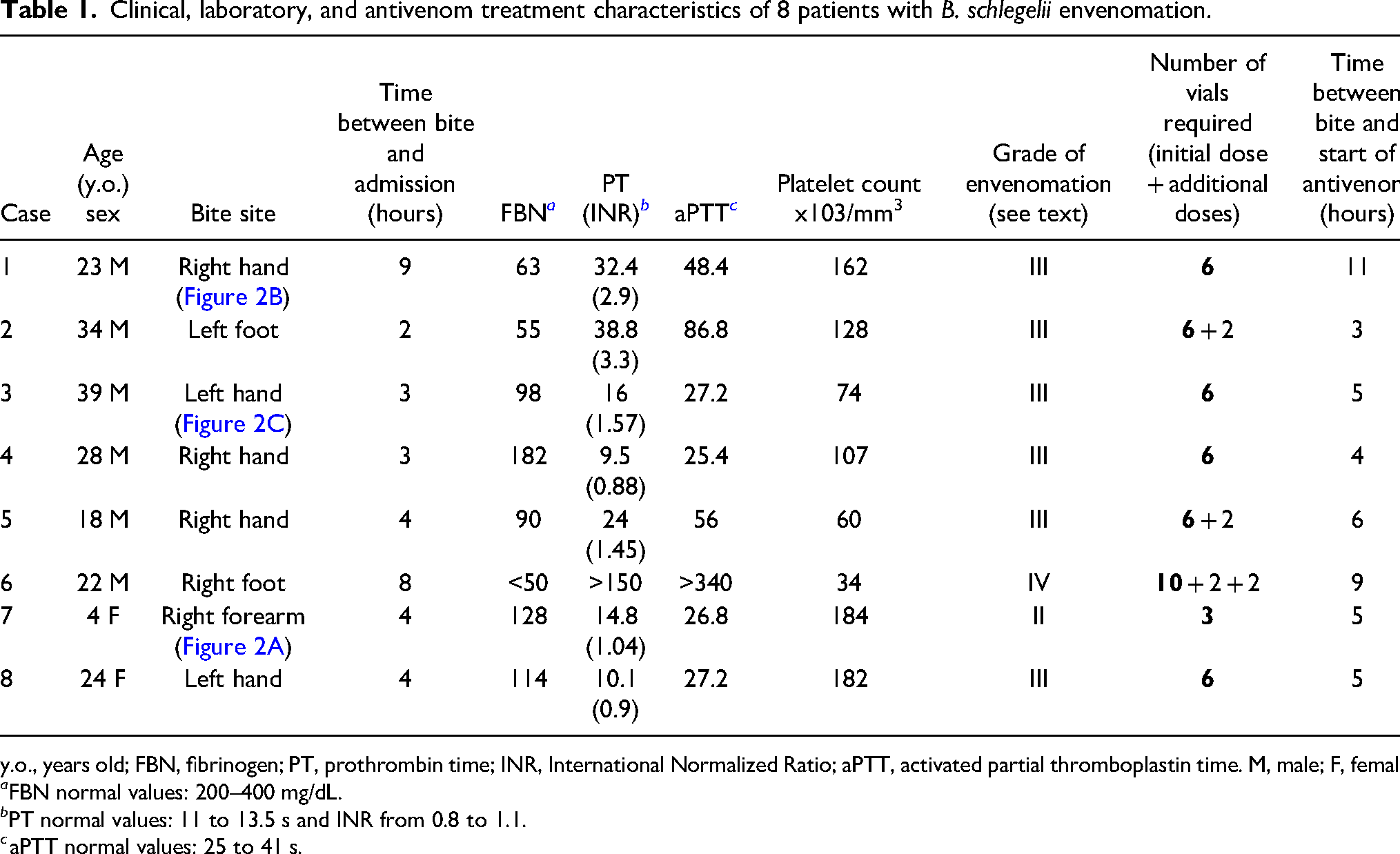
y.o., years old; FBN, fibrinogen; PT, prothrombin time; INR, International Normalized Ratio; aPTT, activated partial thromboplastin time. M, male; F, female.
FBN normal values: 200–400 mg/dL.
PT normal values: 11 to 13.5 s and INR from 0.8 to 1.1.
aPTT normal values: 25 to 41 s.
Results
Patient's General and Clinical Characteristics
Seventy-two cases of snakebites were admitted to the FVL emergency department between 2011 and 2022, of which 58 patients had envenomation by viperid species. Eight (12.9%) cases were caused by B. schlegelii, 12 patients were bitten by B. asper, 10 by B. rhombeatus, 3 by B. punctatus, 2 by B. colombianus, 1 by B. myersi, and 1 by P. nasutum; in 21 patients, the species causing the envenoming was not identified.
The patients were relatively young—the youngest patient was 4 years old and the oldest 39 (mean: 26.9 and SD: 6.77)—and were primarily male (N = 6, 75%). Snakebites occurred in rural areas while recipients engaged in farming or agriculture (N = 2, 25%), trekking (N = 1, 12.5%), working as a professional soldier (N = 1, 12.5%), camping (N = 1, 12.5%), and working as a biologist on a field trip (N = 1, 12.5%). Half of the patients were bitten in the Valle del Cauca territory and the other half were bitten in the Cauca territory. Six (75%) patients were bitten on the upper extremities, and 2 (25%) were bitten on the lower extremities.
All patients were hospitalized. The most common clinical symptoms among these patients were local pain and edema (N = 8, 100%), followed by ecchymoses (N = 2, 25%) and paresthesia (N = 2, 25%). The puncture wound caused by the snakebite may reflect the introduction of one or both fangs; sometimes, these fang marks cannot be located, mainly in patients with callused hands (Figure 2). The edema was variable and related to the severity of envenomation. The most common systemic symptoms were hypofibrinogenemia (N = 8, 100%), prolonged PT (N = 5, 62.5%), and prolonged aPTT (N = 3, 37.5%). Most patients had neutrophilia with a mean neutrophil count of 7430/mm3. Mean platelet count was 116375/mm3 (SD: 53000/mm3). Thrombocytopenia (platelet count less than 100 × 103/mm3) occurred in 3 patients (37.5%). Platelet count was not decisive for calculating the antivenom dose.
One patient (12.5%) was classified as having Grade II envenomation, 6 (75%) as Grade III, and 1 (12.5%) as Grade IV.
Treatment and Outcomes
All patients were treated with Probiol polyvalent antivenom. The median number of vials received was 6.8 (3–14), and the median time between snakebite and antivenom administration was 6 (3–11) h. The mean length of hospital stay was 4.6 (SD: 1.7) days. Mean normalization time of the coagulation tests (fibrinogen, PT, and aPTT) was 23 (SD: 8.2) h after starting the antivenom treatment. Patients were observed for at least 72 h after this correction, which additionally allowed monitoring of the bite site. These patients came from distant locations with few means of transportation.
No complications such as wound infection, need for surgical procedures, or deaths related to envenomation were reported.
Table 1 summarizes the clinical and laboratory characteristics, as well as the grade of envenomation and antivenom doses administered to each patient.
Discussion
According to the World Health Organization, snakebites affect more than 5.4 million people annually, and snake envenomation occurs in 2.7 million cases. 13 The mortality rate ranges from 81,000 to 138,000 deaths annually, with approximately 400,000 people experiencing permanent disabilities such as amputations and long-term psychological impacts. 14 The general underreporting of snake envenomation is well known. 15 The specific occurrence of B. schlegelii envenomation is difficult to determine, particularly when members of the health care team do not know how to identify it. Envenomation by B. schlegelii is grouped into so-called “bothropic envenomation,” which includes bites from species of the Bothrops, Porthidium, and Bothrocophias genera, without being able to precisely determine the type of specific clinical involvement by species. 16 An approximation was made by our group by cataloging various clinical syndromes; however, the number of patients was too small to draw definitive conclusions. 7 Given the occurrence of 8 patients with relatively exotic envenomation, such as that caused by B. schlegelii, we decided to submit this report confirming a clinical presentation that included the development of local pain, edema, and hemorrhagic tendencies. Development of blister, myotoxicity, or dermonecrosis were not observed in envenomation by B. schlegelii, which were most common in B. asper or B. rhombeatus bites.
Based on proteomic studies of venom, as well as the assessment of in vivo effects in experimental animals, it has been found that B. schlegelii venom is composed of bradykinin-potentiating peptides, metalloproteinases, phospholipase A2, serine proteases, and L-amino oxidase,17,18 with significant variations in their concentrations, as has been observed in other species of this genus.19,20 A Kazal-type serine protease inhibitor-like protein has been directly implicated in causing the development of edema. 21 The presence of a myotoxin 22 and a neurotoxic component 23 have been reported.
Variations in snake venom composition are controlled by multiple factors, including genetic, geographic, seasonal, sexual, and dietary factors.
12
Studies carried out in experimental animals with venoms of B. schlegelii from Central America show a hemorrhagic effect,
8
and those carried out in Ecuador
24
were related to a procoagulant effect. A clinical difference in envenoming in humans has not been reported. Our patients came from the center and south of the Colombian Pacific zone, which is an intermediate site between those of these two reports. A study on experimental animals in the same area did not show a more significant hemorrhagic effect than those in Central America or Ecuador.
25
Investigations evaluating the coagulotoxic effects of B. schlegelii venom have revealed a pseudo-procoagulant effect, forming weak clots that break down rapidly, thus depleting fibrinogen levels and contributing to an anticoagulant state. Additionally, an anticoagulant effect can be achieved by inhibiting the activity of thrombin and factor Xa as well as by toxins that accelerate fibrinogen cleavage.
26
Limitations
The small number of cases in the present study did not allow definitive conclusions about clinical presentation of envenomation caused by B. schlegelii. The guidelines for antivenom use provide a general direction that can be modified in relation to characteristics of severity, age of the patient (pediatric or geriatric), or comorbidities, among others.
Conclusions
Most bite sites from B. schlegelii were on the upper limbs. All patients had local reactions, including edema, pain, and systemic effects including hypofibrinogenemia, but none had systemic bleeding. Every patient received antivenom and had favorable outcomes.
Footnotes
Author Contribution(s)
Study concept and design, data acquisition, data analysis, drafting, and critical revision of the manuscript (CC); data acquisition, data analysis, drafting, and critical revision of the manuscript (VE-M); data analysis, drafting, and critical revision of the manuscript (VP-U); data analysis, drafting, and critical revision of the manuscript (SC-V); data analysis, drafting, and critical revision of the manuscript (FC-H); all authors approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Data Accessibility Statement
Data cannot be shared for ethical/privacy reasons. The data underlying this article cannot be shared publicly for the privacy of individuals that participated in the study. The data will be shared on reasonable request to the corresponding author.
Institutional Review Board Statement
The study was conducted according to the guidelines of the Declaration of Helsinki. The photos used were authorized by the patients for publication after signing informed consent. The project was approved by the Fundación Valle del Lilís ethics committee with protocol number 599.
