Abstract
To provide guidance to medical providers, wilderness users, and travelers, the Wilderness Medical Society convened an expert panel to develop evidence-based guidelines for treating water in situations where the potability of available water is not assured, including wilderness and international travel, areas impacted by disaster, and other areas without adequate sanitation. The guidelines present the available methods for reducing or eliminating microbiological contamination of water for individuals, groups, or households; evaluation of their effectiveness; and practical considerations. The evidence base includes both laboratory and clinical publications. The panel graded the recommendations based on the quality of supporting evidence and the balance between benefits and risks/burdens according to the criteria published by the American College of Chest Physicians.
Keywords
Introduction
Safe and efficient treatment of drinking water is among the major public health advances of the last century. Without it, waterborne diseases can spread rapidly, resulting in large-scale disease and death.1,2 In high-income countries, the population is generally protected from waterborne disease by sophisticated water supply systems that disinfect water and provide continuous monitoring. In contrast, travelers to wilderness and recreational areas anywhere in the world and to low- and middle-income regions of some countries may be confronted with untreated or contaminated water that poses a risk of acquiring enteric disease. Wilderness visitors and international travelers have no reliable resources to evaluate local water system quality. Less information is available for remote surface water sources. Appearance, smell, and taste are not reliable indicators to estimate water safety from infectious organisms. In addition, disaster situations, such as earthquakes, hurricanes, and flooding events, may result in a breakdown of municipal water systems, exposing victims to nonpotable water. These situations necessitate knowledge of how to disinfect water at the point-of-use, prior to drinking.
Methods of water treatment that can be applied in the field include the use of heat, ultraviolet (UV) light, clarification, filtration, and chemical disinfection. The choices for the wilderness hiker or international traveler are increasing as new technology is applied to field applications. Different microorganisms have varying susceptibilities to these methods. The risk of waterborne illness depends on the number and type of organisms consumed, host factors, and the efficacy of the treatment system.
The Wilderness Medical Society Practice Guidelines for Water Treatment were first published in the Wilderness Environmental Medicine Journal in 2019. 3 The current guideline updates this earlier one with additional information and sources. Three evidence grades were changed due to additional evidence, further evidence evaluation by the authors, or modified scope of the recommendation.
Methods
Our panel of specialists in wilderness medicine, travel medicine, public health, and microbiology was chosen in 2017 based on interest and expertise in drinking water quality, as demonstrated by research and publications to develop the initial guidelines. The same panel authored this revision. We used relevant articles from the 2019 guideline and identified recent publications through PubMed and Google Scholar databases using the following keywords or phrases: water disinfection, water purification or water treatment, waterborne illness, water sanitation and health, plus specific terms in combination with those general search terms: filtration, halogen, pasteurization, ultraviolet, SODIS, nanoparticles, household, and disaster. This was supplemented by a search of references or topics from articles in the initial results. References were not limited by publication year because much of the key research on basic methods applicable to our interest was conducted decades earlier. Websites are cited when they provide unique data, reports, or agency recommendations. Some review articles are cited to provide sources of in-depth information and to augment reference selection.
The evidence base for water treatment has 2 substantial differences from other clinical guidelines. Most of the literature concerning effectiveness of specific treatment methods against various waterborne microorganisms is laboratory-based. Evidence of the benefits of water treatment is either population-based public health research of disease outbreaks or household trials of water treatment. Therefore, the evidence grade is a combination of laboratory, population, and household or community-level studies that can be extrapolated to wilderness settings and international travel. In addition, the information is divided between medical and engineering literature.
The panel used a consensus approach to develop research questions and recommendations. Evidence grades were assigned according to the methodology stipulated by the American College of Chest Physicians guideline methodology, which was updated in 2018 (see online Supplemental Table 1). 4 These recommendations are graded based on the totality of supporting evidence and balance between the benefits and risks or burdens for each modality. Water treatment techniques were primarily evaluated for removal of microbiological contaminants, not for the removal of chemicals or toxins. Laboratory research evaluating treatment impact on microorganisms was considered as high, moderate, or low quality, similar to clinical trials.
Risk and Etiology of Waterborne Infection
Even in high-income countries with low rates of diarrheal illness, regular waterborne disease outbreaks indicate that the microbiologic quality of the water, especially surface water, is not assured.5–9 The environment and activity upstream from the travelers’ surface water source define the risk. Upstream usage by humans, farm animals, or wildlife poses a major risk.10–14 Giardiasis is a zoonotic infection with numerous host species, including farm animals, deer, and other wild ungulates, beavers, and even household animals. 15
Because it is very difficult to exclude animal and human activity in the watershed, the US Centers for Disease Control and Prevention (CDC) recommends treating any surface water before ingestion as a precaution to protect health. 16
While substantial progress has been made in the past 20 y toward the goal of safe drinking water and sanitation worldwide, 25% of the world’s population lacks safely managed drinking water in their homes. A total of 1.7 billion people lack access to basic sanitation services, and nearly 500 million people still practice open defecation. 17 Studies in low- and middle-income regions around the world show high levels of microbes in the environment and water sources.18–22 The combined roles of safe water, adequate sanitation, and hygiene in reducing diarrhea and other diseases are well documented.23–25 In any area of the world, after natural disasters such as hurricanes, tsunamis, and major earthquakes, one of the most immediate public health problems is a lack of potable water.26,27
Infectious agents with the potential for waterborne transmission include bacteria, viruses, protozoa, and nonprotozoan parasites. Most enteric organisms, including Shigella spp, Salmonella enterica serotype Typhi, hepatitis A, and Cryptosporidium spp, can retain viability for long periods in water, including when frozen in water.28–30
The risk of waterborne illness depends on the number of organisms consumed, virulence of the organism, and defenses of the host.31,32 Microorganisms with a low infectious dose (eg, Giardia, Cryptosporidium, Shigella spp, hepatitis A, enterohemorrhagic Escherichia coli, and norovirus) may cause illness even from inadvertent drinking during water-based recreational activities. 33
Although the primary reason for treating and disinfecting drinking water is to destroy microorganisms from animal and human biologic wastes, water may also be contaminated with toxins and chemical pollutants from industrial sources or from the environment. Toxins can be generated by biological organisms. 34 Many cyanobacteria (also known as blue-green algae) may produce toxins such as microcystins under certain conditions that can cause a wide range of symptoms, including stomach pain, vomiting, diarrhea, headache, neurological symptoms such as confusion and seizures, liver damage, and cardiovascular collapse. 35
General Recommendations for Drinking Water Disinfection
Definitions
The term
Water Treatment Methods
Multiple techniques for improving the microbiologic quality of water are available to individuals and small groups while hiking or traveling. Bottled water may be a convenient and popular solution but has logistical challenges (eg, access, bulk, and weight) and creates ecological problems from plastic waste. Furthermore, in some low- and middle-income countries, the quality of packaged water (eg, sold in bottles or sachets) may not meet the standards of high-income countries and may contain pathogenic microbes.37,38
Clarification Techniques
Clarification refers to techniques generally used as a predisinfection step that reduce the cloudiness of water caused by natural organic and inorganic material—known as turbidity and measured in nephelometric turbidity units (NTU). Higher turbidity indicates more suspended solids in the water and creates a dirty appearance. An average person can begin to see turbidity levels starting at approximately 5 NTU, but water treatment standards generally require less than 1 NTU. Cloudy water can rapidly clog filters designed to remove microorganisms. Moreover, cloudy water requires increased levels of chemical treatment, and the combined effects of water contaminants plus chemical disinfectants result in an unpleasant taste. Clarification techniques can markedly improve the appearance and taste of water. They may reduce the number of microorganisms, but not enough to ensure potable water; however, clarifying the water facilitates disinfection by filtration or chemical treatment.
Adsorption
Granular activated carbon is widely used in water treatment. When activated, charcoal’s regular array of carbon bonds is disrupted, making it highly reactive for adsorbing dissolved chemicals.39,40 Granular activated carbon is the best means to remove toxic organic and inorganic chemicals from water (including disinfection byproducts) and to improve odor and taste.41,42 Thus, it is widely used in municipal disinfection plants, in household under-sink devices and water pitchers, and in portable water filters. Granular activated carbon does not kill microorganisms and is not designed for microbial removal.41–43
In field water treatment, granular activated carbon is often used after chemical disinfection to make water safer and more palatable by removing the taste of chemical disinfectants, disinfection byproducts, and pesticides as well as many other organic chemicals and some heavy metals. Activated charcoal will remove most but not all microcystin toxins, so it may decrease the risk or severity of illness but cannot eliminate risk.44,45
Sedimentation
Sedimentation is the separation of suspended particles, such as sand and silt, that are large enough to settle rapidly by gravity. Most microorganisms, especially protozoan cysts, also settle eventually, but this takes much longer. 46 Simply allowing the water to sit undisturbed for about 1 h or until sediment has formed on the bottom of the container—then decanting or filtering the clear water from the top through a coffee filter or finely woven cloth will remove many larger particles from the water. Additional treatment is needed to obtain potable water.
Coagulation–Flocculation
Coagulation–flocculation (C-F) is a technique in use since 2000
The amount of alum added in the field—approximately 1 large pinch (1 mL or 1/8 tsp) in 4 L (approximately 1 gal) of water—need not be precise. The C-F agent is stirred or shaken briskly for 1 min to mix and then agitated gently and frequently for at least 5 min to assist flocculation. If the water is still cloudy, more C-F agent may be added, followed by repeat mixing. After at least 30 min for settling, water is poured through a fine-woven cloth or paper filter. Although most microorganisms are removed with the floc, a final process of microbiologic filtration or chemical disinfection (below) should be completed to ensure disinfection. Several products combine C-F with halogen disinfection, which provide a single-step dual process for low quality water that achieves better water treatment than either alone.49,53–58
Improvisational techniques for clarification
Many traditional plants are used by native peoples as a coagulant, as well as inorganic compounds, including lime (calcium oxide) or potash (from wood ash).59,60 In an emergency, bleaching powder, baking powder, or even the fine white ash from a campfire can be used. 61
Adsorbents such as charcoal and clay and other types of organic matter have been used for water treatment for millennia. 43 These substances are often used as filter media but can also act as coagulants. 23 Clays can decrease turbidity and microbes in water by approximately 90 to 95%, 62 but adsorption is not the main action of ceramic or clay filters.
Disinfection and Filtration Methods
Heat
Heat is the oldest and most reliable means of water disinfection. Heat inactivation of microorganisms is a function of time and temperature (exponential function of first-order kinetics). Thus, the thermal death point is reached in a shorter time at higher temperatures, while lower temperatures are effective if applied for a longer time. Pasteurization uses this principle to kill food pathogens and spoiling organisms at temperatures well below boiling, generally between 60°C (140°F) and 70°C (158°F) within 30 min. Flash pasteurization occurs at 70 to 72°C (158–162°F) within 30 s.63,64
All common enteric pathogens are readily inactivated by heat at pasteurization temperatures, although microorganisms vary in heat sensitivity, with protozoan cysts being the most sensitive to heat, bacteria intermediate, and viruses less sensitive (Table 1).65,66,67–77 Only bacterial spores are more resistant, but they are not generally enteric pathogens. 78 After boiling, water may become contaminated again from storage containers or handling.79,80
Heat inactivation of microorganisms.

aTested in food. Expect lower temperatures and shorter times in water for inactivation.
As enteric pathogens are killed within seconds by boiling water and rapidly at temperatures >60°C (140°F), previous advice, now obsolete, was to boil water for 10 min to ensure potable water (Table 1). The time required to heat water from 55°C (131°F) to a boil works toward disinfection; therefore, any water brought to a rapid boil should be adequately disinfected. 63 Boiling for 1 min is recommended by the CDC to account for user variability in identifying boiling points and to add a margin of safety. The boiling point decreases with increasing altitude, but this is not significant compared with the time required for thermal inactivation at these temperatures (Table 2).
Boiling temperatures at various altitudes.

Improvisational techniques
In wilderness or travel environments, the main limitation for using heat is availability of fuel. Although attaining boiling temperature is not necessary to kill microorganisms, boiling is the only easily recognizable endpoint without using a thermometer. If fuel is scarce, heat water until first sign of simmering (small bubbles rising from the bottom), reduce or remove heat, and leave container covered for 30 min. As a rule of thumb, water too hot to touch falls within the pasteurization range, but tolerance to touch is too variable to be reliable. 81
If no reliable method of water treatment is available, tap water that has been kept hot in a tank for at least 30 min and is too hot to keep a finger immersed for 5 s (estimated 55–65°C; 131–149°F) has been suggested as a means of obtaining potable water for short periods of need based on heat disinfection and microbiological testing.82,83 In the long-term, drinking from water heaters is not advised based on relatively higher levels of metals (eg, lead) and chemical contaminants that can dissolve into water at elevated temperatures. Moreover, this improvisational measure is less useful for hotels that use on-demand water heaters without a hot water tank.
Travelers with access to electricity can boil water with either a small electric heating coil or a lightweight electric beverage warmer brought from home. In austere and desperate situations with hot, sunny climate, pasteurization temperature can be achieved with a solar oven or simple reflectors (see UV–solar UV disinfection [SODIS] below).84–86
UV Light
Ultraviolet radiation (UVR) and UV light disinfection systems are widely used to disinfect drinking water at the community and household levels. At sufficient doses, all waterborne enteric pathogens are inactivated by UVR. 87 Ultraviolet C light in the range of 200 to 280 nm is the most effective. The germicidal effect of UV light is the result of action on the nucleic acids of microorganisms and depends on light intensity and exposure time. Bacteria and protozoan parasites generally require lower doses than do enteric viruses and bacterial spores. However, all viruses, including hepatitis A and norovirus, are susceptible and follow similar kinetics, with relatively minor differences. The vegetative cells of bacteria are significantly more susceptible to UVR than are bacterial spores or viruses. Giardia and Cryptosporidium are susceptible to practical doses of UVR and may be more sensitive because of their relatively large size.88–90 The UV waves must strike the organism, so the water must be free of particles that could act as a shield (ie, not cloudy). 91 The UV waves do not alter the water, but they also do not provide any residual disinfecting power. 92 Both large high-volume units and portable, lightweight battery-operated units for disinfection of small quantities of water are available. The cost of UV devices limits their use in low-income households and communities, but UV-LEDs are effective and show promise when the cost decreases.93,94
Improvisational technique: Solar Disinfection (SODIS) and solar pasteurization (SOPAR)
UV irradiation by sunlight can substantially improve the microbiologic quality of water and reduce diarrheal illness and is widely used in low-income countries and austere settings.60,85,95–103 Similar to powered UV lamps, solar UVR can effectively inactivate bacteria, viruses, and protozoan cysts. The optimal procedure for the SODIS technique is to use transparent bottles (eg, clear plastic beverage bottles), preferably lying on a dark or reflective surface to increase heat and UV, exposed to sunlight for a minimum of 4 to 6 h with intermittent agitation or 2 d under cloudy conditions.102,104 Solar UV disinfection has been studied extensively in mid latitudes, but efficacy has also been demonstrated in cool climates and cold water. 105
Ultraviolet and thermal inactivation are strongly synergistic for the solar disinfection of drinking water, even if pasteurization temperatures are not reached.85,106–109 However, in warm-hot sunny climates, pasteurization temperatures of 65
Filtration
Filters are appealing to outdoor users as well as households without a reliable source of clean water because of their simplicity and suitability for commercial production.112–114 Portable water treatment products are among the most frequently purchased equipment for camping after backpacks and tents. Filtration is a standard step in municipal water treatment and widely used in the food and beverage industry, as well as many other industrial processes. Filtration can be highly effective for removing disease-causing microbes and other particulate contaminants, but unless a disinfectant chemical is incorporated into the filter media, it does not kill or inactivate microbes. Throughout the world’s history, many different types of media, from sand to vegetable products to fabric, have been used for water filtration. Filters have the advantages of being simple and requiring no wait time after passing through the filter. They do not add any unpleasant taste and may improve the taste and appearance of water. All filters eventually clog from suspended particulate matter that is present even in clear streams, requiring cleaning or replacement of the filter. As a filter clogs, it requires increasing pressure to drive the water through, which can force microorganisms through the filter or damage the filter. A crack or eroded channel in a filter will allow passage of unfiltered water. Bacteria can grow on filter media and potentially result in some bacteria in filtered water, but pathogenic bacteria have not been demonstrated. 115 Silver is often incorporated into the filter media to prevent this growth, but it is not totally effective. (See additional information in the section on nanomaterials.)
The primary determinant of a microorganism’s susceptibility to filtration is its size (Table 3 and Figure 1).
Portable filters for water treatment can be divided into microfilters with pore sizes down to 0.1 µm, ultrafilters with pore sizes of 0.002 to 0.01 µm, nanofilters with pore sizes as small as 0.001 µm or less, and reverse osmosis filters with pore sizes of 0.0001 µm or less. 87 The smaller the pore size, the more pressure or greater filter surface required. Waterborne pathogens often adhere to larger particles or clump together, making them easier to remove by physical processes. Therefore, observed reductions are often greater than expected based on their individual sizes.
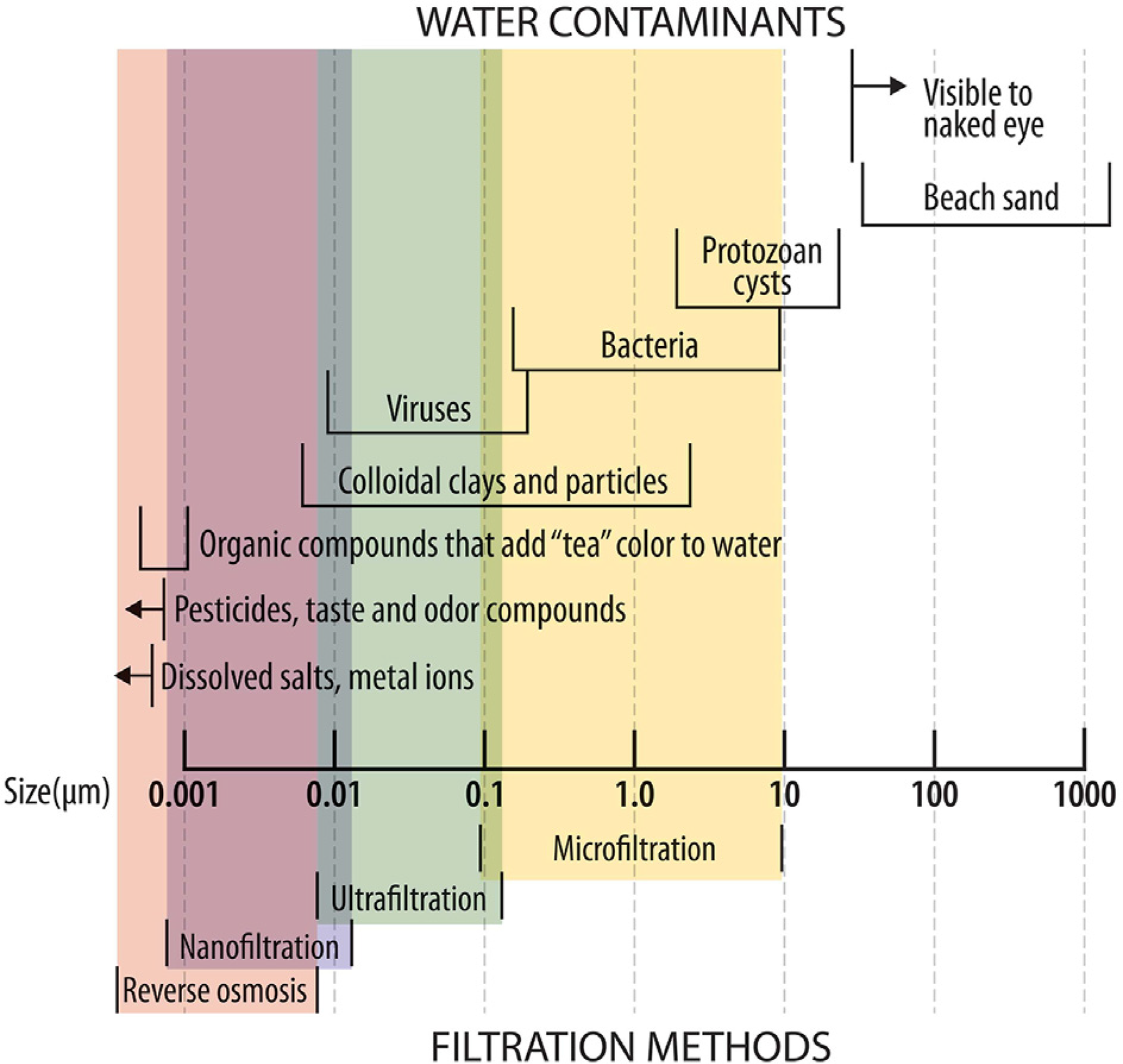
Levels of filtration and susceptibility of common microbial pathogens and other contaminants. Reproduced with permission from the Centers for Disease Control and Prevention. CDC Yellow Book 2024: Health Information for International Travel. New York: Oxford University Press, 2023. Adapted from Backer H. Field Water Disinfection. In: Auerbach PS, ed. Wilderness Medicine, 7th edition. Elsevier; 2017.
Microorganism susceptibility to filtration.

aMicrofilters (most filters with pore size of 0.1–0.2 µm) can filter bacteria and protozoan cysts but are not effective for virus removal unless designed to rely on electrostatic trapping of viruses. Hollow-fiber filters with 0.02 µm pores and reverse osmosis filters are capable of filtering viruses.
Many portable filters are microfilters that can readily remove protozoan cysts and bacteria, but may not remove all viruses, which are much smaller than the pore size of most field filters.116–118 However, viruses often clump together or are attached to other larger particles or organisms, resulting in an aggregate large enough to be trapped by the filter; in addition, electrochemical attraction may cause viruses to adhere to the filter surface.23,62,119 Through these mechanisms, mechanical filters using ceramic elements with a pore size of 0.2 µm can reduce viral loads by 2 to 3 logs (99–99.9%) but are not adequate for complete removal of viruses. 118 Ultrafiltration or nanofiltration is required for complete microbial removal, including viruses; they can also remove colloids, dissolved solids, and some large toxins.120,121 Nanofilters can also remove microcystin toxin. 44
Hollow-fiber technology has been adapted for field use using bundles of tube fibers whose pore size can be engineered to achieve ultrafiltration with viral removal. 122 The large surface area allows these hollow-fiber filters to have relatively high flow rates at low pressure. Hollow-fiber filters that are designed to use gravity, a squeeze bag, or a hand pump are widely available to treat water for individuals or small groups. Compact and lightweight designs have made them popular with long-distance hikers.
Some filters on the market combine filter material with other substances such as iodine or silver to add disinfection to the process. Iodine molecules can be bound in a resin engineered into field products, but the effectiveness of the resin is highly dependent on the product design and function.123–126 Most companies have abandoned iodine resin–containing portable hand-pump filters, water bottles, or drink-through straws due to reports of excess iodine or viral breakthrough in the effluent; however, iodine resins can be highly effective in carefully engineered systems, such as those developed for National Aeronautics and Space Administration space shuttles and the International Space Station. 127
Several factors influence the decision of which filter to use: 1) flow volume sufficient for the number of persons relying on the filter; 2) whether the filter functional claims match the microbiologic demands that will be put on the filter; 3) the preferred means of operation (eg, hand pump or gravity); and 4) cost.
Improvisational filtration techniques
Simple, available products, such as rice hull ash filters, crushed charcoal, sponges, and various fabrics and paper, have been used in low- and middle-income countries and in emergency situations for filtration. Typically, bacteria and viruses can be reduced by as much as 50 to 85% and larger parasites can be reduced by 99%, depending on the media. The effectiveness for decreasing turbidity may be used as an indicator that a filter material will reduce microbiologic contamination.51,128,129
Ceramic filters are a common component in portable water pump filters but are also a cost-effective means of household disinfection in low- and middle-income countries. Ceramic clay is widely available and very inexpensive to locally manufacture in the shape of a sink or flowerpot that is set into a larger container to collect the filtered water.130–132 Extensive epidemiological research has demonstrated efficacy and effectiveness of ceramic filters when properly made and maintained.125,130–134
Biosand filters employ a technology that has been used over centuries and is still used widely in municipal plants and at the household and community level.135–138 Sand filters can be highly effective at removing turbidity (in one study, from 6.2 NTU to 0.9 NTU) and improving microbiologic quality (99% efficacy), depending on their design and operation.135–139 Sand filters are constructed by forming layers of aggregate increasing in size from the top to the bottom. An emergency sand filter can be made in a 20 L (5.3 gal) bucket, composed of a 10 cm (3.9 in) layer of gravel beneath a 23 cm (9.1 in) layer of sand; a layer of cotton cloth, sandwiched between 2 layers of wire mesh, separates the sand and gravel layers. 51 A sand filter also can be improvised with stacked buckets of successive filter layers with holes in the bottom to allow water passage. Many websites provide design and assembly instructions, but there are no data for comparative function. 140
Chemical Disinfection: Halogens (Iodine and Chlorine)
Worldwide, disinfection with chemicals, chiefly halogens, is the most widely used method for improving and maintaining the microbiologic quality of drinking water.87,141 The germicidal activity of chlorine and other halogens is well-established and results from oxidation of essential cellular structures and enzymes. Disinfection effectiveness is determined by characteristics of the microorganism, the disinfectant, contact time, and environmental factors. Given adequate concentrations and contact times, both iodine and chlorine are effective disinfectants with comparable biocidal activity under most conditions (Table 4). Both are widely available in multiple formulations.
Disinfection data for chlorine and iodine to achieve 99.9% kill or inactivation a of select microorganisms.
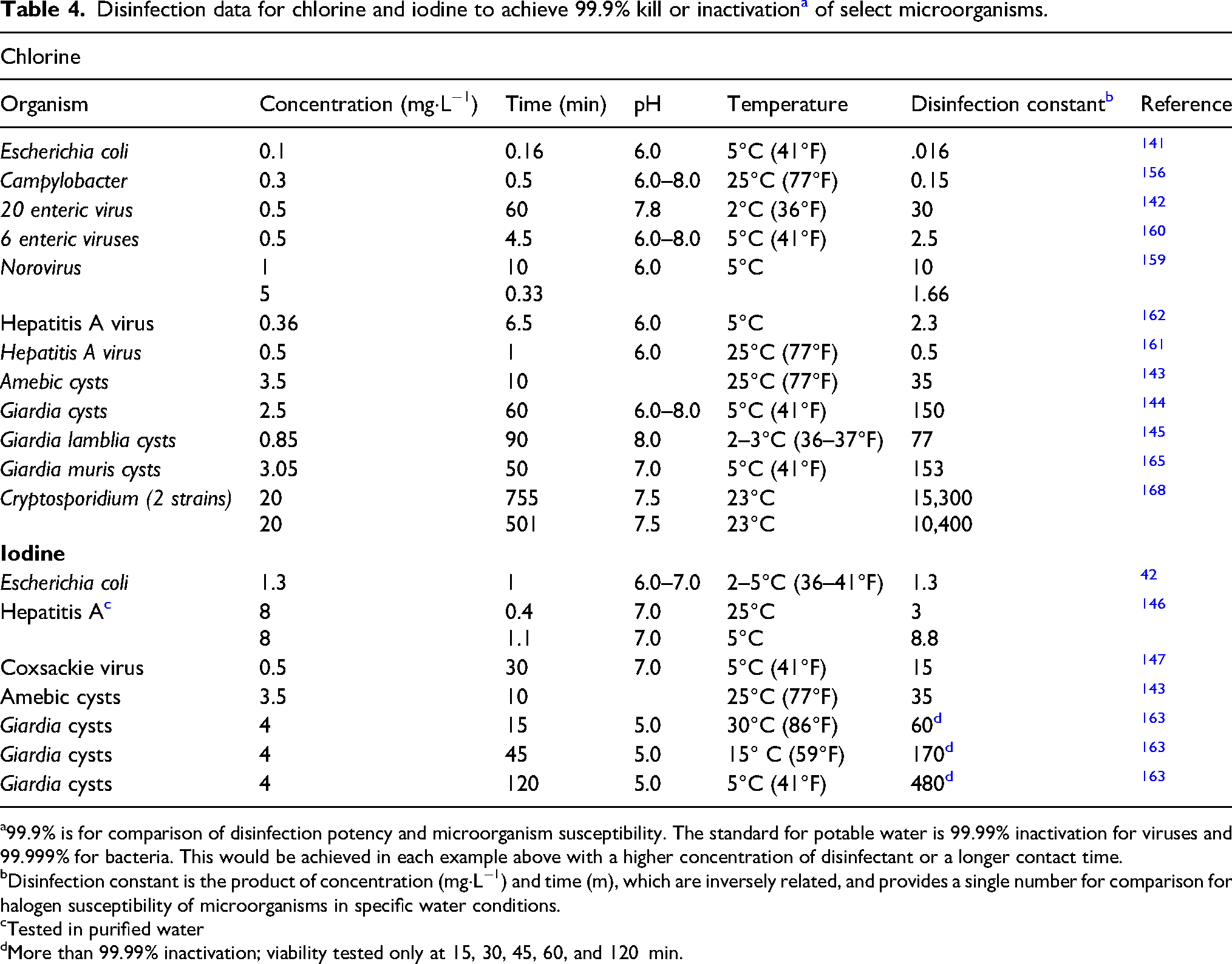
a99.9% is for comparison of disinfection potency and microorganism susceptibility. The standard for potable water is 99.99% inactivation for viruses and 99.999% for bacteria. This would be achieved in each example above with a higher concentration of disinfectant or a longer contact time.
bDisinfection constant is the product of concentration (mg·L−1) and time (m), which are inversely related, and provides a single number for comparison for halogen susceptibility of microorganisms in specific water conditions.
cTested in purified water
dMore than 99.99% inactivation; viability tested only at 15, 30, 45, 60, and 120 min.
One advantage of chemical water disinfection products is flexible dosing that allows use by individual travelers, small or large groups, or communities.61,128 Unlike heat, filtration, and UV, halogens provide a disinfectant residual in water to protect against recontamination during storage.
Chlorine is still advocated by the World Health Organization (WHO) and the CDC as a mainstay of large-scale community, individual household, and emergency use.148,149 Hypochlorite, the major chlorine disinfectant, is currently the preferred means of municipal water disinfection. 141 Calcium hypochlorite and sodium hypochlorite (NaOCl) readily dissociate in water to form hypochlorous acid, the active disinfectant. There are extensive data on effectiveness of chlorine-based disinfectant products in remote settings.80,87,148,150–154
Iodine is also effective in low concentrations for killing bacteria, viruses, and some protozoan cysts, and in higher concentration against fungi and even bacterial spores; however, it is a poor algaecide.124,155 Elemental iodine and hypoiodous acid are the major germicides in an aqueous solution. Of the halogens, iodine reacts least readily with organic compounds and is less affected by pH, indicating that low iodine residuals should be more stable and persistent than corresponding concentrations of chlorine. Despite these advantages, because of its physiological activity, WHO has not established a formal guideline value and recommends iodine only for short-term emergency use. 124 The European Union banned the sale of iodine-based products for water disinfection in 2009.
Halogen efficacy
Vegetative bacteria (non–spore forming) are very sensitive to halogens (Table 4).141,156–158 Viruses, including hepatitis A, have intermediate sensitivity, requiring higher concentrations or longer contact times.125,159–162 SARS-CoV-2, the virus that causes COVID-19, can be found in water; however, it is sensitive to chlorine inactivation, and there is no evidence that COVID-19 can be transmitted through water. Protozoan cysts are more resistant than enteric bacteria and enteric viruses but some cysts (eg, Giardia) can be inactivated by field doses of halogens.163–166 Cryptosporidium oocysts, however, are much more resistant to halogens so inactivation is not practical with common doses of iodine and chlorine used in field water disinfection.167,168 Certain parasitic eggs, such as those of Ascaris, are also resistant, but these are not commonly spread by water. (All of these resistant cysts and eggs are susceptible to heat or filtration.) Bacterial spores, such as Bacillus anthracis, are relatively resistant to halogens, but not much more resistant than are Giardia cysts; furthermore, they do not normally cause waterborne enteric disease.
Halogen disinfection variables in field use
Understanding factors that influence the disinfection reaction allows flexibility with greater reassurance of effectiveness (Table 5). The primary factors of the first-order chemical disinfection reaction are concentration and contact time. 141 Lower concentrations can be used with longer contact times. In field disinfection, this can be used to minimize halogen dose and improve taste or, conversely, to minimize the required contact time with higher doses of halogen.
Factors affecting water disinfection with halogens.
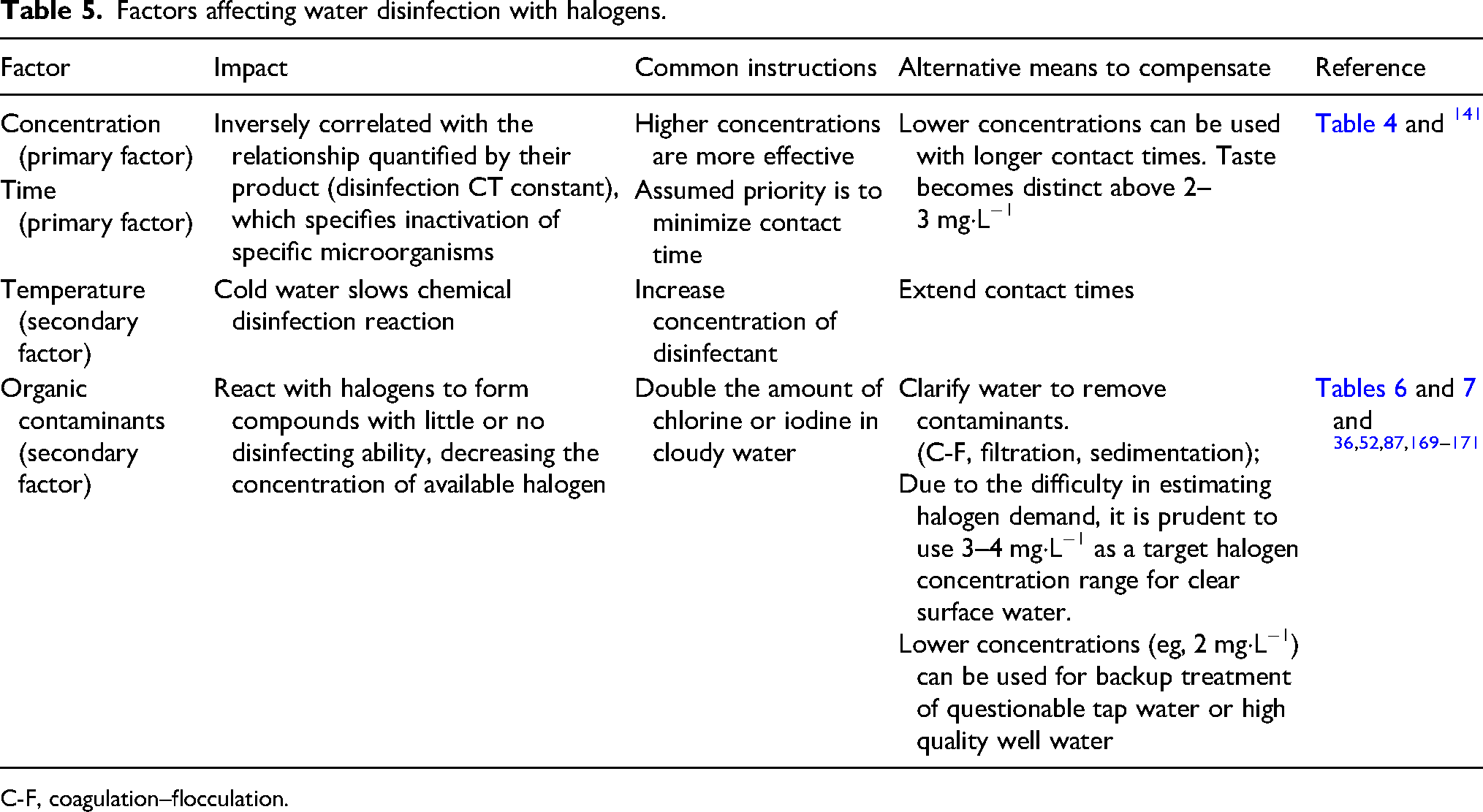
C-F, coagulation–flocculation.
Multiple products combine chlorine with a C-F powder to both clarify and disinfect in 1 step, resulting in decreased turbidity of water, high level of disinfection for bacteria and viruses, increased removal of protozoan cysts over chlorine alone, and significant removal of other water contaminants, such as arsenic.54,55,57,172
Halogen toxicity
Chlorine has no known toxicity at the concentrations used for water disinfection. Reactions of chlorine with certain organic contaminants yield chlorinated hydrocarbons, chloroform, and other trihalomethanes, which are considered to have carcinogenic potential in animal models. Nevertheless, the risk of severe illness or even death from infectious diseases if disinfection is not used is far greater than any risk from byproducts of chlorine disinfection. 173
Iodine has not gained general acceptance for long-term use in large populations because of concern for its physiologic activity. 124 There are some older data that iodination of water with a low residual concentration ≤1 to 2 mg·L−1 appear safe, even for long periods of time, in people with normal thyroid function.174,175 However, no major agencies recommend prolonged use. The WHO did not set a guideline value for iodine in drinking water because of a paucity of data, and because it is not recommended for long-term disinfection. If the typical wilderness/international traveler disinfected 3 L of water each day using 2 to 4 mg·L−1 of iodine, the ingested amount of iodine per day would be 6 to 12 mg, well above US Institute of Medicine recommended dietary allowance levels. Levels produced by the recommended doses of iodine tablets are even higher and would result in ingestion of 16 to 32 mg/d. Therefore, the use of iodine for water disinfection should be limited to short periods (months). Anyone planning to use iodine for prolonged periods should have their thyroid examined and thyroid function tests done to assure that they are initially euthyroid. Certain groups should not use iodine for water treatment: pregnant women (because of concerns of neonatal goiter); those with known hypersensitivity to iodine; persons with a history of thyroid disease, even if controlled on medication; persons with a strong family history of thyroid disease (thyroiditis); and persons from countries with chronic iodine deficiency. 176
Improving halogen taste
Objectionable taste and smell limit the acceptance of halogens, with taste becoming distinctive above 3 mg·L−1. 177 Taste preference between chlorine and iodine is individual and can be improved by several means. One method is to use the minimum necessary dose with a longer contact time (30–60 m) (Table 6). The CDC and Pan American Health Organization developed the Safe Water System for household disinfection, which provides a low dosage of 1.875 or 3.75 mg·L−1 of NaOCl with a contact time of 30 min, sufficient to inactivate most bacteria, viruses, and some protozoa that cause waterborne diseases.148,154
Recommendations for contact time using halogen disinfection in the field.

Concentration and contact time are based on the most resistant target organism, which is the Giardia cyst. These are well beyond the time needed to kill bacteria and viruses. These contact times have been extended from the usual recommendations in cold water to account for the extended inactivation time required in very cold water and for the uncertainty of residual concentration.
Another method is to remove the taste after the disinfection time prior to drinking through chemical reduction of chlorine to chloride or iodine to iodide, which have no color or taste and no disinfectant activity. The best and most readily available agent is ascorbic acid (vitamin C), available in crystalline or powder form. A small pinch in a liter, mixed after the required contact time, will usually suffice. Ascorbic acid is a common ingredient of flavored drink mixes, accounting for their effectiveness in removing the taste of halogens. Granular activated carbon (see above) adsorbs organic and inorganic chemicals, including iodine and chlorine byproducts, thereby improving odor and taste—the reason for its common inclusion in field and household filters. 141 Techniques that reduce residual disinfectant levels should only be used at the time of consumption, not if water will be stored.
Sources of Halogen Disinfectants
There is no comparable substitute for proven chemical disinfectants, but there are many common substances that contain halogens (Table 7). Unscented household bleach containing NaOCl is available in most parts of the world. Products that contain calcium hypochlorite provide much higher concentrations (70%) than household bleach. Iodine is also available in liquid, tablets, or resins. A common household source is tincture of iodine or similar topical disinfectant with an iodine concentration of 2 to 8%. These products also contain iodide, which has no disinfecting power, but does contribute to iodine toxicity. Colorless iodine solution contains only iodide, so it should not be used for water disinfection. Povidone–iodine, a topical disinfectant commonly used in medical settings, contains active iodine bound to a neutral polymer of high molecular weight that gives the iodine greater solubility and stability. In dilute aqueous solution, povidone–iodine provides a sustained-release reservoir, releasing free iodine in a concentration of 2 to 10 mg·L−1 that increases with dilution in a bell-curve manner.178,179
Halogen disinfection products and recommended doses for point-of-use disinfection.
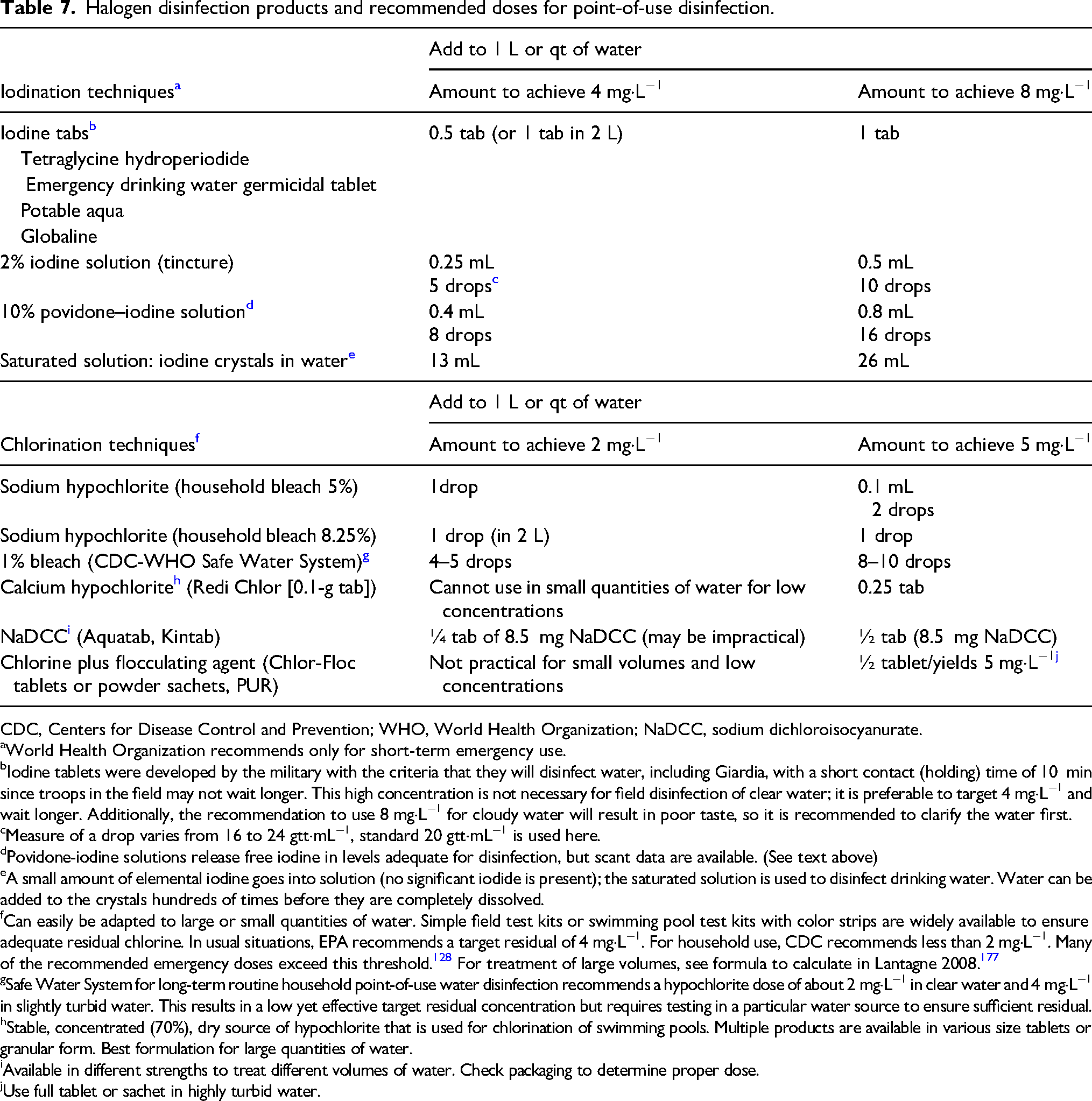
CDC, Centers for Disease Control and Prevention; WHO, World Health Organization; NaDCC, sodium dichloroisocyanurate.
aWorld Health Organization recommends only for short-term emergency use.
bIodine tablets were developed by the military with the criteria that they will disinfect water, including Giardia, with a short contact (holding) time of 10 min since troops in the field may not wait longer. This high concentration is not necessary for field disinfection of clear water; it is preferable to target 4 mg·L−1 and wait longer. Additionally, the recommendation to use 8 mg·L−1 for cloudy water will result in poor taste, so it is recommended to clarify the water first.
cMeasure of a drop varies from 16 to 24 gtt·mL−1, standard 20 gtt·mL−1 is used here.
dPovidone-iodine solutions release free iodine in levels adequate for disinfection, but scant data are available. (See text above)
eA small amount of elemental iodine goes into solution (no significant iodide is present); the saturated solution is used to disinfect drinking water. Water can be added to the crystals hundreds of times before they are completely dissolved.
fCan easily be adapted to large or small quantities of water. Simple field test kits or swimming pool test kits with color strips are widely available to ensure adequate residual chlorine. In usual situations, EPA recommends a target residual of 4 mg·L−1. For household use, CDC recommends less than 2 mg·L−1. Many of the recommended emergency doses exceed this threshold. 128 For treatment of large volumes, see formula to calculate in Lantagne 2008. 177
gSafe Water System for long-term routine household point-of-use water disinfection recommends a hypochlorite dose of about 2 mg·L−1 in clear water and 4 mg·L−1 in slightly turbid water. This results in a low yet effective target residual concentration but requires testing in a particular water source to ensure sufficient residual.
hStable, concentrated (70%), dry source of hypochlorite that is used for chlorination of swimming pools. Multiple products are available in various size tablets or granular form. Best formulation for large quantities of water.
iAvailable in different strengths to treat different volumes of water. Check packaging to determine proper dose.
jUse full tablet or sachet in highly turbid water.
Mixed Species Disinfectant (Electrolysis)
Passing a current through a simple brine salt solution generates free available chlorine as well as other “mixed species” disinfectants, giving the resulting solution greater disinfectant ability than a simple solution of NaOCl.150,151 The process has been engineered for use on both large and small scales. 180
Miscellaneous Disinfectants
Chlorine dioxide and ozone
Both chlorine dioxide (ClO2) and ozone (O3) are potent biocides due to their strong oxidizing potential that have been used for many years to disinfect municipal water and numerous other industrial applications. Until recently, their benefits have been limited to large-scale applications because standard formulations must be made on-site and are associated with a risk for producing volatile or toxic gas. Newer methods may enable cost-effective and portable generation for use in an array of small-scale applications.
Chlorine dioxide has no taste or odor in water at concentrations used for treating drinking water. It is capable of inactivating most waterborne pathogens, including Cryptosporidium parvum oocysts.181,182 It is at least as effective a bactericide as chlorine and far superior for virus and parasite inactivation. There are several commercial point-of-use applications using ClO2 in liquid or tablet form, but relatively few data are available on testing of these products. 183 Chlorine dioxide–production tablets contain 6.4% sodium chlorite as the active ingredient. After a tablet is added to water, a series of complex chemical reactions occur, generating ClO2. Some of the intermediary chemical compounds may also have antimicrobial activity. A major disadvantage for field use of tablets is the long reaction or contact time required, upwards of 2 to 4 h needed to achieve dependable disinfection. Chlorine dioxide does not produce a lasting residual and water undergoing ClO2 disinfection must be protected from sunlight.
Ozone is an unstable form of pure oxygen that is colorless and tasteless in water. It is one of the strongest oxidants and disinfectants used in treating water and wastewater, rapidly killing organisms by oxidizing organic material in the membranes of bacteria, viruses, and parasites.125,141,181,183–186 It also oxidizes metals, facilitating their removal and improving taste, smell, and color. 141 Generally, O3 is generated from air using electrical current or UV light. Small portable products using a low battery current to generate O3 from oxygen in water are now available; however, data on their effectiveness are limited. Ozone breaks down rapidly, so adequate concentration and contact time must be assured.
Silver
Silver ion has bactericidal effects in low doses and some attractive features, including absence of color, taste, and odor. Limited data for disinfection of viruses and protozoan cysts indicate incomplete effect, even at high doses. Moreover, the concentrations are strongly affected by adsorption onto the surface of any container. Silver is physiologically active but not likely to cause a problem in concentrations found in drinking water. Long-term effects are discoloration of the skin (argyria), considered cosmetic and not toxic. The WHO does not recommend silver for primary water disinfection and the US Environmental Protection Agency has not approved it for this use in the United States, but silver is approved as a water preservative to prevent bacterial growth in previously treated and stored water.187,188 In Europe, silver tablets are sold for field water disinfection. One product combines silver with hypochlorite for both effective disinfection and preservation. There is some promise in steady release products and incorporation into nanoparticles, which seem to be more effective than silver ions.141,189,190 Silver is commonly incorporated into filter material to aid in disinfection and to prevent bacterial growth on filter media; however, these effects are limited. 191
Hydrogen Peroxide
Hydrogen peroxide is a strong oxidizing agent that is widely used as a preservative in food, as a sterilant for medical and food equipment, and many other applications. Although hydrogen peroxide can sterilize water, it is not widely used as a field water disinfectant, perhaps because high concentrations that are known to be effective are very caustic, and there is a lack of data for protozoal cysts and quantitative data for dilute solutions. It can be used to remove the taste of hypochlorite and in combination with other processes.192,193
Citrus and Potassium Permanganate
Both citrus juice and potassium permanganate have some demonstrated antibacterial effects in an aqueous solution. 194 However, data are limited and not available for effect on cysts. In municipal water disinfection, potassium permanganate is used primarily for reducing contaminants to improve taste and odor. 188 Either substance could be used in an emergency to reduce bacterial and viral contamination or as an adjunct in combination with another technique but cannot be recommended as a primary means of water disinfection. Citrus can also enhance SODIS. 60
Nanoparticles: Solar Photocatalytic Disinfection
Nanomaterials have structures measuring 100 nm or less that may be in the form of particles, tubes, rods, or fibers and comprised of organic, inorganic, carbon, or composite materials. 195 Several nanomaterials have been shown to have strong antimicrobial properties and are being evaluated for use in water disinfection and purification.190,196–198 Inorganic-based nanomaterials, including different metal and metal oxides, such as titanium, zinc, iron, and silver, are of particular interest for water disinfection applications because they can be activated by UV to produce potent oxidizers that are excellent disinfectants for microorganisms. In addition, they can break down complex organic contaminants and even most heavy metals into nontoxic forms. Titanium dioxide (TiO2) is the most effective photocatalytic substance identified to date. Recent work demonstrated inactivation of Cryptosporidium by TiO2.197,199 These methods are widely used in industry, but few products have incorporated the technology into individual or small group point-of-use products.200,201 Commercial point-of-use products have incorporated silver or TiO2 into filter media or membranes. 190
Preferred Technique
The optimal water treatment technique for an individual or group will depend on the number of persons to be served, space and weight accommodations, quality of source water, personal taste preferences, duration of use, and resources (eg, fuel availability) or devices available. Because halogens are not effective for killing Cryptosporidium at drinking water concentrations and common microfilters are not reliable for virus removal, optimal protection for all situations may require a 2-step process of filtration or C-F followed by chemical disinfection. Heat (boiling) is effective as a one-step process but will not improve the esthetics of the water or provide protective residual disinfection during storage. UVR is an effective 1-step process for clear water. Table 8 summarizes effects of major water disinfection methods on categories of microorganisms. Several authors have reviewed efficacy data for point-of-use methods for household disinfection in low- and middle-income countries24,25,56,113,202 (Table 9).203,204 In practice, there is a difference between laboratory and field application of any water treatment method due to differences in water quality and use of the product or method.
Summary of field water disinfection techniques.
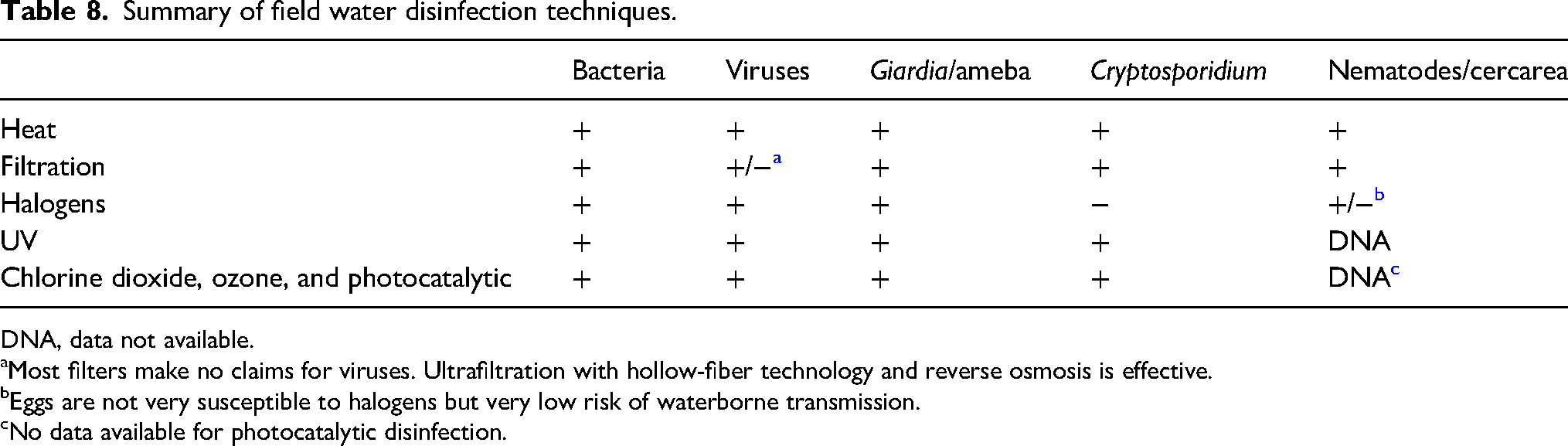
DNA, data not available.
aMost filters make no claims for viruses. Ultrafiltration with hollow-fiber technology and reverse osmosis is effective.
bEggs are not very susceptible to halogens but very low risk of waterborne transmission.
cNo data available for photocatalytic disinfection.
Efficacy and effectiveness of point-of-use technologies for low-income world households.
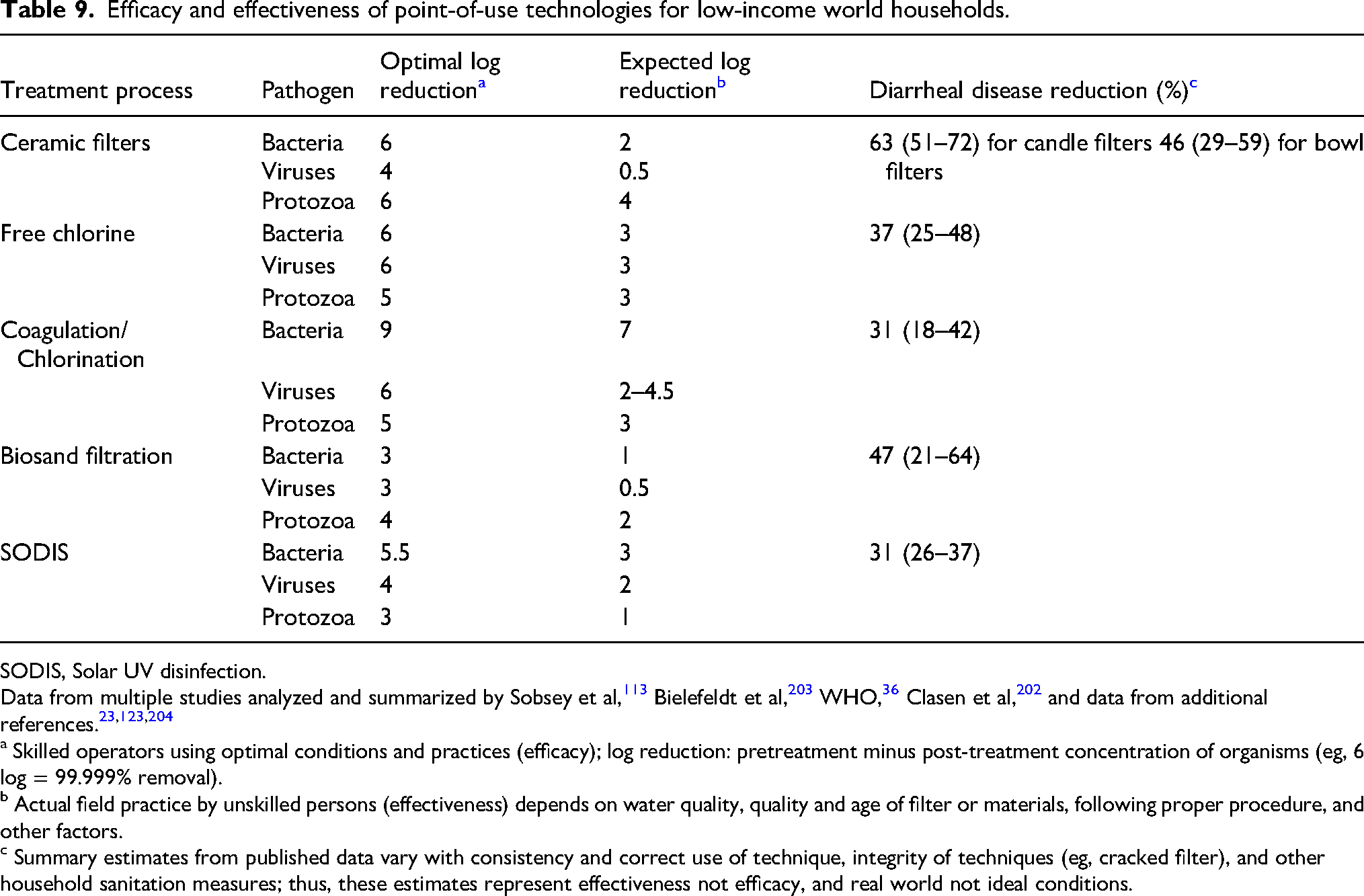
SODIS, Solar UV disinfection.
Data from multiple studies analyzed and summarized by Sobsey et al, 113 Bielefeldt et al, 203 WHO, 36 Clasen et al, 202 and data from additional references.23,123,204
a Skilled operators using optimal conditions and practices (efficacy); log reduction: pretreatment minus post-treatment concentration of organisms (eg, 6 log = 99.999% removal).
b Actual field practice by unskilled persons (effectiveness) depends on water quality, quality and age of filter or materials, following proper procedure, and other factors.
c Summary estimates from published data vary with consistency and correct use of technique, integrity of techniques (eg, cracked filter), and other household sanitation measures; thus, these estimates represent effectiveness not efficacy, and real world not ideal conditions.
In disaster situations, such as floods, hurricanes, and earthquakes, sanitation and water treatment facilities are frequently damaged or inundated, so household or point-of-use water disinfection is advised, as in regions where there is no sanitation or improved water sources. Surface water quality is likely to be poor with turbidity, bacteriological, and chemical contamination. Optimally, cloudy water should first be clarified, followed by application of heat, filtration, or chemical disinfection. Granular activated carbon can remove many chemical contaminants. Chlorination may be the simplest method and sufficient alone for disinfecting contaminated water for most classes of microorganisms (Table 8).26,27,128,177 Other point-of-use methods described above can also improve water quality after a disaster. In sunny climates, if time allows, SODIS requires no special resources other than the container.
On long-distance ocean-going boats where water must be desalinated as well as disinfected during the voyage, only reverse osmosis membrane filters are adequate. Water storage also requires consideration. Iodine will work for short periods only (ie, weeks) because it is a poor algaecide. For prolonged storage, water should be chlorinated and kept in a tightly sealed container to reduce the risk of contamination and to maintain chlorine levels. For daily use, narrow-mouthed jars or containers with water spigots prevent contamination from repeated contact with hands or utensils.92,153,205
Few studies compare multiple techniques or devices.114,118,123,206–210 For additional reviews of water treatment methods, effectiveness, and efficacy data, see the following additional references.56,87,123,202,211
Sanitation
Sanitation and water treatment are inextricably linked.17,132 Wilderness and remote travelers typically lose access to accustomed sanitation with toilets and running water, and their hygiene practices diminish, similar to conditions for many residents in low- and middle-income countries. While there are little to no data on these practices and interventional impacts on the former group, there are extensive studies on the latter. Studies in low- and middle-income countries have demonstrated a clear benefit in the reduction of diarrheal illness and other infections from safe drinking water, hygiene, and adequate sanitation.24,25,212–216 The benefit is greater when all are applied together, especially with appropriate education.17,217,218
Personal hygiene, particularly hand washing, prevents the spread of infection from food contamination during preparation of meals.219–221 Disinfection of dishes and utensils is accomplished by rinsing in water containing enough household bleach to achieve a distinct chlorine odor. Travelers to remote low- and middle-income settings and wilderness areas should practice proper fecal waste disposal to prevent additional contamination of water supplies. Human waste should be buried 8 to 12 in deep, at least 100 ft from any water, and at a location where water run-off is not likely to wash organisms into nearby water sources. Groups of 3 persons or more should dig a common latrine to avoid numerous individual potholes and inadequate disposal. Victims in disaster situations without functional toilets can use latrines or plastic bags for fecal waste.
Conclusions
Wilderness and international travelers should be aware of water and sanitation conditions and plan in advance to use an effective means of disinfecting water. It is important for disaster and medical relief workers to understand the common methods of water treatment as well as improvisational methods. It is not possible for travelers to judge the microbiologic quality of water by sight, smell, or taste alone, and it is prudent to assume that even tap water is nonpotable in many low- and middle-income locations and in emergency situations. Simple and effective field techniques to improve microbiologic water quality are available.
Supplemental Material
sj-pdf-1-wem-10.1177_10806032231218722 - Supplemental material for Wilderness Medical Society Clinical Practice Guidelines on Water Treatment for Wilderness, International Travel, and Austere Situations
Supplemental material, sj-pdf-1-wem-10.1177_10806032231218722 for Wilderness Medical Society Clinical Practice Guidelines on Water Treatment for Wilderness, International Travel, and Austere Situations by Howard D. Backer, MD, MPH, Robert W. Derlet, MD, and Vincent R. Hill, PhD in Wilderness & Environmental Medicine
Footnotes
Author Contribution(s)
study concept and design (HDB, RWD, VRH); drafting of the manuscript (HDB, RWD, VRH); critical revision of the manuscript (HDB, RWD, VRH); and approval of the final manuscript (HDB, RWD, VRH).
Disclaimer
Use of trade names and commercial sources is for identification only and does not imply endorsement by the Centers for Disease Control and Prevention, the US Department of Health and Human Services, the Wilderness Medical Society, or the Wilderness and Environmental Medicine Journal. The findings and conclusions in this report are those of the authors and do not necessarily represent those of the Centers for Disease Control and Prevention, US Department of Health and Human Services, or the University of California.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
