Abstract
Implications for Practice and Research
The balance between the benefits and adverse consequences of screening (improved outcomes versus false-positive tests, overdiagnosis, overtreatment, and cost) must be considered when providing breast health care. Interventions aimed at enhancing patients’ understanding of their cancer risk have had a positive impact across all socioeconomic backgrounds, with low-income patients benefiting the most. A breast cancer risk assessment tool should be included in annual well-person exams in the primary care clinic. Nurses have a unique position in conducting risk assessment screenings and engaging in meaningful conversations with patients, taking into account their personal history, experiences, and expectations to develop a relationship based on trust.
Breast cancer is the most prevalent form of cancer among women in the United States, according to the United States Department of Health and Human Services (HHS), the Centers for Disease Control and Prevention (CED), and the National Cancer Institute (NCI) (2022, hereafter HHS et al., 2022). The lifetime risk of breast cancer for American women is 12.9%, meaning that approximately one in every eight American women will be diagnosed with breast cancer during their lives (NCI, 2020) and remains the second leading cause of cancer-related deaths in women in the United States (HHS et al., 2022). To effectively manage this disease, it is crucial to focus on prevention, risk reduction, and early detection, all of which can be tailored to meet individual patient needs through the implementation of a risk assessment tool.
Context
Problem Description
In 2023, the United States Preventive Services Task Force (USPSTF) released draft revised guidelines related to breast cancer screening. These revisions include recommendations on the appropriate age to start mammograms, the frequency of screenings, guidance for women with dense breasts, and specific considerations for Black women (USPSTF, 2023). The latest recommendation emphasizes that all women should undergo biennial breast cancer screening starting at the age of 40. The USPSTF acknowledges that women with dense breasts face an elevated risk of developing breast cancer and that current screening protocols may not effectively detect breast cancer in this population compared to women with non-dense breasts. Due to insufficient evidence, the task force does not make definitive recommendations either in favor of or against additional screening measures for this group. Additionally, the USPSTF recognizes that Black women are prone to more aggressive forms of breast cancer at an earlier age. The current recommendation to commence screening at age 40 may require supplementary screening strategies for individuals from racial and ethnic minority backgrounds. The new guidelines emphasize the need for further research and evidence to better navigate the optimal methods for identifying and managing breast cancer in these specific populations (USPSTF, 2023).
These updated guidelines from the USPSTF supplement the recommendations regarding risk from the American College of Obstetricians and Gynecologists (ACOG). A systematic review of breast cancer screening guidelines included in an update from ACOG (2017, reaffirmed in 2021) demonstrates a lack of consensus in guiding practice. Guidelines included in the ACOG bulletin include those from USPSTF, ACOG, the American Cancer Society (ACS), and the National Comprehensive Cancer Network (NCCN). As the USPSTF guideline is currently undergoing revision, the final recommendations will differ from what is included in the ACOG bulletin (2017) as indicated above. The analysis of recommendations for performing a clinical breast exam varies from not recommended (ACS) to recommended every 1–3 years for women ages 25–39 and annually for women ages 40 and older (NCCN). The NCCN recommends a clinical breast exam offered “in the context of a shared, informed decision-making approach that recognizes the uncertainty of additional benefits and harms of clinical breast examinations beyond screening mammography” (ACOG, 2017, p. 4).
Additional components of the guidelines from ACOG (2017) include mammography initiation age, screening interval, and mammography stop age. The guidelines again differ significantly, with the recommended initiation of mammography ranging from age 40 to 50. ACOG recommends offering mammograms starting at age 40 after counseling based on risk assessment and education about benefit versus risk of screening. Both the ACS and the NCCN recommend starting mammograms by age 45. The mammogram screening interval ranges from annual to biennial for all guidelines based on risk and assessment. The ACOG bulletin presents evidence-based recommendations to guide health-care providers in conducting breast cancer screenings, including risk assessment, patient education, and screening guidelines (ACOG, 2017). An integral component of the bulletin is the establishment of a shared decision-making framework between patients and providers, aiding in evaluating individual risk factors and striking a balance between the potential benefits and harms associated with various screening options. This framework proves invaluable in formulating appropriate breast cancer screening guidelines within the primary care setting.
According to ACOG (2017), “the goal of screening for cancer is to detect preclinical disease in healthy, asymptomatic patients to prevent adverse outcomes, improve survival, and avoid the need for more intensive treatments” (p. 3). The balance between the benefits and adverse consequences of screening must be considered when providing breast health care. The differing interpretations of the benefits of improved outcomes versus the adverse complications of false-positive tests, overdiagnosis, overtreatment, and cost has led to the development of inconsistent clinical practice guidelines. This lack of consensus has resulted in confusion for patients and providers in providing health care to women.
Available Knowledge
Breast Cancer Risk Assessment in Primary Care
According to Herold and Homer (2016), breast cancer risk assessment should be performed in primary care. “Evaluation of family history is one of the most effective and least expensive tools to identify individuals at increased risk for breast cancer, and yet many providers do not adequately assess their patients’ familial and genetic risk of developing breast cancer” (pp. 37–38). A feasibility study conducted by Anderson et al. (2015) examining the implementation of risk assessment for breast cancer in primary care revealed that nearly 90% of patients expressed a desire to discuss personalized risk results in the primary care setting. Patients who previously received negative mammogram results and those with private health insurance were less inclined to want to know their risk assessment outcomes. Patients over age 50 and those with heightened concerns about breast cancer, and underserved women were more likely to want to be informed about their risk (Anderson et al., 2015).
According to Waters et al. (2016), patients who underestimate their risk of breast cancer are less likely to engage in behaviors that reduce the risk of cancer. The level of uncertainty regarding cancer risk has been correlated with lower adherence to breast cancer screening guidelines. Conversely, patients who perceive themselves as being at higher risk are more inclined to undergo breast cancer screening and adopt health-related behaviors to mitigate their risk. Interventions aimed at enhancing patients’ understanding of their cancer risk have had a positive impact across all socioeconomic backgrounds, with low-income patients benefiting the most (Waters et al., 2016).
Polek and Hardie (2016) suggest that discussions about risk reduction should not be limited to women aged 35 and above. Women of all ages are exposed to messages about breast health from various sources, including family, friends, and the media. Educating women about breast health and breast cancer risk can help dispel common misconceptions about modifiable and non-modifiable risks and allows for shared conversations about adopting healthy breast practices. Early education on the significance of a healthy lifestyle, overall health, and current screening recommendations including clinical breast exams, can serve as a foundation for future discussions as patients age and screening recommendations evolve (Polek & Hardie, 2016).
A cross-sectional study by Guilford et al. (2017) examining the impact of breast cancer knowledge and beliefs on screening behavior among college-age women revealed a significant association between breast cancer knowledge, perceived risk, and behaviors related to risk reduction. Individuals who perceived themselves to be more susceptible to breast cancer were more likely to engage in breast cancer screening. Furthermore, improvements in breast cancer prevention were observed when self-efficacy for screening was enhanced and barriers were reduced (Guilford et al., 2017).
Zheng et al. (2022) investigated the use of breast cancer risk factors to determine the appropriate age to initiate breast cancer screening in China. The study considered risk divisions such as first-degree family history of breast cancer, benign breast disease, breastfeeding, age at menarche, and body mass index. The findings suggested that a risk-adapted starting age for breast cancer screening can inform current screening guidelines and assist in decision making (Zheng et al., 2022).
Breast Cancer Risk
Extensive research has been conducted on breast cancer risk and its correlation with the development of the disease. Risk factors can be categorized as modifiable or non-modifiable, and risk assessment must consider multiple factors. The ACOG practice bulletin (2017) provides guidance on assessing risk factors in clinical practice. Key risk factors for breast cancer include being female (over 99% of breast cancers occur in women) and advancing age.
Nonmodifiable, moderate risk modifiable, and low-risk modifiable factors categorize individual risk factors (Sciaraffa et al., 2020). The highest risk of breast cancer is associated with BReast CAncer (BRCA) gene mutations, or ductal carcinoma in situ (DCIS) or lobular carcinoma in situ (LCIS), although recent research by Strand et al., (2022) called into question what is currently known and accepted about screening and managing these breast tissue cellular changes. Specific familial risks, such as having first-degree relatives with breast cancer, should also be assessed (Solomon et al., 2016). Patients with hereditary breast and ovarian cancer (HBOC) syndrome carry a lifetime risk estimated at 40–80%, linked to known genetic mutations (Solomon et al., 2016).
The risk of hormone receptor-positive breast cancer increases with several reproductive breast cancer risks. Nulliparity and longer intervals between menarche and age of first birth are associated with higher risk. Other risks included younger age at first menarche, or older age at first birth or menopause. Factors that decrease risk include increased parity and breastfeeding. Familial cancer at a younger age is associated with an increased risk of a genetic mutation increasing breast cancer risk. Women with atypical ductal hyperplasia, lobular carcinoma, or atypical lobular hyperplasia have a four-fold increase in risk of subsequent breast cancer. In addition, women who received therapeutic chest radiation between the ages of 10–30 years of age are at increased risk of breast cancer. Girls who received chest radiation between the ages of 10–14 are at the highest risk (ACOG, 2017).
Other risks are identified in the literature as key in guiding breast cancer screening practice. Breast density can be affected by genetic and familial factors, as well as modifying factors including body mass index, parity, hormone replacement therapy, and menstrual cycle phase. The age of the first menarche, first live birth, and menopause are factors in assessing risk (Brown & Kartoz, 2014). It is imperative that the provider understand all possible identified breast cancer risks and an understanding of options for screening. The provider has the role of communicating with the patient her risks of breast cancer. Understanding the patient's own risk serves as a foundation for determining whether the risks of screening mammography outweigh the benefits (Pace & Keating, 2014).
The Risk of Overdiagnosis
The risk of overdiagnosis is an important component needing attention in the discussion of risk versus benefit and screening for breast cancer. A recent retrospective cohort study by Richman et al. (2023) reviewed women ages 70 and older screened for the risk of overdiagnosis, defined by the authors as diagnosing a cancer that would not otherwise cause the patient symptoms in their lifetime. This can be a cancer that is growing slowly enough that it will remain silent during the remainder of the patient's life, or in a patient who has a limited life expectancy. These authors estimated breast cancer overdiagnosis at 30% for women ages 70–74 and more than 50% for women over 85 years of age (Richman et al., 2023). Brawley and Ramalingam (2023), in an editorial about overdiagnosis and the Richman et al. (2023) study, linked overdiagnosis to overtreatment. The authors encouraged a more robust discussion of overdiagnosis as it relates to breast cancer when the risk of screening and treatment overweighs the benefits. Barber (2023) also discussed the overdiagnosis of cancers in America as a topic needing further attention and research. DCIS, a diagnosis already labeling a patient as “high risk” for invasive breast cancer, has more recently been treated with watchful waiting or active surveillance, with little change in the overall chance of dying of breast cancer compared to the general population (Barber, 2023). Strand et al. provided an updated ‘atlas’ to DCIS at the molecular level that increases our understanding of the many variations of these precancer cells which affect the diagnosis and treatment and should be part of the overall conversation about the decision to screen for breast cancer. Barber (2023) called for an improved conversation in our country regarding over testing, over diagnosing, and over treating, and what this means for the individual patient in assessing risk and screening recommendations.
Breast Cancer Risk Assessment Tools
Schrager and Burnside (2019) discussed various risk assessment models available for use in primary care and concluded that none are user-friendly for determining risk. The authors called for the development of a tool that can be seamlessly embedded into the electronic health record. Moreover, the current medical practice model poses challenges in terms of the time required to complete a comprehensive breast cancer risk assessment, provide risk education, and engage in shared decision-making conversations regarding risk reduction and screening (Schrager & Burnside, 2019).
Shared Decision-Making
ACOG recommends the use of a shared decision-making model to facilitate discussions about the risks and benefits of breast cancer screening (ACOG, 2017). Establishing a trusting relationship between the patient and health-care provider is crucial for effective shared decision-making and ensuring the patient's best interests are considered (Go & Sandhu, 2021). Adequate time during appointments is necessary to allow patients to fully understand the risks, benefits, and implications of risk reduction and screening referrals (Rainey et al., 2019). Maes-Carballo et al. (2021) described shared decision-making as a collaborative process that allows the patient to have a voice in health-care discussions, resulting in greater patient satisfaction and engagement.
Martin et al. (2020) studied mammography screening recommendations in primary care, reinforcing that despite differences in protocols, informed decision-making between the patient and nurse is the goal in breast cancer screening. Providing education on individual breast cancer risk should include potential risks, benefits, and harm in the decision to screen or not screen at a given time in the patient's life. The conversation is based on providing education based on risk and respecting the patient as an equal partner in the decision-making process.
A Breast Cancer Risk Assessment Tool
Study Purpose
The purpose of this study was to enhance the knowledge of patients and health-care providers regarding the use of individual breast cancer risk assessment in facilitating shared decision-making for breast cancer screening. This risk assessment guides a shared decision-making practice between the nurse, provider, and patient in determining the need for education about risk reduction, determination of clinical breast exams, and referral for mammography.
Study Site
The study site was an ambulatory care family planning agency in Minnesota offering reproductive and sexual health services for people of all genders. The clinic serves rural and suburban populations in four different locations, employing three nurses and six providers. Key stakeholders included patients, the executive director, the medical director, nurses, laboratory personnel, advanced practice nurse and physician providers, and members of the agency's board of directors.
Before the implementation of this quality improvement project, patients had annual well-person exams including a general health assessment and physical exam. The health assessment included questions on general health and surgical history, not specific to breast cancer risk. Breast exams, referral for mammography, and patient education to reduce breast cancer risk were provided based on general guidelines and were not specific to the patient based on risk.
Methods
Interventions
The goal of the quality improvement program was to implement a breast cancer risk assessment tool to guide practice in a rural family planning program. The research and development of the breast cancer risk assessment protocol resulted in choosing the NCI (2017) Breast Cancer Risk Assessment Tool (see Table 1) available online and used on paper in the clinic. To facilitate patient engagement, the assessment questions were formulated from the patient's perspective, allowing them to answer the risk-related inquiries first. The question regarding race and ethnicity was excluded from the paper version of the risk form as patients had already provided this information on clinic intake paperwork. For patients age 35 and older, a 5-year risk and lifetime risk of breast cancer were calculated using the NCI online tool. For patients under age 35, the questionnaire was used as a teaching tool related to breast cancer risk.
Breast cancer risk assessment tool.
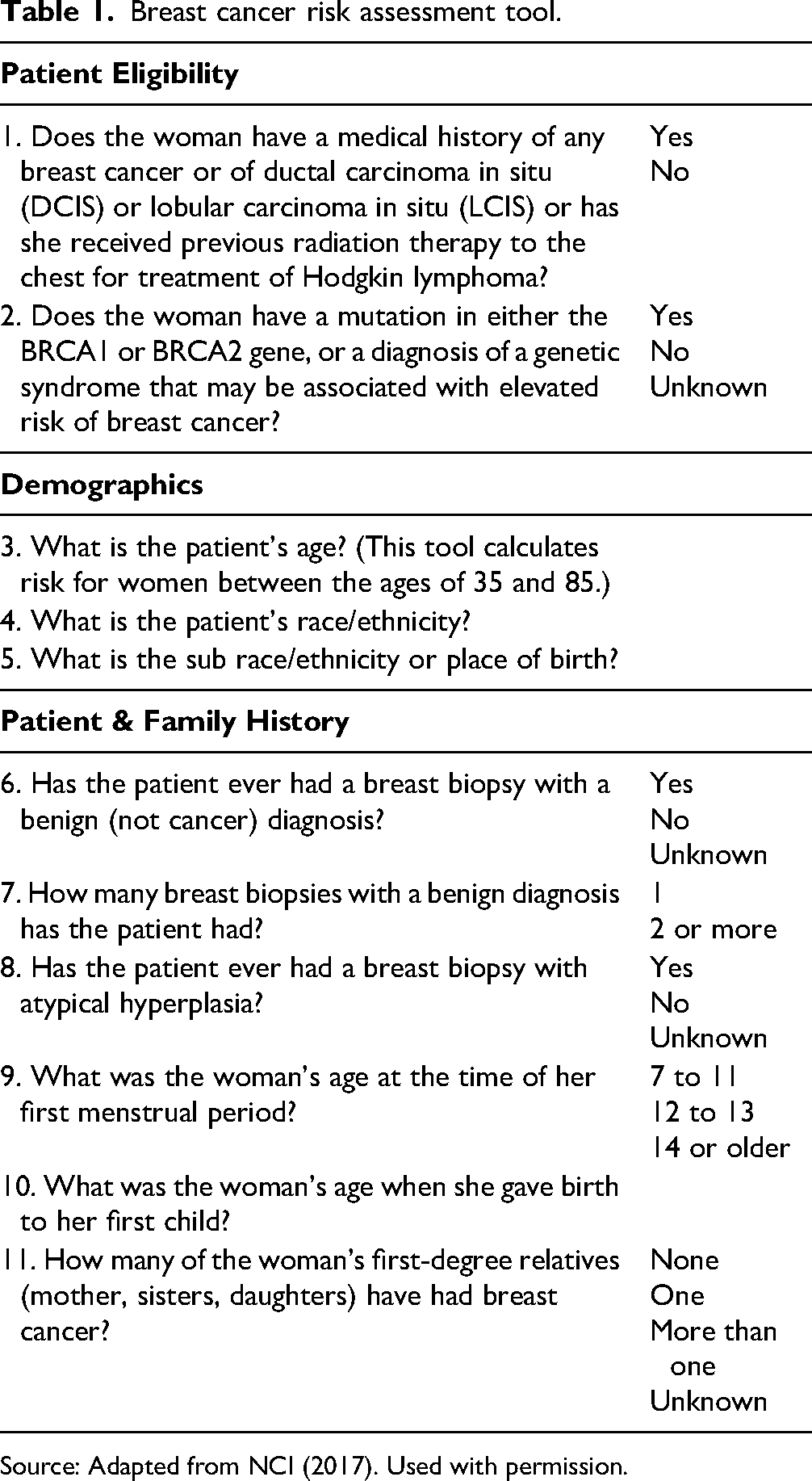
Source: Adapted from NCI (2017). Used with permission.
Patients answering “yes” to either of the first two questions (about personal history of cancer or presence of mutations) are not eligible to use the NCI tool. Patients with a history of breast cancer, DCIS, LCIS, chest radiation for treatment of Hodgkin's lymphoma, a known mutation of BRCA1 or BRCA2 or other genetic syndromes, are all at elevated risk for breast cancer and are not candidates for the use of this risk assessment tool (NCI, 2017). Patients who answer questions one or two as “yes” should be referred to a breast cancer specialist for further discussion in managing breast cancer screening in higher risk patients. The rest of the questionnaire identifies specific areas of risk associated with breast cancer.
Study of the Interventions
During a period of three months, all patients visiting the clinic for their annual exams were requested to complete the Breast Cancer Risk Assessment Tool on paper. The nurse carefully reviewed the form with each patient to ensure that all questions were answered accurately, and that the patient understood them fully. For patients age 35 or above, the nurse entered the questionnaire results from the paper form into the Breast Cancer Risk Assessment Tool website, which generates a specific risk percentage for that patient. This risk percentage was then recorded on the completed paper form for the provider's review.
The information obtained from the paper questionnaire served as a basis for a shared decision-making discussion between the patient and nurse regarding the breast cancer risks identified by the patient. This shared conversation helped guide the nursing practice in recommending appropriate clinical breast exams, referral for mammography if applicable, and providing education aimed at reducing breast cancer risks. These interventions and discussions were documented on the Interventions Form for future reference (see Table 2).
Documentation of interventions provided based on risk.
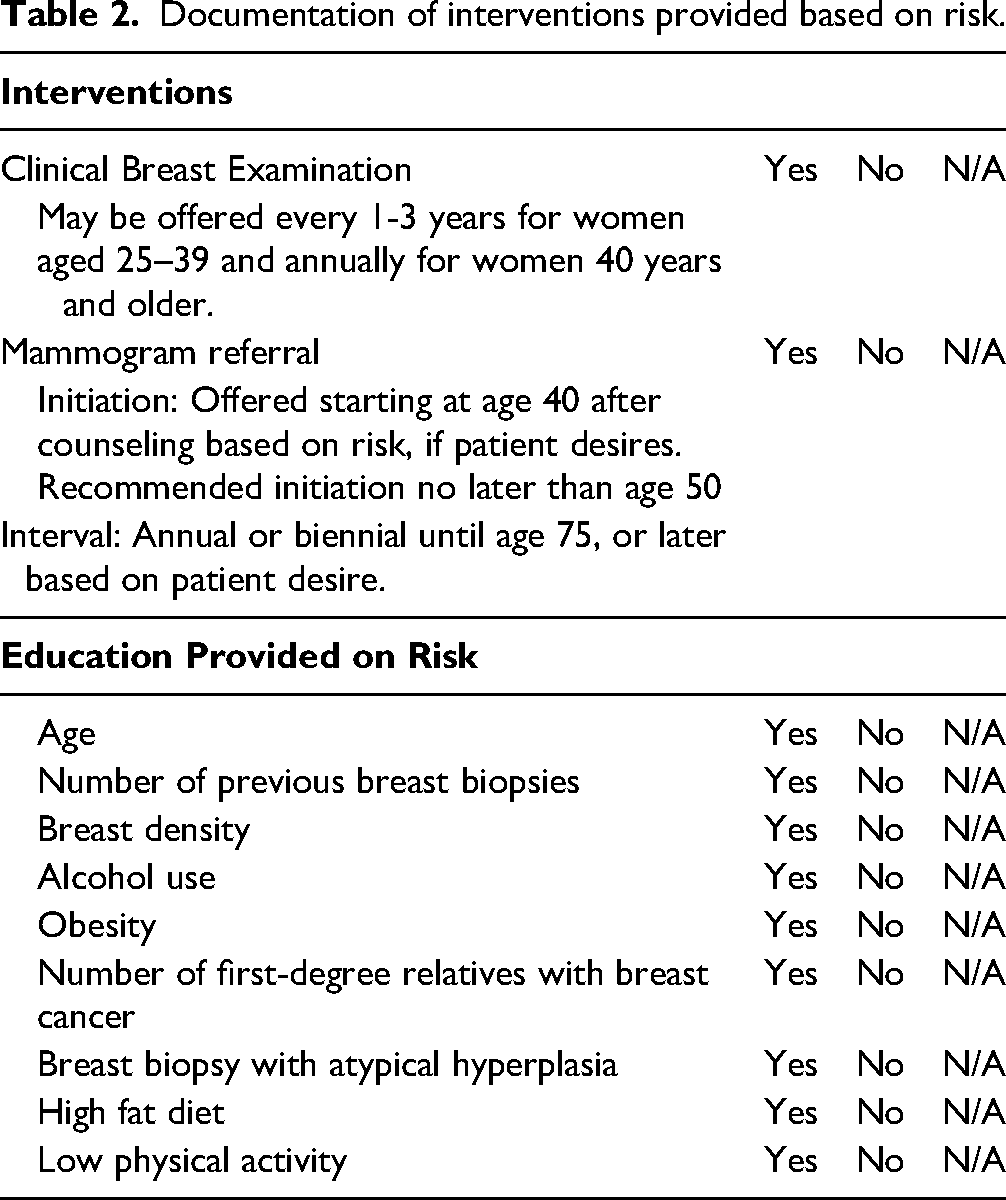
Results
Summary and Implications
During the three-month period, all 14 patients who visited the clinic for a well-woman exam completed the risk assessment on paper. Since none of the patients had a history of breast cancer or identified BRCA mutation, they were all categorized as average risk. The majority of the patients (69.2%, n = 14) were under the age of 35, so their actual risk was not calculated using the NCI website. On average, patients under the age of 35 had 0.3 identified risks per patient, while patients over the age of 35 had an average of 1.75 identified risks per patient. Four patients were over the age of 35 (ages 40, 43, 48, and 51). Three had a 5-year risk of breast cancer lower than the national average compared to women of the same age and race/ethnicity in the general United States population. One patient, aged 48, had a 5-year and lifetime risk of breast cancer over the national average (1.6 patient risk vs 1.2 average 5-year risk; 15.9 patient risk vs 11.5 lifetime risk). The data analysis revealed a correlation between increasing age and increasing risk. Patients over the age of 35 had more identified breast cancer risks compared to those under 35. The identified risks included advancing age, early onset of menstruation, and having first-degree relatives with breast cancer.
The questionnaire also identified additional risks such as increased breast density, high-fat diet, elevated alcohol intake, sedentary lifestyle, and obesity. Patients with zero identified breast cancer risks received an average of two educational units per patient. In contrast, patients with one to two identified risks received an average of 4.6 educational units per patient. This indicates that patients with one or two identified risks received more education regarding their risks, including guidance on reducing modifiable risk factors, compared to patients with no identified risks. See Table 3 for a summary of risks identified by age.
Summary of breast cancer risks identified by age (n = 14).
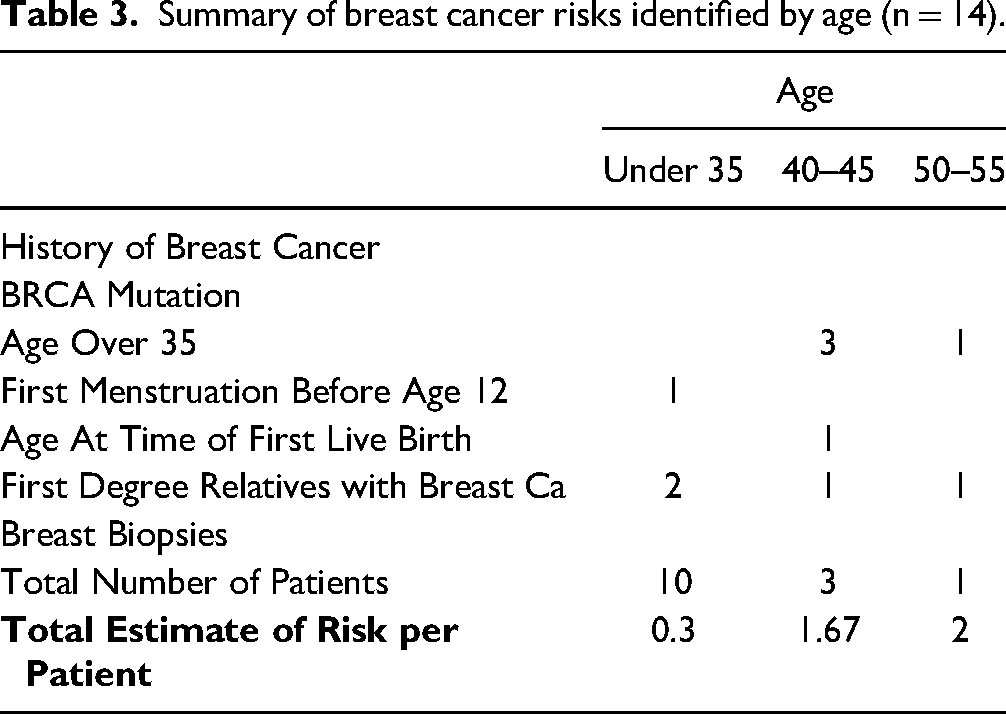
All 14 patients actively engaged in shared decision-making with the nurse and the provider regarding risk reduction education, the indication for a clinical breast exam, and potential referral, based on the results of the risk assessment. For patients over 35, a discussion of their calculated 5-year and lifetime risk of developing breast cancer compared to the general U.S. population was articulated with education provided about what the risk calculation meant for them as an individual. Interestingly, all patients over age 35 identified breast cancer as a concern even if it was below or slightly above the national average for their age and race/ethnicity.
Education was tailored to the specific risks identified, encompassing both non-modifiable factors such as increasing age, having first-degree relatives with breast cancer, previous biopsies, and breast density, as well as modifiable risks. Modifiable risk reduction education focused on strategies such as reducing alcohol consumption, increasing physical activity, maintaining a healthy body weight, and adopting a low-fat diet. A summary of the breast cancer risk assessment education provided, categorized by the number of risks identified, can be found in Table 4.
Summary of breast cancer risk assessment education (n = 14).
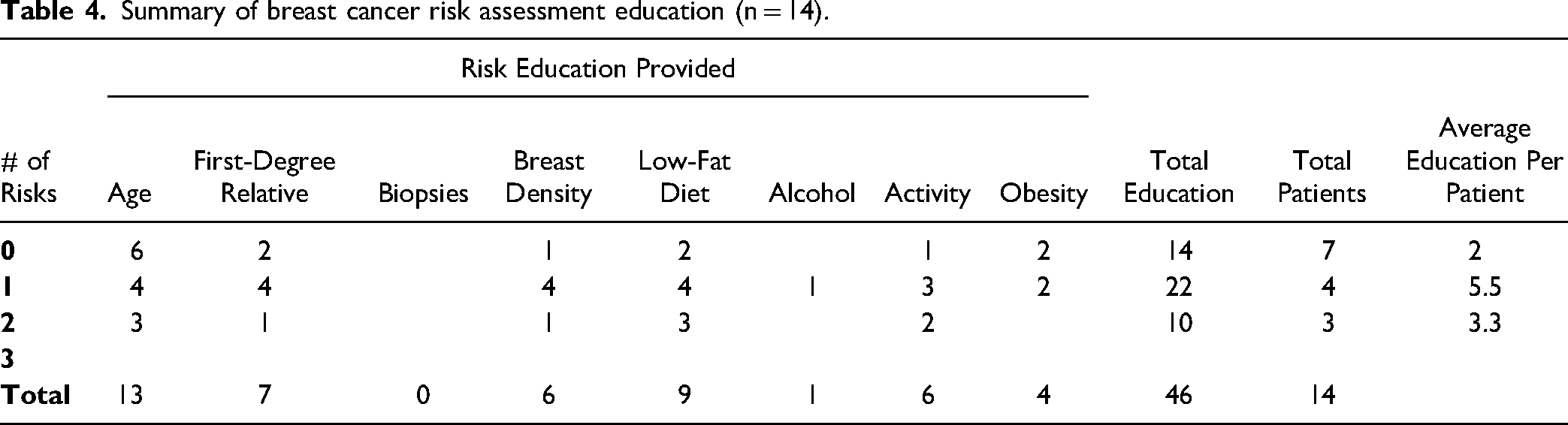
Patients with higher risk profiles were more inclined to choose a clinical breast exam. Among patients with no identified risks, 5 out of 7 opted for a clinical breast exam. For patients with one to two identified risks, all 7 chose to undergo a clinical breast exam after engaging in a conversation with the nurse and provider. Additionally, all 4 patients (100%) who were over the age of 35 elected a mammography referral. Refer to Table 5 for a summary of the interventions provided categorized by the number of risks identified.
Summary of interventions provided based on risk.

Discussion
Nursing
Nursing plays a crucial role in assessing individual risk and providing education on breast cancer screening in primary care. By involving registered nurses (RNs) in the assessment of breast cancer risk, the opportunity for comprehensive evaluation, education, and intervention related to breast health is enhanced. It is essential for the nurse to have a strong understanding of both modifiable and non-modifiable risk factors for breast cancer to develop a risk profile and effectively communicate it to the patient (Solomon et al., 2016). Patients who have a clear understanding of their risks are more likely to actively engage with the education provided to reduce modifiable risks and to participate in clinical breast exams and mammography referrals (ACOG, 2017). Nurses have a unique position in conducting risk assessment screenings and engaging in meaningful conversations with patients, taking into account their personal history, experiences, and expectations, and building trust (Go & Sandhu, 2021).
Advanced Practice Nursing
In partnership with the RN, the advanced practice nurse (APRN) can follow up on the results of the risk assessment and continue the discussion of modifiable and non-modifiable risks for breast cancer (Sciaraffa et al., 2020), reinforcing the education provided and continuing the shared decision-making conversation about clinical breast exams and mammography (ACOG, 2017). The patient can voice their concerns about when to start clinical breast exams, why they are recommended, and what the next steps might be if an irregularity is discovered (ACOG, 2017). The APRN can also reinforce patients’ awareness of their breast tissue norms and when to follow up with concerns or questions. Finally, a shared decision-making conversation can discuss the latest recommendations about when to start mammography for patients over 35, considering the benefits versus risks and outlining the next steps in the individual patient's breast cancer screening journey (ACOG, 2017). This discussion should include the most current information about breast cancer screening recommendations including information about the risks of screening, overdiagnoses, and overtreatment of cancers, especially for women over the age of 50 (Richman et al., 2023).
Limitations
During this quality improvement project, several limitations were identified. The number of patients was small, and their average age was less than 35. Since increasing age is a significant risk factor for breast cancer, it would be valuable to collect additional data from patients over 35 to understand how education, clinical breast exams, and referrals for mammography are related in this age group.
This project was implemented in suburban and rural areas of Minnesota, which may limit its generalizability to more diverse populations. The lack of diversity in the patient population may affect the applicability of the findings and interventions to different cultural, ethnic, and socioeconomic backgrounds. Considering these factors in future projects is recommended, to ensure a more comprehensive understanding of breast cancer risk assessment and education. Addressing these limitations and expanding the project's scope to include a larger and more diverse patient population would strengthen the outcomes and enhance the effectiveness of the interventions in different settings.
Conclusion
Shared decision making is more than just a phrase; it's the practice of listening to the patient and allowing them the opportunity to make decisions alongside the provider (Baenziger, 2017), with a toolkit of known risks and benefits to screen or not screen for breast cancer at any given age. By providing comprehensive care related to breast health, the patient becomes an active participant in their health-care decisions.
Footnotes
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Author Biography
