Abstract
Persistent substance use during pregnancy is in contrast with the behavior of most women, who upon pregnancy awareness attempt to engage in healthier practices (Daley et al., 1998; Kruk & Banga, 2011; World Health Organization, 2016). However, there has been an increase in the rate of illicit substance use among pregnant women compared with nonpregnant women seeking treatment over the past decades. Reports of illicit substance use (and most often polysubstance use) upon treatment admission has increased from 51% in 2000 to 64% in 2010 among pregnant women compared with 38% in 2000 and 49% in 2010 among nonpregnant women of childbearing age (Substance Abuse and Mental Health Services Administration [SAMHSA], 2013). More recent estimates continue to demonstrate an upward trend of illicit substance use among the number of pregnant women entering treatment, from 4.7% in 2015 to 6.3% in 2016 (SAMHSA, 2017). Illicit substance use trends during pregnancy are increasing during a time when specialized treatment resources for this group are limited (Guttmacher Institute, 2021; SAMHSA, 2013, 2017) and numerous barriers exist in accessing care.
Women who use illicit substances during pregnancy experience many barriers to accessing substance use disorder (SUD) treatment and prenatal care (Ashley et al., 2003; Finkelstein, 1996; Haller et al., 2003; Howard, 2015; Jackson & Shannon, 2016; Jansson et al., 1996; Jones & Kaltenbach, 2013; Meixner et al., 2016). External barriers include the lack of comprehensive services in a single location, lack of childcare to enable attendance, lack of transportation, and possible discouragement to attend treatment by a partner or spouse who is using substances (Ashley et al., 2003; Jackson & Shannon, 2016; Jansson et al., 1996; Jones & Kaltenbach, 2013; Meixner et al., 2016). Internal barriers include shame and guilt regarding substance use, fear of losing child custody when entering treatment or upon delivery, and the internalization of social stigma related to substance use among pregnant and parenting women (Ashley et al., 2003; Finkelstein, 1996; Haller et al., 2003; Howard, 2015; Jackson & Shannon, 2016; Meixner et al., 2016). When pregnant women do access care and are enrolled in SUD treatment, they have high rates of psychiatric comorbidity (Covington, 2008; Finkelstein, 1996; Fitzsimons et al., 2007; Jones et al., 2011).
Women in general are more likely than their male counterparts to present to SUD treatment with psychiatric symptoms (Covington, 2008; Greenfield et al., 2007). Specifically, pregnant women with SUDs often experience greater psychiatric deficits compared with nonpregnant women with SUDs (Grella, 1999; SAMHSA, 2009). Regardless of pregnancy status, women with SUDs who enter treatment report social and psychological challenges such as mental, emotional, and behavior disorders; trauma exposure; relationship dysfunction; and homelessness (Alhusen et al., 2013; Benningfield et al., 2012; Brown et al., 2012; Covington, 2008; Fitzsimons et al., 2007; Lee King et al., 2015; McHugh et al., 2013; Storbjörk, 2011; Tuten et al., 2004; Tuten et al., 2009; Tuten et al., 2011). In general, women with SUDs often have extensive trauma histories, from microaggressions of being female in a male-dominated society to macroaggressions of persistent physical and sexual abuse, and have identified being victims of domestic violence with histories of childhood abuse (Brady et al., 2009; Covington, 2008; Greenfield et al., 2007). Furthermore, women with SUDs have been found to have greater abuse histories than men with SUDs, and psychiatric severity was demonstrated as being a better predictor of HIV risk behavior than abuse histories among persons with SUDs (Majer et al., 2014).
Pregnant women with SUDs who reported intimate partner violence have higher rates of cannabis use compared with pregnant women with SUDs who did not report this phenomenon (Alhusen et al., 2003), whereas those admitted to SUD treatment revealed more severe alcohol use, social problems, and psychiatric issues compared with those without intimate partner violence histories (Tuten et al., 2004). Pregnant women with severe psychiatric symptoms upon SUD treatment admission have been found to have greater substance use and less favorable treatment outcomes (Back et al., 2011; Fitzsimons et al., 2007; Grella, 1999; Storbjörk, 2011; Tuten et al., 2009). Psychiatric disorders among pregnant women with SUDs are associated with extended neonatal hospital days (Tuten et al., 2011), decreased SUD treatment participation (Benningfield et al., 2012), and increased social vulnerability (Lee King et al., 2015). Adverse outcomes (preterm delivery, low birthweight, neonatal intensive care unit admission, and stillbirth) were significantly greater for pregnant women who reported any psychosocial factor (lifetime history of mental illness, lifetime SUDs, or interpersonal trauma) compared with pregnant women who did not report any psychosocial factor (adjusted odds ratio = 1.34, 95% confidence interval = [1.04, 1.73], p = .03; McDonald et al., 2020). Additionally, when all psychosocial factors were present, the odds of an adverse outcome were significantly greater (adjusted odds ratio = 2.04, 95% confidence interval = [1.09, 3.81], p = .03) compared with pregnant women who did not report any psychosocial factor (McDonald et al., 2020).
Likewise, research among pregnant women with psychiatric comorbidity receiving medication-based therapy (MBT) is consistent with this body of knowledge. Such women with primary mood disorders in particular were significantly more likely to test positive for illicit substance use during treatment (Fitzsimons et al., 2007). Mood disorders were related to negative delivery outcomes among a group of pregnant women receiving MBT in terms of longer hospital lengths of stay for newborns (Tuten et al., 2009). Pregnant women with comorbid psychiatric and SUDs infrequently attended prenatal visits and had less prenatal visits compared with pregnant women with SUDs only (Oei et al., 2009). Overall, co-occurring psychiatric disorders among pregnant women with SUDs have been found to negatively affect treatment outcomes (Brady et al., 2009). Evidence points to the need for comprehensive care programs for this population to include SUD treatment, perinatal care, and services addressing psychiatric symptoms.
Comprehensive Care Models
Comprehensive care delivery models might increase favorable outcomes for pregnant women with SUDs and comorbid psychiatric conditions because they provide a women-only, “one-stop-shop” environment offering psychiatric and SUD treatment and medical perinatal services in a single location. This model type has been identified as generating greater likelihood of abstinence from licit and illicit substances and increasing child custody at 2-year follow-ups (Jansson et al., 2003; Jones et al., 2001). The Center for Addiction and Pregnancy (CAP) is an exemplary program offering holistic services to pregnant women with SUDs by providing a comprehensive care environment within a community-based center (Johns Hopkins Medicine, 2015). Resources at CAP include substance abuse treatment, psychiatric evaluation and treatment, obstetric and pediatric care, and family planning services (Jansson et al., 2003). Women who attend CAP receive individual and group counseling, case management, obstetric and pediatric care via an outpatient clinic.
One investigation using a comprehensive care model at CAP (Jones et al., 2011) examined differences between comprehensive care versus usual care interventions among pregnant women with SUDs with psychiatric symptoms. This study found that women who received the comprehensive care intervention spent significantly more days in treatment and more days in recovery housing. Additional research in comprehensive care settings is warranted to better understand the impact of this delivery model on pregnant clients with psychiatric comorbidity. Although differences in relation to treatment interventions have been found for this population, it is not clear whether comprehensive care models provide effects comparable to those among pregnant women with SUDs who do not report psychiatric comorbidity. Taken together, previous investigations cited in this report suggest that pregnant women with psychiatric comorbid SUDs might require more than what is provided in comprehensive care models in order to achieve results comparable to pregnant women who do not have psychiatric comorbidity.
Psychiatric Severity
Examining psychiatric severity levels among pregnant women with SUDs receiving comprehensive care who present with varied psychiatric symptoms would address this issue. The Psychiatric Severity Index (PSI) of the Addiction Severity Index (ASI; McLellan et al., 1980; McLellan et al., 1992) is a highly regarded measure used in addiction research to assess psychiatric problem severity irrespective of diagnostic categorization. It has been used to assess psychiatric comorbidity in a national, longitudinal investigation of men and women with SUDs to better understand treatment attendance, abstinence, and mental health outcomes (Majer et al., 2008) and a randomized clinical trial examining the effects of self-run and modified therapeutic communities among those with psychiatric comorbidity (Majer et al., 2016). The ASI has been used extensively for treatment planning, program evaluation, and clinical outcome studies with good internal consistency and predictive validity (Butler et al., 2001; Leonhard et al., 2000; Rosen et al., 2000), and the PSI has been demonstrated as the most reliable subscale of the ASI (Makela, 2004).
Study Purpose
The present investigation used the PSI subscale from the fifth edition of the ASI to measure psychiatric comorbidity among pregnant women with SUDs who reported various psychiatric symptoms (depression, anxiety, hallucinations, trouble concentrating, violent behavior, suicide ideation, and suicide attempt) upon admission to a comprehensive care treatment program. It was important to examine the impact of psychiatric comorbidity in relation to neonatal and maternal delivery outcomes to further our understanding of comprehensive care interventions for this population as findings would have relevant treatment and research implications. Participants in this investigation were originally recruited from the CAP. We hypothesized pregnant women with psychiatric comorbid SUDs would have worse neonatal and maternal outcomes compared with pregnant women with SUDs (and no psychiatric comorbidity), consistent with research studies for this population.
Method
Sample
This analysis focused on 74 participants with neonatal and maternal delivery outcome data. Women were on average 30.1 years old (SD = 6.0), Black (52.9%), and single (76.5%), with a mean education level of 11.6 years (SD = 1.6). They reported histories of homelessness (45.4%) and abuse (40.3%), and most (73.1%) presented with medical comorbidities (gestational diabetes, hypertension, depression, or other/undefined). In terms of substance use, most women (71.4%) reported current cigarette smoking with lifetime histories (any use) of alcohol (53.8%), cannabis (52.1%), stimulants (68.1%), opioids (68.1%), and polysubstance use (67.2%). Stimulants were defined as cocaine or amphetamines; opioids as heroin, methadone misuse, and/or other opioids; and polysubstance use as using more than one of these substances excluding cigarettes. During study participation, women were expected to provide weekly urine toxicology screens with 20.2% receiving MBT of methadone.
Instruments
Three neonatal and two maternal outcome measures were examined at delivery. Neonatal outcomes were birthweight in grams, hospital length of stay in days, and whether newborns were treated for neonatal abstinence syndrome (NAS). Maternal outcomes were hospital length of stay in days and positive urine toxicology on delivery for any traces of opioids (excluding MBT), stimulants, alcohol, and cannabis.
Psychiatric comorbidity was assessed using the PSI (McLellan et al., 1992) that participants completed upon admission. The PSI is a composite score index based on a variety of psychiatric symptoms experienced in the past 30 days, and it is calculated using 11 questions from the psychiatric status section of the ASI (Appendix A; McGahan et al., 1986). This composite score, ranging from .00 to 1.00, is used to assess any significant psychological and emotional problems and current level of willingness to receive treatment for these issues (McGahan et al., 1986). Higher PSI scores indicate greater psychiatric problem severity. McLellan et al. (1983) defined high and low psychiatric severity when PSI scores are ±1 SD from the mean, so we dichotomized PSI scores into two groups: (1) high psychiatric severity (high PSI) and (2) no psychiatric severity (no PSI).
Women in this sample had a mean PSI of .18 (SD = .22); therefore, women with PSI scores ≥.40 were classified as high PSI (n = 22) and those with scores equal to .00 were labeled as no PSI (n = 52). Dichotomizing PSI scores in this manner has been used to assess psychiatric comorbidity in previous investigations (Ball et al., 2004; Cridland et al., 2012; Majer et al., 2008; Majer et al., 2015; Majer et al., 2016). Women in the high PSI group in the present study had a mean of .50 (SD = .08), which is higher than PSI scores reported among persons with severe mental disorders (Carey et al., 1997) and other persons with SUDs with diagnosed co-occurring psychiatric disorders (Franken & Hendriks, 2001; McKay, 2005). Thus, the PSI provided us with a measure of psychiatric comorbidity in the sample (i.e., among those who met the criterion cutoff for placement in the high PSI group). The PSI in the present investigation had very good internal consistency (Cronbach’s α = .86).
Design
The present study is a secondary data analyses of women with SUDs receiving comprehensive care treatment during pregnancy and was deemed not human subjects research by the institutional review board. The parent study (Tuten et al., 2012) was an institutional review board–approved, randomized clinical trial comparing reinforcement-based treatment to usual care treatment on delivery outcomes, and it provides more detailed information on study recruitment and randomization. Reinforcement-based treatment is a comprehensive approach that is used to assist clients with SUDs achieve and maintain abstinence by emphasizing and rewarding their strengths. Based on operant conditioning principles and social learning theory, reinforcement-based treatment is a compilation of contingent incentives, motivational interviewing, relapse prevention strategies, case management, and community outreach efforts (Tuten et al). Women in this study did not significantly differ between high (50.0%) and no (54.9%) PSI grouping based on their parent study assignment, χ2(1) = 0.15, p = .700.
The present investigation used the Mega Interactive Model of Substance Use among Women (MIMSUAW) as its guiding framework (Pagliaro & Pagliaro, 2000). Building on prior research, the MIMSUAW defines four overarching dimensions for women with SUDs: women, societal, time, and substance. These dimensions each house multiple expressions and appropriately represent the complex issues surrounding pregnant women with SUDs. The extensive design of the MIMSUAW is representative of the pervasive features surrounding pregnant women with SUDs and was chosen for its relevance to this work.
Analyses
Preliminary analyses examined descriptive statistics in relation to sociodemographic characteristics, population specifics, substance use, and therapeutic measures. Sociodemographic characteristics were age in years, race, marital status, and years of education. Population specifics were history of homelessness, history of abuse (emotional, physical, and sexual), and medical comorbidities (gestational diabetes, hypertension, depression, and other/undefined). Substance use and therapeutic measures were daily cigarette smoker, history of substance use (opioid, polysubstance, stimulants, cannabis, and alcohol), percentage of positive urine toxicology screens (number of positive toxicology screens divided by the number of urine samples collected), and receiving MBT.
Inferential analyses included a preliminary analysis to identify potential confounding variables that would be controlled for in hypothesis testing by examining differences between PSI groups on categorical (chi-square) and continuous (one-way multiple analysis of variance) sociodemographic and population characteristics. Regression models examined PSI groups in predicting changes in delivery outcomes and hospital lengths of stay; simple linear regression to understand the influence of PSI groups on three continuous outcome variables (birthweight, neonatal, and maternal hospital length of stay) and logistic regression to examine the effect of PSI groups on two dichotomous outcomes (neonatal treatment for NAS and maternal positive urine toxicology). Using a post hoc test in G*Power, power (1 − β err probability) was 0.97 for a large effect size (f2 = 0.26) with α err probability = 0.05, sample size = 57, and number of predictors = 1. A pairwise deletion approach was used to evaluate data and calculate analyses. Participants with missing data on any analytic model variable (36% of the entire sample from the parent study due to attrition rates) were excluded from analyses. A missing values analysis of all the independent and dependent variables indicated that the data were missing completely at random (MCAR); Little’s MCAR test: χ2(49) = 48.42, p = .496. The interpretation of MCAR assumes neither observed values nor missing values of a variable affect probability.
Results
Normal birthweights ranged between 2500 g (5 pounds, 8 ounces) and 4000 g (8 pounds, 13 ounces) for full-term deliveries between 37 and 42 weeks’ gestation. Most women (71.7%) delivered infants with normal birthweights averaging 2808.8 g (SD = 675.5). Neonates spent a mean of 10.7 days (SD = 15.7) in the hospital after delivery predominantly related to NAS. Half the neonates (50.0%) were treated for NAS after birth (NAS typically presents in infants 24 to 72 hours after delivery and manifests as neurological, gastrointestinal, and thermoregulatory deficits). Maternal hospital lengths of stay averaged 2.9 days (SD = 1.4) and 28.6% of women had positive urine toxicology screens at delivery. Data on maternal sociodemographic and population characteristics in relation to PSI group are presented in Table 1. Women in both PSI groups were statistically similar on most of these characteristics with a few exceptions; women in the no PSI group were older, whereas proportionately more women in the high PSI group were White and reported more homelessness and abuse histories.
Maternal Demographic Characteristics According to PSI Group.
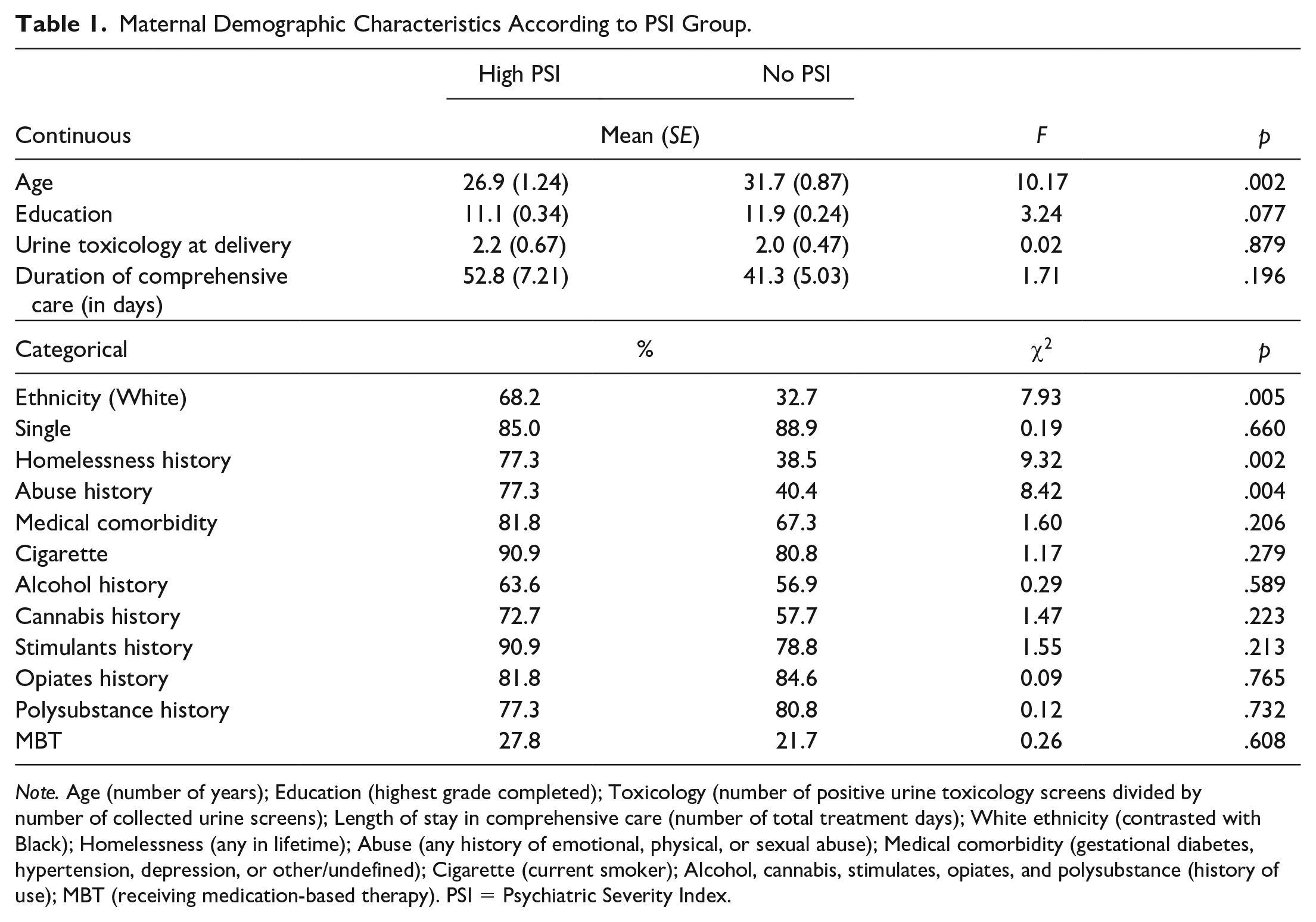
Note. Age (number of years); Education (highest grade completed); Toxicology (number of positive urine toxicology screens divided by number of collected urine screens); Length of stay in comprehensive care (number of total treatment days); White ethnicity (contrasted with Black); Homelessness (any in lifetime); Abuse (any history of emotional, physical, or sexual abuse); Medical comorbidity (gestational diabetes, hypertension, depression, or other/undefined); Cigarette (current smoker); Alcohol, cannabis, stimulates, opiates, and polysubstance (history of use); MBT (receiving medication-based therapy). PSI = Psychiatric Severity Index.
Simple linear regression was used to examine posttreatment changes among PSI groups in relation to neonatal and maternal continuous outcomes (birthweight, neonatal and maternal length of hospital stay), including age, race, and histories of homelessness and abuse in each of these models to control for their potential effects. Individual regression models for each outcome were employed to understand the predictive relationship of high/no PSI groups (coded 0 = no PSI, 1 = high PSI) on outcomes. Findings from each regression model are presented in Table 2. PSI grouping was not a significant predictor of birthweight, F(3, 69) = .04, p < .991; adjusted R2 = −.04, neonatal hospital length of stay, F(3, 30) = 1.24, p < .314; adjusted R2 = .02, or maternal length of stay, F(3, 33) = .93, p < .438; adjusted R2 = −.01.
Regression Analysis Predicting Neonatal and Maternal Delivery Outcomes.

Note. Birthweight in grams. Hospital length of stay in days. NAS is Neonatal Abstinence Syndrome. High PSI is compared to no PSI. PSI = Psychiatric Severity Index.
To better understand these results, we re-ran these analyses without sociodemographic and population characteristics, and the results were statistically similar across models in that PSI grouping was not a significant predictor. In addition, we conducted a one-way multivariate analysis of covariance to control for the potential influence of these characteristics, multicollinearity between predictors, and risk of error associated having conducted multiple models of linear regression, and the results were statistically similar in that neither PSI group nor control variables (age, race, homelessness, and abuse histories) were significantly related to differences in post-treatment outcomes.
Similar findings were observed when examining posttreatment neonatal and maternal categorical outcomes, including sociodemographic and population characteristics (age, race, history of homelessness, and abuse) in each model to control for their potential effects. Logistic regression models were used to test for neonates treated for NAS (coded: 0 = not treated, 1 = treated) and maternal toxicology (coded: 0 = negative, 1 = positive) at delivery. Individual models for each outcome were tested to understand the likelihood of these outcomes in relation to PSI grouping. Findings of these logistic regression models (presented in Table 2) revealed PSI groups did not significantly predict post-treatment changes in terms neonatal treatment, Wald’s χ2(3) = 4.02, p = .259, or maternal toxicology, Wald’s χ2(3) = 1.94, p = .589, outcomes. These results were statistically similar when sociodemographic and population characteristics were not entered in each logistic regression model. Overall, women in this sample had statistically similar neonatal and maternal delivery outcomes regardless of PSI grouping; therefore, our hypothesis was not supported.
Discussion
It was hypothesized that women with psychiatric comorbidity would have worse delivery outcomes, but this was not supported by the data. Psychiatric comorbidity was not a significant predictor of three neonatal and two maternal delivery outcomes, suggesting comprehensive care programs for pregnant women with SUDs are effective for those with co-occurring psychiatric disorders.
In terms of neonatal outcomes, birthweight was found to be within the normal range regardless of psychiatric comorbidity, and although the average birthweight of most newborns is toward the lower end of this range, these findings are consistent with research among pregnant women with SUDs receiving comprehensive care (Jansson et al., 2003; Jansson et al., 2012; Tuten et al., 2009; Tuten et al., 2011). This suggests comprehensive care is an appropriate intervention for preventing low birthweights among newborns of pregnant women with psychiatric comorbid SUDs (Tuten et al., 2009; Tuten et al., 2011). Additionally, neonatal length of hospital stay was not predicted by psychiatric comorbidity, a result that is somewhat inconsistent with findings from one investigation (Tuten et al., 2011) that found increased neonatal length of hospital stays in relation to other markers of psychiatric comorbidity. The discrepancy between these findings might be attributed to how comorbidity was operationalized, pointing to the need for additional research to examine the impact of psychiatric severity regardless of diagnostic categorization because pregnant women with SUDs present with various psychiatric symptomology (Tuten et al., 2011). Furthermore, although psychiatric comorbidity did not significantly predict the third neonatal outcome (NAS treatment), the comparable results across neonatal outcomes in the present study are consistent with research that has demonstrated links between comprehensive care and favorable neonatal outcomes (Jansson et al., 2012; Jones et al., 2011; Tuten et al., 2011).
In terms of maternal outcomes, psychiatric comorbidity was not a significant predictor of maternal length of hospital stay. This finding is consistent with previous research (Tuten et al., 2011) that found no differences in maternal length of hospital stay among pregnant women with SUDs with regard to their psychiatric diagnoses. In addition, this finding suggests that comprehensive care for pregnant women with psychiatric comorbid SUDs would reduce longer hospital lengths of stay (Maeda et al., 2014). Positive urine toxicology results at delivery in the present study were not significantly different among groups, consistent with previous investigations involving pregnant women with SUDs receiving comprehensive care treatment (Jansson et al., 1996; Jones et al., 2011; Krans et al., 2018).
Although significant differences were observed among women with psychiatric comorbidity in terms of their sociodemographic and population characteristics, these did not have an impact on outcomes. The prevalence of historic homelessness and any emotional/physical/sexual abuse in the present study reflects gender disparity among persons with SUDs (Covington, 2008; Tuten et al., 2003), and is representative of pregnant women with SUDs who present to treatment (Brown et al., 2012; Tuten et al., 2003). It is likely that prevalence rates of historic homelessness and abuse reported by women with psychiatric comorbidity in the present study reflects a relationship between these two characteristics (Tuten et al., 2004) that does not impede engagement in comprehensive care. In addition, more women with psychiatric comorbidity in the present investigation were White and younger, pointing to the need for researchers to consider cultural and possibly developmental factors that impact women’s prenatal experience.
When considering implications for practice, additional factors to examine are state policies and treatment availability regarding substance use during pregnancy. Policies for this population vary extensively, from mandatory testing and reporting to incarceration for child abuse (Guttmacher Institute, 2021). These women are categorized as high-risk clients requiring specialized treatment using a multi-professional approach (World Health Organization, 2016) and yet some policies may inadvertently create additional barriers to accessing this care. Furthermore, specialized treatment offering comprehensive care is in limited supply. While pregnant women with SUDs may enter women-only treatment programs or gain priority access in mixed-gender facilities, these resources may fail to adequately address their complex needs. In addition to providing prenatal care and SUD treatment, comprehensive care programs may also include case management services, childcare, pediatric care, social services, and transportation all at a single location.
It is also important to consider research regarding the efficacy of comprehensive care programs for pregnant women with SUDs is limited in part due to high attrition rates. Attrition is likely influenced by ecological factors such as lack of childcare to enable treatment attendance, lack of transportation, and possible discouragement to attend treatment by a partner or spouse who is using substances (Ashley et al., 2003; Jackson & Shannon, 2016; Jansson et al., 1996; Jones & Kaltenbach, 2013; Meixner et al., 2016). Internal barriers related to attrition include one’s shame and guilt regarding substance use, fear of losing child custody when entering treatment or upon delivery, and the internalization of social stigma related to substance use among pregnant and parenting women (Ashley et al., 2003; Finkelstein, 1996; Haller et al., 2003; Howard, 2015; Jackson & Shannon, 2016; Meixner et al., 2016). These influences were not assessed in the present study and should be considered when evaluating the impact of comprehensive care programs for women with SUDs who have psychiatric comorbidity.
Although a comparative analysis of pregnant women with SUDs might help explain the effectiveness of comprehensive care programs for those who have psychiatric comorbidity, there are some limitations in the present study. Medication use was not controlled and might have had an impact on psychiatric severity. Longer (e.g., 3 months) and repeated follow-up assessment intervals would help us better understand the impact of treatment related to neonatal development, whereas multisite clinical trials might identify key treatment components across various comprehensive care programs. Although some missing data were observed with comparable effects between groups, the inclusion of ecological and individual barriers to treatment might have helped us identify missing data attributes. Finally, attrition most likely had an effect on our sample size that could limit generalizability of our findings. Nonetheless, results of the present investigation have policy and research implications, suggesting that comprehensive care programs are effective for pregnant women with SUDs who have psychiatric comorbidity.
Pregnant women with SUDs and co-occurring psychiatric disorders are at high risk for negative neonatal and maternal outcomes. The present study investigated a sample of pregnant women with SUDs and psychiatric comorbidity, and our comparative design permitted analyses that yielded intriguing findings in relation to neonatal and maternal outcomes in response to receiving a comprehensive care program. In future investigations, with repeated measures over greater lengths of time, we can better understand how to improve outcomes for such a high-risk population. Clinical practice implications include a holistic focus on women-only treatment centers offering resources for both SUDs and perinatal care in a single location. Delivery care models with these components have shown improved treatment and medical plan compliance and have reported greater patient satisfaction (Greenfield et al., 2007; Meixner et al., 2016; Tarasoff et al., 2018). Overall, findings from the present study suggest comprehensive care programs are effective interventions for pregnant women recovering from SUDs who have psychiatric comorbidity.
Footnotes
Appendix A
Acknowledgements
Celeste Seger would like to thank Michelle Tuten for use of the dataset.
Author Roles
All authors contributed to the conception or design of the study or to the acquisition, analysis, or interpretation of the data. All authors drafted the manuscript, or critically revised the manuscript, and gave final approval of the version that was submitted for publication. All authors agree to be accountable for all aspects of the work, ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
