Abstract
Introduction
Cancer and Leukemia Group B (CALGB) 10403 is a pediatric-inspired treatment protocol for adolescent and young adult (AYA) acute lymphoblastic leukemia (ALL) patients. During Course V maintenance, patients receive mercaptopurine (6MP) and oral methotrexate (MTX), which require frequent laboratory monitoring and dose adjustments. The purpose of this study was to characterize the impact of pharmacist intervention on the safe implementation of Course V.
Methods
This single-center, retrospective chart review included AYA patients with B- or T-cell ALL who initiated treatment with CALGB 10403 at UNCMC between April 2014 and April 2024. The primary endpoint was the rate of pharmacist-driven 6MP and MTX holds per protocol during Course V. The key secondary endpoint was the rate of pharmacist-driven 6MP and MTX dose adjustments during Course V.
Results
All 18 patients required chemotherapy dose adjustments. Pharmacists led 6MP and MTX dose interruptions in 25 of 35 (71.4%) instances and dose modifications in 85 of the 140 (60.7%) instances. With the addition of a second pharmacist in the leukemia clinic, the monthly rate of pharmacist-led dose holds and modifications for 6MP and MTX increased by over four-fold.
Conclusion
Pharmacists played a significant role in the safe administration of 6MP and MTX, particularly with the addition of a second clinic pharmacist. This study demonstrates the value of pharmacist involvement in optimizing patient safety during treatment of ALL.
Background
Cancer and Leukemia Group B (CALGB) 10403 is a pediatric-inspired treatment protocol for adolescent and young adult (AYA) patients with acute lymphoblastic leukemia (ALL) that is comprised of five courses. 1 Clinical trials have demonstrated that maintenance improves the chance of long-term remission.2–4 Maintenance therapy consists of 2–3 years of therapy, including two oral chemotherapies, 6-mercaptopurine (6MP) and methotrexate (MTX), which require frequent laboratory monitoring and dose adjustments to maintain safety and efficacy. Dose interruptions, reductions, or escalations of 6MP and MTX are required per the CALGB 10403 protocol to maintain a target range of absolute neutrophil (ANC) and/or platelet (PLT) count. 1 Specifically, per protocol, 6MP or MTX doses should be increased for an ANC > 1.5 × 109/L maintained over six to eight consecutive weeks. 1 Alternatively, 6MP and MTX administration must be interrupted for an ANC < 0.5 × 109/L and/or PLT count < 75 × 109/L, and may only be restarted upon count recovery at a reduced dose. 1 Maintenance therapy also includes corticosteroids which come with a risk of serious adverse effects such as avascular necrosis.
The ability to maintain ANC within target range is crucial to optimizing treatment efficacy and limit complications of severe myelosuppression. The importance of dose intensity in maintenance was demonstrated in a randomized trial in which children with ALL were randomized to receive full-dosage or half-dosage chemotherapy of 4 maintenance drugs (vincristine, cyclophosphamide, methotrexate, and mercaptopurine). 5 Those who received half-dosage had shorter durations of remission and more CNS relapse. The dosing of 6MP and MTX is further complicated by large variation in bioavailability and pharmacokinetics, as well as pharmacogenetic differences, which can lead to significant interpatient variability. Loss of function variants in the genes responsible for metabolism of these drugs (i.e., TPMT and NUDT15), lead to variations in 6MP bioavailability and metabolism. 6 This interpatient variability in dosing and the complex treatment make Course V both a time- and resource-intensive regimen with significant safety concerns if not managed appropriately. Furthermore, physician compliance to recommended dosing of 6MP and methotrexate in maintenance has been shown to directly impact clinical outcomes for these patients. 7
There is minimal data addressing the impact of pharmacist intervention specifically in ALL patients. A previous study by Shah et al. evaluating real world management of patients receiving Course V of CALGB 10403 identified that 92% of patients required 6MP and MTX dose modifications due to adverse events, with each patient requiring up to 14 dose interruptions and 13 dose escalations throughout Course V. 8 Shah et al. also identified that 6MP and MTX dose adjustments in their study varied from that recommended by the CALGB 10403 protocol, suggesting the need for enhanced monitoring and clinician engagement to minimize protocol discrepancies during this treatment course. 8 Despite highlighting the overall high rates of required, per protocol dose adjustments and significant toxicities that can occur during Course V, this study did not explicitly characterize pharmacist involvement in the care of these patients. This data gap underrepresents the work pharmacists do in clinical practice to ensure patient safety and protocol adherence throughout this complex treatment regimen. Clinical pharmacist resources may be a barrier for some institutions’ ability to maintain patient safety and protocol adherence during Course V treatment.
At the UNCMC, each hematology/oncology clinic has an embedded clinical pharmacist. These pharmacists are licensed as Clinical Pharmacist Practitioners (CPPs) and practice under a collaborative practice agreement that allows them to independently prescribe oral oncolytics and supportive care medications in agreement with a supervising physician. In efforts to optimize patient safety and protocol adherence, the pharmacists on the leukemia team currently lead management of patients receiving Course V treatment. For patients initiating Course V, pharmacists provide in-depth patient education highlighting the treatment schedule, dosing and administration, side effects, monitoring, and follow up plan. Pharmacists will typically see patients every 4 weeks at a minimum while receiving Course V. At these visits, the pharmacist will review medications, adherence, assess treatment-related adverse effects, and address any supportive care needs. Pharmacists will also review labs and determine necessary dose adjustments to 6MP and MTX based on ANC and PLT count thresholds per protocol. Due to an increasing patient volume, the growth of oral oncolytics in leukemia, and with a goal of expanding outpatient acute leukemia services, a second pharmacist was added to the leukemia team in July of 2022.
Currently available literature does not address the role of pharmacists in managing patients during Course V of CALGB 10403. The purpose of this study was to characterize the impact of pharmacist intervention on the safe implementation of Course V of the CALGB 10403 protocol.
Methods
This was a single-center, retrospective study conducted at UNCMC. The research protocol was reviewed and deemed as non-human subjects research by UNCMC's Institutional Review Board. Eligible patients were AYA patients aged greater than or equal to 18 years and diagnosed with Ph-negative or Ph-like B-cell ALL, T-cell ALL, or lymphoblastic lymphoma (LLy) who initiated Course I of CALGB 10403 between April 2014 and April 2024 under the care of adult oncologists. Patients were excluded if they had a diagnosis of mixed phenotype acute leukemia, initiated CALGB 10403 at an outside hospital, received previous ALL or LLy directed therapy prior to CALGB 10403 initiation, or were treated on a study protocol. Electronic health records (EHR) were reviewed for collection of patient baseline characteristics, oral chemotherapy treatment modifications, and pharmacist interventions including change in antimicrobial prophylaxis or supportive care measures during Course V of CALGB 10403.
The primary outcome of this study was to characterize the frequency of pharmacist-driven 6MP and MTX holds per protocol during Course V. The primary outcome was chosen as a surrogate for patient safety. The secondary outcome was the number of pharmacist-driven 6MP and MTX dose adjustments per protocol during Course V. Other exploratory outcomes include the reason for 6MP and MTX dose holds and modifications, number of pharmacist visits, number of pharmacist-led changes in antimicrobial prophylaxis and supportive care interventions, and the change in the number of interventions before and after July 2022, corresponding with the addition of another pharmacist to the outpatient leukemia team.
The patient's baseline characteristics were collected at the initiation of Course I. Patients were followed throughout the duration of Course V or until disease relapse. Baseline characteristics and all other outcomes were described using descriptive statistics. All outcomes were summarized by median (range) values for continuous variables and frequency (percentage) for categorical variables. The percent change in the number of interventions before and after July 2022 was determined by calculating the monthly rate of each intervention type before and after July 2022, and reporting the percent change. Statistical analyses were performed using R™ (2019).
Results
A total of 70 patients were identified via EHR as initiating CALGB 10403 therapy during the specified time frame. A total of 39 patients met inclusion criteria and were included in the analysis. Following the completion of Courses I-IV of CALGB 10403, 18 patients went on to initiate Course V.
Baseline characteristics are described in Table 1. In the population of patients who received Course V, the median age was 29 years old. Patients were predominantly female (61%), white (44%), English speaking (72%), and insured (67%). A total of 12 patients were diagnosed with B-cell ALL (67%) and six patients with T-cell ALL or LLy (33%). Only 17% of patients were identified as Philadelphia chromosome-like, and a majority of patients were categorized with standard risk cytogenetics (61%). TMPT and NUDT15 status was not available in 44% and 56% of patients receiving Course V, respectively. TPMT and NUDT genotyping were only assessed when dictated by the treatment protocol until March 2022 at which time the assay was added to all patients’ labs to guide 6MP dosing and monitoring. Of those with known TPMT and NUDT15 status, 50% of patients were normal TPMT metabolizers and 39% were normal NUDT15 metabolizers. A total of 12 of the 18 patients who initiated Course V proceeded to complete Course V (and therefore the entire CALGB 10403 protocol), with an average of 7.7 cycles of Course V treatment.
Baseline characteristics.

During Course V, 18 of 18 patients required 6MP or MTX dose holds or dose modifications. A total of 216 dose holds or dose modifications of 6MP and MTX were conducted across the patients. Patients that required dose holds subsequently would have had dose reductions, whereas dose increases would not require a dose hold per protocol. Five patients experienced avascular necrosis with subsequent omission of steroids. Pharmacists led 6MP and MTX dose holds in 25 of the 35 (71.4%) instances of PLT count < 75 109/L and/or ANC < 0.5 109/L, as instructed per protocol. For the other six instances of PLT count and/or ANC below threshold, 6MP and MTX dose holds were conducted by a physician or advanced practice provider (APP; nurse practitioner or physician's assistant) or not held at all. Prior to the addition of the second pharmacist in clinic, pharmacists led 11 of the 18 (61%) 6MP or MTX dose holds. Following the pharmacist addition, pharmacists led 14 of the 17 (82%) 6MP or MTX dose holds. The predominant reasons for 6MP and MTX dose holds made by both pharmacists and other providers were per protocol for myelosuppression (71.6%) or non-hematologic treatment-related toxicity (13.5%), as shown in Table 2. Treatment-related toxicities included transaminitis and hyperbilirubinemia.
Reasons for oral chemotherapy dose holds and dose modifications.
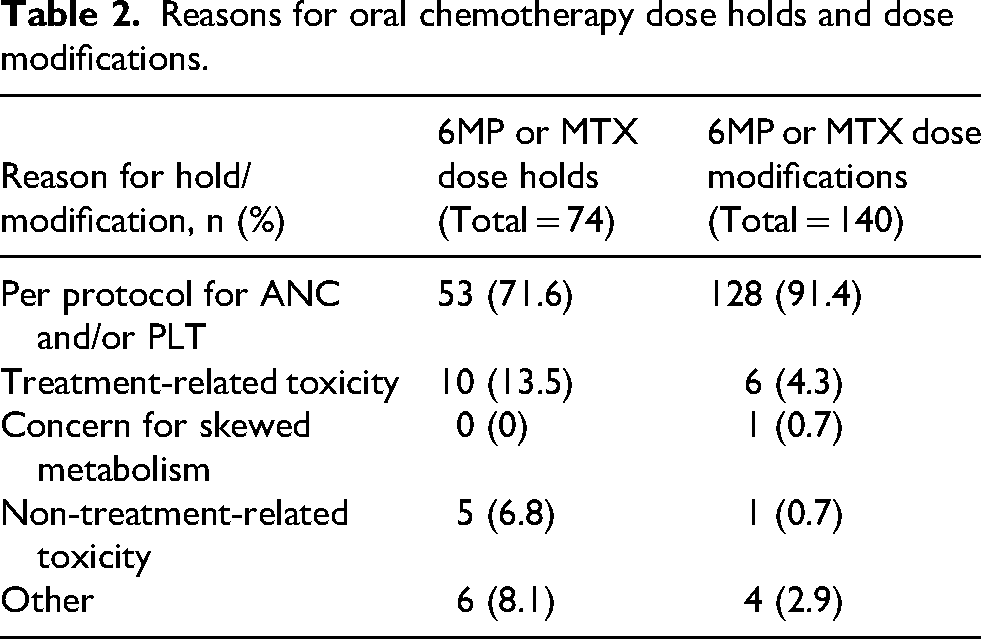
Dose modifications of 6MP and MTX were led by pharmacists in 85 of the 140 (60.7%) total dose modifications conducted. The other 55 dose modifications were conducted by physicians or APPs. The most common reasons for dose modifications were per protocol for PLT count and/or ANC outside goal range (91.4%) and treatment-related toxicity (4.3%), as shown in Table 2. Treatment related toxicities included transaminitis and hyperbilirubinemia. Other reasons for dose modifications included taking incorrect dose or lack of insurance coverage resulting in high-cost medications.
Pharmacist-driven interventions evaluated throughout the study period are listed in Table 3. A total of 216 pharmacist visits were conducted, with 108 visits during the 96-month period with one CPP in leukemia clinic and another 108 visits during the 22-month period with two CPPs. Compared to the single pharmacist model, the rate of monthly pharmacist visits increased by 445% with the addition of a second clinic CPP. The rate of 6MP and MTX dose holds increased by 555% and the rate of 6MP and MTX dose modifications increased by 407% after July 2022. A 561% increase in the monthly rate of antimicrobial prophylaxis modifications was observed, capturing initiations and discontinuations of prophylaxis based on ANC. In addition, there was a 367% increase in the monthly rate of supportive care interventions, which included management of nausea/vomiting, constipation, pain, peripheral neuropathy, and other chemotherapy-related toxicities.
Pharmacist-driven interventions over time.
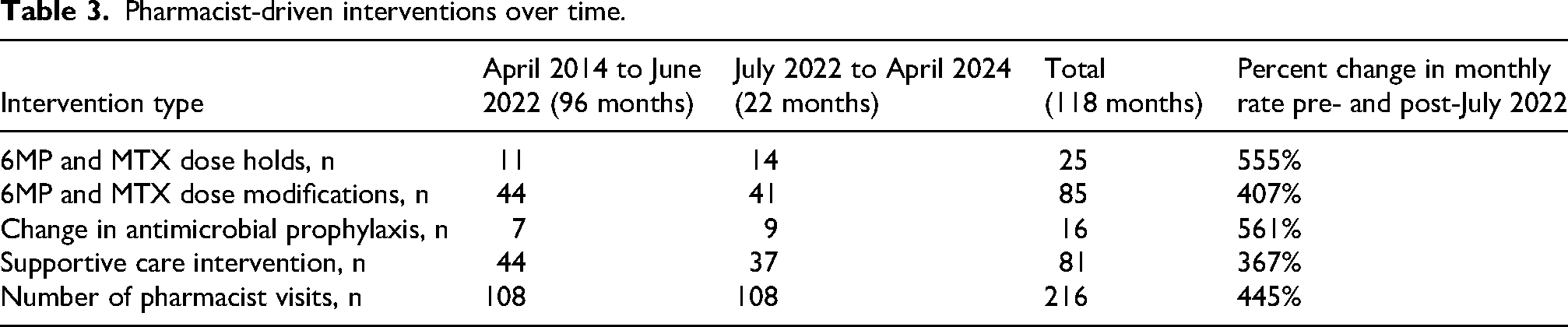
Discussion
The results of this study highlight the significant role pharmacists play in the safe and appropriate administration of 6MP and MTX during Course V of CALGB 10403. The high frequency of oral chemotherapy dose interruptions and adjustments in our study was similar to that previously reported in a real-world analysis of Course V treatment. 8 There is no previously described literature on the impact of pharmacist intervention in maintaining adherence to the 10403 protocol. In our study, pharmacists performed the majority of oral chemotherapy dose adjustments, including over 70% of 6MP and MTX per protocol dose holds and over 60% of 6MP and MTX per protocol dose modifications. The primary reason for 6MP and MTX dose holds and modifications were per protocol based on parameters for ANC and PLT values. This demonstrates the key role of pharmacists in preventing complications secondary to myelosuppression while also maintaining therapeutic dosing for each individual patient. In our study, 66.7% of patients were able to complete maintenance therapy. In the 10-year follow-up of CALGB 10403 regimen, patients who completed all maintenance therapy had a significantly lower risk of relapse than patients who did not complete maintenance. 2
In addition to ensuring patient safety, pharmacists were also heavily involved in supportive care interventions and antimicrobial prophylaxis adjustments. Pharmacists’ involvement in providing supportive care measures, especially given the wide range of toxicities associated with Course V chemotherapy, highlights their diverse skillset and value in proactively managing treatment-related toxicities and subsequently improving treatment tolerability, which is an essential factor for maintaining treatment intensity. A third of patients in our study were uninsured, which can be a barrier to care and medication adherence. Despite that, only one patient required a dose interruption or modification due to financial toxicity, likely owing to pharmacists’ active involvement in promoting patient adherence and referral to medication assistance programs. Given the known adherence and social barriers AYAs face with prolonged courses of treatment, the frequent pharmacist engagement may have helped bridge this gap in care. 9 Five patients in this study experienced avascular necrosis during maintenance, highlighting the implications of long-term steroid use. This illustrates the need for survivorship practices, which is an area pharmacists may be well-positioned to lead.
The ASCO Quality Oncology Practice Initiative (QOPI) outlines measures to maintain quality in cancer care. Vulaj and colleagues reviewed these QOPI measures for pharmacist involvement and determined that over one-third of these measures included areas of pharmacist impact, particularly those focused on patient education and management of oral chemotherapy. 10 Pharmacists are in an excellent position to provide support to the oncology care team and improve quality of care. This may be particularly true for centers that are providing care for acute leukemia, which is a high-risk disease state that requires specialized expertise.
Given the central role of pharmacists in managing dose modifications during Course V, a key secondary outcome was to evaluate the number of pharmacist-driven interventions following the addition of a second leukemia clinic CPP to characterize the impact of this additional position on patient care. Following the addition of the second pharmacist in July 2022, over four times as many pharmacist-led visits were conducted per month. Substantial increases in the monthly rate of all intervention types were also observed. The addition of a single pharmacist to the leukemia clinic team resulted in more than a doubling in the number of pharmacist visits and opportunities for pharmacist-led interventions, broadening the team's impact on patient care. Since baseline service needs were met with an initial pharmacist, the addition of the second pharmacist allowed for the development of standardized pharmacists visits and monitoring for patients on 10403. Each patient was seen at a minimum of every 4 weeks, or more as needed for lab monitoring or supportive care. These results provide justification for expanding clinical pharmacist involvement in the clinic setting patients with ALL.
Previous studies have shown the impact of clinical pharmacists in the outpatient setting on outcomes of cancer patients. One study evaluating a novel pharmacist-led oral chemotherapy program for patients with chronic myeloid leukemia demonstrated improved patient knowledge regarding oral chemotherapy, improved adherence rates, and superior major molecular responses compared to published literature, highlighting the definitive impact of clinical pharmacists in the management of oral chemotherapy-based regimens. 11 Trinidad and colleagues showed that the involvement of pharmacists in caring for leukemia and lymphoma patients in the outpatient setting resulted in over 500 clinical interventions translating to over $300,000 of cost-savings in a two-year time period. 12 Previous literature has also described the role of pharmacists in managing complex venetoclax-based regimens in both acute myeloid leukemia and chronic lymphocytic leukemia.13,14
A review by Gatwood and colleagues describe a clear benefit of integrating pharmacists into oncology care models through identifying treatment and medication-related issues and even symptom improvement for patients on cancer-directed therapy. 15 There is no literature, however, describing the role a pharmacist can have in the care of acute lymphoblastic leukemia patients. This study highlights the remarkable role a pharmacist can have for these patients. The treatment of ALL consists of intricate, multi-agent chemotherapy regimens and with the evolving treatment paradigm of ALL and the continued inclusion of drugs such as mercaptopurine and methotrexate, the role of the pharmacist in interpreting these complicated protocols and implementation of these therapies will be even more critical. 16
Knapp and colleagues reported that with the number of board-certified oncology pharmacists (BCOP) in the United States, the growing number of patients requiring cancer treatment, and predicted shortages of oncology physicians, pharmacists are positioned to bridging this gap in providing patient care. They projected that the number of BCOPs anticipated by the year 2025 could provide 2.9–4.1 million 30-min visits annually. 17 This supports our finding that the addition of a second pharmacist embedded in our leukemia clinic resulted in over four times the number of visits and could alleviate physician shortages.
There are several limitations to this study. The retrospective design of this study restricts data to that documented in the EHR and collected via manual chart review. Any missing documentation may have limited our ability to fully characterize pharmacist-led interventions. For dose holds that were made by a physician or other provider, the study design limits our ability to tell if this may have been at a time when a pharmacist was not available or if a pharmacist may have been informally consulted without any documentation in the EHR of a pharmacist visit. Additionally, this was a single center study with a small sample size of patients. When comparing data before and after the addition of a second clinical pharmacist, it is important to note the difference in duration of data collection before and after July 2022 (96 months versus 22 months), which limits direct comparisons and interpretation of the data. However, a broad date range was utilized in this study to maximize the number of AYA patients eligible for inclusion at our institution. Changes in clinical practice (i.e., evaluating TPMT and NUDT15 status at diagnosis), practicing physicians, physician preferences, and detail of EHR documentation over this 10-year study period may have also impacted the standardization of data collected and evaluated in this study. Additionally, interpretation of 6MP and MTX dose modifications according to the CALGB 10403 protocol is complex and difficult to apply to all patient cases, allowing room for clinical interpretation, which may be clinician or institution specific.
Despite these limitations, the findings of this study demonstrate the definitive impact pharmacists have on the safe implementation of Course V treatment and continues to highlight the importance of clinical pharmacists embedded in the clinic setting, especially for ALL. This study also demonstrates the complexities of this protocol and burden of toxicity of the CALGB 10403 regimen. The real-world efficacy and safety of the patients treated in our study will be published separately. Additional research is needed to better understand pharmacist impact during Course V on survival outcomes of patients treated with CALGB 10403, which could further demonstrate the instrumental role of pharmacists in the clinic setting during maintenance therapy and in other ALL regimens.
Footnotes
Acknowledgements
N/A
Ethical approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Author contributions
TD, JA, KMB, SMC, HH, ABC, LK, and BM designed study; BP, JA, HH, TD, CA and BL collected data; WZ completed statistical data analysis; BP, HH, CA, and BL interpreted the results; BP and CA drafted the manuscript; all authors reviewed and approved the final version of the manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Data availability statement
Data utilized for this study is not currently publicly available but may be provided upon reasonable request.
