Abstract
Background
In acute lymphoblastic leukemia (ALL), pegaspargase is included in the backbone of numerous chemotherapy regimens. Treatment-related toxicities can be substantial, often resulting in treatment delays, dose reductions, or early discontinuation. This study aimed to evaluate whether differences in efficacy, safety, and economic outcomes exist between standard dosing and modified approaches involving dose-reduced and delayed administration of pegaspargase.
Methods
This retrospective, single-center study evaluated hospitalized adult patients who received pegaspargase as part of CALGB 10403 induction course I. Patients were stratified into two groups: 1) full pegaspargase dose 2500 units/m2 and standard administration on day 4 (Early PEG group), or 2) dose reduced pegaspargase 1000 units/m2 and delayed administration on day 15 (Delayed PEG group).
Results
Eight patients (27%) were treated with a reduced dose of pegaspargase on day 15 of induction (Delayed PEG). Median age of the study population was 27 years, and median BMI was 29.6 kg/m2. The majority of patients were male (60%), Hispanic (53%), and had a diagnosis of Philadelphia chromosome negative B-ALL (70%). Incidence of grade 3 or higher toxicities was not significantly different between groups. MRD status at the end of induction was similar between groups [Early PEG 12 (55%) vs Delayed PEG 4 (50%), p = 0.51]. Economic outcomes within 30 days were also similar.
Conclusion
Our study demonstrates comparable incidence of high-grade toxicities and MRD negative status, suggesting that concomitant dose reduction and delay of pegaspargase administration during CALGB 10403 induction does not significantly impact effectiveness, safety, or economic outcomes.
Introduction
L-asparaginase is a bacterial-derived enzyme that catalyzes the degradation of serum asparagine into L-aspartic acid and ammonia.1,2 There are currently two available formulations with differences existing in their derived bacterial species, pharmacokinetics, and toxicity profiles: pegylated Escherichia coli-asparaginase (pegaspargase and calaspargase pegol-mknl) and Erwinia recombinant asparaginase.1,3 Pegylation extends the plasma half-life of the enzyme, enhancing its pharmacokinetic profile; this modification offers advantages including decreased administration frequency, lower incidence of hypersensitivity reactions, and comparable therapeutic efficacy.4,5
In acute lymphoblastic leukemia (ALL), pegaspargase has long been the backbone of many pediatric-based chemotherapy regimens, with expanding usage to include the treatment of adults. 4 Adapted from the Children's Oncology Group study AALL0232, CALGB 10403 is a recommended regimen for adult and young adolescent (AYA) patients who qualify for intensive chemotherapy.4,6–8 Due to its mechanism of action, pegaspargase has a distinct and extensive toxicity profile including hypersensitivity reactions, hypertriglyceridemia, pancreatitis, coagulopathy, and most notably hepatotoxicity which is observed at a higher rate in adults compared to pediatric patients.2,3,7 Despite their reversibility, these toxicities may necessitate treatment delays or omissions, which can compromise overall treatment efficacy and lead to inferior outcomes.3,4,9,10
Institutions utilize different approaches to reduce the incidence of toxicities, and corresponding treatment delays, with asparagine depleting therapies. Examples of strategies during induction include reducing pegaspargase dose (from 2500 units/m2 to 1000 units/m2) or delaying pegaspargase administration (from day 4 to day 15 of induction).4,8,11,12 However, there is limited data evaluating the effectiveness and safety of combining these strategies. The adaptation of delayed pegaspargase has more recently been validated with the E1910 protocol with a dose of 2000 units/m2 on day 18 of induction. 13 Despite this, wide variation continues to exist even within regimens amongst providers.
At UC San Diego Health, we utilize a combined approach of reducing the dose and delaying administration of pegaspargase with CALGB 10403 induction in our AYA patients. The San Diego AYA population is largely represented by Hispanic patients characterized by higher rates of obesity and nonalcoholic steatohepatitis (NASH).14,15 This population is possibly more prone to hepatotoxicity and worse outcomes, given the higher incidence of Philadelpha chromosome-like (Ph-like) cytogenetics in this ethnic group. 3 We hypothesized that delayed administration and dose reduction of pegaspargase is associated with less frequent treatment delays and lower incidence of high grade toxicities, without significantly affecting survival and response rates. The economic implications of limiting pegaspargase-related toxicities have not been evaluated but may manifest as decreased need for costly supportive care such as antithrombin III and cryoprecipitate repletion. Other significant economic outcomes or inpatient quality metrics impacted could be reduced need for intensive care unit (ICU) admission, re-hospitalization within 30 days, and length of hospitalization. The objective of this study is to assess if differences in efficacy, safety, and economic outcomes exist between standard and dose reduced, delayed pegaspargase.
Methods
A retrospective cohort study was performed among adult patients who received pegaspargase in the inpatient setting at UC San Diego Health between January 1, 2011 and August 1, 2024. Patients were eligible if they were at least 18 years of age and received pegaspargase during their CALGB 10403 induction cycle (course I) and extended induction cycle (course Ia), if applicable. Patients were excluded from the study if they were 1) enrolled in a clinical trial or received an induction regimen other than CALGB 10403; 2) incarcerated; or 3) pregnant.
Patients were stratified into two groups: 1) those who received the full pegaspargase dose of 2500 units/m2 with standard administration on day 4 (Early PEG), and 2) those who received a reduced dose of pegaspargase 1000 units/m2 with delayed administration on day 15 (Delayed PEG). Both groups had a maximum allowable pegaspargase dose of 3750 units. The toxicities investigated include hypertriglyceridemia, pancreatitis, hepatotoxicity, hypofibrinogenemia, significant bleeding, antithrombin III (ATIII) deficiency, and thrombotic events. Only pegaspargase was included due to its frequent usage compared to the other commercially available asparaginase products. The primary endpoint for efficacy was evidence of minimal residual disease (MRD) at the end of induction, defined as <0.01% leukemic cells in bone marrow as assessed on flow cytometry based on the reporting assay at the time. Patients were classified into cytogenetic risk groups based on the criteria utilized in the CALGB 19802 study, consistent with the CALGB 10403 protocol. 15 Adverse events were graded according to the National Cancer Institute (NCI) Common Toxicity Criteria for Adverse Events (CTCAE) version 5.0.
Statistical analysis
Statistical analyses were performed in SPSS v29.0 (IBM Corp. Armonk, NY). Categorical variables were compared between early vs delayed PEG groups using the chi-square or Fisher's exact tests, and continuous variables were compared using the Mann-Whitney U test.
Results
A total of 30 patients met inclusion criteria during the study period. Of these, 22 patients received the standard full dose of pegaspargase (2500 units/m2) administered on day 4 of CALGB 10403 induction (Early PEG). Eight patients received a reduced dose of pegaspargase (1000 units/m2) administered on day 15 of induction (Delayed PEG). Notably, one patient received a full dose of pegaspargase (2500 units/m2) with delayed administration on day 15 before institutional standardization; this patient was evaluated in the Delayed PEG group.
Baseline characteristics were comparable between the Early PEG and Delayed PEG groups (Table 1). The median age of the cohort was 27 years. The majority of patients were male (60%), Hispanic (53%), diagnosed with Ph-like or Ph-negative B-ALL (70%), and classified as intermediate risk cytogenetics (67%). The median BMI was 29.6 kg/m2 (interquartile range (IQR) = 25.5–38.7 kg/m2). A notable difference between groups was observed in median body surface area (BSA), which was significantly higher in the delayed PEG group compared to the Early PEG group (Early PEG: 2.09 m2, (IQR 1.88–2.43 m2) versus Delayed PEG: 2.19 m2, (IQR 2.00–2.55 m2), p = 0.001). The use of anticoagulation for venous thromboembolism prophylaxis at the time of pegaspargase administration did not differ significantly; the majority of patients (57%) received prophylactic enoxaparin concurrently with pegaspargase initiation.
Baseline demographics.
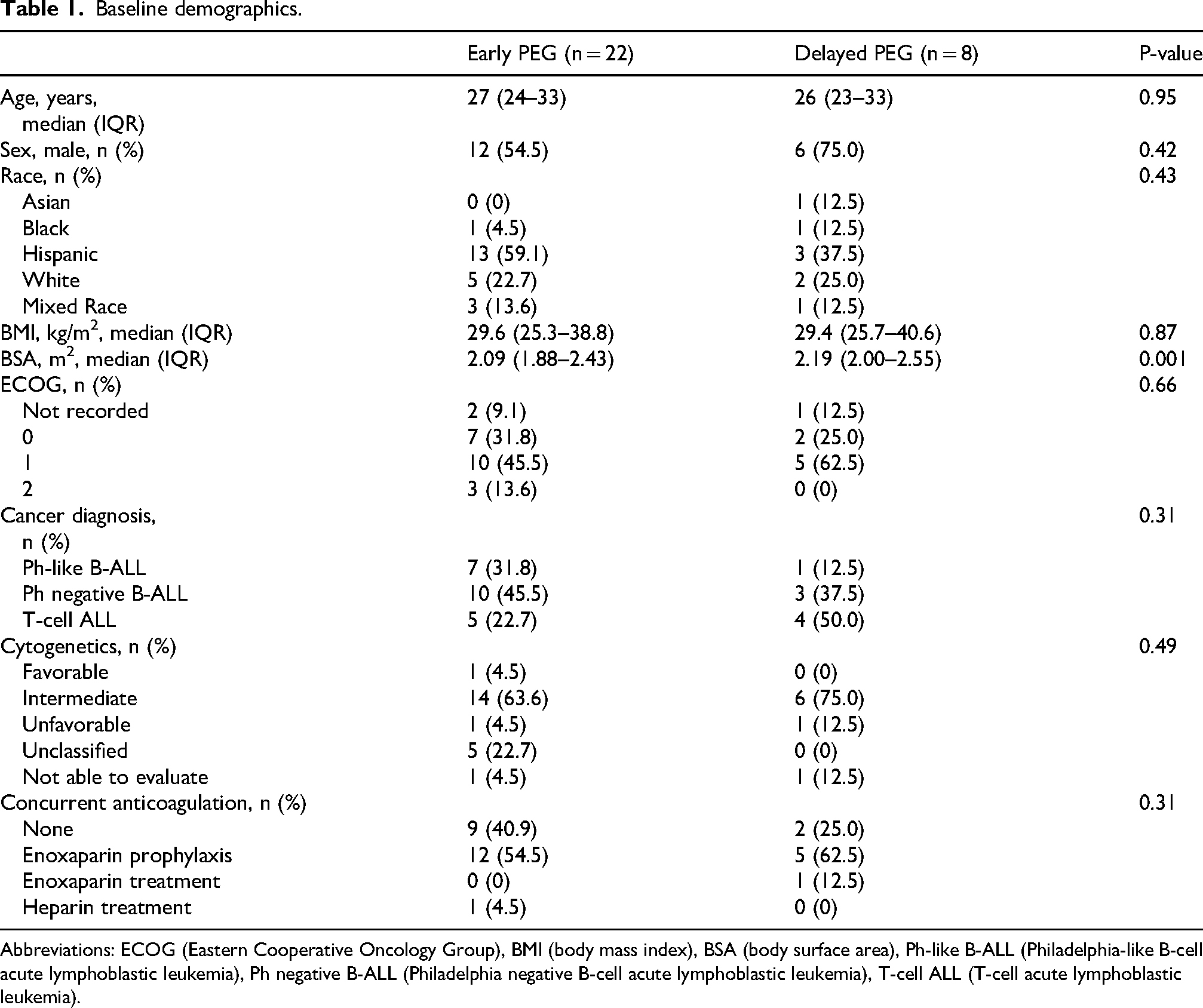
Abbreviations: ECOG (Eastern Cooperative Oncology Group), BMI (body mass index), BSA (body surface area), Ph-like B-ALL (Philadelphia-like B-cell acute lymphoblastic leukemia), Ph negative B-ALL (Philadelphia negative B-cell acute lymphoblastic leukemia), T-cell ALL (T-cell acute lymphoblastic leukemia).
Baseline laboratory values were assessed within the two-week period preceding pegaspargase administration up to the day of administration and showed no significant differences between the Early PEG and Delayed PEG groups (Table 2). Most patients presented with baseline laboratory values within normal limits; the most common abnormalities were transaminitis and/or hyperbilirubinemia (30%) with one patient who presented with baseline grade 4 hepatic dysfunction in the delayed PEG group. Baseline measurements of triglycerides, amylase, lipase, and antithrombin III were not routinely obtained. Cryoprecipitate supplementation was administered per physician discretion to three patients (10%) with baseline hypofibrinogenemia. Two patients in the Early PEG group had significant bleeding at baseline (9%), and one patient in the early PEG group had a prior history of pulmonary embolism.
Baseline laboratory values.
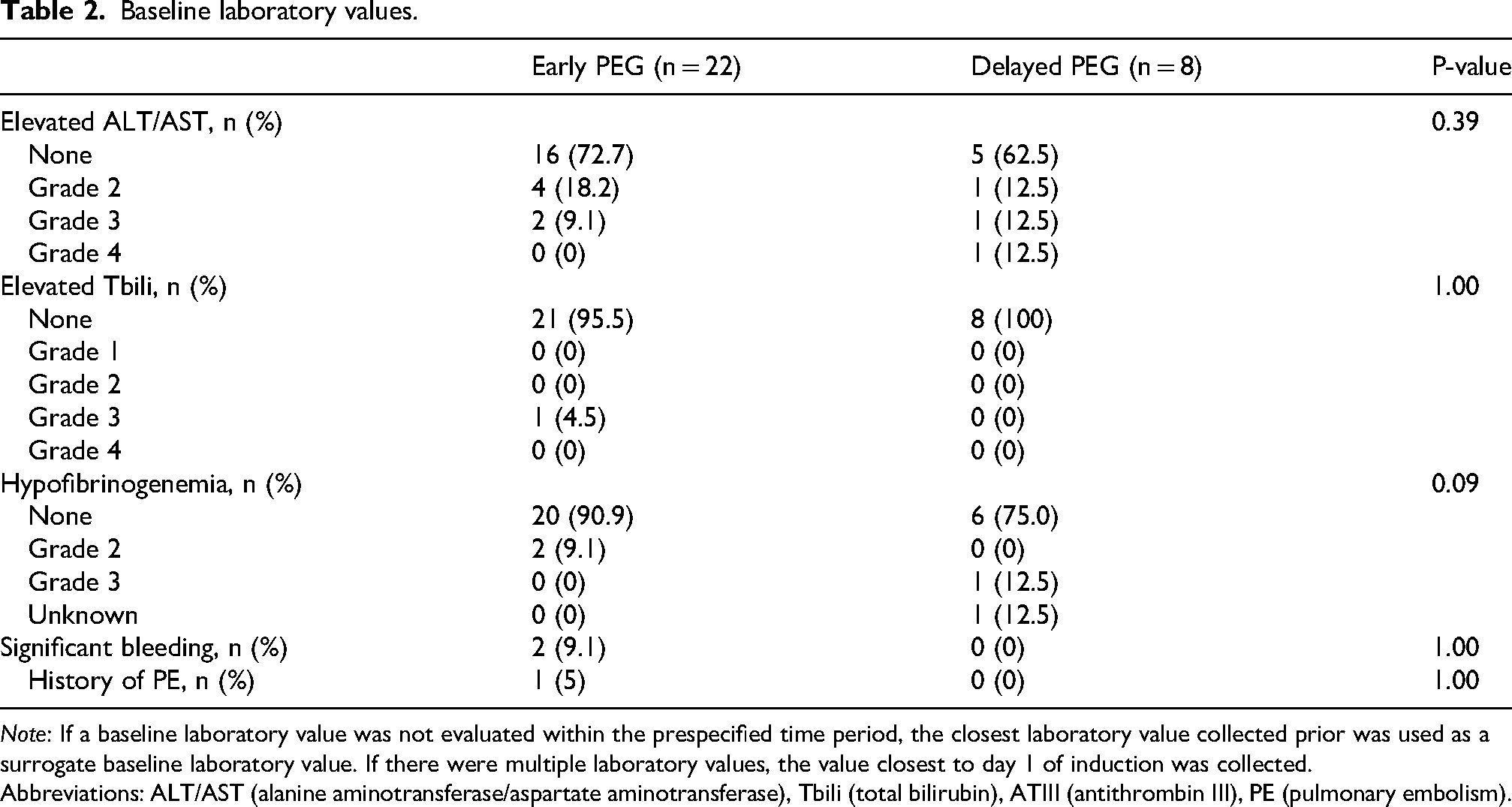
Note: If a baseline laboratory value was not evaluated within the prespecified time period, the closest laboratory value collected prior was used as a surrogate baseline laboratory value. If there were multiple laboratory values, the value closest to day 1 of induction was collected. Abbreviations: ALT/AST (alanine aminotransferase/aspartate aminotransferase), Tbili (total bilirubin), ATIII (antithrombin III), PE (pulmonary embolism).
Safety and clinical outcomes are reported in Table 3. There were no statistically significant differences in the incidence of grade ≥3 toxicities between groups. A total of 64 toxicities were observed, with the majority occurring in the Early PEG cohort (81%). Hypofibrinogenemia and antithrombin III deficiency were the most commonly observed toxicities in both groups. The median number of antithrombin III doses did not differ significantly between groups (Early PEG: 3 doses, (IQR 0–4 doses) versus Delayed PEG: 1 dose, (IQR 0–2 doses), p = 0.12). Similarly, the median time to first pegaspargase toxicity was comparable (Early PEG: 5 days (IQR 2–9 days) versus Delayed PEG: 5 days (IQR 3–7 days), p = 0.84). Ninety-day survival did not differ significantly between cohorts.
Safety and clinical outcomes (within cycle I induction).
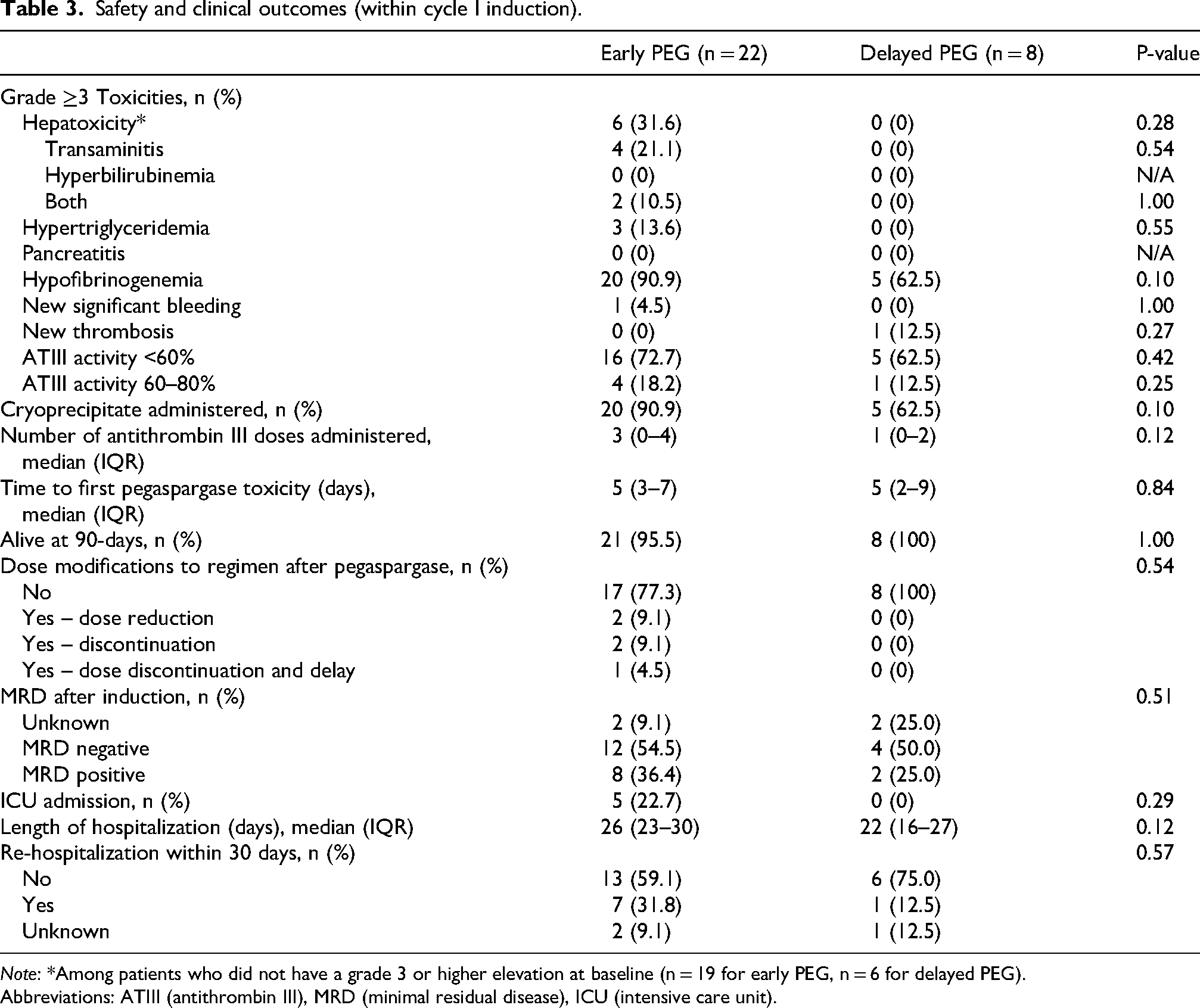
Note: *Among patients who did not have a grade 3 or higher elevation at baseline (n = 19 for early PEG, n = 6 for delayed PEG).
Abbreviations: ATIII (antithrombin III), MRD (minimal residual disease), ICU (intensive care unit).
Toxicity-related modifications including dose reduction, treatment delay, or discontinuation were required in 5 patients; all instances occurred in the Early PEG group. The majority of patients in both groups achieved MRD-negative status following induction, with no significant differences detected. One patient required extended induction course Ia, and their MRD status was not available at the end of that period. Rates of ICU admission, length of hospitalization, and 30-day readmission were similar between groups.
Discussion
While previous studies have examined the effects of either delayed administration or dose reduction of pegaspargase individually, data supporting the use of a dual-modality approach remains limited.12,16,17 In this retrospective study of 30 patients who received pegaspargase as part of CALGB 10403 induction, the combined strategy of dose reduction and delayed administration of pegaspargase did not lead to significant differences in safety or efficacy outcomes.
Effective management of pegaspargase-associated toxicities is essential to limiting early treatment discontinuation and optimizing long-term outcomes in patients with ALL. 10 Tinajero et al. evaluated delayed pegaspargase administration during induction and reported a reduction in high-grade hepatotoxicity without compromising complete remission rates or achievement of MRD. 12 Our findings are consistent with these results and expand upon the adverse event profile by including additional toxicities such as hypofibrinogenemia and antithrombin III deficiency. Tiao et al. examined a capped dosing strategy of pegaspargase 3750 units in an elderly patient population, reporting no significant difference in adverse event rates, though the overall incidence of toxicities was low in that cohort. 16 No differences in overall response rate, complete response, or MRD negativity were reported between groups. 16 In our study, after excluding patients with baseline aberrations, the incidence of grade ≥3 hepatoxicity was 24%, aligning with the rates reported in both prior studies.12,16 Notably, the median time to first pegaspargase-related toxicity was 5 days in both groups, which is shorter than previously reported. 16 This discrepancy may be attributed to the predominance of hypofibrinogenemia and antithrombin III deficiency in our cohort, toxicities that may manifest earlier than hepatic or other organ dysfunction.18–20 Variability in dose intensity and induction regimens may also contribute to the observed differences across studies since our inclusion criteria restricted induction to the CALGB 10403 regimen.12,16
The combined strategy of reducing the pegaspargase dose to 1000 units/m2 and delaying its administration to day 15, as implemented in this study, may raise concerns regarding potential reductions in treatment efficacy. 7 MRD negativity at the end of induction was selected as the primary efficacy endpoint given its established prognostic indicator for remission, long-term survival, and transplant outcomes.7,21 In our cohort, approximately 50% of patients achieved MRD negativity at the conclusion of CALGB 10403 induction, with no significant difference observed between groups receiving full-dose or modified pegaspargase administration. These findings align with prior studies that evaluated either full-dose or delayed administration approaches during induction, including more recent data from the E1910 trial.7,12,13 Acute mortality, defined as within 90 days from the start of induction, was low in both groups and did not differ significantly, consistent with previously published reports. 12
The secondary objective of this study was to assess the economic impact of delayed and dose-reduced pegaspargase administration within our institution. Cost-related factors evaluated included length of hospitalization, ICU admission, hospital readmission, and incidence of significant toxicities which may prolong hospitalization or necessitate treatment modifications or delays. No statistically significant differences were observed between the Early PEG and Delayed PEG groups for any of these outcomes. However, the limited sample size may have reduced the power to detect a meaningful difference if one existed. Despite the absence of significant findings, important economic considerations remain. For example, each additional day of hospitalization is estimated to cost approximately $5,000, and our approach may influence the need for supportive interventions such as cryoprecipitate and antithrombin III repletion.17,22 To date, no studies specifically address the cost-utility of a combined delayed and reduced-dose pegaspargase strategy. The data reported here provides early institutional insight and continues to support the feasibility of this approach.
Several limitations should be considered when interpreting these findings. This was a retrospective cohort study of 30 patients, and the inherent limitations of this study design including the potential for selection bias, confounding, and limited generalizability are applicable. Additionally, variations in institutional practice patterns over time introduced some inconsistencies. Two patients included in the analysis had deviations from the predefined group stratifications. One patient, who was treated prior to the implementation of the institutional maximum pegaspargase dose (3750 units in 2012), received 4725 units. Excluding this patient did not meaningfully alter the study's findings. Before standardization of the reduced-dose, delayed administration strategy in February 2023, three patients received pegaspargase at the discretion of the treating physician; one of these patients received the maximum dose of 3750 units (based on a weight-based dose of 2500 units/m2) on day 15. These patients did not contribute to the primary, safety, clinical, or economic outcomes of interest, and were retained in the analysis to preserve statistical power and increase the likelihood of detecting a true difference, if one existed.
It is also important to acknowledge that several baseline laboratory values, most notably antithrombin III activity and triglyceride levels, were unavailable for a subset of patients. In response, institutional protocols have been updated to ensure these values are routinely obtained prior to pegaspargase administration. Additionally, the timing of pegaspargase administration has been standardized to noon to permit appropriate monitoring and management in the event of an infusion-related anaphylactic reaction. Only one patient in this cohort experienced an anaphylactic reaction, consistent with the low incidence reported in the literature during first exposure. 23
Although statistical significance was not reached, the absence of meaningful differences in real-world effectiveness between the two pegaspargase administration strategies is reassuring. Most toxicities were observed in the Early PEG group, while no high-grade hepatotoxicity or hypertriglyceridemia occurred in the Delayed PEG group. These clinical trends suggest that the combined approach of dose reduction and delayed administration may mitigate toxicity without compromising efficacy. This strategy is particularly relevant in AYA Hispanic patients, a population known to be at increased risk for poor outcomes due to factors such as obesity, NASH, and Ph-like ALL.3,14,15,24 Despite the small sample size, these high-risk features were generally well-represented in our cohort. Encouraging trends in economic outcomes were also present such as a reduction in cryoprecipitate repletion in the Delayed PEG group. No chemotherapy modifications were required in the Delayed PEG group and a reduction in median hospital length of stay by four days following pegasparagase administration, while not statistically significant, is economically meaningful given the high daily cost of inpatient care. These findings support the feasibility of this approach and underscore the need for further investigation in larger, prospective studies, particularly those focused on high-risk AYA Hispanic populations where this strategy may yield substantial clinical and economic benefits.
Conclusion
Our retrospective, single-center study suggests that the combined approach of dose reduction and delayed administration of pegaspargase during CALGB 10403 induction does not significantly compromise overall effectiveness or safety. The comparable rates of MRD negativity and grade ≥3 toxicities between groups provide real-world evidence to support the feasibility of this modified strategy. While no statistically significant differences were observed in fiscal outcomes, notable trends toward reduced high-grade toxicities and shorter hospital stays highlight the potential clinical and economic benefits. These findings warrant further investigation in larger, prospective studies to validate the impact of this approach, particularly in high-risk populations.
Footnotes
Acknowledgements
Not applicable
Ethical approval and informed consent statements
This study received ethical approval from the UC San Diego IRB (Approval #810831) on August 02, 2024. This is an IRB-approved retrospective study, all patient information was de-identified and patient consent was not required. Patient data will not be shared with third parties.
Author contribution statement
SV: Project administration, Investigation, Data curation, Writing—Original draft preparation, Writing–Review & Editing, JM: Writing—Review & Editing, KM: Writing–Review & Editing, NP: Validation, Formal analysis, Resources, TT: Writing–Review & Editing, YA: Conceptualization, Methodology, Supervision, Writing–Review & Editing. All authors reviewed and approved the final version of the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Tanaka: Consulting or Advisory Role: Bristol Myers Squibb and Servier Pharmaceuticals; Research Funding: Function Oncology
Data availability statement
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
