Abstract
Introduction
Exposure of healthcare workers to hazardous drugs may result in adverse health effects underscoring the importance of validating working procedures and safety precautions to minimise the risk. The objective was to monitor environmental contamination caused by the hazardous drug workflow: from drug vials, compounding process, to patient administration.
Methods
Surface wipe samples were collected from potentially contaminated surfaces in the compounding department and in the administration department. The outside of drug vials, compounded syringes, bags, elastomeric pumps, and gloves used by the nurses for administration were also monitored. Stationary air samples were collected near the isolators and above the bench top. Personal air samples were collected from pharmacy technicians, pharmacists, and nurses. Monitoring was performed in three trials during two-months. Samples were analysed for cyclophosphamide, 5-fluorouracil, docetaxel, and paclitaxel using liquid chromatography tandem mass spectrometry.
Results
Contamination was mainly found for 5-fluorouracil and cyclophosphamide on isolator surfaces, bench top, trays, and compounded products. Lower levels of contamination were measured in the administration department on trays, trolley arms and gloves of the nurses. Paclitaxel and docetaxel were incidentally detected. Air contamination was found for paclitaxel in the compounding department in one trial, and 5-fluorouracil was detected once in front of an isolator. Docetaxel was found in one air sample of a nurse.
Conclusions
Contamination was mainly found for 5-fluorouracil and cyclophosphamide on the products compounded in the isolators. Contamination was further spread along the workflow towards the administration department causing surfaces in between being contaminated too.
Introduction
Exposure of healthcare workers to hazardous drugs may cause adverse health effects such as cancer and reproductive effects.1–5 Guidelines and recommendations have been implemented to prevent healthcare workers from being exposed.6–9 These include organisational measures such as protocols, education and training, technical measures such as clean-rooms, biological safety cabinets, isolators, and devices, as well as Personal Protective Equipment (PPE). However, the potential risk of being exposed remains as surface wipe sampling studies still show environmental contamination with hazardous drugs in the healthcare sector where these drugs are compounded and administered to patients.10,11
The objective of this study was to measure surface and air contamination with hazardous drugs in the pharmacy aseptic unit and in the day care oncology unit within the Edinburgh Cancer Centre, part of the Western General Hospital in Edinburgh, UK. Monitoring was repeated three times over a period of two months. In contrast with other published studies the whole workflow from compounding of the drugs until administration to the patient was monitored. This includes contamination on the outside of hazardous drugs vials, on compounded and outsourced products such as syringes, infusion bags, and elastomeric pumps, work surfaces, inside transport bags, trays, trolley handles, and on gloves of the nurses. In addition, environmental air sampling was included to evaluate potential exposure of the healthcare workers by inhalation, an exposure route that is often neglected. Although dermal uptake is considered as the main exposure route, it cannot be excluded that exposure via inhalation of particles and vapours occur. The combination of surface wipe sampling and air monitoring has not been frequently investigated and most studies are outdated.12–15
Methods
The study was performed by Exposure Control Sweden AB.
Monitoring site
The study was performed between October 18th and December 12th, 2023, in the Western General Hospital in Edinburgh, UK. Two departments were involved: a recently refurbished pharmacy aseptic unit where hazardous drugs are compounded, and adjacent a day care chemotherapy unit where hazardous drugs are administered to patients. Sampling in both departments was performed in three trials with a 4-week interval in between. Annually about 15,000 doses of hazardous drugs are issued from the pharmacy aseptic unit for administration in the day case oncology unit. Approximately 5500 of these are externally compounded, with the remainder compounded in-house within the pharmacy aseptic unit.
Workflow
The pharmacy aseptic unit is an unlicensed facility, operating under a Section 10 exemption to the Medicines Act 1968. It consists of three main rooms, with associated change rooms, under controlled ventilation creating a clean room environment that is compliant with EU Good Manufacturing Practice (GMP) and UK Department of Health requirements. To access the rooms, clean room clothing requirements should be fulfilled by the pharmacy staff and Personal Protective Equipment (PPE) appropriate to the environment always worn.
In the clean support room (EU GMP Grade D), product assembly takes place based on aseptic worksheets specific to the patient and their drug regime. Hazardous drug vials as well as infusion bags are stored in secondary packaging at the required temperature. For each compounding, the patient specific worksheet and all necessary materials such as syringes, infusion bags and the hazardous drug vials are placed on a plastic tray. Dust caps and secondary packaging are removed from infusion bags and vials, and the tray and contents undergoes the first stage (wipe and spray) disinfection using a sporicidal wipe (Contec SAT wipes with Prochlor) followed by a 70% industrial methylated spirit/denatured ethanol (IMS) spray, before being transferred via an interlocked pass-through hatch into the pharmacy preparation room for compounding.
In the clean preparation room (EU GMP Grade C), four negative pressure isolators are installed. Depending on the workload, one or more isolators are in function. During a daily shift, each isolator is used by only one pharmacy technician. The tray and all materials within undergo a further disinfection step (wipe and spray with 70% IMS) during transfer into the entrance hatch of the isolator. Compounding is performed by pharmacy technicians using needle and syringe technique (manual pressure equalisation). When the product (infusion bag, syringe, or elastomeric pump) is ready, it is placed back in the original plastic tray together with the worksheet and all the materials that were used. A pharmacy support worker removes the tray from the isolator exit hatch and passes through a second interlocked pass-through hatch to the pharmacy aseptic checking room.
In the pharmacy aseptic checking room, a final check and release is performed by a pharmacist. During the final check, the products are labelled and packed into clean, clear plastic mini bags (outer bags), with an additional protect from light bag for light sensitive product. Next, the compounded product is transported in a yellow transport bag and delivered to the day care oncology unit for administration to the patients.
A proportion of the infusion bags and syringes issued for administration are outsourced and purchased in pre-compounded, ready to administer fixed dosages. These are stored in the pharmacy aseptic checking room at room temperature or in the fridge depending on the requirements of the compounding company. After a check by a pharmacist against a patient specific order, they are also labelled, packed, and transported in a yellow transport bag following a comparable process to that followed for in-house compounded products.
In the day care oncology unit, compounded products, and all other materials needed to perform an administration are put on a separate tray and are checked by two nurses before administration to the patient. The tray is placed on a trolley.
Study design
The aim of the study was to monitor environmental contamination during the whole workflow: from drug vials, compounding process, prepared products to administration to the patients. Environmental contamination was measured by surface wipe sampling and personal and stationary air sampling. Workers involved were pharmacy technicians (compounding), pharmacists (check), and nurses (administration).
As different drugs were compounded and administered, and practically not all can be measured, a selection was made representing in-house compounded and outsourced products as well as different type of products (infusion bags, syringes and elastomeric pumps). This has resulted in the selection of 5-fluorouracil (5FU), cyclophosphamide (CP), docetaxel (DOC), and paclitaxel (PAC) as hazardous drugs to be monitored. Over the study period, there were 327 administrations of PAC, 166 of 5FU, 164 of CP, and 93 of DOC.
Sample collection compounding department
For each of the drugs, surface wipe samples were taken from the outside of four drug vials and four outsourced infusion bags or syringes. In the clean preparation room, surface wipe samples were collected from the working surfaces of the isolators in use. In the pharmacy aseptic checking room, surface wipe samples were taken from the outside of four to nine compounded syringes, elastomeric pumps, and infusion bags. After check and packing, wipe samples were taken from the corresponding four outer bags, and from the top side of the four yellow trays used for compounding. The bench top used by the pharmacist to check and label the compounded products was also monitored for contamination. Stationary air samples were collected in the clean preparation room in front of the exit hatch of the isolators in function, and on the bench top in the pharmacy aseptic checking room where the compounded products are labelled and checked. Personal air samples were collected from the pharmacists performing the checks.
Sample collection administration department
Surface wipe samples were collected from the inside of four yellow transport bags sent by the pharmacy department and containing compounded products. Yellow transport bags were selected at random but should have contained at least one compounded product of the four drugs monitored. Surface wipe samples were taken twice from the topside of three or four trays: after cleaning and before use, and after the administration when all materials are discarded. Next, wipe samples were taken from both arms of the trolley, and the gloves of the nurses used for the administration of the drugs were collected. Personal air samples were collected from nurses performing administration of hazardous drugs including the four drugs monitored.
Surface wipe sampling procedure and glove collection
Cyto Wipe Kits from Exposure Control Sweden AB were used for surface wipe sampling (www.exposurecontrol.net). The kits contain materials to take wipe samples from several types of surfaces and include paper tissues, droppers with 17 mL 0.1% formic acid, containers for sample storage, gloves for personal protection, labels, and a registration form. The wipe samples were taken by dripping 17 mL 0.1% formic acid on the surfaces. Next, one paper tissue was used to spread the liquid over the whole surface. The second paper tissue was used to dry the surface. Both tissues were collected in the storage container. For the vials, syringes, pumps, infusion bags, and outer bags, both tissues were split in 3–5 equal parts as 3–5 surfaces (subsamples) were combined to one sample. A part of the liquid was dripped on the smaller tissues, and these prewetted tissues were used to wipe the smaller surfaces. A second small paper tissue was used to dry the surfaces. The dimensions of the surfaces have been measured and the areas were calculated. Pairs of gloves were collected in storage containers. Nurse and drug administered were registered. Positive control samples (tissues and gloves spiked with 5FU, CP, DOC, and PAC) and negative control samples (non-used tissues and non-used gloves) were also collected.
Air sampling procedure
The air samples were collected with IOM samplers connected to VSS-5 Buck pumps. For personal sampling, the IOM sampler was attached to the worker's protective clothing, near the breathing zone. The sampling pump was placed in a carrying bag with shoulder strap. For stationary sampling, the IOM samplers and the sampling pumps were fixed at specific locations near potential emission sources: in front of the exit sluice of the isolators in the clean room, and on the bench top in the back ante room. Total particulate matter was collected on polytetrafluoroethylene filters (Whatman, 25-mm diameter and 1.0-µm pore size). The flow rate was 2.0 L/min, and the sampling time was registered. Positive control samples (filters spiked with 5FU, CP, DOC, and PAC) and negative control samples (non-used filters) were also collected.
Sample storage, sample preparation for analysis, and lc-ms/ms analysis
All samples were stored at 4°C after sampling and at room temperature during transport until arrival at the laboratory. In the laboratory, they were stored frozen until sample preparation and analysis.
The wipe samples and the gloves were prepared for analysis by adding a 0.1% formic acid solution up to a total volume of 100 ml. After extraction, a part of the extract was used for analysis. For the air filters, the preparation was similar except that 10 ml distilled water was used. Samples were analysed using liquid chromatography tandem mass spectrometry (LC-MS/MS) as recently published. 16 The detection limit is 0.2 ng/mL for cyclophosphamide and 2 ng/mL for 5-fluorouracil, docetaxel and paclitaxel. Finally, the contamination per cm2 for wipe samples and gloves, and per m3 for the air samples is calculated.
Results
The air concentration (ng/m3) was calculated by dividing the amount of drug measured on the filter (ng) by the air volume sucked (m3) which is equal to the sampling time (min) multiplied with the flow rate of 2 L/min. For comparison, the air concentrations were converted into Time-Weighted Averages (TWAs) over 8 h. The TWAs were calculated by multiplying the air concentrations (ng/m3) by the sampling time as percentage of an 8-h working day (480 min).
The contamination on the surfaces (ng) is corrected for the surface area wiped (cm2) and is presented in ng/cm2. The surface area of a pair of gloves is 1250 cm2. Next, the contamination is classified according to the colour indication of the ‘‘traffic-light’’ model linked to alert and action levels. 15 Surface and glove contamination indicated as ND are also indicated as ‘‘green’’. Drug recovery efficiency of the surface wipe samples is in general >80% based on laboratory experiments on comparable surfaces.
Compounding department - surface contamination
During the three trials, twelve surface wipe samples were collected from the outer surface of 5FU, CP, PAC, and DOC drug vials as specified in Table 1. Each sample was taken from four identical vials. Contamination was found in only one sample of PAC vials (2.1 ng/cm2). Cross-contamination of a drug vial with the other drugs was not observed.
Contamination with 5-fluorouracil (5FU), cyclophosphamide (CP), docetaxel (DOC), and paclitaxel (PAC) on the outer surface of drug vials (n = 4) (ng/cm2).
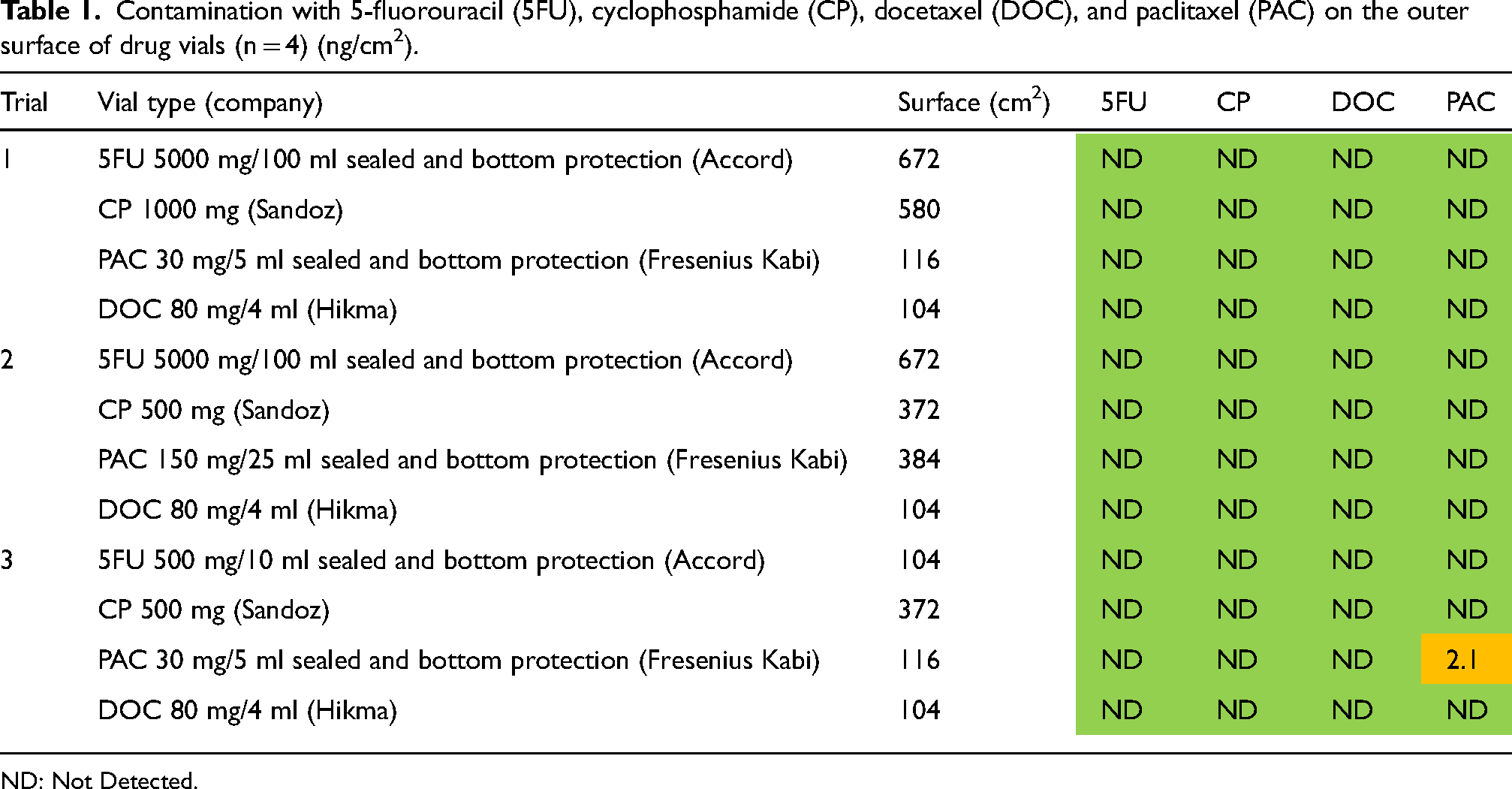
ND: Not Detected.
During the three trials, eleven surface wipe samples were collected from the outer surface of outsourced (prefilled) compounded 5FU and CP syringes, and PAC and DOC bags as specified in Table 2. Each sample was taken from four identical syringes and bags unless otherwise indicated. Contamination was mainly found for CP and to a lesser extent for DOC and 5FU. Contamination with PAC was not detected. High levels of contamination, indicated in orange, were found for CP in three samples. Lower levels of contamination were measured for DOC in two samples, and for 5FU in one sample. Cross-contamination of a syringe or bag with the other drugs was not observed.
Surface contamination with 5-fluorouracil (5FU), cyclophosphamide (CP), docetaxel (DOC), and paclitaxel (PAC) on outsourced compounded syringes and infusion bags a (ng/cm2).

ND: Not Detected.
Outsourced compounded drugs: DOC and PAC only available in infusion bags and 5FU and CP only available in syringes.
DOC infusion bags temporarily not available.
Only two syringes CP available.
At the end of the shift, and before daily cleaning, surface wipe samples were collected from the work surfaces in the isolators used for drug compounding, and from the bench top where the final check is performed by the pharmacists (Table 3). Contamination was mainly found for 5FU and CP, and to a lesser extent for PAC and DOC. High levels of contamination, indicated in red and orange, were measured for 5FU and CP on the bench top and the working surfaces in the isolators during the three trials (up to 21 ng/cm2).
Surface contamination with 5-fluorouracil (5FU), cyclophosphamide (CP), docetaxel (DOC), and paclitaxel (PAC) on bench top and on working surfaces in the isolators in the compounding department (ng/cm2).
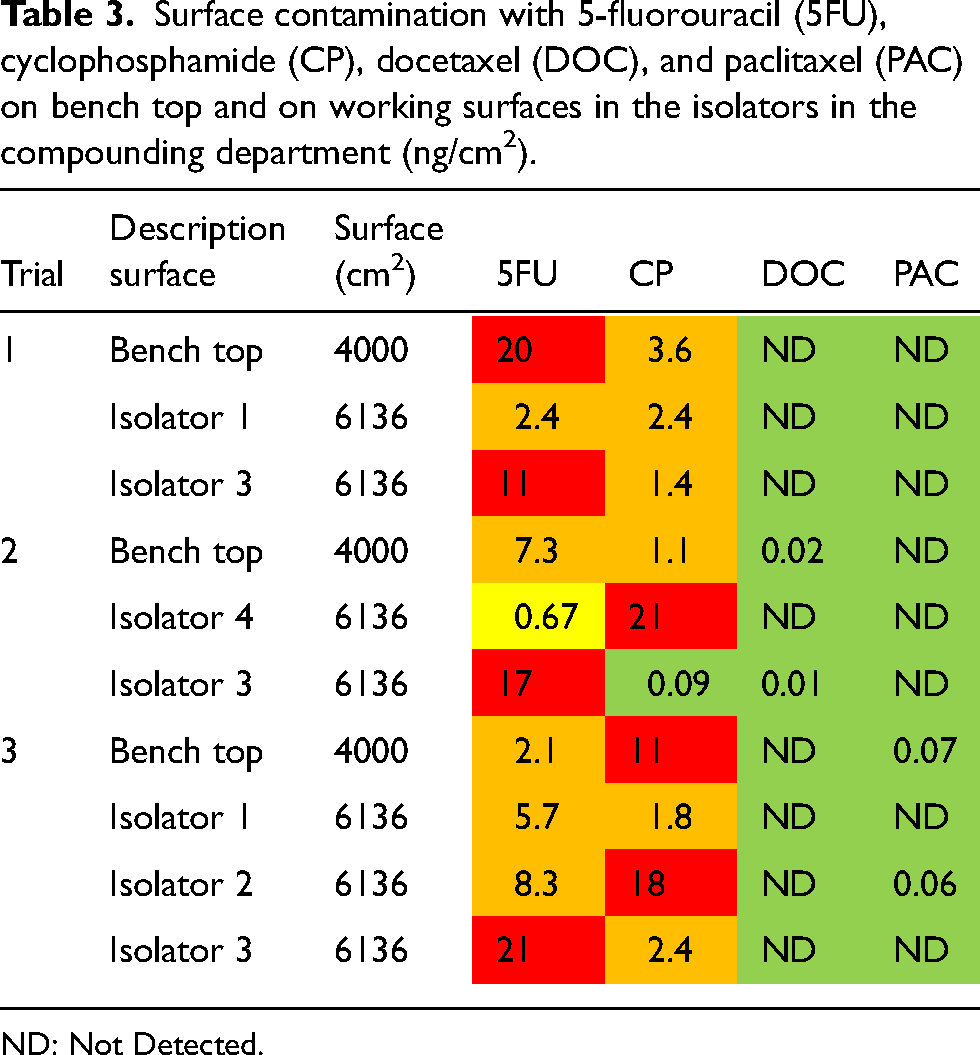
ND: Not Detected.
During the three trials, six sets of surface wipe samples were collected in the compounding department (Table 4). High levels of contamination, indicated in red and orange, were found for 5FU and CP on the yellow trays (up to 855 ng/cm2) and on the compounded products (up to 90 ng/cm2). Lower levels of contamination were measured on the clear outer bags (up to 6.1 ng/cm2). Some contamination with PAC was detected on the compounded products and on the accompanying yellow trays (up to 1.6 ng/cm2). Within most sets, especially when the contamination is high, the contamination in the samples is linked i.e., the contamination is found on the compounded products, on the clear outer bags and on the yellow trays. High levels of CP and 5FU contamination are found when CP syringes and 5FU bags are compounded.
Surface contamination with 5-fluorouracil (5FU), cyclophosphamide (CP), docetaxel (DOC), and paclitaxel (PAC) on compounded products (syringes, infusion bags and elastomeric pumps), clear outer bags, and yellow trays in the compounding department (ng/cm2).

ND: Not Detected.
One clear outer bag missing (not sampled) due to wrong dose in syringes (discarded).
Administration department - surface contamination and gloves
During the three trials, nine sets of surface wipe samples including nurses’ gloves were collected in the administration department. (Table 5). In addition, three surface wipe samples were collected from the inside of twelve yellow transport bags containing CP, 5FU, DOC, and PAC products for administration. High levels of contamination, indicated in red and orange, were found for 5FU and CP on trays after administration, trolley arms, and gloves in four sets of samples when 5FU was administered with Baxter pumps and CP was administered with syringes. On the contrary, contamination with DOC was only observed on one pair of gloves (just measurable) and PAC was not detected at all. All DOC and PAC was administered in bags. All trays at start except one were free of contamination indicating an effective cleaning procedure. After administration, four tray samples were contaminated with 5FU and/or CP, the drugs administered. Contamination of the trolley arms was measured in seven samples. The highest contamination was found in the five samples when the 5FU and CP were administered indicating contamination caused by handling. Lower 5FU and CP contamination was found in the two samples when the drugs were not administered, indicating insufficient cleaning. Within most sets, especially when the contamination is high, the contamination in the samples is linked i.e., the contamination is found on the trays after administration, on the trolley arms and on the gloves of the nurses.
Surface contamination with 5-fluorouracil (5FU), cyclophosphamide (CP), docetaxel (DOC), and paclitaxel (PAC) inside yellow transport bags, on trays at start and after administration, on trolley arms, and on gloves worn by the nurses during administration of the drugs (ng/cm2).
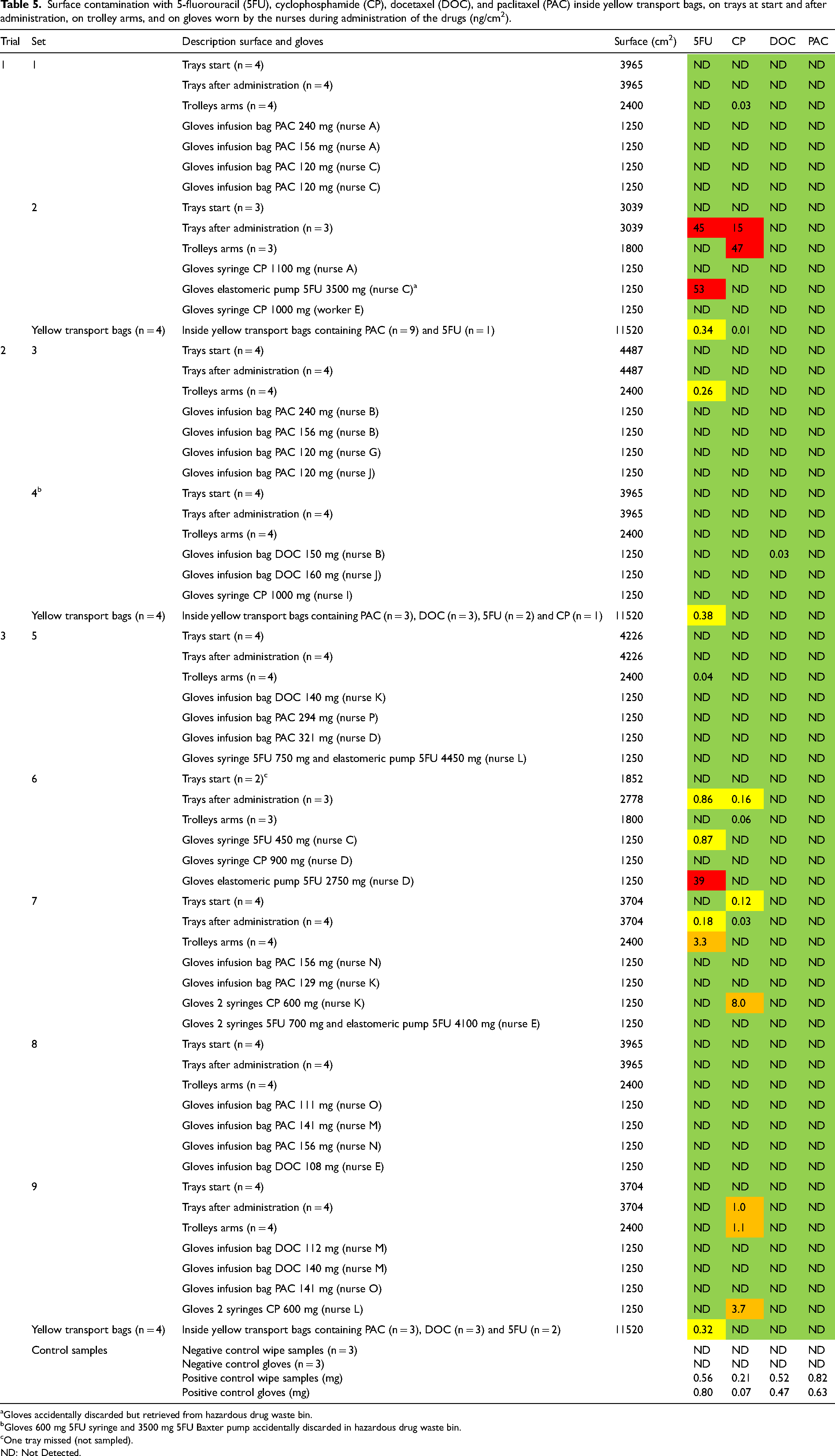
Gloves accidentally discarded but retrieved from hazardous drug waste bin.
Gloves 600 mg 5FU syringe and 3500 mg 5FU Baxter pump accidentally discarded in hazardous drug waste bin.
One tray missed (not sampled).
ND: Not Detected.
Thirty-three pairs of gloves were collected. Six pairs of gloves were contaminated with the drug administered indicating contamination caused by handling. The highest contamination was found for 5FU administered via elastomeric pumps followed by CP in syringes, 5FU in syringes, and DOC in a bag.
Three surface wipe samples were collected from the inside of twelve yellow transport bags. The highest contamination was measured for 5FU in all samples of the yellow transport bags which did contain 5FU products, while CP was just measurable in one sample of a yellow transport bag who did not contain a CP product.
The positive control wipe samples and gloves contained 5FU, CP, DOC, and PAC and the negative control wipe samples and gloves did not.
Pharmacy department - environmental air contamination
Stationary air samples were collected in front of the exit hatch of the isolators in function, above the bench top, and personal air samples were collected from the pharmacists performing the final check (Table 6). Different sampling times were registered due to different compounding and check periods of the pharmacists. Each personal air sample was collected during the whole shift and concerns up to four pharmacists working during the shift (job rotation/sampler rotation). During compounding, 5FU was detected in the air in front of one isolator in one trial, and in another trial, DOC was detected in front of all isolators. In the same trial, DOC was also measured above the bench top and in the personal air sample of the pharmacist performing the checks. CP and PAC were not detected in environmental air during the three trials.
Environmental air contamination with 5-fluorouracil (5FU), cyclophosphamide (CP), docetaxel (DOC), and paclitaxel (PAC) in the compounding department.
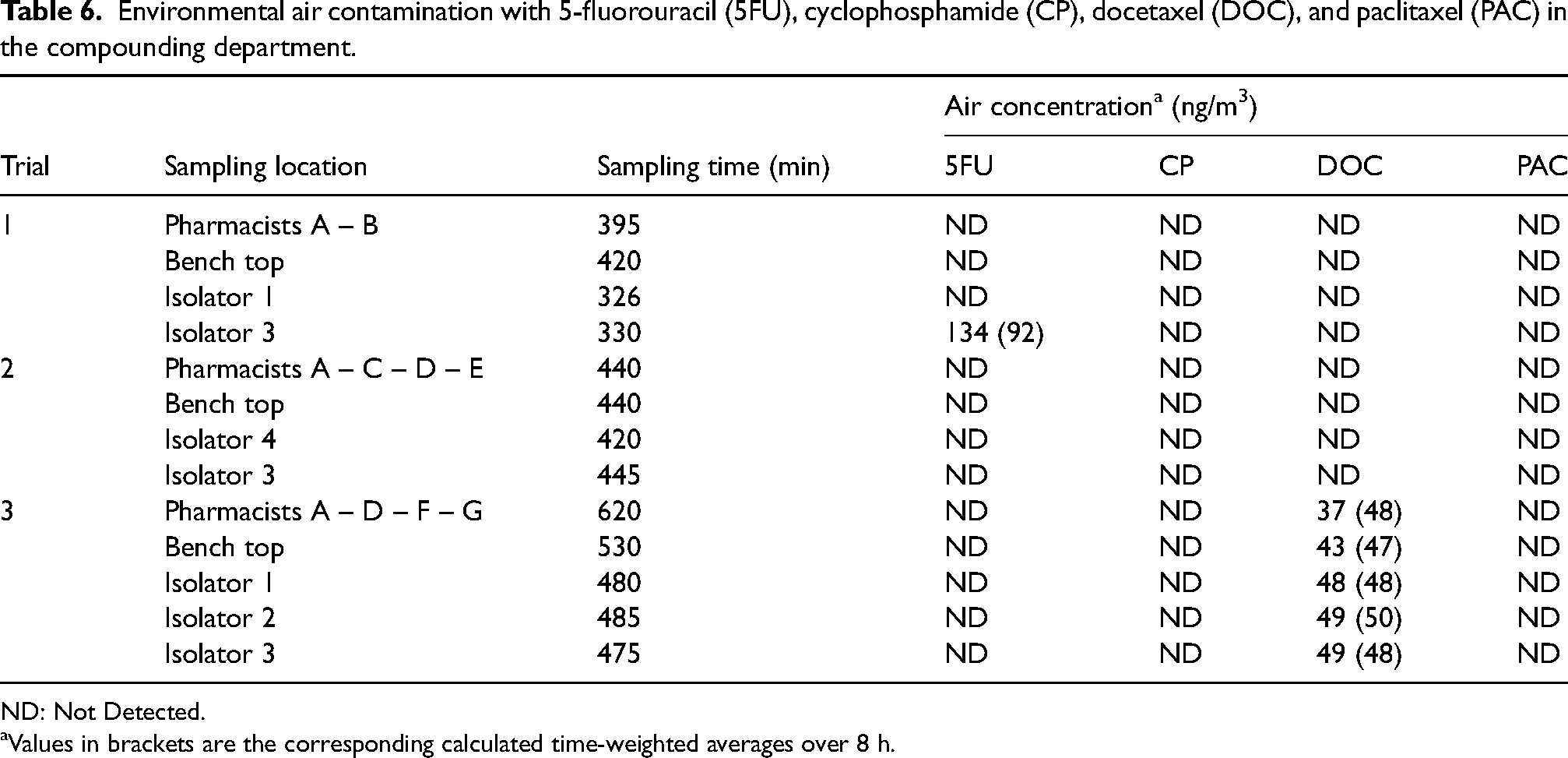
ND: Not Detected.
Values in brackets are the corresponding calculated time-weighted averages over 8 h.
Administration department – environmental air contamination
Personal air samples were collected from fifteen nurses administrating different hazardous drugs including 5FU, CP, DOC, and PAC (Table 7). Three nurses participated in two trials, and one nurse in three trials. DOC was the only drug detected in one personal air sample of a nurse who has administered different drugs including two bags DOC. 5FU, CP, and PAC were not detected in environmental air during the three trials.
Environmental air contamination with 5-fluorouracil (5FU), cyclophosphamide (CP), docetaxel (DOC), and paclitaxel (PAC) in the administration department.
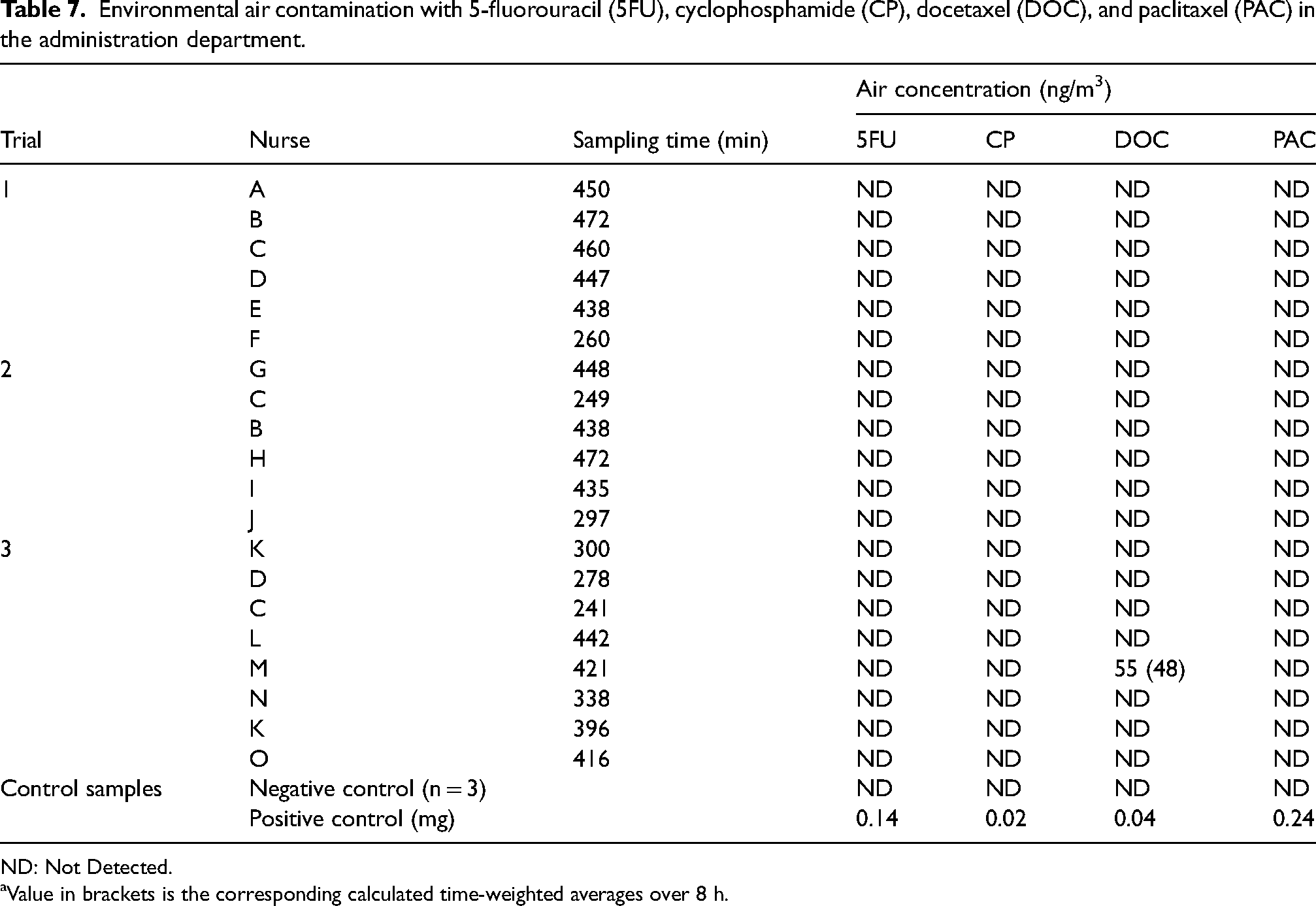
ND: Not Detected.
Value in brackets is the corresponding calculated time-weighted averages over 8 h.
The positive control air samples contained 5FU, CP, DOC, and PAC and the negative control air samples did not.
Discussion
The results show environmental contamination in the compounding department and to a lesser extend in the administration department. Contamination is mainly found for CP and 5FU. It is obvious that the contamination is caused by the compounding process as high levels of contamination were found on the working surfaces in the isolators. Vials entering the compounding process were hardly contaminated and the outsourced compounded products show substantially lower levels of contamination compared to the in-house compounded products. The results indicate that contamination caused by contaminated vials and outsourced products is neglectable compared to the in-house compounded products. However, CP contamination on the outsourced compounded syringes needs some attention and must be prevented.
The use of needle and syringe technique for compounding is considered as an open technique causing spillage by overpressure in drug vials and during withdrawal of the needle-syringe from the vials and can only be removed by effective cleaning. However, it is recommended to focus on preventing contamination to be achieved by applying closed handling techniques such as closed-system drug transfer devices (CSTDs). Several studies have shown a substantial reduction in environmental contamination by using a CSTD.16–20
The contamination on the working surfaces in the isolators will be transferred to the compounded products contaminating all surfaces in between such as the bench top, the yellow trays and to a lesser extent the clear outer bags when checked by the pharmacists. Contamination is mainly observed for 5FU and CP. This can be explained by higher dosages and differences in compounded products (syringes and elastomeric pumps versus infusion bags). Finally, the contamination on the compounded products will be transferred to the inside of the yellow transport bags when they are transported to the administration department, where further spread of contamination will occur.
At administration, contamination was incidentally observed. CP and 5FU were detected on trays after administration, trolley arms, and corresponding gloves worn by the nurses. It concerns CP in syringes and 5FU in elastomeric pumps corresponding with the results observed in the compounding department.
DOC and 5FU were incidentally detected in environmental air in front of the exit hatches of the isolators, above the bench top, and in a personal air sample of a pharmacist performing the checks. DOC was also detected in a personal air sample of a nurse. The result indicates release of the drugs during compounding and administration. Official Occupational Exposure Limits (OELs) for hazardous drugs do not exist in the UK. If not available, one may also use Occupational Exposure Bands (OEBs). As CP is a genotoxic (non-threshold) carcinogen, it is obvious to classify the drug in OEB 5 with a corresponding OEL < 1 µg/m3.21,22 Although 5FU, DOC and PAC are non-carcinogens, they are anyhow considered as hazardous and can, as a worst-case approach, also be classified in OEB 5. The air sampling results show that the TWA-corrected air concentrations are all below 1 µg/m3 indicating that inhalation is a low risk for being exposed and that worker's safety is guaranteed.
Limitations of the study concern the actual levels of surface contamination. Wipe efficiency is depending on many factors, and it is practically impossible to determine the exact recovery for each drug on all types of surfaces. Hence, the presented results are not corrected for recovery and present in fact an underestimation of the actual contamination. In addition, threshold limit values for surface contamination and OELs for hazardous drugs do not exist. As contamination may vary over time, one may debate whether a single trial can be considered as a representative study. Hence, three trials were performed to obtain more information and being more representative. For some parts of the study, the results are comparable between the three trials while other parts of the study show different results supporting the relevance of performing more trials.
The study results have been shared with the team, and the need to consistently adhere with PPE requirements and appropriately segregate checking to the designated benches with pharmacy aseptic unit is emphasised. Venting spikes will be introduced within the pharmacy aseptic unit and will be used for standard drug compounding. This will negate the need for manual equalisation of vial pressure using the needle and syringe technique, thus preventing aerosol generation which is the likely source of the contamination seen in these trials. The implemented changes will be validated in a follow up study.
Conclusions
The results of the three trials show spread of surface contamination mainly with CP and 5FU in the compounding department. Drug release in the isolators during compounding by using the open needle and syringe technique has resulted in contamination of the compounded syringes, infusion bags, and elastomeric pumps. Contamination is further spread and finally transferred to the administration department. Contamination with DOC and 5FU was also found in environmental air in front of the isolators, above the bench top and around pharmacists and a nurse but the concentrations are all below the OEL. In general, the results show that if contamination starts at the beginning of the workflow (compounding) it will be found on all following stages of the workflow supporting the need of prevention of contamination during compounding.
Footnotes
Acknowledgements
The authors thank the nurses, pharmacy technicians, and pharmacists at Western General Hospital in Edinburgh, UK who have participated in the study.
Author contributions
All authors designed the study; PS, collected the samples, performed the analysis, interpreted the results, and drafted the manuscript. All authors reviewed and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this study was provided by ICU Medical, Inc., San Clemente, USA.
