Abstract
Introduction
Exposure of healthcare workers to hazardous drugs can lead to adverse health effects supporting the importance of a continuous monitoring program, for example, by taking surface wipe samples. The objective was to describe the results of repeated monitoring of contamination with hazardous drugs on multiple surfaces in a hospital pharmacy and at two wards using standardized preparation techniques and cleaning procedures.
Methods
Twelve surfaces in the hospital pharmacy and at two wards were sampled and analyzed for contamination with the hazardous drugs cyclophosphamide, doxorubicin, 5-fluorouracil, gemcitabine, methotrexate, and paclitaxel. The drugs were prepared with a closed-system drug transfer device (CSTD). Sampling of the drugs was performed in four trials during eight months. Liquid chromatography tandem mass spectrometry was used for the analysis of the drugs.
Results
During the four trials, contamination with five of the six hazardous drugs was found on half of the surfaces in the pharmacy and in a ward. Seventeen out of 288 possible outcomes were positive (6%), with the biological safety cabinet grate (n = 6) and scanner (n = 5) most frequently contaminated. The highest level of contamination was observed on the pass-thru window (cyclophosphamide: 2.90 ng/cm2) and the touch screen of the Diana device (5-fluorouracil: 2.38 ng/cm2). Both levels were below the action level of 10 ng/cm2.
Conclusions
The long-term use of a CSTD in combination with appropriate cleaning has proven effective in achieving low levels of surface contamination with hazardous drugs.
Introduction
It has been documented in several studies that exposure of healthcare workers to hazardous drugs may cause adverse health effects such as cancer and reproductive effects.1–5 To prevent healthcare workers from being exposed, authorities and organizations have developed guidelines and recommendations that have been implemented in daily practice.6–8 These include organizational measures such as protocols, education, and training, technical measures such as clean rooms, biological safety cabinets (BSCs), isolators, and closed-system drug transfer devices (CSTDs), as well as personal protective equipment (PPE).
Despite the implementation of guidelines and recommendations, the potential risk of being exposed remains.9,10 Surface wipe sampling studies still show environmental contamination with hazardous drugs in hospital pharmacies and inpatient and outpatient departments where these drugs are prepared and administered to patients.9,11
The objective of this study was to measure surface contamination with hazardous drugs at different locations in the hospital pharmacy and at two wards of the University Hospital Leuven in Belgium. Monitoring was repeated four times over a period of eight months which is in contrast with other published studies mostly presenting a single measurement.
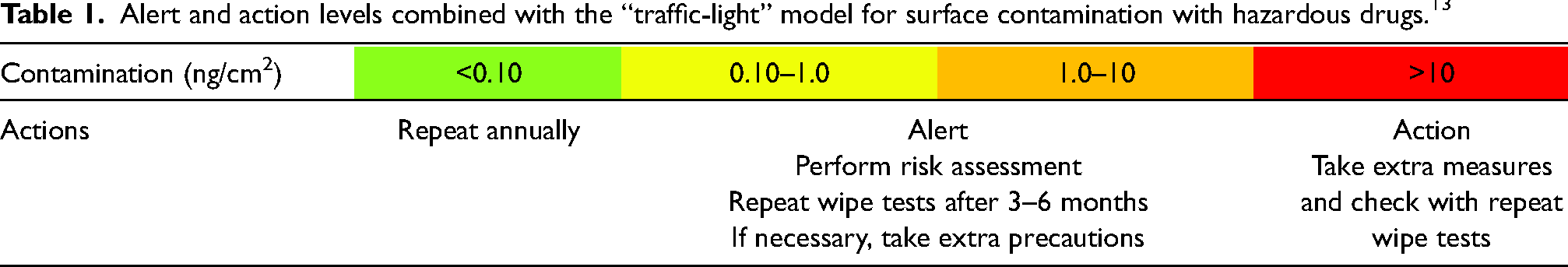
In addition, a new model for the interpretation of the surface wipe sampling results was presented based on alert and action levels and a color indication. Alert and action levels of surface contamination with hazardous drugs have been developed in The Netherlands. 12 Based on a benchmark, the alert level was set at 0.1 ng/cm2, and the action level was set at 10 ng/cm2. The levels were combined with actions to be performed. The “traffic-light” model presented seven years before, showed the same format but added the color indication green, yellow, orange, and red. 13 Green indicates “safe,” while red indicates “not acceptable.” Yellow and orange are intermediate steps. The color indication makes comparison of the results easier and quickly identifies the areas where action is needed. Alert and action levels, combined with the “traffic-light” model are presented in Table 1.
Alert and action levels combined with the “traffic-light” model for surface contamination with hazardous drugs. 13
Methods
Surface contamination
Contamination with the hazardous drugs cyclophosphamide, doxorubicin, 5-fluorouracil, gemcitabine, methotrexate, and paclitaxel, was measured by surface wipe sampling in the pharmacy and at two wards. The wipe samples were taken from six surfaces in the pharmacy and three surfaces at both wards 1 and 2. An overview is presented in Table 2. The wipe samples were taken with Cyto Wipe Kits from Exposure Control Sweden AB (www.exposurecontrol.net). Wipe samples were taken in the morning before the start of the shift (trial 1), and in the afternoon at the end of the shift before cleaning (trial 2). The end-of-shift wipe sampling before cleaning was repeated 2 to 3 months later (trial 3), and eight months later (trial 4). Between trials 2, 3, and 4, preparation protocols and procedures, including cleaning were not changed.
Surface contamination with six hazardous drugs in three departments during four trials (ng/cm2).
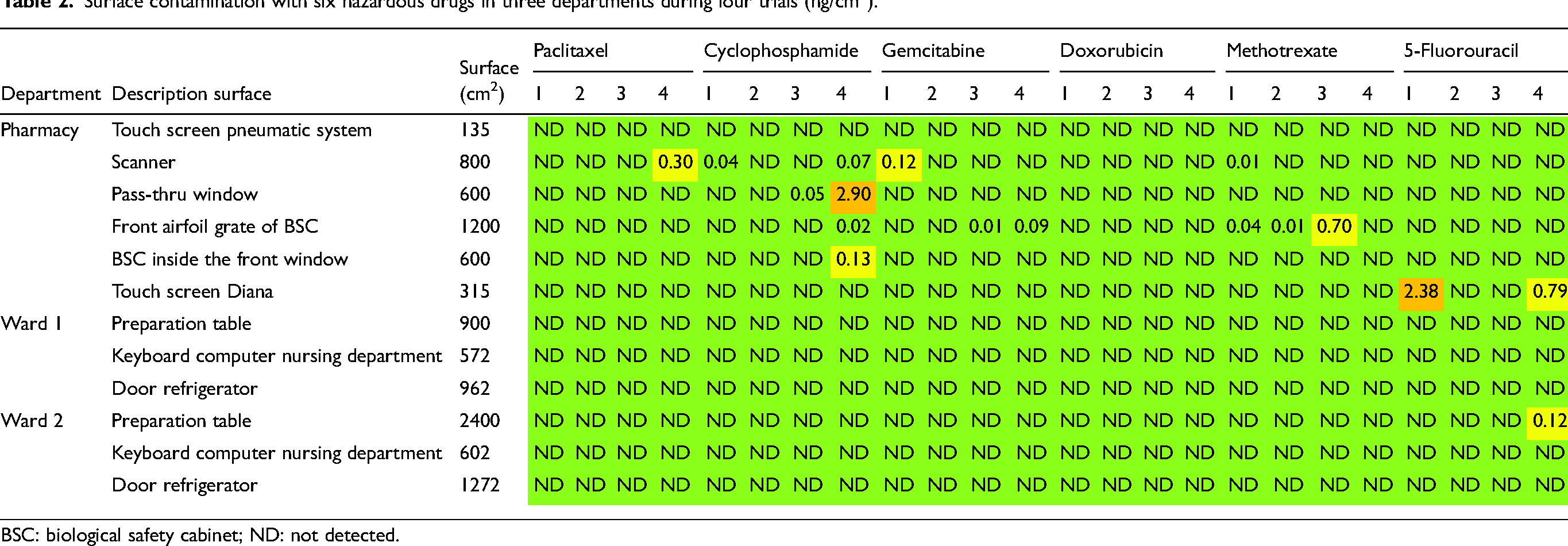
BSC: biological safety cabinet; ND: not detected.
About 60,000 units are prepared annually and 38% is accounted for by the six measured drugs. The hazardous drugs were prepared in a BSC installed in a cleanroom using a CSTD (ChemoClave, ICU Medical, San Clemente, USA). The technicians wore PPE including a protective gown, a surgical mask, two pairs of gloves (nitrile and latex), cover shoes, and a hair/beard cover.
Daily cleaning is performed several times a day with Klercide Neutral detergent followed by 70% alcohol. Weekly cleaning is performed with Klercide Sporicide peroxide (both products from Ecolab, Belgium).
Surface wipe sampling
The wipe samples were taken by an experienced hospital pharmacist. The dimensions of the surfaces have been measured and the areas were calculated. All samples were stored at −20°C after sampling, during transport, and at the laboratory until sample preparation and analysis. The wipe samples were prepared for analysis by adding a 0.1% formic acid solution. The total extraction volume was 100 mL. After extraction, a part of the extract was used for analysis. The drugs were analyzed with liquid chromatography tandem mass spectrometry. Finally, the contamination per cm2 is calculated and presented in combination with the corresponding color indication of the “traffic-light” model. Drug recovery efficiency is in general >80% based on laboratory experiments performed on comparable surfaces.
Liquid chromatography with tandem mass spectrometry analysis
Analysis was performed on a Xevo TQ-S micro mass spectrometer combined with an Acquity UPLC H-class sample manager and quaternary solvent manager controlled by Masslynx software (Waters, Milford, USA). An Acquity BEH C18, 1.7 µm, 2.1×100 mm separation column (Waters, Milford, USA) operated at 40°C was used.
Elution (0.4 mL/min) started with a composition of 100% solvent A (100% MilliQ RO-water with 0.1% formic acid) and 0% solvent B (100% acetonitrile with 0.1% formic acid) with a delay of 1 min. Between 1 and 3 min, the composition changed to 50% B, and from 6.25 min ramped to 100% B. Starting conditions were restored between 7.00 and 7.25 min. The total runtime was 10 min, and the injection volume was 5 µL. The mass spectrometer was operated with a capillary voltage of +1.5 kV, a desolvation temperature of 600°C, and a nitrogen flow of 1100 L/hr. The cone gas flow was set at 50 L/min (nitrogen). Argon was used as collision gas. All transitions were measured in positive mode, except for 5-fluorouracil in negative mode. The drugs have a linear calibration curve up to 100–500 ng/mL. The detection limit is 0.1 ng/mL for cyclophosphamide, doxorubicin, gemcitabine, and methotrexate, 1 ng/mL for 5-fluorouracil, and 2 ng/mL for paclitaxel.
Results
For trials 1 and 2, contamination was only found in the pharmacy (Table 2). The highest contamination was measured for 5-fluorouracil on the touch screen of the Diana automated compounding device (Diana type 1, ICU Medical, San Clemente, USA) followed by gemcitabine on the scanner. The scanner was also contaminated with cyclophosphamide and methotrexate, and methotrexate was also detected on the airfoil of the BSC, but the contamination was just above the detection limit. Contamination with paclitaxel and doxorubicin was not found. In trial 1, the scanner, airfoil BSC, and touch screen of the Diana device were contaminated while in trial 2, contamination was only found on the airfoil BSC, and was somewhat lower than in trial 1.
For trial 3, contamination was again only found in the pharmacy. The highest contamination was measured for methotrexate on the airfoil of the BSC. The airfoil was also contaminated with gemcitabine, and cyclophosphamide was detected on the pass-thru window, but the contamination was very low and just measurable. Contamination with paclitaxel, doxorubicin, and 5-fluorouracil was not found. The results of trials 2 and 3 are comparable.
For trial 4, contamination was mainly found in the pharmacy. The highest contamination was observed for cyclophosphamide on the pass-thru window followed by 5-fluorouracil on the touch screen of the Diana device. Lower levels of contamination were observed on the scanner and on the BSC grate and window indicating some spread of contamination with cyclophosphamide. The scanner was also contaminated with paclitaxel and the BSC grate was also contaminated with gemcitabine, but the levels of contamination are low. At ward 2, contamination was only found for 5-fluorouracil on the preparation table. The results of trial 4 compared to trial 3 show a slight increase in the number of contaminated surfaces, the number of drugs detected, and the level of contamination.
The overall results of the four trials show some contamination with the hazardous drugs up to 2.90 ng/cm2. Seventeen out of 288 possible outcomes were positive indicating a contamination score of 6%. The score is 100% if all six hazardous drugs have been found on all 12 surfaces during the four trials (six drugs multiplied with 12 surfaces multiplied with four trials gains 288 outcomes). Only eight outcomes were in the alert level range with the color indication orange (n = 2) and yellow (n = 6).
Discussion
Implementation of guidelines and protocols in combination with the use of a CSTD and appropriate cleaning has proven to be effective in minimizing contamination with hazardous drugs. Despite these measures, some contamination was still found. The sources of contamination were not established and contamination from drug vials cannot be excluded.14,15 Vials were not checked for contamination on the outside and it is possible that contamination on the vials has been transferred, for example, by hand or gloves to other surfaces. This means that the contamination on the surfaces can also be caused by vial contamination.
Reduction of surface contamination has frequently been found for hazardous drugs after implementation of a CSTD.16–19 Implementation comparison mainly consists of pre- and post-implementation measurements. On the contrary, the present study has monitored contamination over a longer period using a CSTD. Monitoring was performed four times over a period of eight months. The present study is in accordance with the recommended workplace monitoring presented in the “Guidance for the safe management of hazardous medicinal products at work” recently published by the European Commission. 20 The study can serve as a good example for hospitals that want to start with the implementation of workplace monitoring.
Conclusions
The results clearly show that it is possible to prevent the spread of contamination, and if contamination is present, to keep the levels of contamination low for a long period despite substantial numbers of hazardous drugs being compounded. Appropriate cleaning and the use of a CSTD have proven to be effective in achieving low levels of surface contamination with hazardous drugs.
Footnotes
Author contributions
All authors designed the study; PS and BT, collected the data; PS, performed the analysis and interpreted the results; PS, drafted the manuscript. All authors reviewed and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Financial support for this study was provided by ICU Medical, Inc., San Clemente, USA.
