Abstract
Introduction
The aim of this study was to analyze real-life data from a cohort of adult patients receiving atezolizumab in combination with carboplatin and etoposide for first-line treatment of ES-SCLC, in order to assess relative dose intensity (RDI), time-to-treatment discontinuation (TTD), time-to-treatment failure (TTF), progression-free survival (PFS), overall survival (OS) of treatments as well as the correlation between these outcomes.
Methods
An observational retrospective study was conducted. All patients treated with atezolizumab combined with carboplatin and etoposide for first-line treatment of ES-SCLC were included. Median TTD, TTF, PFS and OS were calculated in our cohort of patient by the Kaplan Meier method.
Results
The curves obtained with the Kaplan Meier method of TTF and TTD are substantially similar, indicating a good concordance of the information extracted by the two different data sources. This tendency was confirmed also when the TTD versus PFS curves were compared. The median OS registered was 11.8 months. Patients with no liver metastases showed a longer median time of OS than patients with liver metastases. The mean value of RDI for the entire cohort was 87.4%.
Conclusions
Our study showed that TTD, calculated from the administration data is a useful proxy of TTF as registered in the clinical chart. TTD is a real-world outcome that can be used to demonstrate the efficacy of drugs used for administered therapies. It can be used as an end point for RWE studies, where the evaluation is less structured and standardized.
Keywords
Introduction
Small cell lung cancer (SCLC) is an aggressive disease accounting for 13% of all lung cancers. Since it is characterized by rapid tumour growth and early development of widespread metastases, most patients (60–70%) have extensive-stage (ES) disease at the time of diagnosis. The prognosis of these patients is associated with a five-year survival rate of approximately 3%.1,2
ES-SCLC represents a major challenge for clinicians. After several years without witnessing significant clinical progress, the addition of the more recent immunotherapy agents (e.g. atezolizumab, durvalumab based on two double-blind, phase III Randomised Clinical Trials IMpower133 and CASPIAN) to platinum-based chemotherapy has shown significant improvement in overall survival (OS) and represents the current standard of care in the first-line setting. 3
Atezolizumab is a humanized monoclonal antibody targeting programmed death ligand 1 (PD-L1), which is an inhibitory ligand that negatively regulates T cell activation and proliferation by binding to the PD-1 receptor. In particular, atezolizumab selectively binds to PD-L1 to impede the interaction between PD-1 and B7, still allowing PD-L2 and PD-1 to interact. 4 When used in combination with platinum-based chemotherapy in a randomized phase I/III study of 403 chemotherapy-in naïve ES-SCLC patients, atezolizumab showed an OS of 12.3 months and a progression-free survival (PFS) of 5.2 months. 5
Recently, in order to gain a better assessment of real clinical practice outcomes, which in turn also supports the clinician's decision in the decision-making process, the use of more pragmatic endpoints such as time-to-treatment discontinuation (TTD) has been proposed. 6 TTD did in fact correlate with strong endpoints of efficacy such as OS and PFS 7 which was found to be useful owing to several aspects, such as an increased sample size, concluding studies more rapidly and, consequently lower costs. It is easy to calculate TTD from administrative data of prescriptions.
Real-world time on treatment with first-line immunotherapy for advanced NSCLC has been analysed by Velcheti V et al. 8
Atezolizumab in combination with carboplatin and etoposide has been authorized in Italy for first-line treatment of adult patients with ES-SCLC since July 2020. According to the European public assessment report, duration of treatment with atezolizumab is administered until disease recurrence or unacceptable toxicity. Treatment ends for two main reasons, progression of disease or drug-related toxicity. Therefore, TTD is defined as the time from the first drug administration until the discontinuation of treatment due to disease progression or drug toxicity. 9
Since 2021 several authors have described atezolizumab-related TTD and PFS in clinical practice,10–13 but none of these studies have evaluated the correlation between TTD and strong endpoints like PFS and time-to-treatment failure (TTF). In a recent study, Lasala et al. 14 have reviewed all TTD values of drugs used in first-line NSCLC.
In a retrospective observational study, 15 the PFS and OS were longer in patients who received immune checkpoint inhibitor (ICI) therapy (atezolizumab or durvalumab) than in the Impower133 study.
Relative dose intensity (RDI) is the ratio of the dose intensity delivered to the reference standard dose intensity for a chemotherapy regimen. 16
None of the published studies evaluated the RDI versus the standard treatment schedule and consequently nor the reason why treatments received significant interruptions.
The aim of this study was to analyze real-life data from a cohort of adult patients receiving atezolizumab in combination with carboplatin and etoposide for first-line treatment of ES-SCLC, in order to assess RDI, TTD, TTF, PFS, OS of treatments as well as the correlation between these outcomes. Our primary scope was to assess whether the TTD calculated from administrative data (date and milligrams of doses received from each patient) could be a potential proxy of the TTF and PFS as reported in the clinical ward charts.
Methods
Study population
An observational retrospective study was conducted during the period between 1 September 2020 to 31 May 2022. All adult patients treated with atezolizumab combined with carboplatin and etoposide for first-line treatment of ES-SCLC in accordance with Italian Medicine Agency (AIFA) eligibility criteria were included in the study. The date of first administration of atezolizumab was defined as the index date (ID).
Patients who received atezolizumab after 31 October 2022 were considered continuers; among these, patients who discontinued therapy with atezolizumab, reasons of drug discontinuation were further investigated. The follow-up period for each patient closed on 31 December 2022 (cut-off date).
Data sources
Real-world data were extracted from all patients treated for ES-SCLC in the oncology departments of the Regina Elena Institute and Policlinico Gemelli of Rome, Italy.
In this study, we included patients with ES-SCLC based on eligibility for atezolizumab from the Italian Medicine Agency (AIFA) monitoring registries 17 ; dose administration, schedule and clinical data were extracted from the electronic clinical charts and prescriptions software (administrative data).
The data collected at baseline included: (a) demographic data, including age (registered at the date of first administration of atezolizumab) and sex, performance status (PS) based on ECOG (Eastern Cooperative Oncology Group) PS scale, number and type of metastatic sites; (b) atezolizumab administration data, including dose in milligrams, reason and date of drug discontinuation, and survival status of each patient.
Study drugs and administered protocol
The drugs of interest in this study were atezolizumab combined with chemotherapy (i.e. carboplatin and etoposide) approved in Europe by the European Medicine Agency and in Italy for first-line treatment of adult patient with ES-SCLC since July 2020.
Based on the AIFA registry criteria of eligibility, therapy was administered by considering two consecutive phases:
induction phase (4 cycles recurring every 3 weeks): at day 1 of every cycle, patients receive carboplatin (area under the curve of 5 mg per millilitres per minute, administered intravenously on day 1 of each cycle), etoposide (100 mg per square meter of body-surface area, administered intravenously on days 1 through 3 of each cycle) and atezolizumab (at a dose of 1200 mg, administered intravenously on day 1 of each cycle); maintenance phase: atezolizumab was administered as monotherapy (1200 mg given by IV infusion) every 3 weeks.
Duration of treatment with atezolizumab is administered until disease recurrence or unacceptable toxicity.
Statistical analysis
All patients included in our study were characterized in terms of sex, median age with its relative range, PS and metastatic sites involved at the time of diagnosis (type and number of metastasis). Variables were shown as frequencies and percentage values.
Median OS, TTD, TTF and PFS were calculated by the Kaplan Meier (KM) method and we respectively considered as event death, drug discontinuation, treatment failure, progression of disease.
More specifically, to include the data in the KM analysis, for each treatment TTD was calculated considering the time (months) from the first to the last dose of atezolizumab administered plus 21 days concerning the drug coverage period of the administration cycle; patients who did not receive doses of atezolizumab after 31 October 2022 were considered as discontinuers.
TTF was calculated as the time between the first dose of atezolizumab administered to the time of treatment failure registered by clinicians in the electronic chart; as for the protocol, the reasons behind ending treatment were expressed as:
progression of disease (PD), considered in case of an increase in tumour volume registered at baseline or onset of new metastatic tumour lesions (not yet registered at baseline); failure for clinical worsening of patient due to medical decision without progression of disease; failure for drug-related toxicity or other adverse events, like infections;
PFS was calculated considering the time between the first dose of atezolizumab administered until to disease progression or death from any cause.
OS was calculated considering the time between the first dose of atezolizumab administered until death if it occurred before 31 December 2022 and no later.
Patients who were still alive after 31 December 2022 were censored. The log rank test was used in order to assess potential differences between subgroups.
RDI 18 was calculated as the ratio between the administered dose and the standard dose of atezolizumab. RDI was used to define the dose of drug administered per unit of time, considering the administration of 1200 mg every 3 weeks (i.e. 21 days) as the standard in this study.
Evaluation of TTD and its relation to PFS and TTF caused by PD, discontinuation or temporary suspension of therapy due to clinical worsening, toxicity or death of the patient were conducted.
Data regarding patient's death, clinical progression/worsening or drug toxicity were extracted from the electronic clinical chart. All of the analyses and plots were performed using SPSS version 28.1. A p-value <0.05 was considered statistically significant.
Results
Overall, N 79 patients with ES-SCLC treated with atezolizumab were included in our analysis. The median age was 67.4 (min–max = 44.4–84.8). The number of males was slightly higher than females (N = 46; 58.2%) (Table 1). Regarding the ECOG PS, we found a higher percentage of patients with PS = 1 (N = 39; 49.4%), followed by PS = 0 (N = 37; 46.8%); only 3 patients (3.8%) had PS = 2. Liver metastases were registered in 34.2% (N = 27) of cases, while 11.4% (N = 9) of patients had brain metastases. More than half of the patients (N = 43; 54.4%) had other sites of metastases other than brain and liver (Table 1).
Baseline characteristics of patients in the cohort studied.
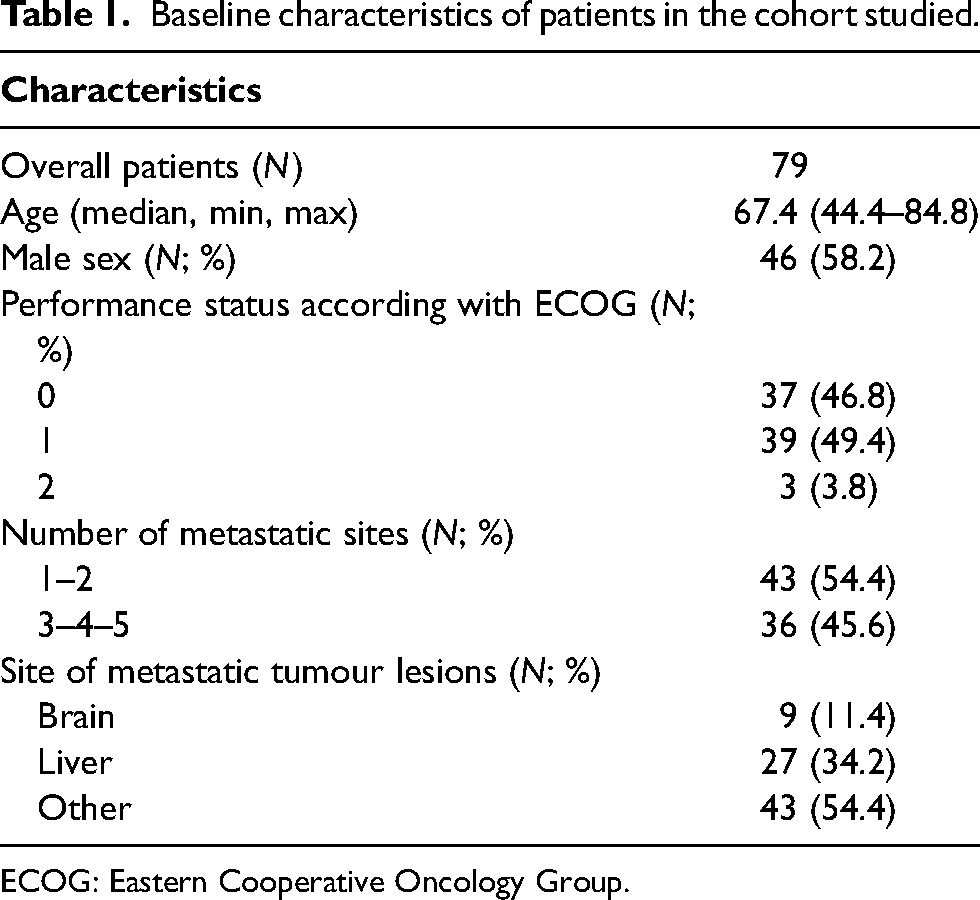
ECOG: Eastern Cooperative Oncology Group.
Treatment data registered on 31 December 2022 were shown in Supplemental Table 1: 38.0% of patients were still alive at the end of observation, only five patients were lost to follow up for information regarding OS before the end of observation. Therapeutic failure was the reason for interrupting treatment for most of the patients (N = 72; 91.1%); among these, 46 (63.9%) discontinued treatment for PD, 16 (22.2%) for toxicity/infection and 8 (11.1%) for clinical worsening/death. In two cases (2.8%), the reason for interrupting treatment was not registered. N 62 patients (78.5%) progressed or died and 17 (21.5%) were censored from the PFS analysis (Supplemental Table 1).
The recruitment curve for cohort patients and duration of treatments over time were shown in Figure 1. Only seven patients were still on treatment by the end of the year 2022 where there were very few treatments (8; 10.1%) that exceeded 400 days (Y right scale, black dashes above the 400 days’ line).
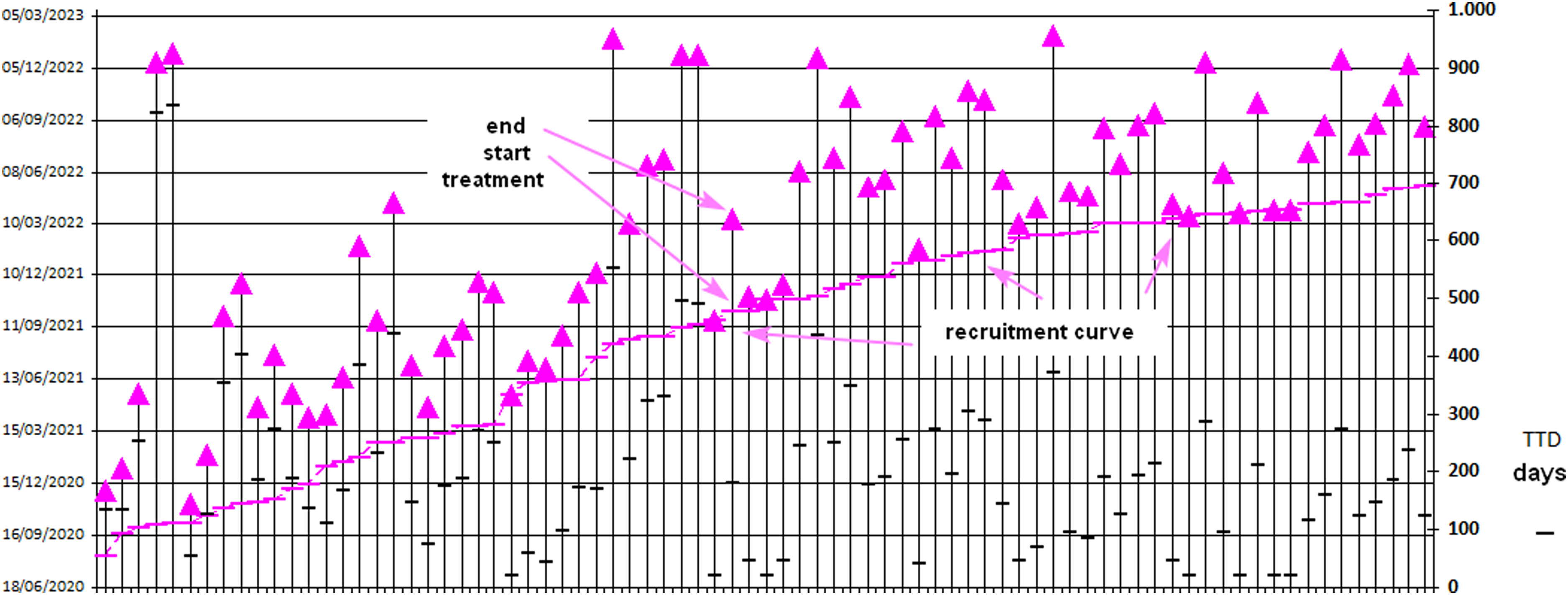
Recruitment curve of the cohort of patients treated with atezolizumab and duration of treatment.
TTD and relations with PFS and discontinuation for treatment failure
Figure 2 shows the KM curves of TTD calculated from administrative data versus discontinuation due to treatment failure, as recorded in the medical record for PD, clinical worsening or toxicity (
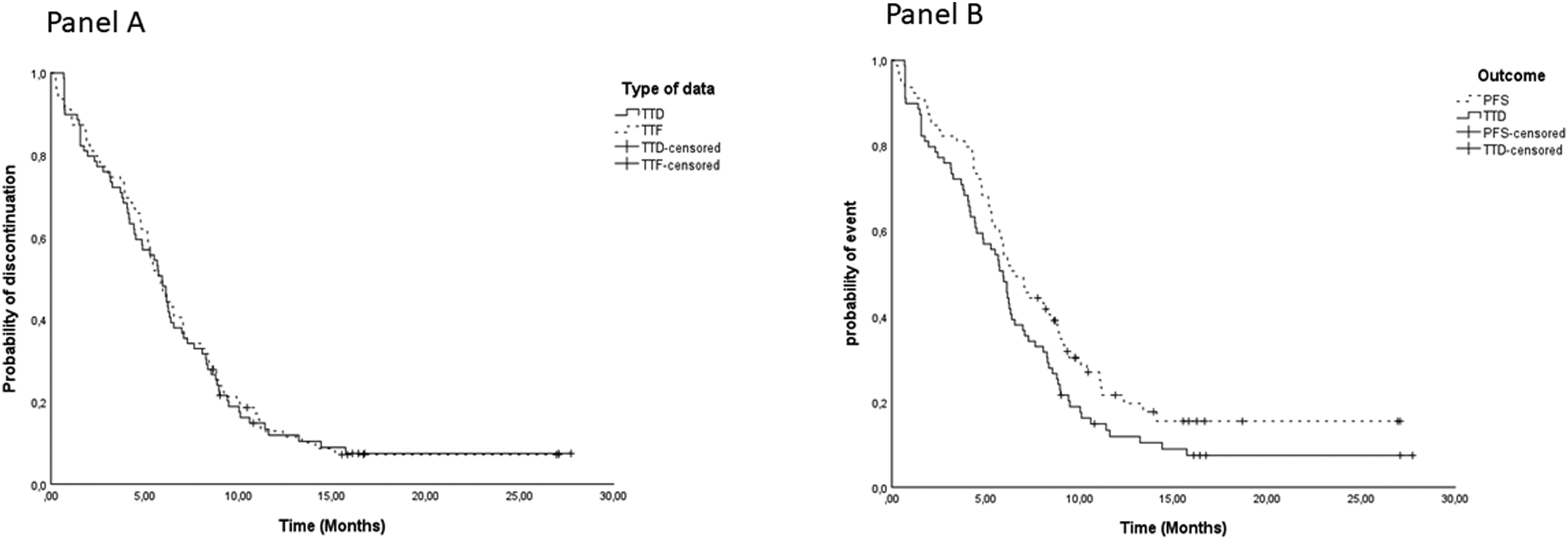
Time-to-discontinuation (TTD, administrative data) in comparison to time-to-failure (TTF, chart data) (Panel A) and to progression-free survival (PFS) (Panel B).
Overall survival
The median OS registered on 31 December 2022 was 11.8 months (95%CI: 8.6 −15) (Supplemental Figure 1) with a median observed follow-up of all patients of 9.3 months (Supplemental Table 1). We also investigated the correlation between TTD and OS, including N = 74 patients in the analysis (Table 2). The median time to discontinuation was longer for patients with PS = 0 than PS = 1–2 (TTD = 6.4 months vs 4.5 months, respectively, p = 0.026). Patients with liver metastases discontinued treatment earlier than those with brain metastases (TTD = 4.4 months vs 4.8 months, respectively). No metastases at baseline (liver or brain sites) were associated with a longer TTD in both cases. In regards to OS, the median time was quite similar for patients with a PS score 0 and 1–2 (11.8 months vs 11.0, respectively, p = 0.486). Patients with no liver metastases showed a longer median time of OS than patients with liver metastases: 15.9 months versus 6.6 months (p < 0.001), respectively (Table 2 and Figure 3, Panel A). The duration of treatment was similar, slightly longer for those without liver metastases (6.2 vs 4.4 months, p = 0.019) (Figure 3, Panel B). Surprisingly, those without brain metastases had a shorter time median of OS (10.4 months) than patients with brain metastases (15.9 months), but without any significant differences (p = 0.605) (Table 2).
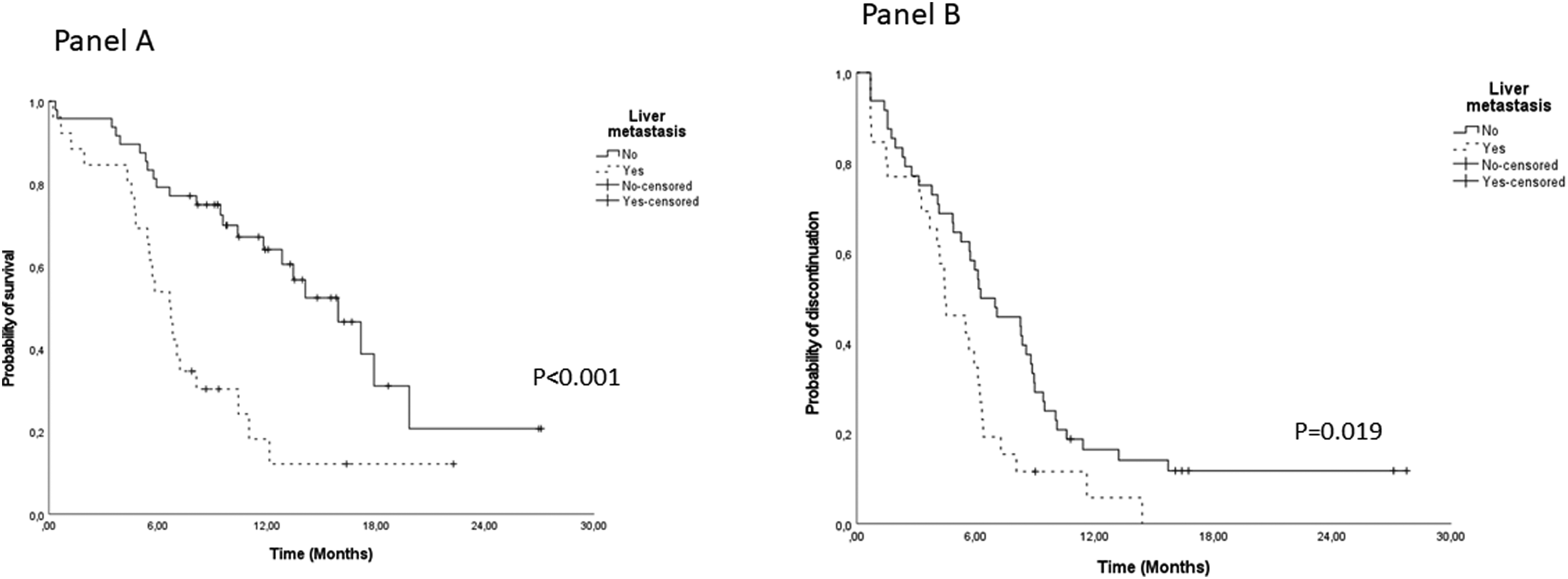
Overall survival (Panel A) and TTD (Panel B) by liver metastasis status.
Time-to-discontinuation and overall survival of cohort of patients stratified by different parameters.
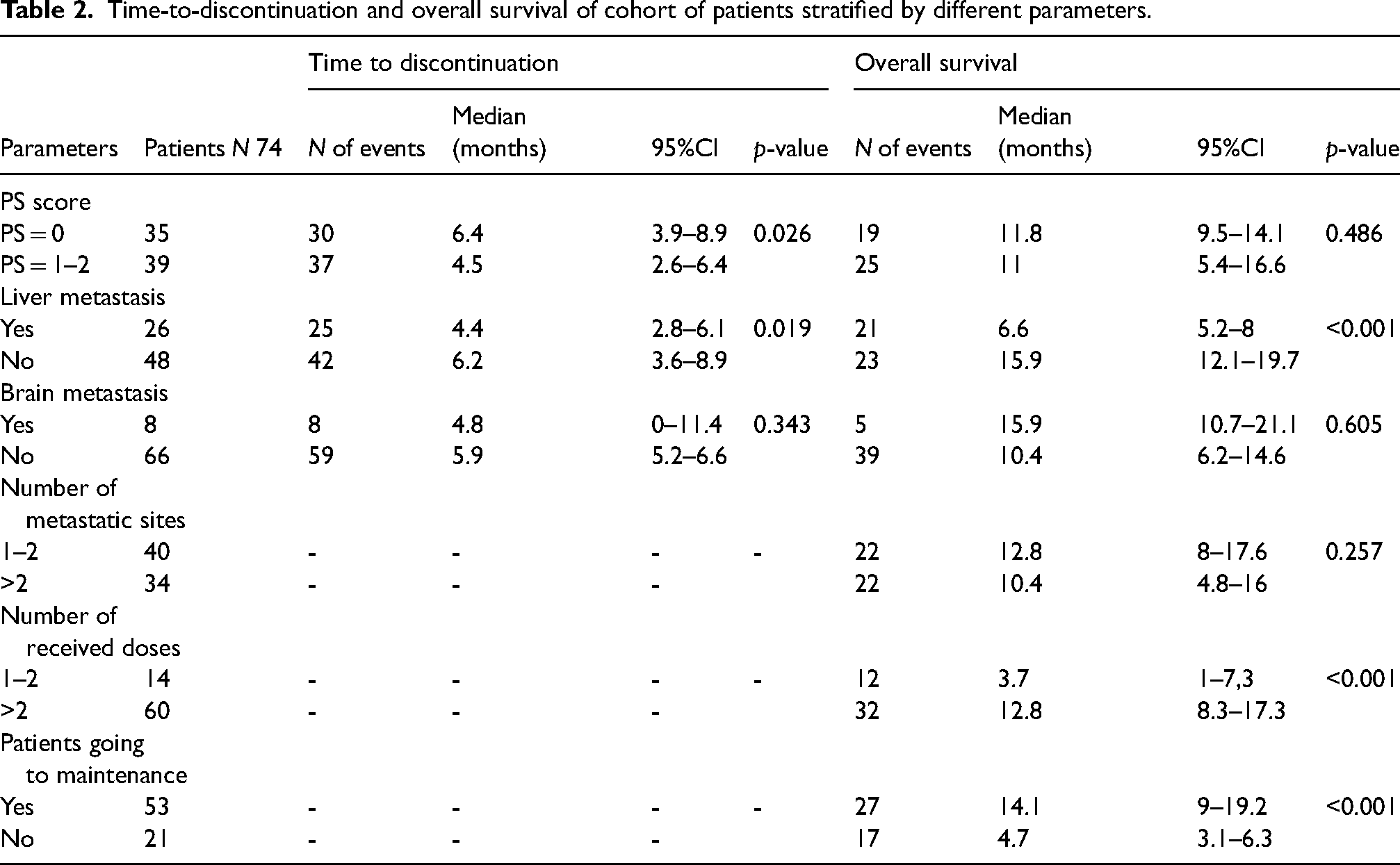
Patients with only one or two metastatic sites at baseline had better OS versus all the others with ≥3 metastatic sites (12.8 vs 10.4, p = 0.257). We evaluated OS in patients who received very few doses. Twelve patients received only one or two doses and OS was 3.7 months. In all other patients who received more than two doses of treatment, OS was 14.1 with p < 0.001.
The ECOG PS also plays a role in clinical outcomes. Patients with better ECOG PS (0–1) had slightly better results both for OS and TTD (median 11.8 months and 6.4 months, p = 0.486 and p = 0.026).
RDI and temporary drug suspension
The reasons for temporary drug suspension of treatments using RDI levels (dots on the green line in Supplemental Figure 2 with patients ordered by increasing treatment duration represented by the dots on the fuchsia line) was investigated. The mean value of RDI for the entire cohort was 87.4%. We therefore investigated the reasons why treatment was interrupted for patients with an RDI less than 80% and those who lost more than one treatment cycle (Supplemental Figure 3). In these seven treatments the main reason for treatment discontinuation and low RDI was pneumonia.
Discussion
Our real-world population was compared to the IMpower133 trial population 19 where most of the results were found to be somewhat similar. The median age of our cohort patients was 67 years (range: 44–85 years), slightly higher than that reported in the registration trial IMpower133 study 18 : 64 years (range: 26–90 years). Accordingly, the number of males reported was higher than females also in our data. Baseline PS = 1 was prevalent in our study (49.4%), which is in line with IMpower133 study (65%). 19 Moreover, in agreement with the previous study, more liver metastases were reported than brain metastases in the cohort of patients.
In our cohort of patients, we found a significant concordance in terms of median time to event among TTD, TTF and PFS (5.9 vs 5.8 vs 6.5 months, respectively). Specifically, TTF overlaps the TTD curve (p = 0.874). In an updated analysis of IMpower133 5 the median duration of treatment was 4.7 months (range: 0–29 months) where patients received a median of 7 doses (range: 1–39 doses). Results from our study were quite similar, with a median TTD of 5.9 months (range 1–27.5 months) and a median of 8 doses (range 1–39 doses) were received by patients.
The median PFS in IMpower133 at the updated analysis was 5.2 months (95%CI: 4.4–5.6) in the atezolizumab plus CP/ET arm, which was slightly lower than the median PFS registered in our cohort (6.5 months (95%CI: 5.2–7.8). Similarly, in regards to TTD, our results were quite similar to those reported by a previous study 20 evaluating TTD for reasons in a real-world cohort of 267 patients (5.9 months vs 4.7 months, respectively).
The OS in our research study was similar to previous findings from clinical trials and observational studies. In IMpower133, the median OS was 12.3 months (95% CI: 10.8 −15.8) and also in a retrospective cohort by Patel et al. 21 consisting of 65 patients (with no report of baseline characteristics) treated with atezolizumab plus standard chemotherapy, a median OS of 12.3 months (95% CI: 8–15.1 months) was registered. In our study, the OS was lower (11.8 months, 95% CI:8.6–15.0), but the median follow up was also lower compared to IMpower133 (9.3 vs 22.9 months, respectively).
In our study, patients with 1–2 or more metastatic sites observed at diagnosis, median OS were respectively, 12.8 and 10.4 months. The number of patients enrolled with brain metastases was small (N = 9) and surprisingly, the median OS was 15.9 vs 10.4 months in patients with brain metastases versus patients without brain metastases (p not significant = 0.605). Brain metastases are usually associated with poor clinical outcomes, even if in a retrospective study by Patel 19 the risk of death for patients with brain metastases was 82%, lower than those without brain metastases.
However, further clinical trials are needed to investigate the role of immunotherapy in patients with SCLC who have brain metastases to confirm these results. Conversely, the role of liver metastasis in reducing survival seems to be confirmed in our study. We observed a difference in OS for patients with liver metastases who presented lower OS versus patients without liver metastases (6.6 vs 15.9 months, p=<0.001). According to our results, updated results from IMpower133 showed that OS was better in patients without liver metastases than that without brain metastases.
Real-world studies may better estimate the dose received by patients in real clinical practice. The mean RDI of atezolizumab received in this study was 87.4% and the median calculated was 89%; in IMpower133 the authors observed a median dose intensity of 94.9%.
A delay in the sequence of treatment cycles decreased the dose intensity. Patients who received less than 80% of dose intensity and those who lost more than 1 cycle of 21 days were investigated in order to identify the reasons for the discontinuation of the study drug. Seven patients were identified and, among these, six patients had infections, mainly represented by pneumonia (N = 5) associated with interstitial lung disease and neutropenia requiring antibiotic treatments. In patients with relevant temporary treatment cessation, adverse events such as pneumonia, immune-related or non-related, were responsible for these drug suspensions. Identifying risk factors for ICI-pneumonia and interstitial lung disease could help avoid these adverse events and discontinuation of atezolizumab. However, these risk factors associated with ICI-pneumonia are still unclear thus calling for an urgent need to identify them using reliable and accessible parameters. 22 In one case, a herpes zoster infection was reported.
Finally, we had 18.9% of patients receiving only one or two doses. The median OS in these patients was only 3.7 months vs 12.8 months in all the others receiving longer treatments (p < 0.001); in poor prognosis patients, etoposide plus carboplatin or best palliative care, could be considered alternative options.
Like every study, in our study there are some strengths and limitations. First, we provide a more realistic overview of a patient population, which is more heterogeneous than those enrolled in clinical trials. For example, our cohort included patients of all ECOG PS levels. Moreover, we included patients that died within the first three months following the first dose in the survival analysis, who were deemed to be unlikely to benefit from immunotherapy (n = 9). We included all patients treated in a real-world setting of two different Italian hospitals which represents a further strength of our study.
A study limitation may refer to the cohort size. In fact, our study population included only 79 patients, where very few patients had brain metastases and/or low RDI, so results should be considered in relation to this aspect.
Conclusions
Our study showed that TTD, calculated from the administration data is a useful proxy of TTF (as registered in the clinical chart) which includes PFS, toxicity and clinical worsening. TTD is a real-world outcome that can be used to demonstrate the efficacy of drugs used for administered therapies when duration of treatment is until disease recurrence or unacceptable toxicity. It can be used as an end point for RWE studies, where the evaluation is less structured and standardized.
Overall, the data suggest that real-world clinical benefits were observed in patients with ES-SCLC treated with first-line atezolizumab plus CP/ET.
The benefit of immunotherapy in patients with liver metastases seems to be less pronounced, but given that some of these patients also appear to benefit, highlights the need for further research to identify better predictive biomarkers.
Supplemental Material
sj-pptx-1-opp-10.1177_10781552241242096 - Supplemental material for A real-world multicentre evaluation of atezolizumab plus platinum-etoposide chemotherapy as first-line treatment in patients with extensive-stage small cell lung cancer in Italy
Supplemental material, sj-pptx-1-opp-10.1177_10781552241242096 for A real-world multicentre evaluation of atezolizumab plus platinum-etoposide chemotherapy as first-line treatment in patients with extensive-stage small cell lung cancer in Italy by Felice Musicco, Chiara Fulgenzio, Antonia La Malfa, Nicoletta Jannitti, Antonietta Vitiello, Silvia Carpano, Francesca Fusco, Federico Cappuzzo, Irene Terrenato, Isabella Sperduti, Piera Polidori, Domenico Tarantino, Lidia Di Cerbo, Marcello Pani, Valentina Isgrò and Ruggero Lasala in Journal of Oncology Pharmacy Practice
Footnotes
Author Contributions
FM was involved in protocol development, literature research, conceptualization, methodology, data curation, writing, review & editing, supervision, gaining ethical approval and conceived the study; CF and VI wrote the first draft of the manuscript and were involved in review & editing and literature research; ALM, NJ, FF, FC, PP, LDC, MP were involved in review and editing; AV, SC in data curation; IT and IS were involved in formal and data analysis, DT in gaining ethical approval and data curation; RL researched literature and was involved in conceptualization, methodology, review & editing. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
