Abstract
Introduction
The use of immune checkpoint inhibitors, which have an important role in the treatment of malignant tumors, is increasing. Although rarely observed, neurological immune-related adverse events associated with immune checkpoint inhibitors result in high morbidity and mortality. Small cell lung cancer is a common cause of neurological paraneoplastic syndromes. The differentiation between paraneoplastic syndromes and neurological immune-related adverse events is important in patients using immune checkpoint inhibitors. Cerebellar ataxia caused by atezolizumab is a rare immune-related adverse event.
Case report
In this context, we present a 66-year-old man with small cell lung cancer who developed immune-mediated cerebellar ataxia after three cycles of atezolizumab, a programmed cell death ligand-1 inhibitor. The admission of brain and spinal gadolinium-based contrast-enhanced magnetic resonance imaging supported the preliminary diagnosis and indicated leptomeningeal involvement. However, the blood tests and a lumbar puncture did not reveal any structural, biochemical, paraneoplastic, or infectious cause.
Management and outcome
High-dose steroid treatment resulted in an improvement in the radiological involvement, as evidenced both clinically and on follow-up whole spine magnetic resonance imaging. Therefore, the immunotherapy was discontinued. The patient was discharged on day 20 without neurological sequelae.
Discussion
In light of this, we present this case to emphasize the differential diagnosis of neurological immune-related adverse events originating from immune checkpoint inhibitors, which require rapid diagnosis and treatment, and clinically similar paraneoplastic syndromes and radiologically similar leptomeningeal involvement, in a case of small cell lung cancer.
Keywords
Introduction
The U.S. Food and Drug Administration has approved three different groups of immune checkpoint inhibitors (ICIs) for the treatment of various cancers: programmed cell death protein-1 (PD-1) inhibitors (nivolumab, pembrolizumab, and cemiplimab), programmed cell death ligand-1 (PD-L1) inhibitors (atezolizumab, durvalumab, and avelumab), and a cytotoxic T-lymphocyte-associated protein-4 (CTLA-4) inhibitor (Ipilimumab). 1 Despite their clinical benefits, ICIs can impair the immune tolerance to self-antigens and induce off-target toxicities called immune-related adverse events (irAEs). 2
Although any organ system can be affected, the main irAEs include diarrhea, colitis, hepatitis, skin toxicities, and endocrinopathies such as hypophysitis and thyroid dysfunction. 3 Although early studies indicated that the incidence of neurological toxicity associated with ICIs, which is less common, was around 1%, recent studies have shown higher rates. 4 Neurological irAEs include myasthenia gravis or myasthenic syndrome, myasthenia gravis with myositis overlap, aseptic meningitis, encephalitis, Guillain-Barré-like syndrome, and other peripheral neuropathy phenotypes and demyelinating disorders. 5 The median time to the onset of nervous system toxicity is 4 (range 1–68) weeks. 4 irAEs can affect any part of the nervous system and should be included in the differential diagnosis of new neurological symptoms in patients receiving ICIs. Cerebellar ataxia or impaired balance and coordination, is commonly seen in neurology practice. Known causes of cerebellar ataxia include stroke, infectious diseases, immune-mediated damage, paraneoplastic syndromes, vitamin deficiency, structural lesions, metabolic diseases, and toxins. 6 A definitive diagnosis of cerebellar ataxia is based on a comprehensive anamnesis and physical examination, serum and cerebrospinal fluid analysis, and neuroimaging studies. 7
In this context, this report presents a 66-year-old man who presented to the emergency department with a headache, inability to sleep, and involuntary limb movements and was diagnosed with atezolizumab-related cerebellar ataxia.
Case report
A 66-year-old man presented to the emergency department with a headache of gradually increasing severity, inability to sleep, and involuntary limb movements he had experienced for about 10 h. The movement disorder was diagnosed as trunk ataxia and extremity myoclonus compatible with cerebellar ataxia. The patient had a history of diabetes mellitus, hypertension, coronary artery disease, and metastatic lung small cell neuroendocrine carcinoma metastasized to the liver without brain metastasis at the time of diagnosis. He had no history of autoimmune diseases. His symptoms had started 3 weeks after the third cycle of carboplatin treatment (area under the curve (AUC) of 5) with 1200 mg atezolizumab on day 1 and 100 mg/m2 etoposide on days 1–3.
Investigations
The hemogram, renal, liver, and thyroid function tests and the vitamin B12 test were unremarkable. Contrast-enhanced brain magnetic resonance imaging (MRI) revealed minimal hyperintense cerebrospinal fluid (CSF) in the subarachnoid space, adjacent to the cerebral hemispheres. Leptomeningeal involvement was suggested by contrast enhancement seen in cervical nerve roots, thickening at the distal thoracic spinal cord level, conus medullaris localization and cauda equina fibers, and diffuse contrast enhancement in the whole spine MRI. The CSF contained glucose 117 mg/dL, protein 122 mg/dL, and no neutrophils or organisms. CSF was negative on bacterial culture and polymerase chain reaction for herpes simplex viruses 1 and 2 and cytomegalovirus. All parameters related to paraneoplastic syndromes, that is, anti-amphiphysin, anti-CV2, anti-Ma2/Ta, anti-Ri/Anna-2, anti-Yo/Pca-1, anti-Hu/Anna-1, anti-Recoverin, Sox1/Agna, Zic4, Gad65, Tr (Dner), and anti-Titin antibodies, were negative. Blood and urine cultures were also negative. Positron emission tomography-computed tomography (PET-CT) indicated a nearly complete treatment response.
Differential diagnosis
Bacterial meningitis, herpes simplex encephalitis, and the paraneoplastic neurological syndrome were ruled out. The differential diagnosis focused mainly on treatment toxicities and leptomeningeal metastases. It was thought that the cerebellar ataxia might be an irAE, given that the patient was suspected to have leptomeningeal involvement on MRI, yet had a near-complete treatment response on PET-CT, no malignant cells in the CSF, a negative paraneoplastic panel test, high CFS protein, and glucose levels, and the patient was using an ICI. His Naranjo Probability Score was + 8, supporting the diagnosis of adverse drug reaction. 8 So, he was considered as an irAEs.
The treatment and outcome
The patient was initially given 120 mg (2 mg/kg) of methylprednisolone for 3 days. Then, he was given 60 mg (1 mg/kg) methylprednisolone, which was tapered as his involuntary movements improved and muscle strength increased. The patient improved markedly within a week, suggesting an irAE. On a follow-up MRI taken on day 15, the previous findings had resolved (Figure 1). The patient was discharged on day 20 without any neurological sequelae. He has prescribed 10 mg of methylprednisolone, which was to be tapered depending on his treatment response. Subsequently, the atezolizumab was discontinued in the fourth treatment cycle, while carboplatin plus etoposide was continued.
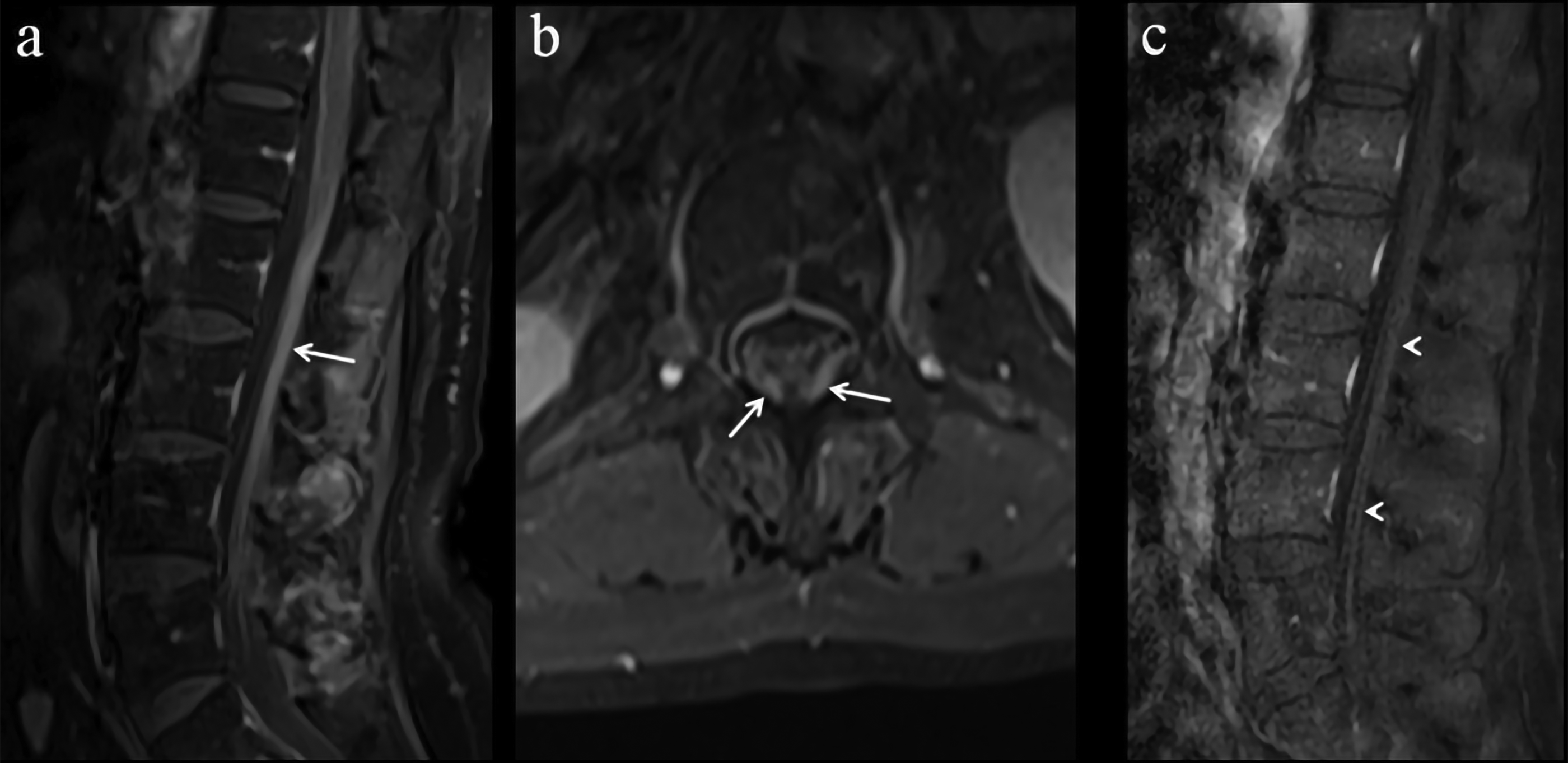
On initial presentation, post-contrast fat-saturated sagittal (a) and axial (b) T1-weighted images reveal thickening and diffuse contrast enhancement of the conus medullaris and nerve roots of the cauda equina (arrows), which indicate leptomeningeal metastases. Three weeks after the initial presentation, a sagittal fat-saturated post-contrast T1-weighted image (c) shows that the conus medullaris and nerve roots of the cauda equina have returned to normal thickness, with no contrast enhancement (arrowheads), indicating a positive response to treatment.
Discussion
This case report features a patient who developed cerebellar ataxia 3 weeks after the third cycle of atezolizumab plus carboplatin and etoposide. An early diagnosis followed by high-dose steroid therapy led to a marked improvement in the patient's symptoms within a week.
The clinical benefits of ICIs have resulted in their increased use, which in turn has led to an increase in related irAEs. 9 Rare side effects, such as encephalitis,10–14 cerebellar ataxia,15,16 aseptic meningitis, 17 posterior reversible encephalopathy syndrome, 18 opsoclonus, and myoclonus 19 have been reported with atezolizumab-induced central nervous system (CNS) involvement. Several case reports have featured encephalitis as a CNS neurotoxicity. 11 Similar to our case, two patients with cerebellar ataxia associated with atezolizumab have been described. In addition, three patients who developed encephalitis after atezolizumab treatment had diffuse leptomeningeal involvement on MRI, as in our case, and resolution we observed after steroid therapy11,14–16,20 (Table 1).
Cases of cerebellar ataxia associated with atezolizumab reported in the literature.
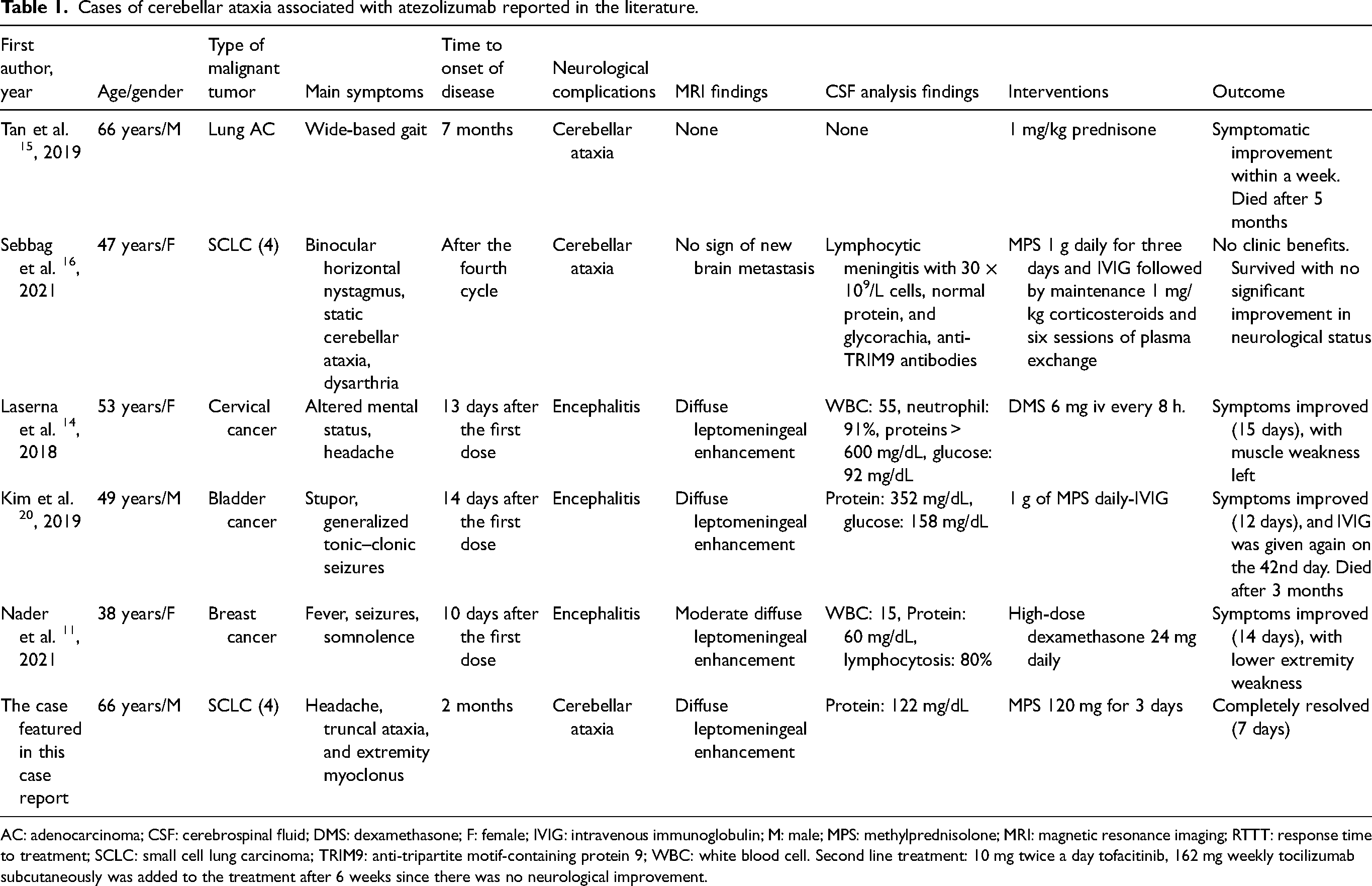
AC: adenocarcinoma; CSF: cerebrospinal fluid; DMS: dexamethasone; F: female; IVIG: intravenous immunoglobulin; M: male; MPS: methylprednisolone; MRI: magnetic resonance imaging; RTTT: response time to treatment; SCLC: small cell lung carcinoma; TRIM9: anti-tripartite motif-containing protein 9; WBC: white blood cell. Second line treatment: 10 mg twice a day tofacitinib, 162 mg weekly tocilizumab subcutaneously was added to the treatment after 6 weeks since there was no neurological improvement.
In most reports, encephalitis developed within 14 days after the first atezolizumab dose.10–14 In comparison, cerebellar ataxia occurred 8 or more weeks after the first dose of atezolizumab, as in our case.15,16 Our patient presented with a headache and movement disorder compatible with cerebellar ataxia. Neurotoxicity with ICI-associated CNS involvement was diagnosed after other possibilities, such as cancer metastasizing to the CNS, seizure activity, infection, and metabolic derangement, was ruled out based on the clinical evaluation, serum, and CSF analyses, and detailed radiological imaging. 4 Blood, urine, sputum, and CSF samples from the patient grew no microorganisms. In addition, the sudden onset of cranial symptoms with a near-complete response after the third treatment cycle, rapid response to steroid therapy, and negative paraneoplastic antibodies are not typical of paraneoplastic syndromes. 21 Leptomeningeal carcinomatosis was ruled out by the negative CSF cytology and loss of leptomeningeal involvement after corticosteroid therapy. The patient's CSF contained high total protein levels, as seen in some cases of encephalitis after immunotherapy.12–14
The patient was started on high-dose steroid therapy, as recommended in the American Society of Clinical Oncology guidelines for the treatment of irAEs. 4 His complaints regressed significantly within 2 weeks in line with the literature11,15,22; therefore, the atezolizumab was discontinued permanently. Some studies have concluded that the development of neurological irAEs may be considered a positive predictor of treatment response.23,24 In parallel, our patient had a nearly complete treatment response.
In conclusion, when any unexplained CNS-related finding is detected in a patient receiving immunotherapy, ICI-related neurotoxicity, a rare but potentially fatal condition, should be considered. When ICI-related neurotoxicity is suspected, the immunotherapy should be discontinued and corticosteroids administered as the first treatment. Early diagnosis and treatment are important for treatment success. In addition, other immunomodulators, such as plasma exchange, intravenous immunoglobulin, 20 or infliximab may help to treat steroid-resistant symptoms. 2
Authors’ contribution
TK, FA, SY, NB, and GUE: Literature review, conceptualization, formal analysis, data collection. TK, GUE: Visualization, writing the original draft. SY: Radiology images. TK, FA, SY, NB, and GUE: Approved the final version of the manuscript.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical statement
Written informed consent was obtained from the individual for the publication of any potentially identifiable images or data included in this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
