Abstract
Introduction
Second-generation Bruton's tyrosine kinase (BTK) inhibitors, acalabrutinib and zanubrutinib, are preferred agents for the treatment of relapsed and/or refractory mantle cell lymphoma (MCL) over first-generation BTK inhibitor, ibrutinib. The comparative safety and efficacy of these two agents have not been studied. Currently, the decision between using one second-generation BTK inhibitor over the other is largely dependent on provider preference, cost, organ dysfunction, presence of drug–drug interactions, adherence considerations, and theorized differences in safety outcomes due to the lack of head-to-head trials in MCL.
Methods
This retrospective, observational study seeks to provide real-world data on the safety and efficacy of second-generation BTK inhibitors in the setting of relapsed and/or refractory MCL.
Results
Thirty-eight patients treated with a second-generation BTK inhibitor were evaluated. Ten percent of patients experienced a select adverse drug event (ADE) in the acalabrutinib group that included hypertension and major hemorrhage with no patients experiencing a select ADE in the zanubrutinib group.
Conclusions
Results support historical data that acalabrutinib and zanubrutinib have a more favorable safety profile compared to ibrutinib in MCL.
Introduction
Bruton's tyrosine kinase (BTK) is an important protein involved in the proliferation, maturation, and differentiation of B-cells. Due to its important role in the B-cell antigen signaling cascade, BTK has been identified as a targetable protein in the development of treatment options for B-cell malignancies. 1 Historically, ibrutinib was the first covalent BTK inhibitor approved in 2013 for mantle cell lymphoma (MCL), a B-cell lymphoma, in the second-line setting. 2 Ibrutinib also had a role in the first-line setting as a lead in to decrease chemotherapy exposure and in combination with rituximab for elderly patients 65 years or older.3,4 Recently, ibrutinib's indication for MCL was voluntarily withdrawn due to failure to meet the requirements related to its accelerated approval for MCL with results from the SHINE study showing increased adverse reactions in the ibrutinib arm compared with placebo.5,6 Following ibrutinib, acalabrutinib, a second-generation covalent BTK inhibitor, was approved in 2017 for MCL in the relapsed and/or refractory setting. Shortly after in 2019, another second-generation covalent BTK inhibitor, zanubrutinib, was also approved for MCL in the relapsed and/or refractory setting.1,7 While ibrutinib has been studied extensively in MCL, there is lack of guidance informing the selection of one second-generation BTK inhibitor over the other for MCL in the relapsed and/or refractory setting. To date, there is a lack of head-to-head trials comparing the safety and efficacy of acalabrutinib and zanubrutinib in MCL. It is also unknown how resistance or intolerance to ibrutinib affects response to subsequent therapy with a second-generation BTK inhibitor.
The decision between using one second-generation BTK inhibitor over the other is largely dependent on provider preference, cost, organ dysfunction, presence of drug–drug interactions, adherence considerations, and theorized differences in safety outcomes due to the lack of head-to-head trials in MCL. Second-generation BTK inhibitors have been shown to have a better tolerability profile in head-to-head trials that compared ibrutinib to acalabrutinib in chronic lymphocytic leukemia (CLL) and ibrutinib to zanubrutinib in Waldenstrom's macroglobinemia (WM).8,9
Randomized trials for ibrutinib have demonstrated increased risk of atrial fibrillation, hypertension, bleeding, and infections which are likely due to off-target inhibitions of various kinases and pathways. 10 The cardiovascular side effects of ibrutinib such as atrial fibrillation is suggested to be due to the inhibition of BTK as well as Tec protein tyrosine kinase (TEC) which causes decreased phosphoinositide 3-kinase (PI3 K)/protein kinase B (Akt) signaling. Irreversible binding to human epidermal growth factor receptor 2 (HER2) by ibrutinib is another mechanism of cardiomyocyte inhibition that leads to cardiac side effects such as hypertension and atrial fibrillation. In comparison, acalabrutinib and zanubrutinib do not have inhibitory effects on HER2 and acalabrutinib does not exhibit inhibitory effects on TEC whereas zanubrutinib weakly inhibits TEC. Off-target inhibition of TEC also contributes to increased incidence of bleeding as well as opportunistic infections. These differences in off-target effects contribute to the improved side effect profiles of second-generation BTK inhibitors. In the ELEVATE-RR trial comparing acalabrutinib to ibrutinib in patients with relapsed or refractory CLL, there were fewer incidences of atrial fibrillation, hypertension, and bleeding events in the acalabrutinib group compared to the ibrutinib group. 8 Similar results were seen in the ASPEN trial comparing zanubrutinib and ibrutinib in patients with WM and MYD88L265P disease. 9 There were fewer incidences of atrial fibrillation or atrial flutter, hypertension, bleeding, and pneumonia in the zanubrutinib group compared to the ibrutinib group. While second-generation BTK inhibitors are postulated to be better tolerated than ibrutinib in MCL, the comparative safety profiles as well as the efficacy of the second-generation BTK inhibitors are unknown.
Acalabrutinib and zanubrutinib are approved by the U.S. Food and Drug Administration for the treatment of relapsed and/or refractory MCL.11,12 This retrospective study seeks to describe the safety and efficacy of second-generation BTK inhibitors in patients with MCL in the real-world setting.
Methods
This was a single-center, retrospective, observational study of patients treated with acalabrutinib or zanubrutinib between October 2017 and August 2022. Patients were followed up for a minimum of nine months following initiation of the BTK inhibitor. The time frame was chosen based on the median onset of atrial fibrillation, hypertension, hemorrhagic events, and opportunistic infections from pooled safety analyses of patients taking acalabrutinib or zanubrutinib.13,14 Patients were included if they were at least 18 years of age with diagnosis of MCL, treated with at least one dose of acalabrutinib or zanubrutinib, with regular follow-up, and had available prescription dispense history in the electronic medical record (EMR). Patients were excluded if they received a BTK inhibitor in combination with an agent other than rituximab or if they were treated on protocol.
The primary endpoint of this study was to determine the incidence of atrial fibrillation, hypertension, major hemorrhage, and invasive fungal infections. Diagnosis of atrial fibrillation was determined if there was confirmation with electrocardiogram (ECG) or new rate or rhythm medication started after initiation of acalabrutinib or zanubrutinib in accordance with the Common Terminology Criteria for Adverse Events (CTCAE) definition. 15 Hypertension was identified if a new diagnosis of hypertension was added to the problem list or if patient started new antihypertensive therapy. Major hemorrhage was defined as diagnosis of major hemorrhage including severe bleeding, severe epistaxis, upper or lower gastrointestinal (GI) bleeding according to the International Society on Thrombosis and Hemostasis (ISTH) definitions and grade 3 or 4 thrombocytopenia. 16 Invasive fungal infection was identified if a patient experienced an aspergillus or pneumocystis jiroveci infection or started a new antifungal medication. 17 The secondary endpoint was to evaluate the efficacy of acalabrutinib and zanubrutinib using time to treatment failure defined as date of initiation to date of discontinuation due to disease progression, toxicity, or death.
All data collection was conducted by one investigator through chart review which included assessment of patients’ fill history of prescription medications, review of note documentation at each visit with the primary medical oncologist, past medical history as documented in the EMR, and results from laboratory draws and tests such as electrocardiograms (ECG). The other two investigators reviewed the data collected and intermittently met with the investigator who collected the data to ensure that all data was appropriate. The data was evaluated with descriptive statistics.
Results
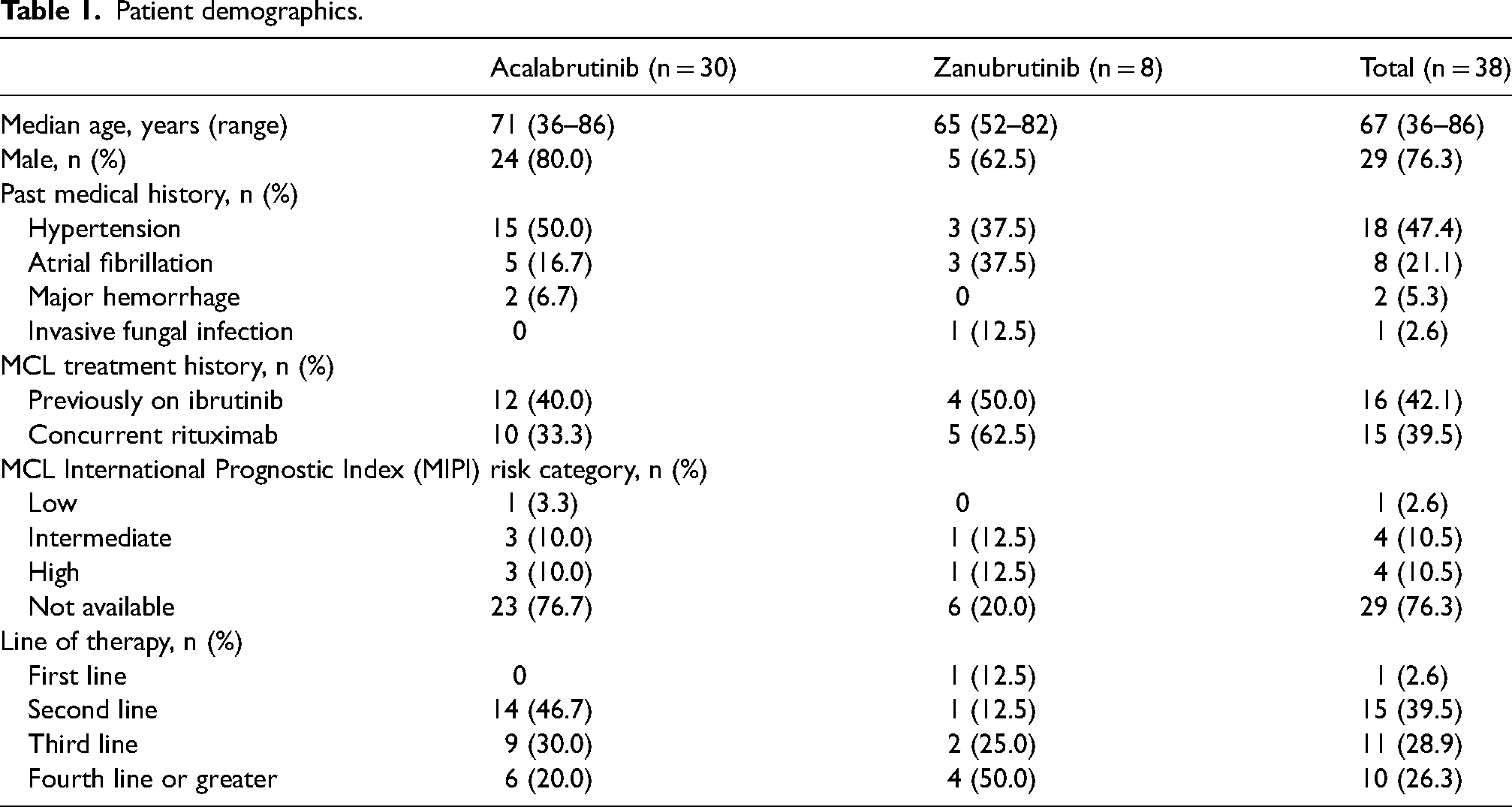
A total of 255 patients were screened with 30 patients included in the acalabrutinib group and eight patients included in the zanubrutinib group at a single NCI-designated cancer center (Figure 1). The baseline characteristics of the patients were consistent with the MCL population with a median age of 67 years and 76.3% male (Table 1). Baseline past medical history was collected and 47.4% of patients had pre-existing hypertension and 21.1% had a history of atrial fibrillation. Most patients received a second-generation BTK inhibitor in the second-line setting (39.5%) followed by third-line (28.9%) and fourth-line or greater (26.3%). One patient received zanubrutinib as first-line treatment.
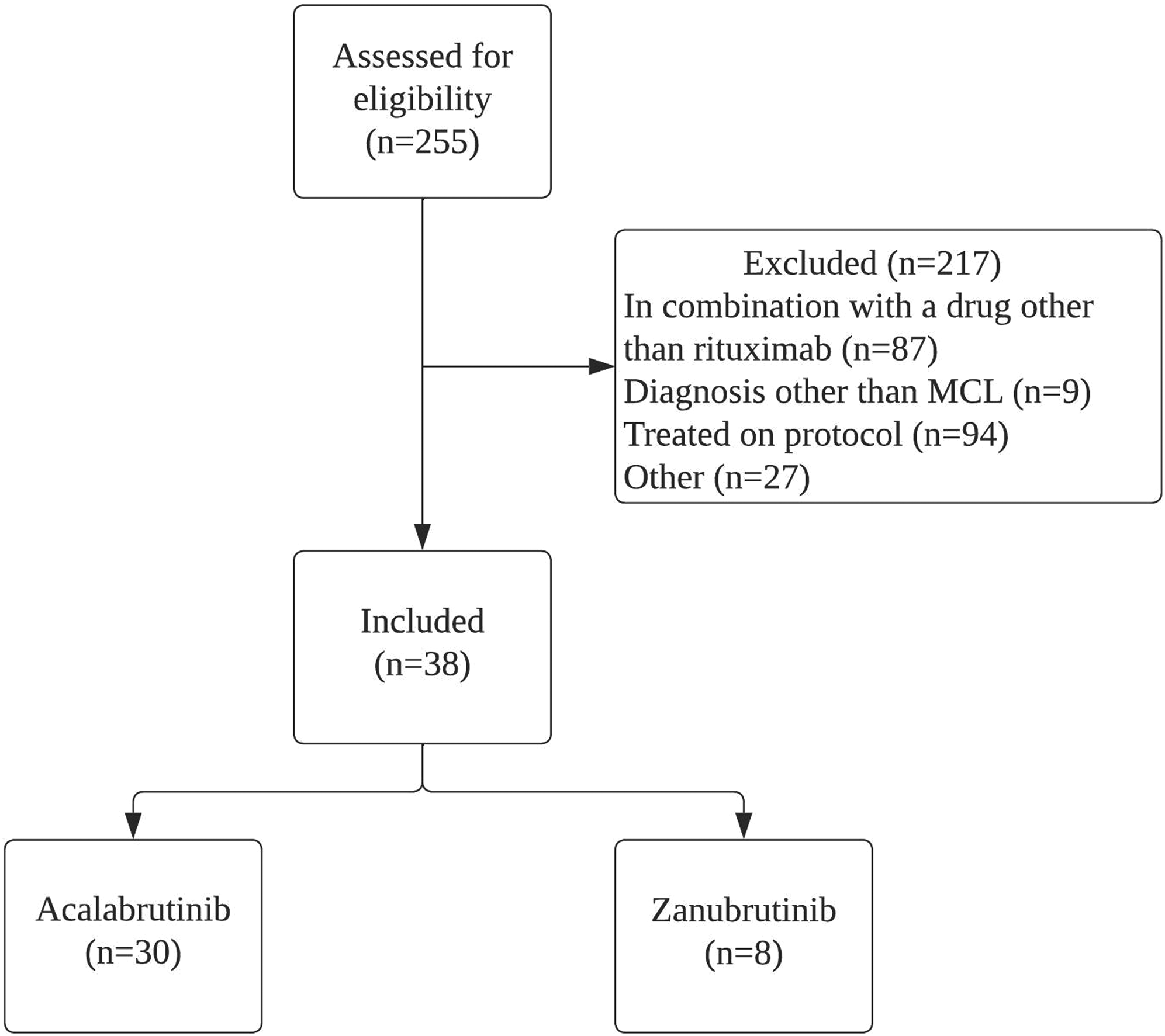
Patient enrollment.
Patient demographics.
Select treatment ADEs leading to drug discontinuation occurred in 10% of patients who received acalabrutinib with two (6.7%) patients experiencing a grade 3 or higher adverse effect (atrial fibrillation, symptomatic bleed) with patient characteristics detailed in Table 2. None of the patients in the zanubrutinib group experienced a select treatment ADE. No patients in either group experienced hypertension or an invasive fungal infection.
Characteristics of patients with grade ≥2 adverse drug events.
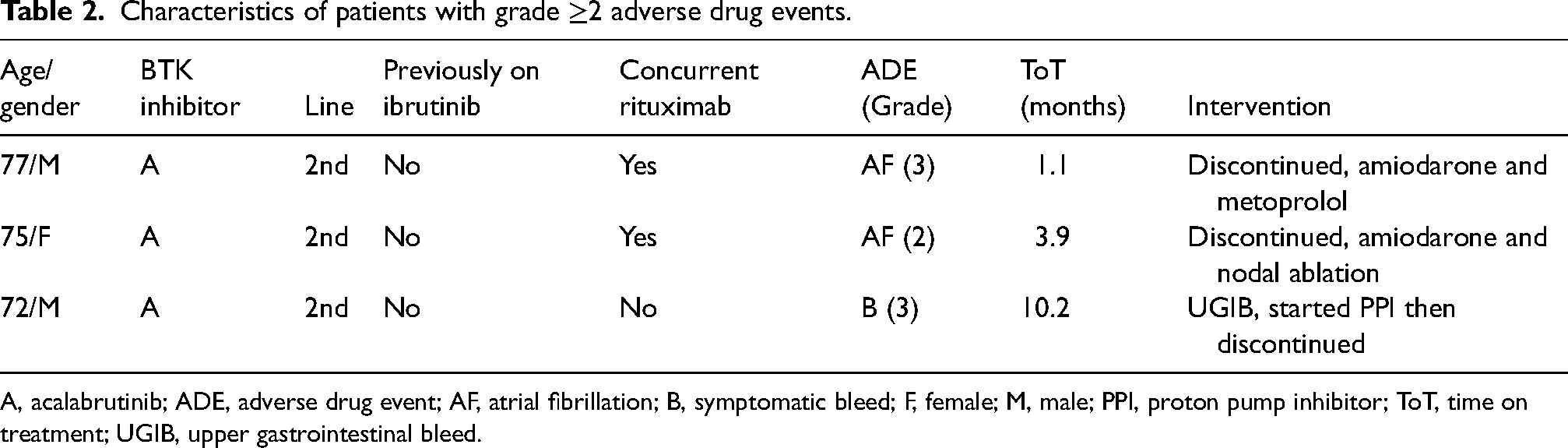
A, acalabrutinib; ADE, adverse drug event; AF, atrial fibrillation; B, symptomatic bleed; F, female; M, male; PPI, proton pump inhibitor; ToT, time on treatment; UGIB, upper gastrointestinal bleed.
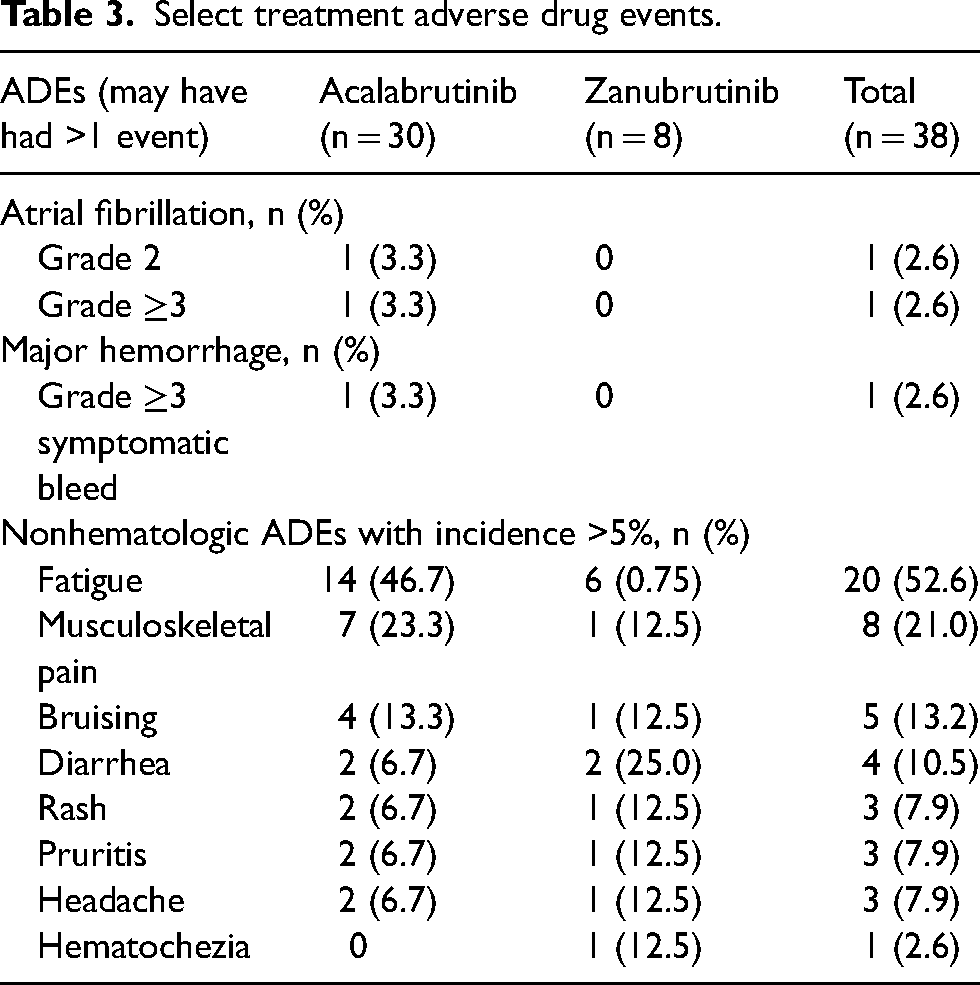
Any-grade nonhematologic ADEs occurred in 83.3% and 87.5% of patients in the acalabrutinib and zanubrutinib groups, respectively. The most common nonhematologic ADEs included fatigue (52.6%), musculoskeletal pain (21.0%), and bruising (13.2%) (Table 3). In the acalabrutinib group, 20% of patients discontinued therapy due to an ADE while no patients discontinued therapy in the zanubrutinib group due to an ADE. One patient in the acalabrutinib arm with a history of arthritis had therapy held and dose reduced to 100 mg once daily due to arthralgias and myalgias which initially improved with dose reduction. Acalabrutinib was eventually discontinued after 8 months of therapy due to worsening arthralgias and myalgias.
Select treatment adverse drug events.
The median time on treatment for acalabrutinib and zanubrutinib was 4.2 (range, 0.3–44.1) and 10.6 (range, 0.2–29.9) months, respectively (Table 4). Three (10.0%) of the acalabrutinib patients and 5 (62.5%) of the zanubrutinib patients were still on treatment at time of data evaluation. Of the 12 patients on acalabrutinib and 4 patients on zanubrutinib previously treated with ibrutinib, the median time on treatment for patients was 3.3 (range, 0.3–32.0) and 14.0 (range, 0.2–29.9) months, respectively. Patients were switched from ibrutinib to second-generation BTK inhibitor primarily due to adverse effects in six patients (diarrhea, fatigue, GI infection, thrombocytopenia, subconjunctival hemorrhage, and conjunctival hemorrhage) or disease progression in six patients. Of the six patients who switched from ibrutinib to a second-generation BTK inhibitor due to disease progression, median time on treatment was 3.2 (range, 0.2–14.6) months.
Treatment characteristics.

Discussion
The National Comprehensive Cancer Network (NCCN) Guidelines on B-cell lymphomas were updated to now recommend second-generation covalent BTK inhibitors, acalabrutinib and zanubrutinib, as preferred agents of choice for relapsed and/or refractory MCL over the combination of ibrutinib with rituximab. 7 This recommendation was made based on the more favorable side effect profile of second-generation BTK inhibitors in head-to-head trials for other B-cell malignancies without compromising efficacy.
While acalabrutinib and zanubrutinib are both preferred agents for second-line and subsequent therapy in relapsed and/or refractory MCL, there are no head-to-head studies to inform choice of one agent over the other based on safety or efficacy considerations. Reported side effects in this study were consistent with known side effect profiles of the second-generation BTK inhibitors with low rates of select ADEs such as hypertension and major bleeding. This data supports the previously reported lower incidence of ADEs with second-generation BTK inhibitor in the MCL population.
Limitations of this study include the single-center retrospective design which may not capture patients who may have experienced an ADE at an outside facility that does not use the same EMR system if not reported by the patient, low patient numbers due to patients excluded for being treated on protocol or in combination with a drug other than rituximab, and potential for underreporting of adverse events. While the results of this study demonstrate tolerability of acalabrutinib and zanubrutinib, consistent with results from previous studies comparing the second-generation BTK inhibitors with ibrutinib, prospective head-to-head studies still need to be conducted to determine the comparative safety and efficacy of second-generation BTK inhibitors in MCL.
Footnotes
Author Contributions
JL researched the literature and wrote the manuscript including figure and tables. BD and BP conceived the research idea and assisted with the study design, reviewing, and editing of the manuscript. All authors reviewed and approved the final version of the manuscript.
Acknowledgement
Chun Feng, MA, Certified ASQ Six Sigma Black Belt, Sr. Informatics Analyst—Department of Pharmacy—Quality and Regulatory, Division of Pharmacy, University of Texas M.D. Anderson Cancer Center.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
