Abstract
Objective
To provide a comprehensive review of the pharmacokinetics, pharmacodynamics, safety, and efficacy of a new Food and Drug Administration (FDA) approved Bruton's tyrosine kinase inhibitor (BTKi), pirtobrutinib for relapsed/refractory mantle cell lymphoma (r/r MCL).
Data sources
A literature search was conducted through PubMed MEDLINE, ClinicalTrials.gov, and the FDA website (January 2018-January 2023) using the following key terms: lymphoma, non-covalent, Bruton's tyrosine kinase (BTK), and relapse. Relevant English language monographs, studies, and abstracts conducted in humans were reviewed and considered.
Data summary
Pirtobrutinib, a novel non-covalent BTKi, was granted accelerated approval for treatment of r/r MCL on January 27th, 2023, based on an open-label, multi-center phase 1/2 BRUIN trial. In phase l, 61 patients with r/r MCL received seven dose levels of pirtobrutinib (25–300 mg). There was no reported maximum tolerated dose or dose-limiting toxicities during this study period. In phase 2, 56 r/r MCL evaluable efficacy patients received pirtobrutinib 200 mg daily. The overall response rate (ORR) was 52% (95% CI 38–65). Additionally, patients who received a previous covalent BTKi, ORR was 52% (95% CI 38–66). Neutropenia was the most common adverse reaction reported as a grade 3 or higher.
Conclusion
Pirtobrutinib has demonstrated safety and efficacy in heavily pre-treated adult patients with r/r MCL. Advantages of this drug include its usage in patients whose malignancy is resistant to current BTKi, tolerability, and response rate. Multiple clinical trials are underway to determine the efficacy of pirtobrutinib in other B-cell malignancies
Keywords
Introduction
B-cell lymphomas make up approximately 85% of non-Hodgkin lymphomas (NHL). 1 The different types of B-cell malignancies include chronic lymphocytic leukemia/small lymphocytic lymphoma (CLL/SLL), follicular lymphoma (FL) and mantle cell lymphoma (MCL). MCL is a rare and aggressive type of lymphoma, forming in the mantle zone and comprising only 5% of lymphomas. 1 It is more common in men than women and occurs more often in patients > 60 years of age. 1 MCL is characterized by translocation (11;14) and (q13;q32), resulting in overexpression of cyclin D1 (CCND1), which is significant for cell cycle progression and poor clinical outcomes. Cyclin-dependent kinase inhibitor (CDKN) 2A deletions and cyclin-dependent kinase (CDK) 4 amplification are associated with aggressive histology, and CCND1 gene mutations are more common with ibrutinib resistance. 2 Ataxia-telangiectasia mutated (ATM), a tumor suppressor gene, is seen in 30–50% of patients, and tumor protein (TP) 53 mutations are predictors for aggressive disease and inferior outcomes. 2 Despite recent advances in treatment, including chemotherapy, immunotherapy, and autologous stem cell transplantation (ASCT), MCL remains incurable, and many patients experience relapse.2,3 Individuals who are older, frail, or with preexisting comorbidities are unable to tolerate intensive therapy due to treatment-related complications. Unfortunately, this leads to poor long-term outcomes and disease progression. 3
Currently, there are no definitive causative environmental factors for developing MCL; however, familial risks and pesticide exposure can be predisposing factors. 2 Patients with MCL clinically present with pancytopenia or extensive leukocytosis. 4 Disease progression involves crowding out normal blood cells, resulting in neutropenia, anemia, and thrombocytopenia. Initially, chemotherapy was the primary treatment option; but MCL quickly became resistant to standard therapy. To optimize treatment outcomes with existing systemic therapies, researchers developed novel oral targeted agents such as Bruton's tyrosine kinase inhibitors (BTKi). BTKi have revolutionized the treatment landscape of various hematologic malignancies due to their ability to block the B cell receptor (BCR) signaling cascade reversibly or irreversibly by binding to the Bruton's tyrosine kinase (BTK) enzyme. 5 This intracellular process inhibits cell function, survival, and proliferation of normal and malignant B cells. Utilizing these oral agents allows for the continuation of treatment when other options are limited.
The first BTKi approved for relapse/refractory (r/r) MCL was ibrutinib, which binds to the C-terminal Src kinases. 5 This off-target binding effect led to many side effects, such as atrial fibrillation, bleeding, bruising, and fatigue. More recently, newer second-generation BTKi therapies such as acalabrutinib and zanubrutinib are more selective, specifically targeting the BTK C481 protein. Unfortunately, adverse effects were still reported with both drugs. Upper respiratory tract infections, diarrhea, and headaches are associated with acalabrutinib, and neutropenia, contusions, respiratory infections, and diarrhea associated with zanubrutinib. 5 As researchers began to study these agents, it became apparent that mutations at the BTK C481 residues caused resistance to pharmacological inhibitors. 5 The mechanism of resistance is not well understood. However, developing a new non-covalent binding agent may overcome this barrier in patients previously exposed to a BTKi. 5
Data selection
A literature search was conducted through PubMed MEDLINE, ClinicalTrials.gov, and the FDA website from January 2018 to January 2023 using the following key terms: lymphoma, non-covalent, BTK, and relapse. Complete human trials evaluating the efficacy and safety of pirtobrutinib in patients with r/r MCL, and the prescribing information was assessed. More clinical data is emerging for pirtobrutinib and will come to light in the coming years. Data from the BRUIN trial was used with manufacturer-published data and the FDA package insert to encompass all aspects of the drug.
Indication and usage
Pirtobrutinib is a selective non-covalent BTKi that received accelerated approval based on positive response rates for treating adult patients with r/r MCL after receiving at least two lines of systemic therapy, including previous BTKi therapy. 6
Pharmacology
The BCR is a transmembrane receptor located on the cell surface of normal and malignant B-lymphocytes. Antigen stimulation of the BCR is a critical mechanism contributing to lymphoid malignancies. The binding of the antigen induces the aggregation of the BCR, which initiates a cascade of intracellular signaling leading to cellular proliferation, differentiation, adhesion, survival, anergy, and apoptosis. 7 Once activated, recruitment of multiple intracellular protein tyrosine kinases (Src-family kinases Syk and BTK) becomes phosphorylated and initiates a downstream of events. 7 In MCL, the BTK enzyme and other kinases are persistently activated, allowing uncontrolled cell growth. These proteins specifically have become distinctive therapeutic targets and are used in combination with other drug therapies.
Pirtobrutinib is an orally administered, highly selective, non-covalent (reversible) BTKi (Figure 1). It is an adenosine triphosphate (ATP) competitive inhibitor that binds to the wildtype BTK and BTK C481 mutations with equal low nM potency. 8 Additionally, pirtobrutinib inhibits B-cell CD69 expression and malignant B-cell proliferation. 6 Pirtobrutinib shows dose-dependent anti-tumor activities in BTK wild type and BTK C481. 6
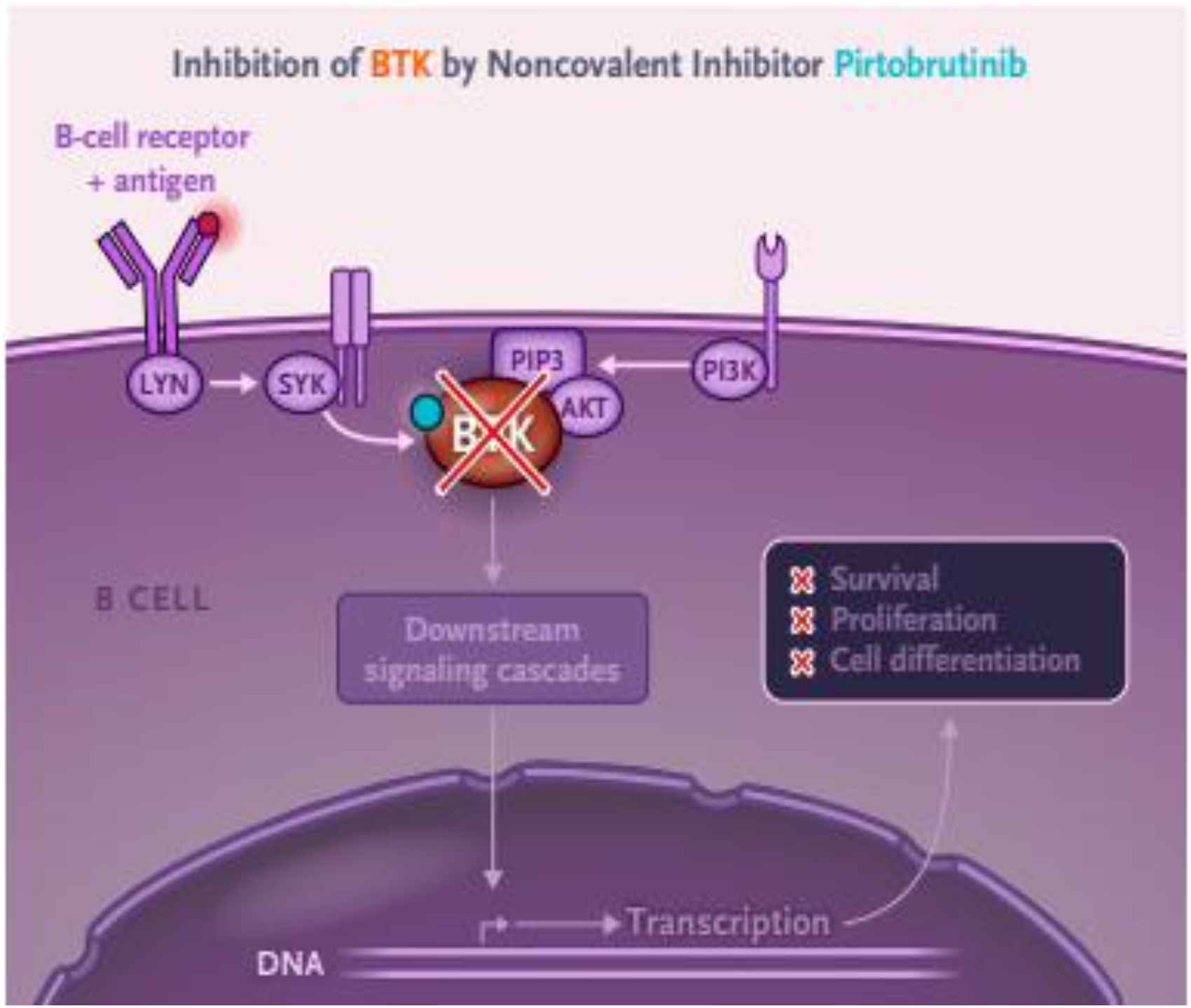
Mechanism of pirtobrutinib: inhibition of BTK signaling pathway. 9
Pharmacodynamics/pharmacokinetics
Like other BTKi agents, pirtobrutinib inhibits the BTK enzyme by interfering with intracellular signaling. However, due to its non-covalent binding capabilities, it has more than 300-fold selectivity for the BTK than other kinases. 8 Thereby potentially reducing toxicities, as well as potency against wild-type and BTK C481. 8 Additionally, it is effective in treating B-cell malignancies in patients that have developed resistance or intolerance to previous BTK therapy. 8 Trough concentration has shown to exceed BTK IC96 with administration of the 200 mg daily dose. 6 Even at a higher dose, no clinical effect on QTc was reported, which concluded that pirtobrutinib exposure was not correlated with changes in QTc interval. 6
Absorption
Administration of a single 200 mg dose has 85.5% bioavailability. The median time to reach peak plasma concentration is approximately 2 hours. 6
Distribution
The mean apparent central volume of distribution of pirtobrutinib is 32.8 L. Human protein binding of pirtobrutinib is 96% and is independent of concentration in vitro. Mean blood-to-plasma ratio is 0.79. 6
Metabolism
Pirtobrutinib is primarily metabolized by CYP3A4 and direct glucuronidation by UGT1A8 and UGT1A9. 6
Excretion
Healthy subjects received a 200 mg dose of pirtobrutinib. From the 200 mg dose, 37% of the dose was recovered in feces (18% unchanged) and 57% in urine (10% unchanged). 6 See Table 1 for a summary of key pharmacokinetic (PK) properties.
Select pharmacokinetic properties of pirtobrutinib. 6

Abbreviations: Cmax is the highest concentration of a drug in the blood; Tmax time required for a drug to reach peak concentration in plasma.
Therapeutic uses 8
BRUIN was a global, open-label, multi-center phase 1/2 trial evaluating pirtobrutinib in patients (n = 323) previously treated for B-cell malignancies who failed or were intolerant to the standard of care. The study was conducted from March 21st, 2019 to September 27th, 2020. Phase 1 of the study consisted of 203 patients (61 with r/r MCL) assigned to receive seven dose levels (25 mg, 50 mg, 100 mg, 150 mg, 200 mg, 250 mg, and 300 mg) of pirtobrutinib daily with an accelerated dose escalation design. The median age of participants was 69 years (range: 63–75). Forty-two percent of participants received an Eastern Cooperative Oncology Group (ECOG) score of 0, while 31% received a score ≥1. The total number of MCL participants previously exposed to other therapies included 57 (93%) BTKi; 56 (92%) chemotherapy; 60 (98%) anti-CD20 antibody; 9 (15%) B-cell lymphoma (BCL) 2 inhibitor; 1 (2%) phosphoinositide 3-kinase (PI3 K) inhibitor; and 12 (20%) lenalidomide. A total of 21 participants received either an autologous stem cell transplant, allogenic stem cell, or chimeric antigen receptor (CAR) T-cell therapy. Patients with pre-existing cardiovascular issues (i.e., atrial fibrillation) who were not on warfarin but on other oral anticoagulants and antiplatelet therapy were allowed enrollment.
The primary endpoint was to determine the maximum tolerated dose and recommended dose for phase 2. The secondary endpoint was an overall response rate (ORR), pharmacokinetics, and safety. There was no reported maximum tolerated dose or dose-limiting toxicities during this study period; however, the recommended phase 2 dose was 200 mg daily. Among all patients evaluated for safety, neutropenia was the most common adverse event with a grade 3 or higher (10%). Majority of adverse events (grade 1 or 2) included fatigue (20%), diarrhea (17%), and contusion (13%). Atrial fibrillation or atrial flutter was observed as a grade 2 in two patients but unrelated to pirtobrutinib due to preexisting atrial fibrillation in both patients.
During phase 2 of the trial, 120 patients were assigned to receive pirtobrutinib 200 mg daily for 28 days. Evaluable efficacy was observed in 56 patients with MCL. Changes in tumor burden from baseline were determined by computerized tomography (CT) images. The primary endpoint was ORR (assessed by an independent review committee), and the secondary endpoint was ORR (assessed by investigators), overall response, duration of response, progression-free survival, overall survival, safety, and pharmacokinetics. In the efficacy evaluable patients with MCL, the ORR was 52% (95% CI 38–65), including 14 patients with complete response, 15 with partial response, 10 with stable disease, 12 with progressive disease, and five not evaluable. Among the 52 patients who received a previous covalent BTKi, the ORR was 52% (95% CI 38–66). Responses were also observed in patients who received previous cellular therapy, including 14 patients with previous autologous or allogeneic transplants and 2 with prior CAR T-cell therapy. The median time to first response was 1·8 months (IQR 1·8–1·9). Fifty-seven percent of patients continued therapy with a median efficacy follow-up of 6 months. Of the 29 responding patients, 5 discontinued treatment (four for progressive disease and one in complete response).
In this initial phase 1/2 trial, pirtobrutinib showed safety and efficacy in treating multiple B-cell neoplasms, including patients with MCL. Additionally, effectiveness was seen in patients previously treated with first-line therapies, including those who received a covalent BTKi and patients with resistance mediated by BTK C481 mutations. The data presented in this clinical trial showed promising effects, and more global randomized trials are underway. Currently, there are 11 ongoing studies on pirtobrutinib and its efficacy. A head-to-head study comparing pirtobrutinib monotherapy versus the investigator's choice of covalent BTKi (ibrutinib, acalabrutinib, or zanubrutib) in patients with r/r MCL is currently being investigated. 10 The primary endpoint is progression-free survival and determining the superiority of pirtobrutinib monotherapy over other BTKi. Furthermore, other studies are recruiting patients to evaluate the efficacy and ORR of pirtobrutinib versus other first-line therapies in B-cell malignancies (Table 2).
Ongoing clinical trials with pirtobrutinib. 10
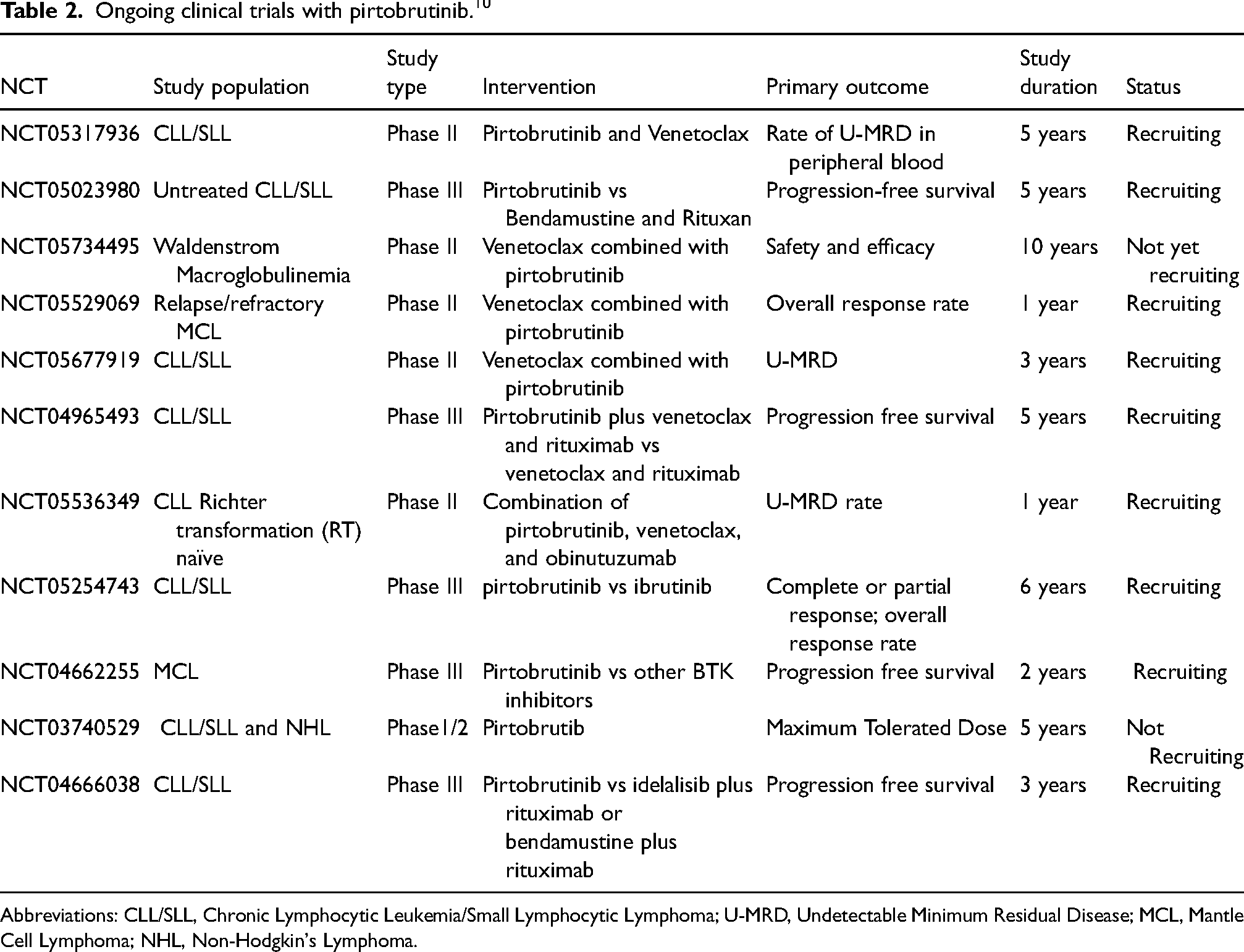
Abbreviations: CLL/SLL, Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma; U-MRD, Undetectable Minimum Residual Disease; MCL, Mantle Cell Lymphoma; NHL, Non-Hodgkin's Lymphoma.
A 2022 update of the BRUIN study on 90 patients with MCL previously exposed to a median number of 3 prior therapies showed an ORR of 58% (95% CI, 47–68), including 18 (20%) complete remissions. At a median follow-up of 12 months, the duration of response among 52 patients who responded to treatment was 22 months. For patients who discontinued prior BTKi due to progression, ORR was 50%, and the median duration of response was 14.8 months. 11
Safety
Adverse events 6
Treatment with pirtobrutinib should be interrupted for any grade 3 or higher non-hematological toxicities. Hematologic toxicities are defined as one of the following: absolute neutrophil count (ANC) < 1000 mm3 to 500 mm3 with fever and infection, ANC <500/mm3 lasting ≥ 7 days, platelets 25,000 to <50,000/mm3 with bleeding or platelets <25,000/mm3. Non-hematological toxicity includes gastrointestinal pain, musculoskeletal pain, and connective tissue disorders. Refer to Table 3 for recommended dose modifications.
Dosage adjustment for pirtobrutinib toxicities. 6

Dosing and administration 6
Pirtobrutinib is available in 50 mg and 100 mg tablets. The recommended dose is 200 mg once daily until disease progression or unacceptable toxicity. Each dose is administered orally with or without food at approximately the same time each day. If a dose is missed by more than 12 hours, administer the next dose as scheduled. Tablets should be administered whole without cutting, crushing, or chewing. Pirtobrutinib is a hazardous agent, and authorized personnel must take appropriate precautions for handling and disposing this agent.
Special populations
Pregnancy and lactation 6
During an embryo-fetal development study in pregnant rats, approximately 500 mg/kg of pirtobrutinib was administered twice daily. Doses given at ≥375 mg/kg twice daily correlated with decreased fetal body weights and increased incidence of malformations and variations in the urinary tract, reproductive tract, and bones. Developmental outcomes, such as structural abnormalities, altered fetal growth, and embryo-fetal mortality, were noticed during the study. Based on the findings of animal reproduction studies, pirtobrutinib can result in fetal harm when administered to pregnant women. Initiating a pregnancy test before starting treatment is recommended. Females of reproductive potential should use effective contraception during treatment and for at least one week after the last dose of pirtobrutinib. No data is available on the presence of pirtobrutinib in human milk or its effects on milk production.
Geriatric use 6
Clinical trials included patients 65 years of age and older. Unfortunately, patients < 65 years old were not included in the study to determine whether older patients respond differently from younger patients. However, in the pooled safety population, older patients experienced higher rates of grade 3 and higher adverse reactions compared to patients who were less than 65 years of age.
Renal impairment 6
Patients with severe renal impairment (eGFR15–29 mL/min) require a 50% dose reduction from the starting dose. However, discontinue treatment if the starting dose is 50 mg daily. No dosage adjustment for pirtobrutinib is recommended in patients with mild (60–89 mL/min) and moderate (30–59 mL/min) renal impairment. Currently, there are no dosage adjustments for patients with an eGFR < 15 or for those receiving dialysis.
Hepatic impairment 6
Studies showed no clinically significant difference in pharmacokinetics; therefore, no dose adjustment are recommended in mild, moderate, or severe hepatic impairment.
Drug and food interactions 6
Pirtobrutinib is primarily metabolized by CYP3A4. Co-administration of pirtobrutinib with a strong CYP3A inhibitor (itraconazole) increases the AUC of pirtobrutinib by 49%, consequently increasing the risk of adverse effects and toxicities. If a strong CYP3A inhibitor is unavoidable, a dose reduction of 50 mg is recommended. If the current dose is 50 mg, interrupt treatment during CYP3A inhibitor use and resume treatment after discontinuing the CYP3A for five half-lives. Moderate CYP3A inhibitors such as verapamil and diltiazem increase the AUC of pirtobrutinib by 30% and 20%, respectively. Co-administration of strong CYP3A inducers (rifampin) decreased the AUC of pirtobrutinib by 71%. If concomitant use of a strong CYP3A inducer is required, increase the dose by 50 mg or 100 mg, depending on the baseline dose.
Clinical trials demonstrated no clinically significant difference in the pharmacokinetics of pirtobrutinib following administration of a high-fat or high-calorie meal in healthy subjects. High-fat meals consist of 800 to 1000 calories, with 150 calories from protein, 250 calories from carbohydrates, and 500 to 600 calories from fat.
Contraindications
There are no contraindications to pirtobrutinib listed in the manufacturer's labeling.
Warnings and precautions 6
Although the drug manufacturing label lists no contraindications to pirtobrutinib; infections, hemorrhaging, cytopenias, and cardiovascular-related issues are possible side effects. In clinical trials, grade 3 or higher infections occurred in 17% of 583 patients, including pneumonia (9%), sepsis (4.5%), and febrile neutropenia (2.9%). Due to the risk of developing an infection, antimicrobial prophylaxis and vaccination may be considered for patients with increased risk. Major hemorrhaging, considered as grade 3 or higher bleeding or any CNS bleeding, was reported in 2.4% of 583 patients. Therefore, clinicians should assess the risk versus benefit of antithrombotic use when co-administered with pirtobrutinib. In addition, consider the risk versus benefit of withholding pirtobrutinib 3 to 7 days pre- and post-surgery. Cytopenias constitute a significant concern as neutropenia, thrombocytopenia, and anemia can develop. Because of these effects, monitoring complete blood counts regularly during treatment is essential.
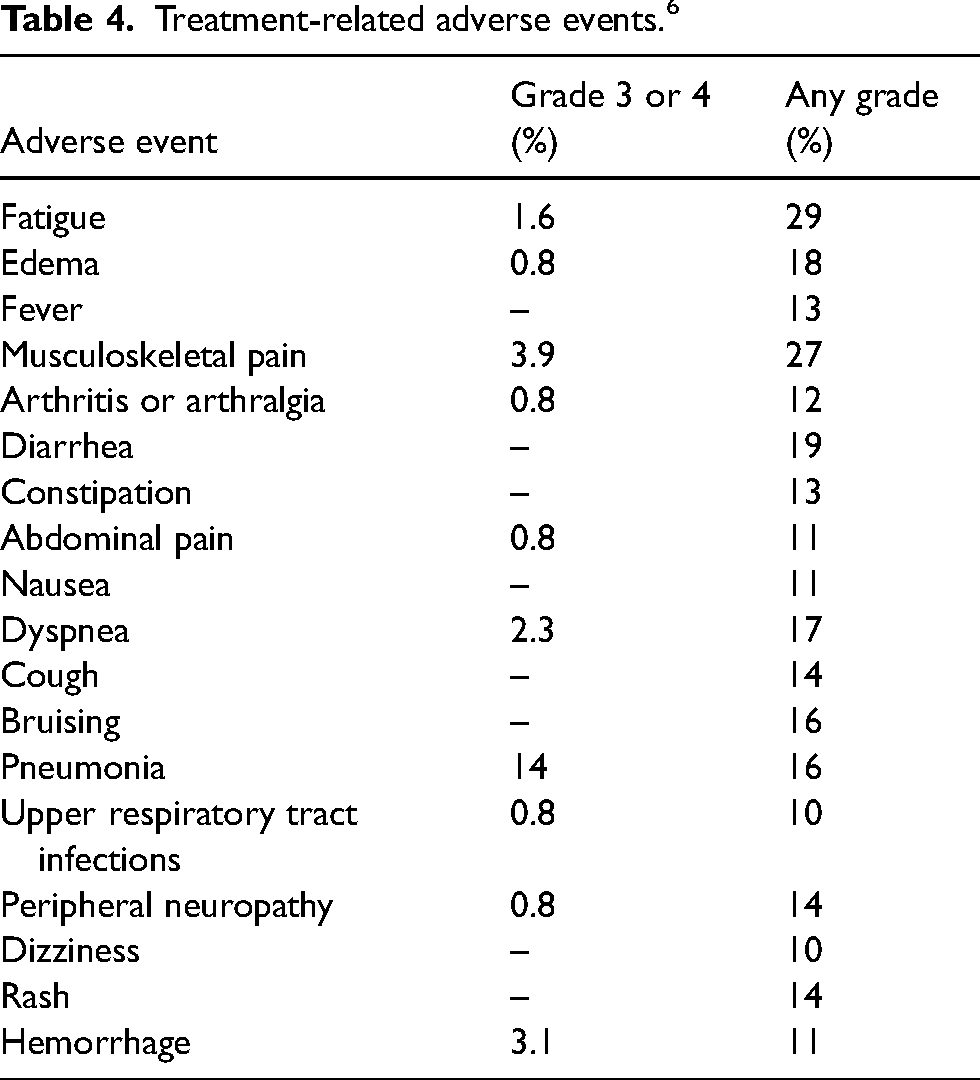
Patients with pre-existing cardiovascular diseases (hypertension and arrhythmias) should be monitored closely for signs and symptoms of arrhythmias, including dizziness, dyspnea, palpitations, and syncope. Clinical data reported atrial fibrillation or atrial flutter in 2.7% of patients, and grade 3 or 4 was reported in 1% of patients. Based on the severity of symptoms, treatment with pirtobrutinib may be dose reduced, temporarily withheld, or permanently discontinued. Moreover, due to the risk of developing skin and subcutaneous disorders, patients should be advised to use sun screen protection, as non-melanoma skin cancer was reported in 3.8% of 583 patients receiving treatment. Refer to Table 4 for a summative report of adverse events.
Treatment-related adverse events. 6
Clinical use controversies
Currently, pirtoburtinib is considered third-line therapy for patients with MCL and used for disease progression or intolerance to covalent BTKi therapy. 12 Ongoing clinical trials can assist with establishing pirtobrutinib's role as a first-line option for treating B-cell malignancies. Regarding formulary inclusion, insurance companies will cover treatment for patients who are intolerant or resistant to other BTKi as there are no studies of efficacy or appropriateness in the first-line setting.
Product description
Pirtobrutinib is available in two different strengths of 50 mg and 100 mg tablets. The 50 mg tablet is a blue film-coated, triangle-shaped tablet with “Lilly 50” on one side and “6902” on the other. 13 The 100 mg tablet is also a blue film-coated, round-shaped tablet, with “Lilly 100” on one side and “7027” on the other. 13 The wholesale acquisition price of pirtobrutinib is $280.00 per 50 mg tablet or $420.00 per 100 mg tablet. 13 The estimated monthly cost of 50 mg tablets is $8,400 and $12,600 for the 100 mg tablets. 13 The manufacturer of pirtobrutinib, Eli Lilly Inc., offer patients an interim access program that may provide a temporary supply of pirtobrutinib at no cost to patients who have been prescribed treatment for the first time and are experiencing a delay in their insurance coverage. Additionally, the manufacturer offers a savings card for commercially insured patients with a co-pay as little as $0 a month.
Discussion
Pirtobrutinib is a novel, non-covalent BTKi approved therapy for adults with r/r MCL. Although many patients diagnosed with MCL typically respond well to initial treatment, some patients will relapse or experience refractory. Due to the aggressive nature of MCL, certain patients with preexisting comorbidities may be unable to tolerate intensive therapy. Additionally, the off-target binding capacity of current BTKi and mutations at the BTK C481 residues make existing treatments ineffective for patients with progressive disease. Pirotbrutinib shows promising, durable response and an effective ORR. The most common adverse events grade 3 or higher include pneumonia, musculoskeletal pain, bleeding, dyspnea, and fatigue. Given the limited data presented, pirtobrutinib is a viable option for patients who have failed other BTKi agents. Additional data from clinical trials is needed to determine the role of pirtobrutinib as a first-line option in MCL, and its role in treating other B-cell malignancies.
Footnotes
Authorship
Authors DD, ZO, and HP conducted primary literature searches and contributed to the concept and design of the paper. DD and HP wrote the first draft of the manuscript. ZO revised the manuscript for intellectual content and accuracy. All authors reviewed and edited the manuscript and approved the final version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
