Abstract
Introduction
Etoposide is a key component of many pediatric chemotherapy regimens for both hematologic and solid tumors. It is well documented that patients receiving etoposide may experience infusion-related reactions.
Methods
In this study, total doses of etoposide and etoposide phosphate were identified, and infusion-related reactions were retrospectively evaluated at a large pediatric oncology ambulatory clinic. The primary outcome was to determine the rate of acute infusion-related reactions to etoposide. A secondary objective was to identify potential risk factors associated with such reactions.
Results
Overall, 1463 doses of etoposide were dispensed to 150 unique patients and 15 patients (10%) experienced etoposide infusion-related reactions. Of the 15 patients that experienced etoposide infusion-related reaction, two were successfully able to tolerate subsequent infusions with a slower etoposide infusion rate and pre-medications. The remaining patients changed drug formulation to etoposide phosphate (n = 12) or continued therapy without any formulation of etoposide (n = 1). Patients with classical Hodgkin's lymphoma experienced a higher incidence of infusion-related reactions compared to the overall patient population (30.5% vs. 10%) and made up 73.3% of all infusion-related reactions during the study period.
Conclusions
During the examined study period, 10% of patients experienced an acute infusion-related reaction to etoposide. This review builds on previous literature concluding that additional monitoring may be warranted in patients with classical Hodgkin's lymphoma receiving etoposide.
Introduction
Etoposide is an antineoplastic agent which has been in use in the United States since 1983. This drug is a key component of many pediatric chemotherapy regimens for both hematologic and solid tumors.1,2 It is well documented in both the primary literature and package insert that patients receiving etoposide may experience anaphylaxis (0.7%–2%) or non-anaphylactic (1.3%–27.1%) infusion-related reactions (IRRs).1–3 The cause of IRR to etoposide (majority of which are type I hypersensitivity reactions) is thought to be mainly due to the solvent polysorbate-80. 4 Etoposide phosphate is a water-soluble prodrug of etoposide that does not contain the solvent polysorbate-80. Patients who experience IRRs to etoposide have been switched to etoposide phosphate without any additional occurrence of hypersensitivity reactions. Previous studies have looked to determine if there are any known risk factors for developing an IRR to etoposide including history of drug allergy, concentration of drug, and rate of infusion.5,6 This evaluation looks to build on the literature, specifically examining the pediatric oncology population at a large tertiary cancer center.
Methods
After institutional review board approval, total doses of etoposide and etoposide phosphate were identified, and IRRs were retrospectively evaluated from January 1, 2021, to June 10, 2023, at Dana-Farber Cancer Institute (DFCI), Boston, MA, pediatric oncology ambulatory clinic. All patients, irrespective of age, who received a dose of etoposide or etoposide phosphate in clinic were included in the study. Patients who experienced an IRR to etoposide at an outside hospital before receiving treatment were excluded. Only doses given by intravenous route were examined. Etoposide concentration varied across patients but was generally compounded to yield 0.4 mg per milliliter (mg/ml). The drug was infused over 1 to 6 h. An in-line filter was not used for any drug preparations. Use of etoposide or etoposide phosphate triggered an electronic medical record review to collect data related to the patient demographic and etoposide infusion reaction. The primary outcome was to determine the rate of acute IRRs to etoposide. A secondary objective was to identify potential risk factors associated with acute IRRs to etoposide including patient age, allergies, oncologic diagnosis, body surface area, selected laboratory values indicating inflammation, concomitant medications, date of reaction, infusion rate, and drug preparation characteristics (concentration and diluent type).
Results
During the study period, we identified 15 patients who experienced an etoposide IRR. Overall, 1463 doses of etoposide were dispensed to 150 unique patients, and 15 patients (10%) experienced etoposide IRRs. Of the 15 patients that experienced etoposide IRR, two were successfully able to tolerate subsequent infusions with a slower etoposide infusion rate and pre-medications. Both patients had experienced a type I hypersensitivity reaction (shortness of breath, dizziness, and nausea being presenting symptoms) within minutes of starting the initial etoposide infusion. The remaining patients changed drug formulation to etoposide phosphate (n = 12) or continued therapy without any formulation of etoposide (n = 1). No subsequent etoposide phosphate IRRs were identified in these patients. The highest incidence (n = 6) of IRRs occurred between January 1st to June 10th of 2023 (Figure 1).
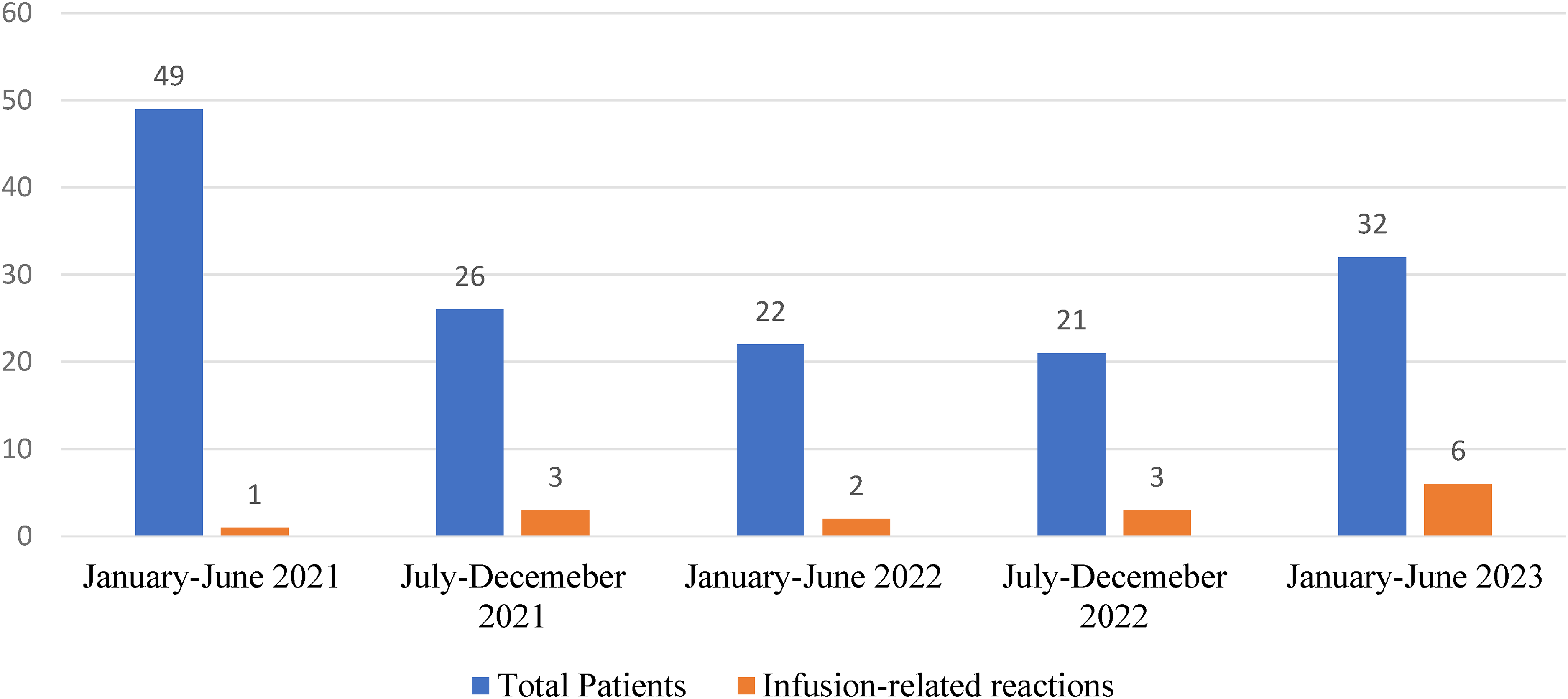
New etoposide patients (n) and infusion-related reactions (n).
The mean age of patients experiencing etoposide IRRs was 195 months (16 years and 3 months) (range 61 months (6 years and 1 month)–247 months (20 years and 7 months)), with 10 male and 5 female patients (Table 1). Five patients had a documented history of a previous drug (n = 4) or food (n = 1) allergy. One patient had a history of seasonal allergies. Patients were identified across 18 unique oncologic disease states; however, only four of these groups had patients which experienced an IRR. These disease groups were classical Hodgkin's lymphoma (n = 11), acute lymphoblastic leukemia (n = 2), osteosarcoma (n = 1), and germ cell tumors (n = 1) (Table 2). Patients with classical Hodgkin's lymphoma (cHL) experienced a higher incidence of IRRs compared to the overall patient population (30.5% vs. 10%) and made up 73.3% of all IRRs during the study period. Nine of the cHL patients who reacted were identified to have the nodular sclerosing subtype of disease.
Patient demographics and selected lab values*.

*Comparison of mean values in patients who experienced an IRR compared with those who did not. ESR: erythrocyte sedimentation rate; CRP: C-reactive protein; IRR: infusion-related reaction.
Infusion-related reaction rate in relation to diagnosis.
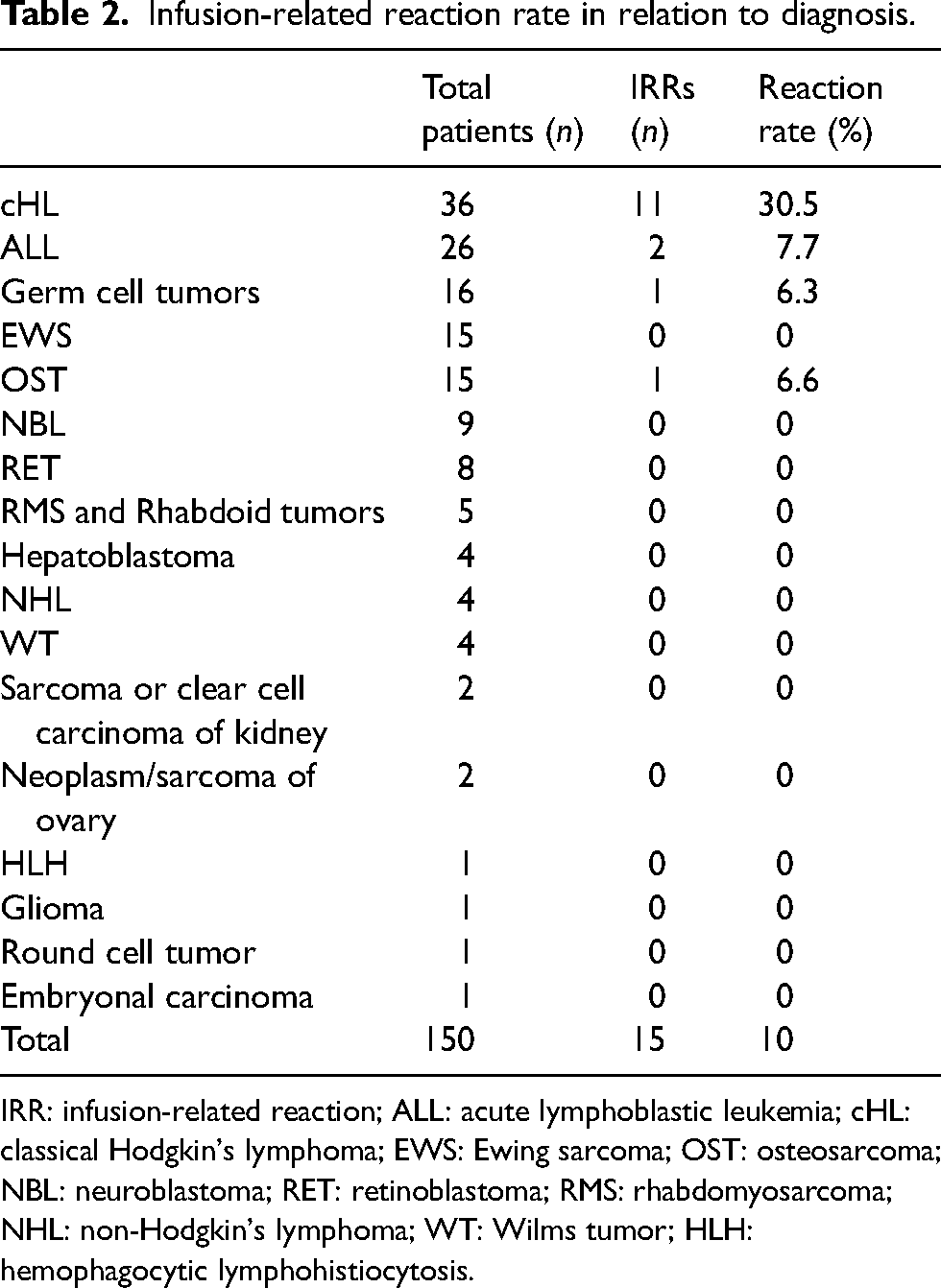
IRR: infusion-related reaction; ALL: acute lymphoblastic leukemia; cHL: classical Hodgkin's lymphoma; EWS: Ewing sarcoma; OST: osteosarcoma; NBL: neuroblastoma; RET: retinoblastoma; RMS: rhabdomyosarcoma; NHL: non-Hodgkin's lymphoma; WT: Wilms tumor; HLH: hemophagocytic lymphohistiocytosis.
For etoposide, the mean initial infusion rate was 111.1 mg/m2/h for patients that experienced etoposide IRR and 101.2 mg/m2/h for patients without reaction (Table 3). The dose in milligrams (mg) of etoposide was greater in patients that experienced an IRR compared to those that did not (202.1 mg vs. 152.7 mg); however, the mean drug concentration was 0.4 mg/ml for both patient groups. In 15 patients where an IRR occurred, 11 of the etoposide preparations used a dextrose/sodium chloride 5%/0.9% (D5NS) diluent, two used a dextrose/sodium chloride 5%/0.45% (D51/2NS) diluent, and two used sodium chloride 0.9% (NS) diluent.
Etoposide infusion and preparation.

IRR: infusion-related reaction; D5NS: dextrose/sodium chloride 5%/0.9%; NS: sodium chloride 0.9%.
Discussion
IRRs are a well-documented adverse effect associated with the use of the chemotherapeutic agent etoposide. Primary literature and package insert data suggest that non-anaphylactic IRRs can occur in 1.3%–27.1% of patients with one study treating pediatric patients with acute leukemia reporting IRRs in up to 33% of patients.1–3,6 The rate of etoposide IRRs reported in this analysis (10%) is consistent with the package insert and previous literature. The results of this study do highlight the following findings: (1) Most reactions occurred in patients with classical Hodgkin's lymphoma, (2) higher rate of infusion was associated with an increased reaction rate, (3) an increased incidence of IRRs was observed at the end of the study period.
Patients with cHL experienced a higher rate of IRRs compared to the overall patient population (30.5% vs. 10%) and made up 73.3% of all IRRs during the study period. Higher rate of etoposide IRRs has been reported in this population before. In one 1993 study, conducted in pediatric patients treated with the etoposide-containing VEPA regimen for Hodgkin's disease, Hudson et al. found that 23 of the 45 patients (51%) had one or more acute IRR to etoposide. 7 The authors hypothesized that this could be due to immunologic disturbances caused by Hodgkin's disease. In the current study, markers of inflammation were examined where data was available, including erythrocyte sedimentation rate (ESR), C-reactive protein (CRP), platelet count, and albumin. These markers were chosen as they had been previously studied in patients with cHL. 8 It was confirmed that patients with cHL did have elevated ESR, CRP, and platelet count compared to patients in other disease groups. However, there did not appear to be differences in the values between the subset of cHL that experienced an IRR and those that did not (Table 4). The presence of brentuximab in the treatment regimen, which was hypothesized to have a potential impact on the IRR rate, also did not appear to have an impact on the incidence of IRRs (56.0% in the no IRR group vs 63.6% in the IRR group).
Classical Hodgkin's lymphoma subset analysis.

ESR: erythrocyte sedimentation rate; CRP: C-reactive protein; IRR: infusion-related reaction.
A higher rate of etoposide infusion may be associated with an increased reaction rate. The mean initial infusion rate was 111.1 mg/m2/h for patients that experienced etoposide IRR and 101.2 mg/m2/h for patients without reaction. In the 1993 study previously mentioned, patients were given a higher dose (200 mg/m2), faster infusion rate (given over 1–2 h), and more concentrated preparation (0.6–1.0 mg/ml) compared to patients in our analysis. Patients in this study experienced a higher incidence of IRR compared to the subset of cHL patients in our analysis (51% vs 30.5%). When changes were made to drug administration (addition of premedication with corticosteroid +/- antihistamine; slower and less concentrated etoposide infusion) 78% of patients were able to successfully resume etoposide administration. 7 Another study, conducted in a pediatric population, found that the mean initial infusion rate of etoposide was higher in patients who experienced hypersensitivity compared with those who did not (64.6 ± 40.9 mg/m2/h vs. 49.5 ± 33.4 mg/m2/h). 9 Our observational findings are consistent with the previous literature that faster infusion rates are a potential risk factor for etoposide IRRs.
During the last 6 months of the study period there was an increased number of etoposide IRRs (n = 6). The mean initial infusion rate was 100.5 mg/m2/h for patients treated during this period, which led investigators to believe it was not a significant factor. The etoposide product used in clinic did change on March 9, 2023. Four IRRs occurred, after this product formulation change, in a 3-month period. It is possible this change may have been a contributing factor. However, no difference in active or inactive ingredients was identified between the two etoposide formulations. This includes polysorbate-80 concentration, which was 80 mg in one milliliter of drug solution for both products.10,11
We conclude that additional monitoring may be warranted in patients with cHL receiving etoposide. Etoposide phosphate may be offered as a suitable alternative to conventional etoposide formulations in patients that experience an IRR.
Footnotes
Authors’ contributions
PM and BD researched literature, developed the study, and were involved in protocol development. PM gained ethical approval, performed data collection/analysis, and wrote the first draft of the manuscript. All authors reviewed and edited the manuscript and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
