Abstract
Introduction
A case series of hypersensitivity reactions (HSRs) during intravenous administration of etoposide was observed following the introduction of in-line filters (ILFs) at a specialized university-affiliated center. This raised questions about the possible involvement of filters in these reactions. Despite there being very little published evidence to inform clinical decision making in this potentially clinically significant situation, the use of ILFs was discontinued at this center pending further investigation. The aims of this study were to evaluate the cumulative incidence of etoposide-related HSR with and without the use of ILF and to describe the reactions in adult and pediatric patients with cancer.
Methods
A retrospective cohort study was performed among all pediatric and adult patients treated with intravenous etoposide at a maximal concentration of 0.4 mg/mL at our center between 30 September 2015 and 16 August 2018. This covered periods of time during which ILFs were used, as well as 6 months before their implementation and after their withdrawal. Data were extracted from medical records and cumulative incidence was calculated for each of the time periods (pre-ILF, ILF, and post-ILF) as the proportion of patients who recorded an HSR (one or more). Confidence intervals were calculated for each proportion using Fisher's Exact 95%. Comparisons of proportions between time periods were performed using Exact Pearson Chi-squared tests. Data were stratified by a number of perfusion cycles (single cycle or multiple cycles) and by patient population (adult and pediatric).
Results
A total of 284 patients were included in the study. The overall cumulative incidence of etoposide HSR was 9.9%. The cumulative incidence of HSR tended to be higher during ILF use when compared with combined pre- and post-ILF periods (12.2% [95% CI: 7.9–17.8] vs. 5.2% [95% CI: 1.7–11.7], p = 0.09). In patients who received multiple cycles of etoposide, the cumulative incidence of HSRs was higher during ILF use when compared with combined pre- and post-ILF periods (15.0% [95% CI: 9.6–21.8] vs. 3.9% [95% CI: 0.8–11.0], p = 0.01). The majority of HSRs' maximal severity were grade 1 or 2 (85.7%) according to Common Terminology Criteria for Adverse Events (CTCAE) version 4.0.
Conclusions
This study suggests a link between the use of ILFs and increased incidence of HSR during etoposide perfusion.
Introduction
Etoposide is a semi-synthetic podophyllotoxin derivative that is used as an antineoplastic agent in the treatment of several cancers, such as lung cancer, testicular cancer, and lymphoma. 1
Anaphylactic-like reactions with etoposide are rather rare and are reported in 0.7% to 3% of patients.1,2 The incidence is higher in children with acute lymphoblastic leukemia. 3 Etoposide hypersensitivity reactions (HSRs) are described as type 1 reactions (immediate). 2 However, the mechanism behind these reactions is unclear.2,4 Moreover, HSRs are defined as any drug-related reaction with manifestations of any of the four major types of drug-induced allergy, as originally defined by Gell and Coombs, and are usually considered to be mediated by immunoglobulin E (IgE).2,5,6
The most common symptoms associated with etoposide HSR include chills, fever, tachycardia, bronchospasm, dyspnea, and hypotension.1,2,4,7 Other manifestations reported include flushing, exanthema, urticaria, cyanosis, or hypertension. In most patients, these reactions occur within the first 5 to 10 min following the initiation of the infusion and rapidly resolve upon discontinuation of etoposide.3,8–11 Such reactions can generally be managed by premedication with corticosteroid and anti-histaminic drugs and by slowing down the infusion rate.4,8,11
Etoposide-related HSRs have been reported in both adult and pediatric patients. Although some causes have been proposed, factors associated with HSRs remain unclear. Among these, etoposide dose and infusion rate, ingredients used in the compounding of injectable formulations such as polysorbate 80, a non-medical additive in etoposide formulation,4,12–14 some clinical conditions (e.g. pediatric leukemia) and more rarely reaction to etoposide itself are discussed in the literature.1–4,7–12,15–17
Etoposide can be administered undiluted or diluted. In the latter case, it should be diluted in dextrose 5% or normal saline to obtain a concentration between 0.2 and 0.4 mg/mL. Concentrations higher than 0.4 mg/mL are not recommended due to the risk of precipitate formation.1,18,19 This risk is unpredictable and is affected by a number of factors including solution concentration and time elapsed since dilution. 19
This has led agencies providing guidance on high-quality cancer care delivery in Canada, including Cancer Care Ontario and British Columbia Cancer Agency, to encourage the use of a 0.22-micron in-line filter (ILF) for the administration of diluted etoposide. Otherwise, undiluted etoposide is administered with no filter to prevent the cracking of the filter. 19
Following these recommendations, ILFs were implemented with diluted etoposide for adults at our center in 2016 and pediatric patients in 2017. Due to a perceived increase in the frequency of HSRs with ILF, by the clinical oncology team, the use of ILFs was discontinued following a risk–benefit analysis and additional experience of another hospital. 20 The aims of this study were to evaluate the cumulative incidence of etoposide-related HSR with and without the use of ILF and to describe the reactions in adult and pediatric patients with cancer.
Methods
Study design
A retrospective comparative cohort study was conducted among patients treated with etoposide at CHU de Québec-Université Laval; a university hospital center of five hospitals in Quebec City, Canada. At the time of study, there were four oncology clinics, including one pediatric clinic, across which the administration of etoposide was standardized. Medical records of adult and pediatric patients who began etoposide-based chemotherapy for cancer between 30 September 2015 and 16 August 2018 were reviewed. This corresponds to three distinct study periods, namely 6 months before the implementation of ILF (pre-ILF), the period during which ILF were used (ILF), and 6 months following the withdrawal of ILFs from etoposide perfusion protocols (post-ILF) (Figure 1). Two brands of etoposide were available. During the study period, one of these brands was used for the majority of treatment episodes. The other brand was used only during periods of shortage of the preferred brand. No other changes in routine etoposide administration procedures occurred throughout the study periods, including premedication procedures and perfusion durations. Primarily, etoposide was given over 60 min at concentrations of 0.2–0.4 mg/mL. Patients were excluded if data were missing for the primary outcome (HSRs). Patients were identified by the pharmacy computer software and data were manually extracted from medical files. The occurrence of HSR with etoposide was assessed for the first three cycles of etoposide. HSR cases were defined as a patient with one or more HSRs during a perfusion protocol. The severity of the perfusion reaction was determined based on the Common Terminology Criteria for Adverse Events (CTCAE) version 4.0 and evaluated independently by two research team members to minimize the misinterpretation of retrospective data. Discrepancies were minor and resolved by consensus. In brief, mild and transient reactions that resolved without symptomatic treatment were classified as grade 1. Reactions requiring a symptomatic treatment (e.g. antihistamines) that promptly responded were defined as grade 2. Reactions that persisted despite an appropriate symptomatic treatment were grade 3, while life-threatening reactions were grade 4. In the latter case, hospitalization is indicated. In case the use of ILF changed during the first three cycles of chemotherapy, the assignment period was based on the second cycle, with the exception of patients who underwent an infusion reaction at the first cycle, in which case the period was assigned based on the first cycle. This study was approved by the human research ethics committee of the CHU de Québec-Université Laval (#2019-4366).
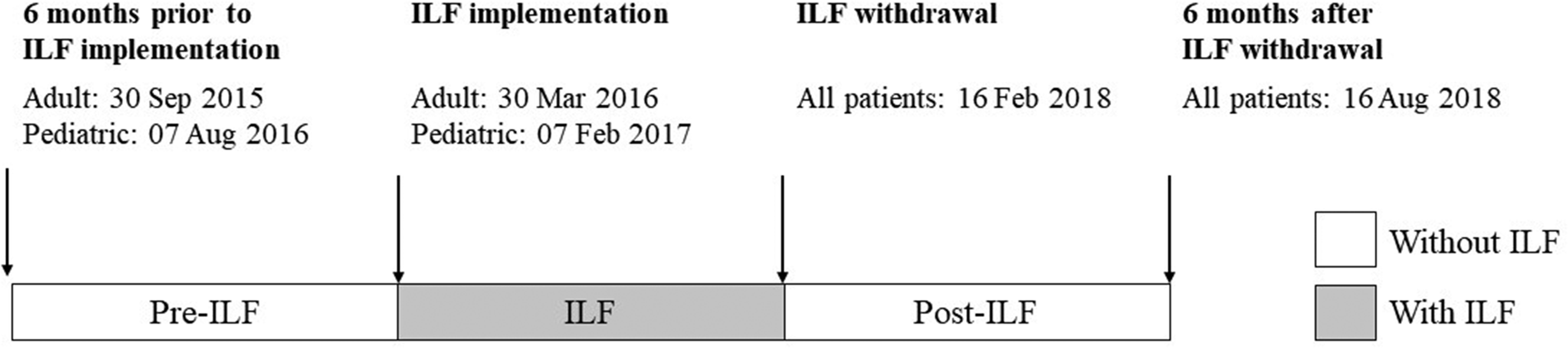
Study timeline.
Statistical analysis
The primary outcome was cumulative incidence of HSR to etoposide. Continuous and categorical data have been represented respectively as mean ± standard deviation and proportion (% [95% CI]). The primary outcome was stratified by the number of cycles of etoposide administered (single cycle or multiple cycles) and by patient population (adult: ≥ 18 years, pediatric: < 18 years). Reasons for receiving only one cycle of etoposide included: a single dose as planned per protocol, change of protocol for disease progression, unacceptable toxicity, or grade 2 or 3 HSR. Categorical variables were compared using the Chi-squared test and continuous variables using Student's t-test or Wilcoxon Mann–Whitney test. A statistically significant difference was defined as p-value <0.05. The sample size was estimated, a priori, up to 335 patients assuming estimated incidence rates of HSR between 0.7% and 2.0% with an error margin of ± 1.5% and 95% confidence interval. Statistical analyses were performed with SAS software, Version 9.4 (Copyright © 2016 SAS Institute Inc., Cary, NC, USA) and WinPepi Version 11.65 (Copyright J.H. Abramson, Aug. 23, 2016).
Results
Characteristics of patients
Among the 303 patients who met the inclusion criteria, 19 were excluded due to missing data. A total of 284 patients were included in the study (n = 46 pre-ILF, n = 188 during ILF, and n = 50 post-ILF) (Table 1).
Patient characteristics and etoposide infusion, by ILF exposure.
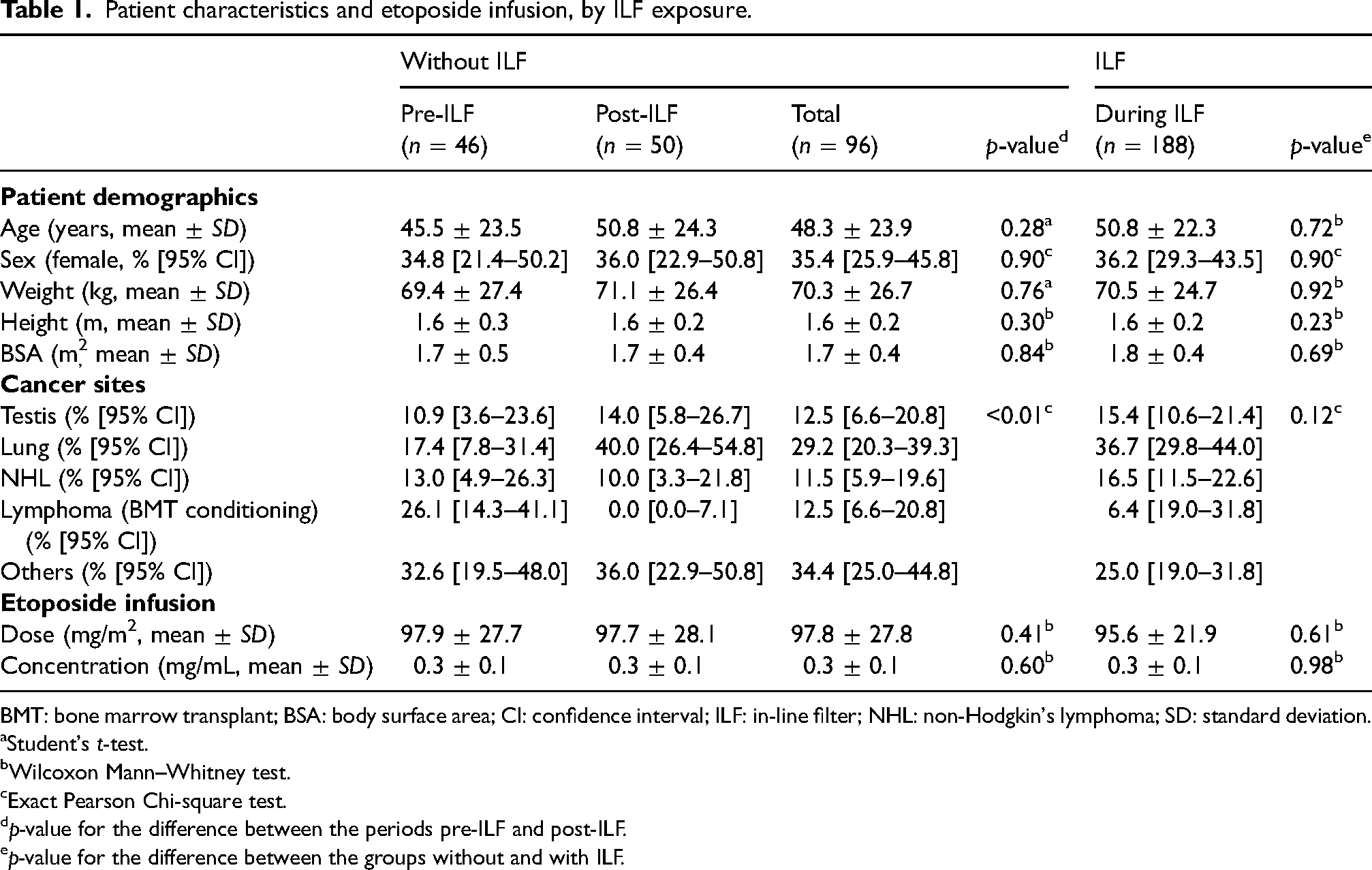
BMT: bone marrow transplant; BSA: body surface area; CI: confidence interval; ILF: in-line filter; NHL: non-Hodgkin's lymphoma; SD: standard deviation.
Student's t-test.
Wilcoxon Mann–Whitney test.
Exact Pearson Chi-square test.
p-value for the difference between the periods pre-ILF and post-ILF.
p-value for the difference between the groups without and with ILF.
Adult patients
A total of 244 adults were included in the study. Demographic and clinical characteristics of adults who received etoposide with an ILF and without an ILF were similar (Table 2). The characteristics of patients between both periods without ILF (i.e. pre-ILF vs. post-ILF) differ in age and type of cancer treated. Pre-ILF patients were younger than those treated in the post-ILF period (p = 0.03). More patients were treated for lung cancer in the period post-ILF when compared with pre-ILF (p < 0.01). Moreover, no patients received a bone marrow transplant (BMT) conditioning regimen for lymphoma in the post-ILF period, compared with 31.6% in the pre-ILF group.
Adult patient characteristics and etoposide infusion, by ILF exposure.
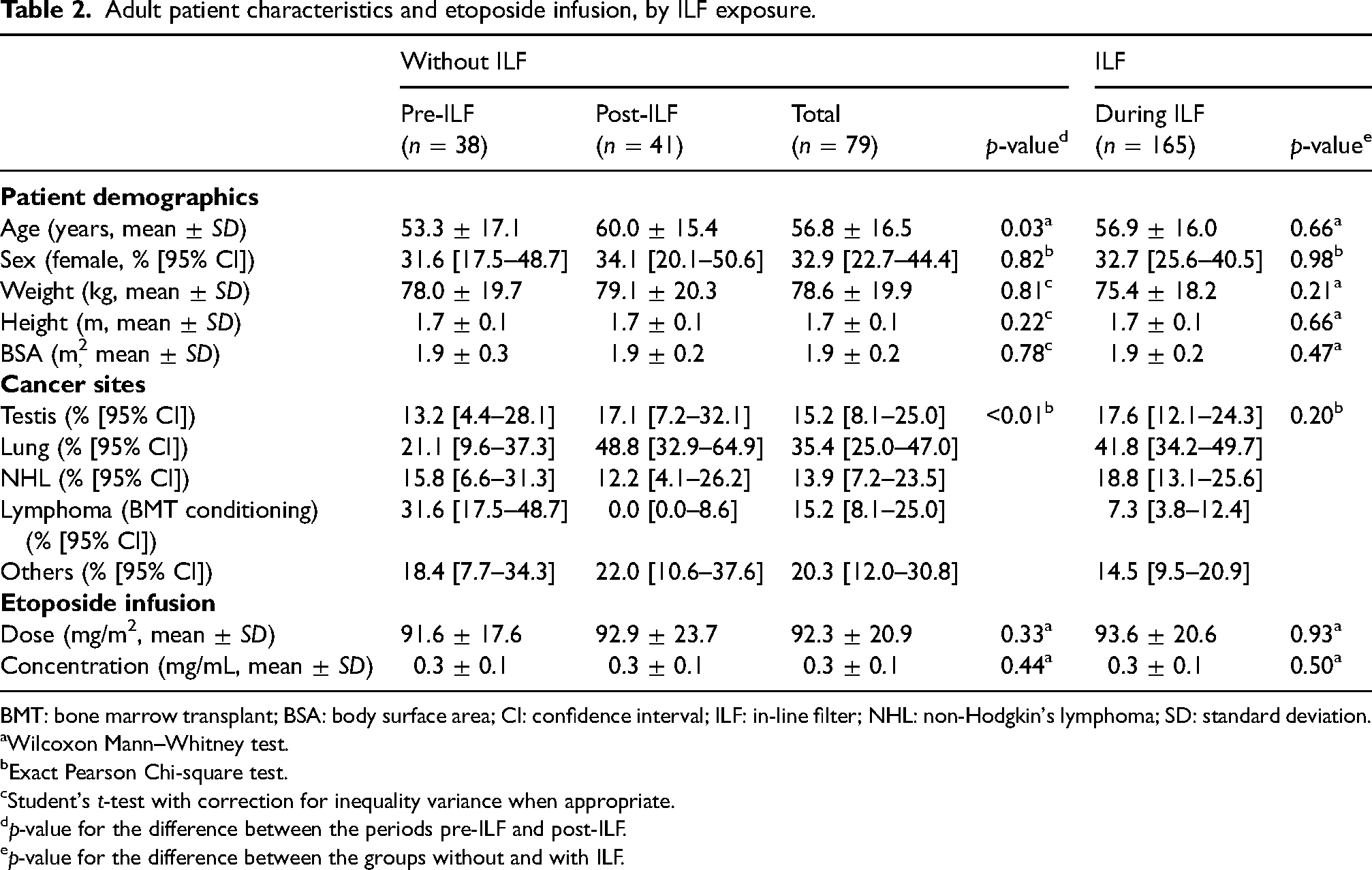
BMT: bone marrow transplant; BSA: body surface area; CI: confidence interval; ILF: in-line filter; NHL: non-Hodgkin's lymphoma; SD: standard deviation.
Wilcoxon Mann–Whitney test.
Exact Pearson Chi-square test.
Student's t-test with correction for inequality variance when appropriate.
p-value for the difference between the periods pre-ILF and post-ILF.
p-value for the difference between the groups without and with ILF.
Pediatric patients
Forty children were included in the study, representing 23 patients treated during ILF use and 17 patients treated pre- and post-ILF use (combined) (Table 3). The mean concentration of etoposide received by patients during the ILF period was significantly lower than that received by patients before or after ILF use (0.30 ± 0.06 mg/mL vs. 0.35 mg/mL ± 0.05, p = 0.03). No other differences were observed between treatment periods.
Pediatric patient characteristics and etoposide infusion, by ILF exposure.
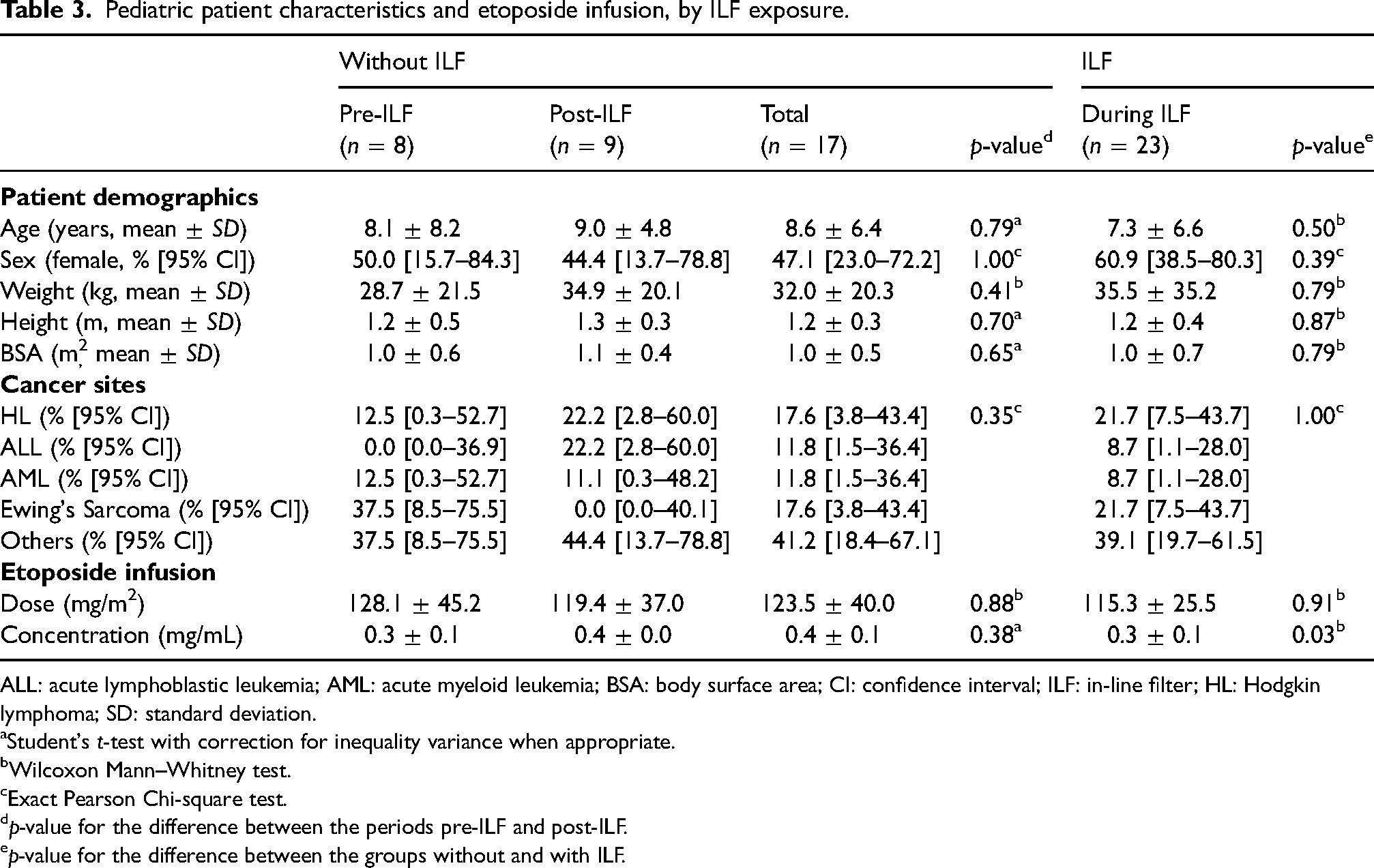
ALL: acute lymphoblastic leukemia; AML: acute myeloid leukemia; BSA: body surface area; CI: confidence interval; ILF: in-line filter; HL: Hodgkin lymphoma; SD: standard deviation.
Student's t-test with correction for inequality variance when appropriate.
Wilcoxon Mann–Whitney test.
Exact Pearson Chi-square test.
p-value for the difference between the periods pre-ILF and post-ILF.
p-value for the difference between the groups without and with ILF.
Primary outcome
Overall, 30 HSRs to etoposide were observed in 28 of the 284 patients (9.9% [95% CI: 6.7–13.4%]). The cumulative incidence for each treatment period is shown in Table 4. The cumulative incidence of HSR tended to be higher during ILF when compared with pre- and post-ILF periods combined, but the threshold for significance was not reached (12.2% [95% CI: 7.9–17.8%] during ILF, 4.3% [95% CI: 5.3–14.8%] pre-ILF, and 6.0 [95% CI: 1.3–16.6%] post-ILF period, p = 0.09). When stratified by the number of etoposide cycles administered (single cycle or multiple cycles), the cumulative incidence of HSRs was higher during ILF when compared with pre- and post-ILF combined in patients who received multiple cycles (15.0% [95% CI: 9.6–21.8%] vs. 3.9% [95% CI: 0.8–11.0%], p = 0.01) (Table 4). This difference appeared more marked in the adult (p = 0.06) versus the pediatric (p = 0.11) population. There was no significant difference attributable to the ILF use period in those treated with a single cycle only (10.5% [95% CI: 1.3–33.1%] vs. 2.4% [95% CI: 0.1–12.9%], p = 0.23).
Cumulative incidence of etoposide HSRs, by number of treatment cycles and patient population.
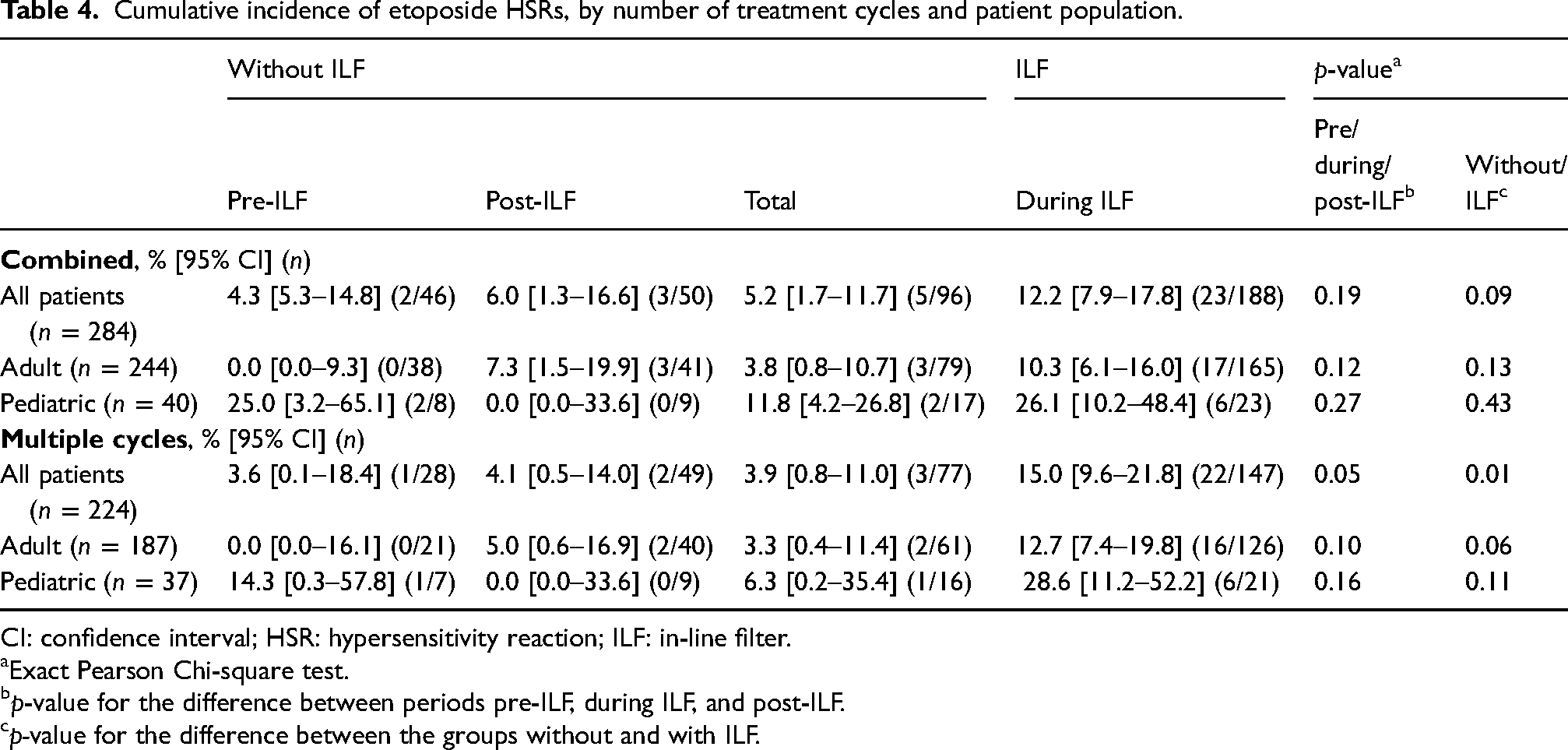
CI: confidence interval; HSR: hypersensitivity reaction; ILF: in-line filter.
Exact Pearson Chi-square test.
p-value for the difference between periods pre-ILF, during ILF, and post-ILF.
p-value for the difference between the groups without and with ILF.
Description of HSRs
Frequency and timing of HSRs
A total of 28 patients (five without ILF and 23 with ILF) had at least one episode of HSR. The frequency of HSR, by the cycle of onset and ILF exposure, is presented in Table 5. The frequency at cycle 1 was similar with or without ILF, however, HSRs at cycles 2 and 3 were observed only in patients exposed to ILFs.
Risk of HSR by the cycle of onset and ILF exposure.

ILF: in-line filter; HSR: hypersensitivity reaction.
p-value is based on an Exact Pearson Chi-square test.
This table shows that the risk of HSR was highest during cycle 2 and that it was significantly associated with ILF exposure. When comparing HSRs in the ILF group itself, cycle 2 was significantly associated with more HSRs (cycle 1 vs. cycle 2 or cycle 2 vs. cycle 3, p < 0.01, based on type 3 generalized estimating equation (GEE) analysis).
Severity and consequences
The majority of HSRs’ maximal severity were grade 1 or 2 (n = 24/28, 85.7%) and were transient or resolved with supportive care (Table 6). All four patients who had a grade 1 reaction were able to continue intravenous etoposide for subsequent chemotherapy cycles.
Maximal severity grade and consequences associated with etoposide HSRs.
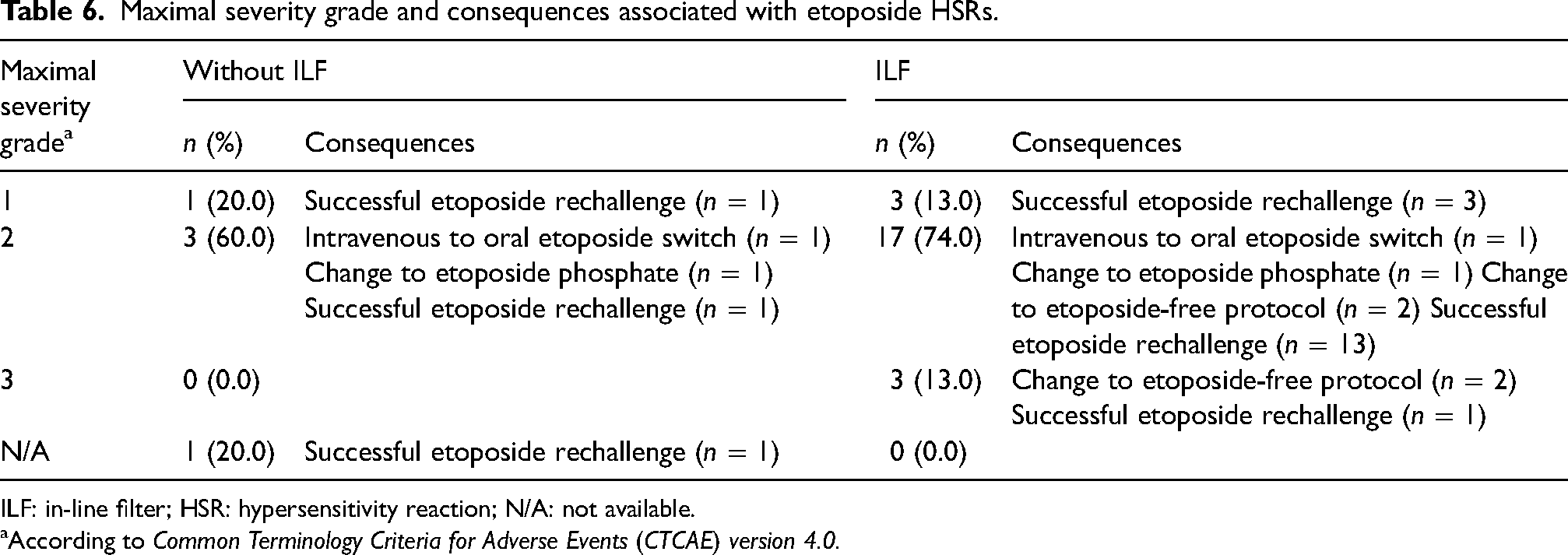
ILF: in-line filter; HSR: hypersensitivity reaction; N/A: not available.
According to Common Terminology Criteria for Adverse Events (CTCAE) version 4.0.
Of the 20 patients (17 exposed to ILFs) who had a grade 2 reaction, two were changed to oral etoposide, two to etoposide phosphate, and two to an etoposide-free protocol. One patient was sent to the emergency room for monitoring, was quickly discharged and received subsequent cycles of etoposide without recurrence of HSR. The remaining 14 patients were successfully rechallenged with etoposide.
Grade 3 reactions were observed in three patients, all of whom had etoposide infusions using ILFs. The evolution of HSR symptoms was monitored at the oncology outpatient clinic for one patient, the others were hospitalized. In these cases, etoposide was either withdrawn from the treatment (n = 2) or successfully readministered intravenously using a desensitization protocol (n = 1).
Information was missing in one patient for an HSR that occurred during the first cycle received at another hospital. The patient was successfully rechallenged with etoposide infused at a slower rate.
For patients who had an HSR for two consecutive cycles, one patient had a grade 1 reaction followed by a grade 2, while the other patient had grade 2 HSRs on both occasions. In more detail, two developed a reaction during two consecutive cycles of etoposide administered with ILF. One reacted at the first and second cycles, while the other had reactions at the second and third cycles.
HSR outcome
Overall, 20 patients were able to successfully resume subsequent cycles of chemotherapies as planned (n = 17/23 during ILF, n = 3/5 pre- and post-ILF combined). For patients who were re-challenged, etoposide was generally administrated with H1-receptor antagonists and/or steroids and by using an increased flow rate according to tolerance.
Discussion
Recently, two pediatric studies have provided evidence of HSRs related to the use of ILFs during etoposide infusion,20,21 yet, to the best of our knowledge, our study is the first to demonstrate a significant association in a predominantly adult cohort.
We have reported an overall cumulative incidence of etoposide HSR of 12.2% in the presence of ILFs and 5.2% without, which trended toward a significant difference. Interestingly, in patients who received multiple cycles of etoposide, the HSR risk was significantly increased by 3.8-fold with the use of ILF (p = 0.01). This suggests the possibility of additional HSR risk from ILFs as the number of treatment cycles increases.
In this study, all episodes of HSR at the second and third cycles occurred with ILF, while a similar proportion of HSRs were observed during the first cycle between the without ILF and the during ILF periods. This suggests that the first exposure to ILFs may predispose patients to the development of a reaction at subsequent cycles. Nonetheless, these results should be interpreted with caution given that our study did not consider HSR beyond the third cycle. In a study by Tillman and colleagues, approximately half of the 28 HSRs reported during ILF use (out of 32 HSRs overall), occurred during the first dose, and the remainder occurred variably during the fourth to fourteenth doses. 21 Other than this, there is very little data on the timing of the onset of HSR from intravenous etoposide with the use of ILFs. Further studies are warranted to test the hypothesis that the risk of HSR at subsequent doses is increased when ILF is used in the administration of the initial dose.
Other studies in pediatric populations have demonstrated similar associations between ILF use and etoposide HSRs. Among 486 etoposide doses administered to 192 children, Ronsley et al. 20 reported an increase in the incidence of HSRs from 1% when ILFs were not used, to 13% when they were. Tillman et al. also observed a higher rate of HSR with etoposide given over 1 h following the introduction of ILF as standard care in one of two pediatric hospitals included in their study. In the second institution, all etoposide doses were given without filter over 2 h. Combined data from both institutions also showed more reactions with the use of a filter. However, the fact that all the doses were administered over 2 h in the second hospital could have favored the lower HSR risk in the absence of filters. 21
With regard to the severity of the HSRs observed in this study, most were grade 2. Grade 3 HSRs were observed only in patients who received etoposide with ILF. No life-threatening reactions (grades ≥ 4) were experienced. For some patients, the infusion reactions required additional care and monitoring, and/or a change of etoposide formulation or its permanent discontinuation. Changes in administration rates and premedication were not collected in our study.
Our study has some limitations. Firstly, since data collection was limited to the information available in the medical record, the possibility of information bias cannot be excluded. For example, missing data between study periods could indicate the possibility of information bias and it is plausible that the medical team may have been more likely to report HSRs from the moment an increase in HSR cases was suspected.
Secondly, the demographic and clinical characteristics of patients between the three periods of our study were similar, except for cancer type and age in adult patients. This is mainly explained by the change in the standard of care for BMT conditioning regimens in lymphoma. From 2015, the conditioning protocols using diluted etoposide infusion (BEAM and BEAC protocols) were progressively replaced with a protocol with undiluted etoposide (melphalan-etoposide protocol),22–24 which lead to a gradual decrease in the number of lymphoma patients eligible for this study across the timeline being investigated. As patients who are eligible for BMT are usually younger due to expectations of enhanced tolerance to intensive chemotherapy, 25 this has also led to an increase in the average patient age over the same period. However, there is little reason to believe that these younger lymphoma patients would have a different propensity for etoposide HSR than other patients, thus this is not expected to have introduced significant selection bias.
Thirdly, we cannot exclude the possibility that, in rare circumstances, at the discretion of the medical team, some patients received non-routine premedications to prevent the first episode of HSR. Likewise, we cannot exclude the possibility that some patients received, concomitantly, drugs for other indications (e.g. prevention of chemotherapy-induced nausea and vomiting) that may have helped to prevent HSR (e.g. H1-receptor or H2-receptor antagonists, steroids, etc.). 26
Lastly, the study did not achieve the sample size of 335 patients, which was estimated to be required to detect a significant difference in HSR risk between ILF and non-ILF study periods (with an alpha of 5% and 80% power). Moreover, the incidence of HSR in our study was higher than the assumption used in our sample size estimates which leads to an even larger required sample size. This may have limited our ability to demonstrate significant differences where they do indeed exist, for example, in the overall comparison of HSR risk with and without ILF (p = 0.09).
Conclusion
Taken together, these findings suggest that ILFs could be involved in etoposide HSR, especially when an ILF was used during a multi-dose protocol. We recommend that clinicians should be informed that ILFs are a potential contributing factor to etoposide HSR and that vigilance beyond the first infusion cycle is required as the risk of HSR may be increased at subsequent cycles. Studies of larger clinical populations are required to confirm this association in adult patients. This will provide more precise estimates of HSR risk from ILF use and thus allow clinicians to better assess the balance between the risks and benefits of using an ILF in the specific context of etoposide. Lastly, the mechanism by which ILFs would promote the occurrence of HSRs remains to be investigated.
Footnotes
Acknowledgements
Special thanks to Mr. Mahukpe Narcisse Ulrich Singbo for his help with statistical data analysis (Clinical and evaluative research platform at CHU de Québec Research Center - Université Laval). We thank Mr. Etienne Camire and Ms. Stephanie Blais-Lapointe for collecting data and Dr Matthew Haren for manuscript editing.
Authors’ contribution
MJR and KD researched literature and conceived the study. MJR was involved in protocol development and gaining ethical approval. MJR and KD were in charge of data collection. Data analysis was performed by MJR and IL. All authors were involved in writing the manuscript as well as reviewing and editing the final version. All authors approved the final version of the manuscript.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Fondation du CHU de Québec (grant number 2019-4366 to MJR). IL is a clinical research scholar with funding from the Fonds de recherche du Québec-Santé.
