Abstract
Background
Cancer and cancer-related treatments are significant independent risk factors for malignant hematology (MH) patients in developing venous thromboembolism (VTE). Treatment of VTE in MH patients at the Princess Margaret Cancer Centre is predominantly initiated with low molecular weight heparin (LMWH) in accordance with guidelines. While guidelines recommend against LMWH use in patients with thrombocytopenia, prescribers may order LMWH conditionally based on platelet values. Currently, there is a lack of consistent practice with variation in both the use of conditional orders as well as the threshold of platelet values for conditional orders. The objectives of the study were to (a) describe the use of conditionally ordered LMWH based on platelet values; (b) determine its safety by measuring administration concordance with conditional orders and bleeding event rates during inpatient admission; and (c) determine its efficacy by measuring the rate of worsening VTE or recurrence during inpatient admission.
Methods
Electronic records of MH inpatients admitted between January 2017 and December 2019 and who were administered at least one dose of an LMWH for the treatment of VTE were screened.
Results
One hundred and eight patients were screened to obtain 50 eligible patients with a median age of 59 years (SD = ±18.8 years). The most frequent MH diagnosis was acute lymphoblastic leukemia (30%). Sixty percent (n = 30) of patients received conditional orders. Out of 571 administrations, 543 (95%) were administered concordantly (Χ2(1) = 472, p < 0.0001). In this group of patients, 8 patients had either documented bleeding or experienced a drop in hemoglobin >10 g/L within a 72 h time frame. No patients experienced a recurrent VTE during inpatient treatment (for up to 40 days post-admission).
Conclusions
It appears that conditionally ordered LMWH can be concordantly administered and is safe and effective in the treatment of VTE in MH patients experiencing thrombocytopenia. There were no reports of worsening or new VTE in our small sample.
Keywords
Introduction
Venous thromboembolism (VTE) can be a fatal disorder, and cancer and cancer-related treatments are risk factors for VTE.1–4 The American Society of Clinical Oncology, International Initiative on Thrombosis and Cancer, and American Society of Hematology guidelines recommend either low molecular weight heparin (LMWH) or direct oral anticoagulant (DOAC) as initial treatment for cancer patients with an established VTE due to meta-analyses confirming increased efficacy of LMWH or DOACs compared to vitamin K antagonists at reducing recurrent VTEs in patients with cancer.5–7 Although recent trials with direct oral anticoagulants (apixaban, rivaroxaban, and edoxaban) have shown comparable efficacy and safety to LMWH in VTE treatment in cancer, these studies recruited a majority of patients with solid tumors. Hematological malignancies including leukemia and lymphoma were underrepresented (<12% of study populations) and require important considerations.1,8–10 In particular, thrombocytopenia is a common complication in hematological malignancies due in part to the chemotherapy regimens used for treatment, but can also be a result of bone marrow infiltration and infection.11–15 The interplay between thrombocytopenia and VTE is complex as the risk of VTE is not reduced in thrombocytopenia, and extended periods of thrombocytopenia (>30 days) can increase the risk of recurrent VTE more than four-fold. 11 Bleeding rates in this population range from 7% to 33%, while recurrent thrombosis rates range from 10% to 44%. 11 Guidance for the treatment of VTE in hematological malignancy patients with concurrent thrombocytopenia commonly suggests a sliding dosing scale, but there is no universal agreement on cut-offs/thresholds.
Malignant hematology (MH) patients being treated for VTE during inpatient admission at the Princess Margaret Cancer Centre (PM) are often prescribed an LMWH order with administration conditions based on platelet values; a “conditional” order. The evidence to support this practice comes from guidelines that base recommendations on small, single-center retrospective studies, non-randomized prospective studies, or expert consensus.11,15,16 Extracting conclusions from previous large-scale trials examining the efficacy of LMWH in cancer patients is problematic as small subgroups (<12%) of patients had hematological malignancies and LMWH treatments were held (or patients outright excluded from the study) if platelets dropped <50 × 109/L.6,9,17,18 It would therefore be prudent to investigate if this practice, which is utilized for our inpatients, represents a safe and effective strategy to treat VTE in MH patients. This would be an opportunity to add to the limited pool of data, and findings from the study may provide a rationale to conduct a prospective study in implementing a standardized conditional LMWH order based on platelet values.
Materials and methods
Design
A single-site, retrospective chart review was conducted at PM. The study protocol was approved by the University Health Network Research Ethics Board. Individuals being treated for a hematological malignancy who were admitted as inpatients at PM between 1 January 2017 and 15 December 2019 were treated for a VTE with an LMWH were eligible for inclusion. Patients were excluded from the trial if no dose was ever administered during admission (i.e. if platelets were never above the minimum platelet threshold), or if the patient received LMWH for indications other than VTE treatment (VTE prophylaxis, arterial thrombi, atrial fibrillation/stroke prophylaxis). Data were collected from patients’ electronic patient records (EPRs) and pharmacy's electronic dispensing records (Centricity). Centricity was used to search for inpatients prescribed an LMWH by the Drug Use Evaluation function, and patients’ LMWH orders and diagnoses from EPR were assessed for inclusion in the study. The diagnosis of VTE was identified from discharge clinical notes or imaging (including peripheral venous dopplers/ultrasounds/computed tomography). The electronic medical administration record (eMAR) from EPR was used to assess the primary objective of administration concordance, and clinical notes in EPR were used to identify the safety endpoints of bleeding events and hemoglobin decreases as well as the efficacy endpoint of VTE recurrence or worsening. EPR was also used to obtain the baseline characteristics and platelet values of patients.
Outcomes
The primary objective of the study was to describe the use of LMWH for the treatment of VTE in MH patients and evaluate the administration concordance when conditionally ordered based on platelet values during inpatient admission. In this study, the criteria for administration concordance were met when the LMWH dose that was administered matched the prescribed order, which consisted of specific LMWH doses within specific platelet ranges. Administrations were considered discordant when the LMWH dose was given even though the patient's platelet values did not match the prescribed order, or the LMWH dose was not given even though the patient's platelet values permitted a dose of LMWH.
As a secondary objective, the safety of conditionally ordered LMWH based on platelet values for the treatment of VTE in MH patients was assessed by measuring bleeding events during inpatient admission. Bleeding events were initially identified from discharge summaries and categorized based on the World Health Organization (WHO) severity grading system (Appendix 1). However, it was determined that the EPR may not always record bleeding events (including WHO grade 1 bleeding events), so daily hemoglobin decreases were recorded and used as a surrogate marker of bleeding events. Hemoglobin decreases between 10 and 20 g/L within a 72 h time frame were considered minor bleeding events, and hemoglobin decreases >20 g/L within a 72 h time frame were considered major bleeding events. 19
Lastly, to evaluate the efficacy of conditionally ordered LMWH based on platelet values for the treatment of VTE in MH patients, VTE recurrence rates were recorded during inpatient admission. The discharge summary was searched for any statement on VTE recurrence or worsening, and any imaging on discharge (including peripheral venous dopplers/ultrasounds/contrast tomography) was reviewed to search for any comment on VTE recurrence or worsening. Worsening VTE is defined as the worsening of pulmonary vascular or venous obstruction. 20
Statistical analysis
Baseline characteristics including median age, percentage of hematological malignancy diagnosis, and percentage of LMWH use were determined for each group. All statistical calculations were performed using Microsoft Excel software.
Results
The preliminary screening revealed 108 patients with a hematological malignancy who were admitted as an inpatient between January 2017 and December 2019 and were ordered an LMWH. Fifty-eight patients were excluded from the trial if no dose was ever administered during admission (i.e. if platelets were never above the minimum platelet threshold), or if the patient received LMWH for indications other than VTE treatment (VTE prophylaxis, arterial thrombi, and atrial fibrillation/stroke prophylaxis). Out of the 108 patients, 50 patients met eligibility criteria (i.e. had a cancer diagnosis of hematological malignancy, a diagnosis of VTE, and were treated with at least one dose of an LMWH). and were included in the study. Baseline patient characteristics (n = 50) are outlined in Table 1. The median age of all patients was 59 years (SD = ± 18.8 years), with 26 (52%) male patients and 24 (48%) female patients. Of the 50 patients, 4 (8%) had upper extremity VTEs, 10 (20%) had pulmonary embolisms, 17 (34%) had catheter-associated VTEs, 9 (18%) had lower extremity VTEs, and 10 (20%) had other VTEs (cortical thrombi, portal vein thrombi, etc). Of the patients treated with conditional orders (N = 30), 6 (20%) at baseline had platelet levels <50 × 109/L, 8 (26%) had platelet levels between 50 × 109/L and 100 × 109/L, and 16 (53%) had platelet levels >100 × 109 /L. The most frequent diagnosis was acute lymphoblastic leukemia (ALL; 30% of all patients) and tinzaparin was the most commonly prescribed LMWH (52%), while 34% of patients received enoxaparin and 14% of patients received dalteparin.
Patient demographics and characteristics (N = 50).
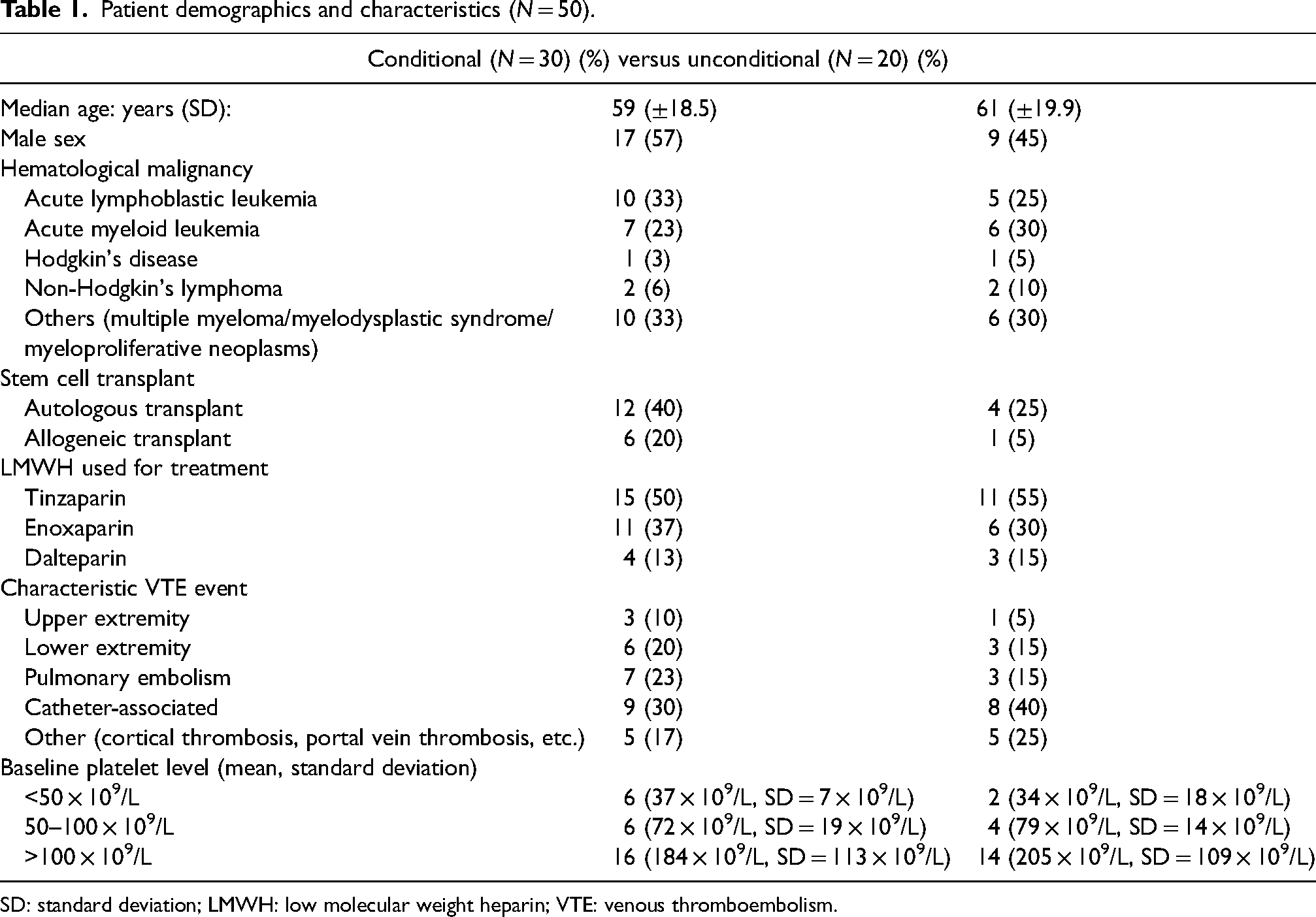
SD: standard deviation; LMWH: low molecular weight heparin; VTE: venous thromboembolism.
Of the 50 patients included in the study, 60% (n = 30) received conditional orders (including 12 patients who were initiated on conditional orders and 18 patients who had their orders changed to conditional orders; Figure 1) and 40% (n = 20) received LMWH without conditional orders. Of the 18 patients with orders changed to conditional orders, 3 patients had platelets below 50 × 109/L at the time of the order change. Only one patient had a conditional order initiated after the patient's platelet levels had decreased below a threshold that would have resulted in a dose adjustment, while the other 17 patients had conditional orders initiated pre-emptively before platelet levels dropped below the thresholds of their conditional orders. For patients with a single conditional order, the most common lower threshold was platelets >50 × 109/L (n = 12), while one patient had a single conditional order for LMWH dose if platelets >40 × 109/L and two patients had a single conditional order for LMWH dose if platelets >30 × 109/L. The characteristics of the conditional orders are listed in Table 2.
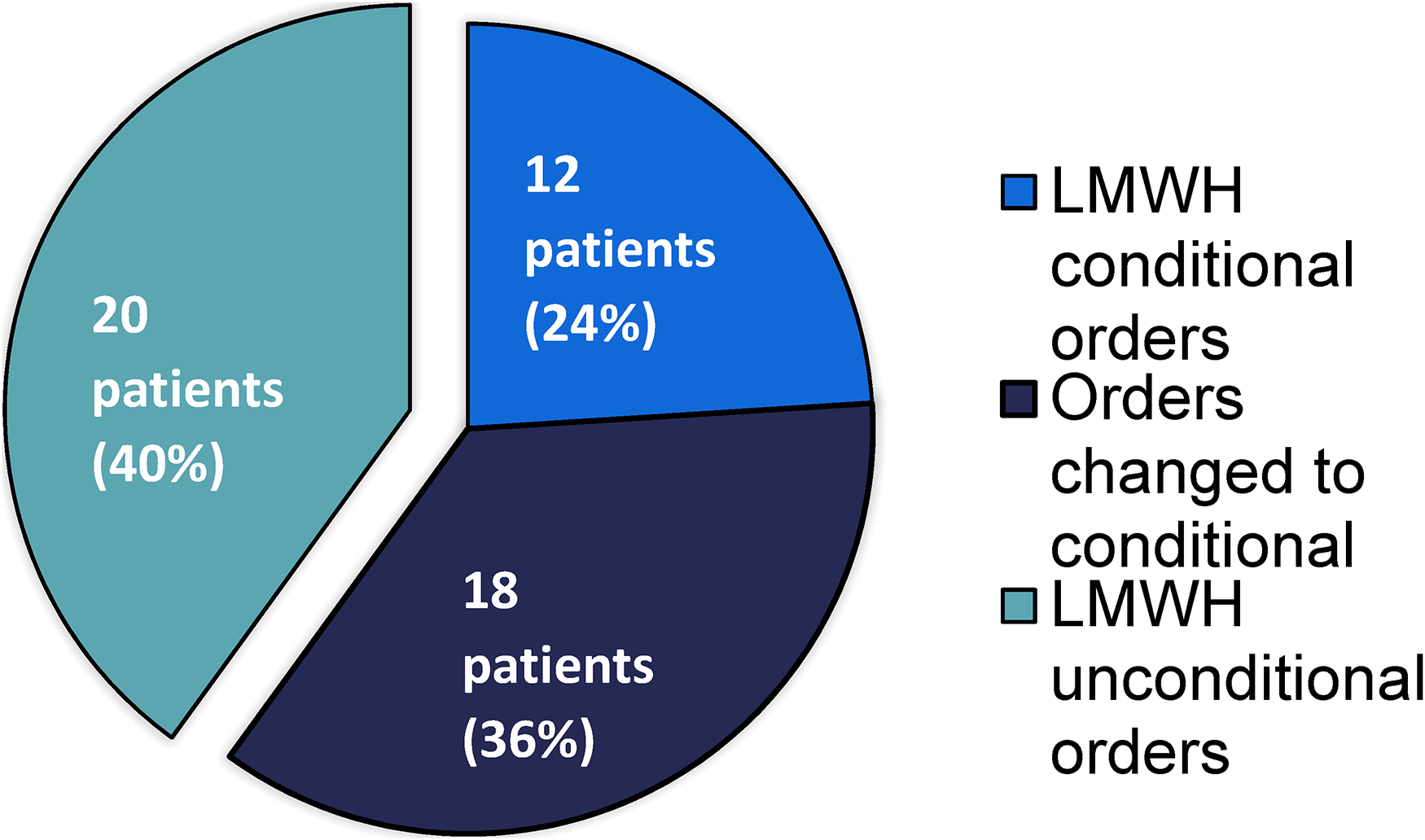
Number of patients prescribed conditional versus unconditional low molecular weight heparin (LMWH) orders.
Characteristics of conditional orders.
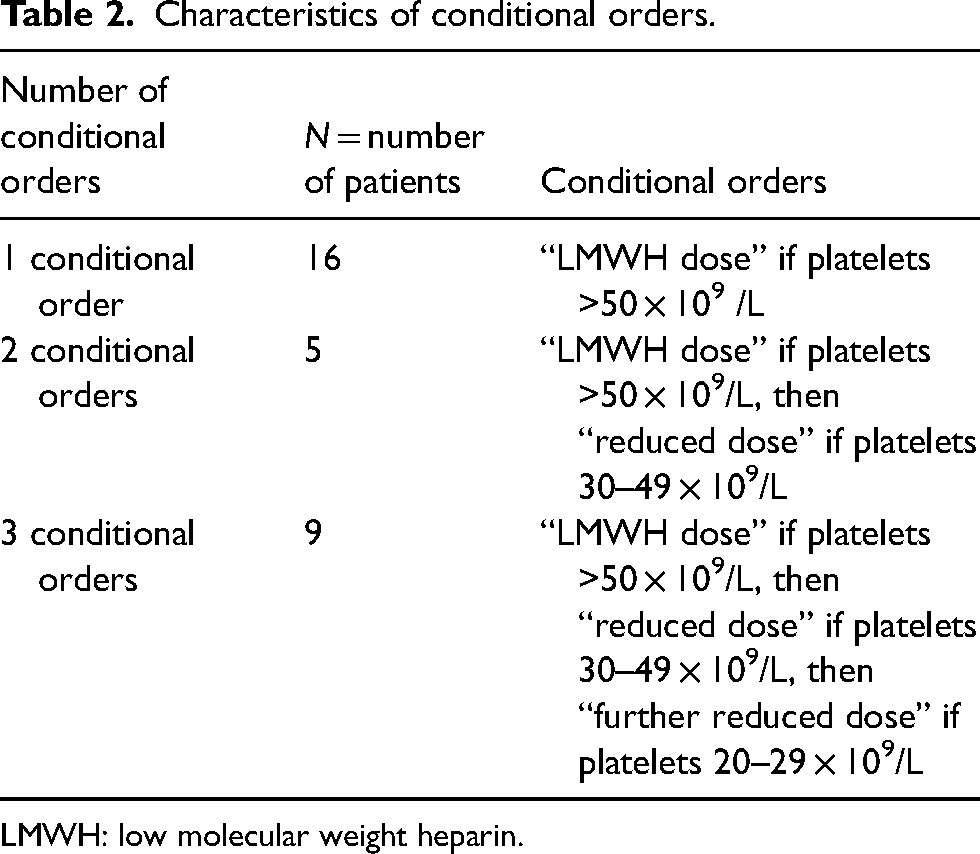
LMWH: low molecular weight heparin.
It may be noted that for unconditional orders initiated by an on-call prescriber, the order was subsequently changed to a conditional order by the attending prescriber or upon review by the unit pharmacist. Out of 571 administrations of LMWH, 543 (95%) were administered concordantly with conditional orders (Figure 2). However, 5% (n = 28) of administrations were administered discordantly in 11 patients (37%). For one patient, the documented reason for two discordant administrations was that the prescriber had ordered to administer the dose despite the platelet values. In four patients, holding LMWH administration due to a procedure was the documented reason for six discordant administrations. In the other six patients with 20 discordant LMWH administrations, no reason was documented. No discordant administration resulted in a documented bleeding event or a drop in hemoglobin.
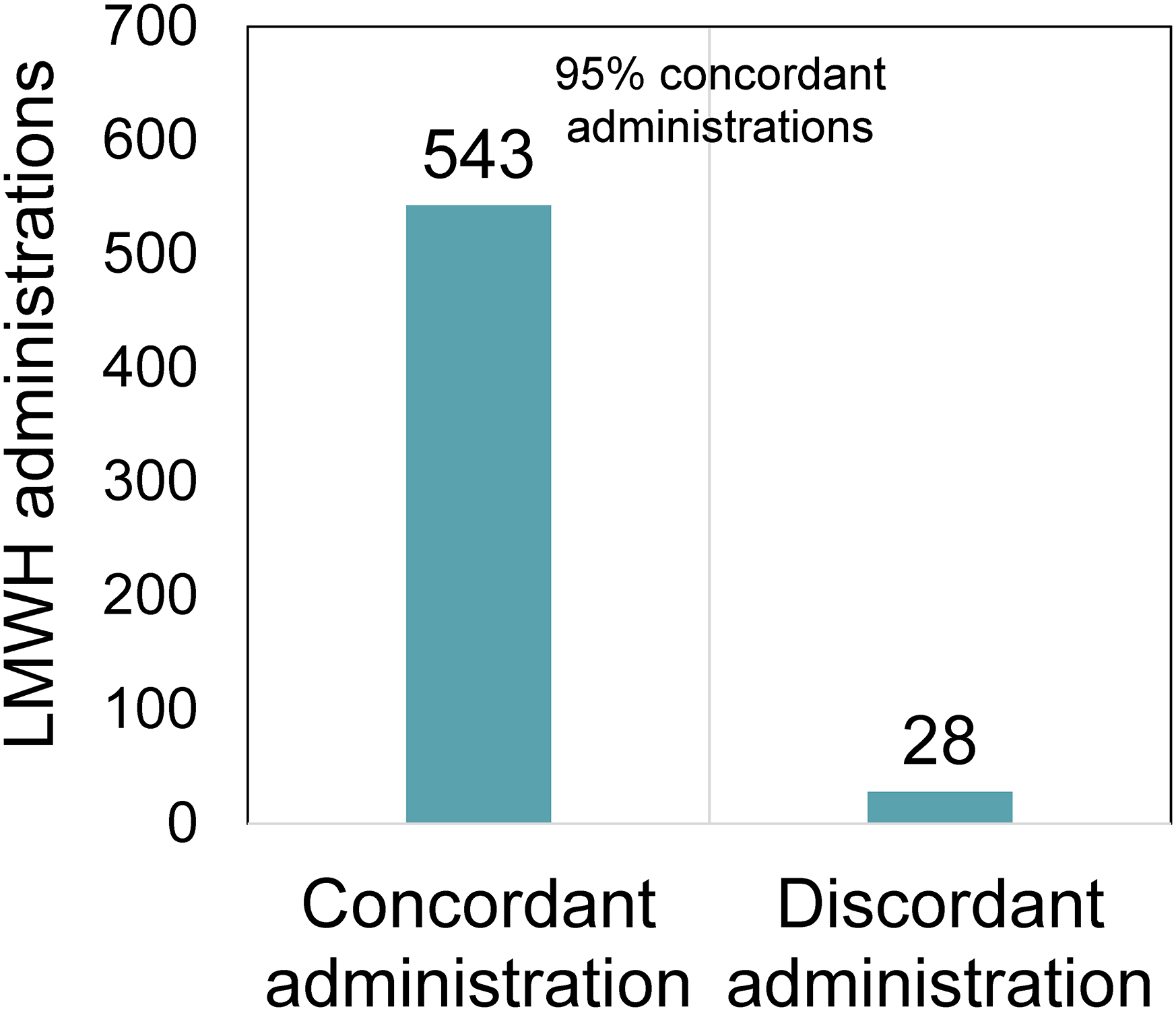
Administration concordance versus discordance of low molecular weight heparins (LMWHs) with conditional orders.
In the group of patients with conditional orders, 8 patients experienced a drop in hemoglobin between 10 and 20 g/L, while 2 patients had both a documented bleeding event and a drop in hemoglobin >20 g/L (Figure 3). Of the two documented bleeding events, one patient experienced a WHO grade 2 bleeding event (melena) when LMWH with unconditional orders was started. After 3 doses of LMWH were held and the resolution of melena, the LMWH was restarted with conditional orders, and no further bleeding events were documented. The other patient experienced a documented WHO grade 2 (bleeding event at an invasive site), but the LMWH was only held for one day before it was continued with no further documented bleeding events. No patients experienced a worsening or recurrent VTE during inpatient treatment (for up to 40 days post-admission).
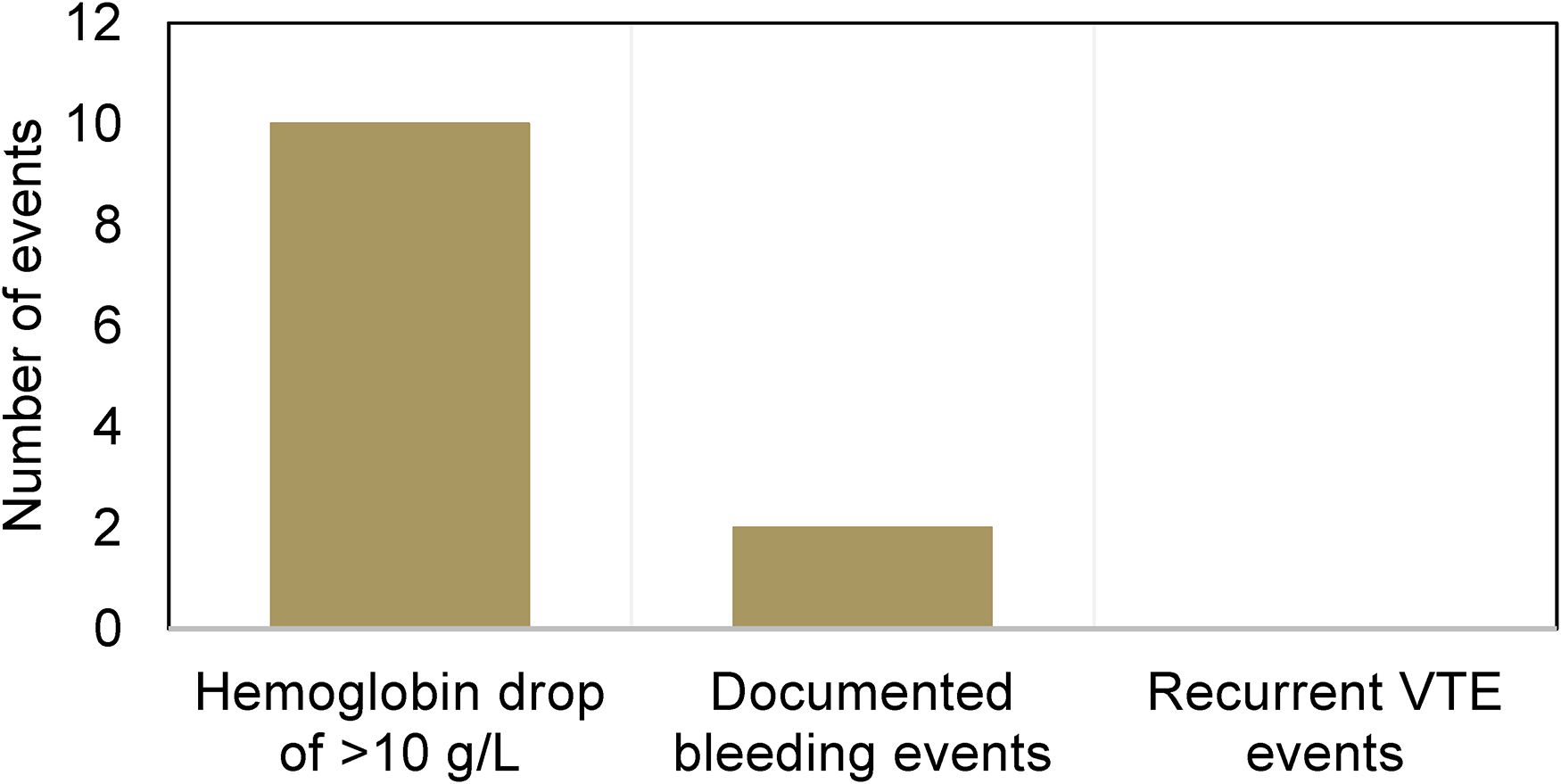
Safety and recurrent events with low molecular weight heparin (LMWH) conditional orders.
Discussion
MH patients being treated for VTE during inpatient admission at PM are often prescribed an LMWH order conditional on platelet values.11,16 This single-site, retrospective chart review was conducted to add to the body of evidence for the safety and efficacy of conditional orders of LMWH based on platelet values.
The most frequent diagnosis encountered in the subjects included in the study was ALL. This is consistent with retrospective studies and reviews reporting a high VTE incidence in ALL (10.6%), acute promyelocytic leukemia (11%), and acute myeloid leukemia (AML; 4.2%), compared with lymphomas (1.4% to 4.2% depending on lymphoma subtype) and multiple myeloma (5%).1,3,21 The frequency of VTE in ALL may be attributed in part to the treatment of ALL with L-asparaginase, which has been shown to increase the risk of VTE by 4.9-fold.1,3,22 Other factors associated with VTE occurrence in ALL include baseline thrombocytopenia, Philadelphia chromosome positivity, age over 40, presence of an additional hematologic malignancy (AML, myeloma, and lymphoma), and concurrent hormonal therapy.1,23
This study revealed the use of conditional orders with a variation in the platelet threshold cut-offs as well as the number of platelet threshold cut-offs for a single conditional order. The most common ordering scenario utilized three conditional orders/platelet threshold cut-offs. Although this gradated conditional order may follow from the available guidelines,11,16 to date there is no published comparison of efficacy or safety between conditional orders with varying numbers of threshold cut-offs (e.g. 1 condition (i.e. “LMWH dose” if platelets >50 × 109/L) versus 2 conditions (i.e. “LMWH dose” if platelets >50 × 109 /L, then “LMWH dose” if platelets 30–50 × 109 /L). Some drawbacks to these gradated orders with additional conditions include increasing the risk of the patient receiving an incorrect dose of LMWH if the order was misinterpreted, more entries for the prescriber to order, and an increase in workload for nurses to document in the eMAR. Although our study did not investigate if there was a difference in outcomes between orders with a single condition versus multiple conditions, it is reasonable to assume that standardizing platelet conditions and dose reductions should reduce administration errors. A study into the benefit of additional conditions for conditional orders may be a future direction.
In this retrospective chart review, 60% of patients were treated with a conditionally ordered LMWH. Although this is a majority of patients, the safety and efficacy of the practice suggested by the results of the study indicate that there could be greater utilization of conditional orders to improve patient outcomes. The vast majority (95%) of conditional orders were concordantly administered, suggesting conditional orders can be safely and effectively implemented by nurses. Within the 5% of conditional orders that were discordant with the prescribed order, eight discordant administrations had a documented reason, leaving only 3.5% of all conditional orders to be discordant and undocumented. No patient with a discordant administration experienced a bleeding event when an LMWH was given despite the platelet level being out of the prescribed range, and no recurrent/worsening VTE occurred in patients where LMWH was not given despite a platelet level permitting an LMWH administration. This gives us more confidence that the low number of discordant administrations did not result in adverse events for patients. Additionally, the low overall number of bleeding events and lack of worsening/recurrent VTE events suggest that conditional orders are a safe and effective strategy to treat VTE in MH patients with thrombocytopenia.
One limitation is our assessment of bleeding, which was defined based on the WHO bleeding scale and recorded through diagnoses in clinical notes or hemoglobin decreases. Minor bleeding episodes, such as epistaxis, may not have been recorded in discharge notes as they may not have impacted clinical decisions on VTE treatment. To account for this, even minor hemoglobin drops between 10 and 20 g/L were recorded as a surrogate, though it is reasonable to assume that bleeding did not occur in these instances as hemoglobin derangements in hematological malignancy are common. 3 However, this would strengthen our argument that the practice was safe, as the incidence of bleeding would then be even lower than the recorded rate. Small retrospective studies of patients with hematological malignancies have demonstrated a clinically significant bleeding risk in 11% to 27% of patients when receiving full dose anticoagulation during periods of thrombocytopenia, 1 which is in line with the bleeding rate recorded in our study (33%).
Another limitation of our study is our assessment of discordant administrations. Administration discordance (including holding doses due to bleeding, procedures, or prescriber decision) may have been appropriate to prevent bleeding but were counted as discordant due to the strict predefined definition of discordance used to decrease observer bias in recording data. Therefore, the true estimation of appropriate LMWH administration with the use of conditional orders may be higher.
Of the discordant administrations, it is interesting to note that 4 out of 28 (14%) were administered due to prescriber insistence despite platelet values that were out of range of the original order. However, this reason for administration discordance suggests that the true rate of inappropriate administration is lower than measured, further supporting the safety of conditional orders.
Although it is promising that there were no recurrences of VTE within the 40 days of inpatient visits in the patients included in the study, this is lower than the rate reported in the literature (4% to 10% in acute leukemia).1,3,21 This may be due to the small sample size of the study as well as the short duration of stay for inpatients.
Our data was collected on inpatients admitted between 2017 and 2019. However, only one new prospective study examining the use of anticoagulation based on platelet counts in patients with thrombocytopenia has been published since 2019, 15 which provided additional safety data supporting this practice. Future directions for this research include expanding the number of patients screened to add to the limited pool of data and conducting a quality improvement study of a standardized conditional order set for LMWH based on platelet values at PM. A quality improvement study with the collaboration of prescribers may ultimately increase the uptake of the practice and improve patient outcomes.
In summary, this single-site, retrospective chart review adds to the body of evidence for the safety and efficacy of conditional orders of LMWH based on platelet values. The safety and efficacy demonstrated in the study provide reassurance for PM's practice of LMWH conditional orders based on platelet values. Future studies may include a larger subject pool and be conducted as quality improvement studies with standardized conditional order sets.
Footnotes
Author contributions
HP contributed to data acquisition and analysis, drafting of the article and revisions, approval for publication, and taking responsibility for the manuscript. IP contributed to the design of the project, article revisions, approval for publication, and taking responsibility of the manuscript. KL contributed to the design of the project, article revisions, approval for publication, and taking responsibility of the manuscript. PD contributed to the design of the project, article revisions, approval for publication, and taking responsibility of the manuscript. CM contributed to the design of the project, article revisions, approval for publication, and taking responsibility of the manuscript. PP contributed to the design of the project, article revisions, approval for publication, and taking responsibility of the manuscript. RK contributed to the design of the project, article revisions, approval for publication, and taking responsibility of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Appendix 1. Modified WHO severity grading system
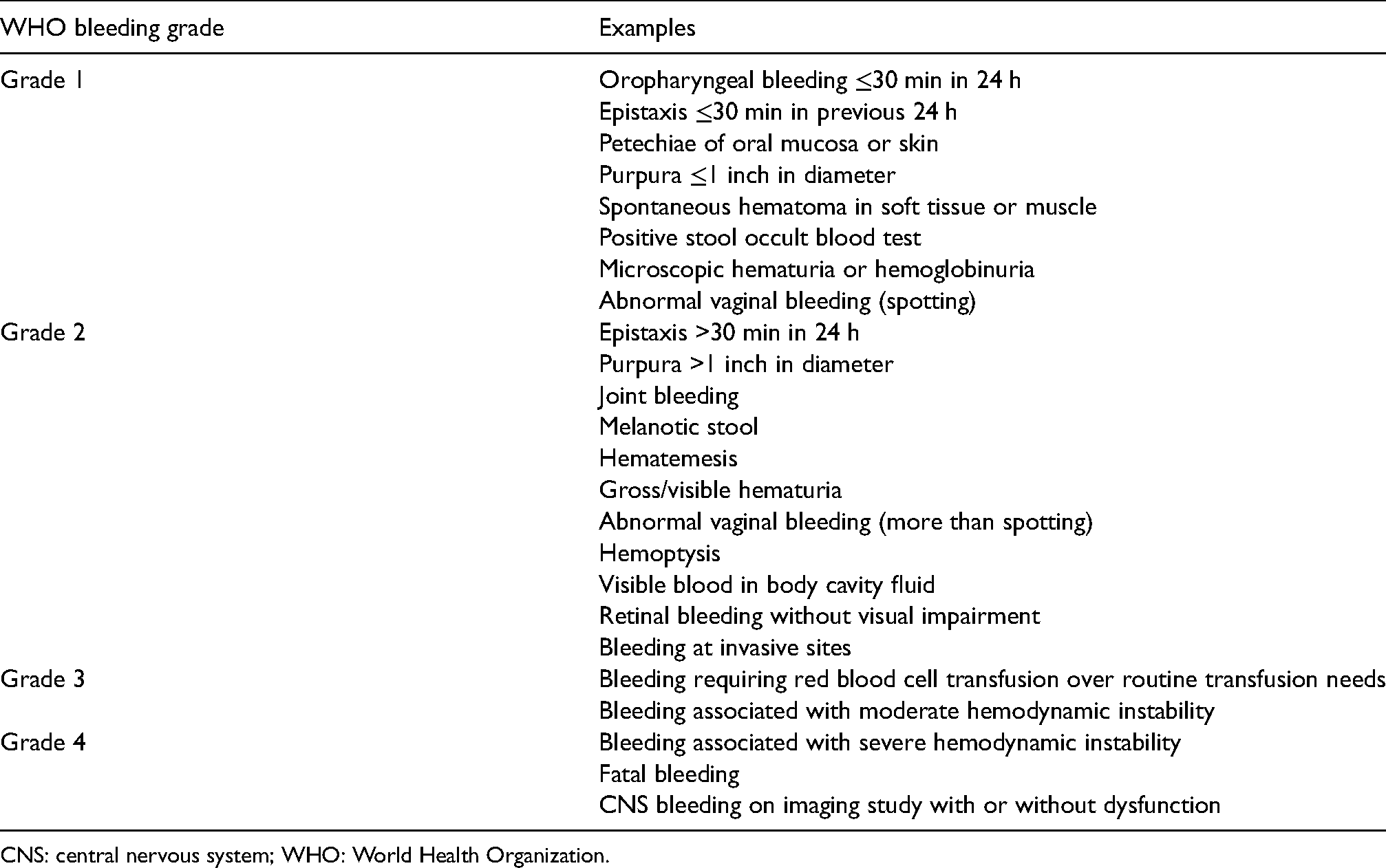
| WHO bleeding grade | Examples |
|---|---|
| Grade 1 | Oropharyngeal bleeding ≤30 min in 24 h Epistaxis ≤30 min in previous 24 h Petechiae of oral mucosa or skin Purpura ≤1 inch in diameter Spontaneous hematoma in soft tissue or muscle Positive stool occult blood test Microscopic hematuria or hemoglobinuria Abnormal vaginal bleeding (spotting) |
| Grade 2 | Epistaxis >30 min in 24 h Purpura >1 inch in diameter Joint bleeding Melanotic stool Hematemesis Gross/visible hematuria Abnormal vaginal bleeding (more than spotting) Hemoptysis Visible blood in body cavity fluid Retinal bleeding without visual impairment Bleeding at invasive sites |
| Grade 3 | Bleeding requiring red blood cell transfusion over routine transfusion needs Bleeding associated with moderate hemodynamic instability |
| Grade 4 | Bleeding associated with severe hemodynamic instability Fatal bleeding CNS bleeding on imaging study with or without dysfunction |
CNS: central nervous system; WHO: World Health Organization.
