Abstract
Inpatient use of inotuzumab ozogamicin (IO) at our academic medical center has increased since its FDA approval in 2017. Administration of IO does not require hospitalization and is appropriate for outpatient use. The aim of this report is to assess the appropriateness of use, overall inpatient utilization, and cost of IO. This is a retrospective chart review of patients that received at least one dose of IO while admitted to the hospital. Data points included rationale for inpatient IO administration, hospital admission reason, number of IO doses and number of vials used, length of stay, in-hospital mortality, percentage of admissions that were new-starts, outpatient continuation of IO, use of concomitant regimens, and CD22 positivity. Between September 1, 2017, and June 30, 2022, 55 doses of IO were identified. Of the 29 unique admissions, common rationales for inpatient IO use included high disease burden/tumor lysis syndrome risk (31%) and use of a regimen requiring hospitalization (28%). The rationale for hospitalization was most commonly ‘chemotherapy administration’ (34%) and ‘relapsed/refractory disease’ (38%). Median length of stay was 23 days, most were new starts (76%), and 5 patients died during the associated admission. Only 63% of patients continued therapy in the outpatient setting. The inpatient use of IO was associated with a prolonged length-of-stay, a 17% in-hospital mortality, and represents a significant cost burden to the health system. As a result of these findings, guidelines for inpatient use of IO were implemented across the health system.
Keywords
Introduction
Health systems nationwide are facing the challenge of providing safe, evidence-based patient care while containing costs in the setting of decreased reimbursement. Acute care hospitals, particularly non-profit academic medical centers, operate on predetermined budgets with small margins. Chemotherapy stewardship programs are increasingly being implemented within health systems to optimize the use of cancer-directed therapy (CDT) and decrease inappropriate inpatient administration.
Inotuzumab ozogamicin (IO) is a CD22-directed monoclonal antibody-drug conjugate (ADC) covalently linked to the cytotoxic agent calicheamicin, that was granted approval by the FDA on August 17, 2017, for the treatment of adults with relapsed or refractory (R/R) B-Cell acute lymphoblastic leukemia (ALL) based on the results of the phase 3 INO-VATE ALL trial.1, 2 At UC San Diego Health, IO was added to formulary with oversight from the Oncology Pharmacy and Therapeutics (P&T) subcommittee. Inpatient use of IO was restricted to the bone marrow transplant (BMT) service and required approval from two BMT attending physicians. The requirement for approval of inpatient use was a result of the discussion surrounding its high-cost and the desire to ensure appropriate use. As of March 2023, the average wholesale acquisition cost (WAC) of each 0.9 mg vial was approximately $25,772.81 based on Micromedex REDBOOK data. 3 Payor billing for inpatient admissions uses the diagnosis-related group (DRG) method and therefore payor coverage for the hospital stay may not cover the expense of medications provided. Administration of IO, in and of itself, does not require hospitalization and is appropriate for outpatient use. The purpose of this review is to assess the appropriateness of use, overall inpatient utilization, and cost of IO.
Methods
The study was designated by the UC San Diego Office of IRB Administration as not human subjects research and exempted from continuing review. The electronic health records (Epic® Systems Corporation) of patients at the University of California, San Diego between September 1, 2017, and June 30, 2022, were reviewed retrospectively. All patients age 18 and above who were treated with IO in the acute care setting at any point in their disease course were included in this study. Baseline demographics were collected and included age, gender assigned at birth, baseline Eastern Cooperative Oncology Group (ECOG) performance status, and CD22 status. Data related to the hospital admission included the reason for hospitalization, route of admission, length of stay (LOS), time from admission to first IO dose, discharge ≤48 h after IO administration, and in-hospital mortality. IO administration data included prescriber-identified rationale for inpatient use, percentage of new-starts, continuation to the outpatient setting, monotherapy vs use with a concomitant regimen, number of doses per patient, total number of vials purchased, and associated costs. All data points were collected into a password-protected excel file, saved on a password-protected computer within the locked Department of Pharmacy. The sample size was small and therefore descriptive statistics were calculated using Microsoft® Excel®.
Results
In total, 55 doses of IO were given across 29 inpatient admissions of 24 unique patients were identified from September 1, 2017, to June 30, 2022. Across the 29 admissions, patients were mostly female (69%) and the median age was 51 years (range 24–73 years) (Table 1). Most patients had a baseline ECOG score ≤ 2 (72%) on admission and all patients had a diagnosis of ALL. One patient had CD22-negative ALL and one patient had newly diagnosed, treatment-naïve B-Cell ALL. The majority of admissions (52%) occurred via the emergency department (ED), and the median length of stay was 23 days (Table 2). The most common prescriber-identified reasons for the associated hospital admission were ‘chemotherapy administration’ (34%) and ‘relapsed/refractory disease’ (38%); other documented reasons were ‘infection’ (21%), ‘new diagnosis’ (3%) and ‘symptoms requiring admission’ (3%). Thirty-four percent of all discharges occurred within 48 h of the last dose of IO and five patients (17%) expired while hospitalized; two were due to respiratory failure, one due to multiorgan failure, one due to progressive anemia and pneumonia, and one due to nonspecific progressive disease. Two of the patients that experienced in-hospital mortality received a dose of IO within 1 week of death.
Baseline characteristics (N = 29 admissions).

*Upon admission.
ECOG: Eastern Cooperative Oncology Group performance status.
Hospitalization data (N = 29 admissions).
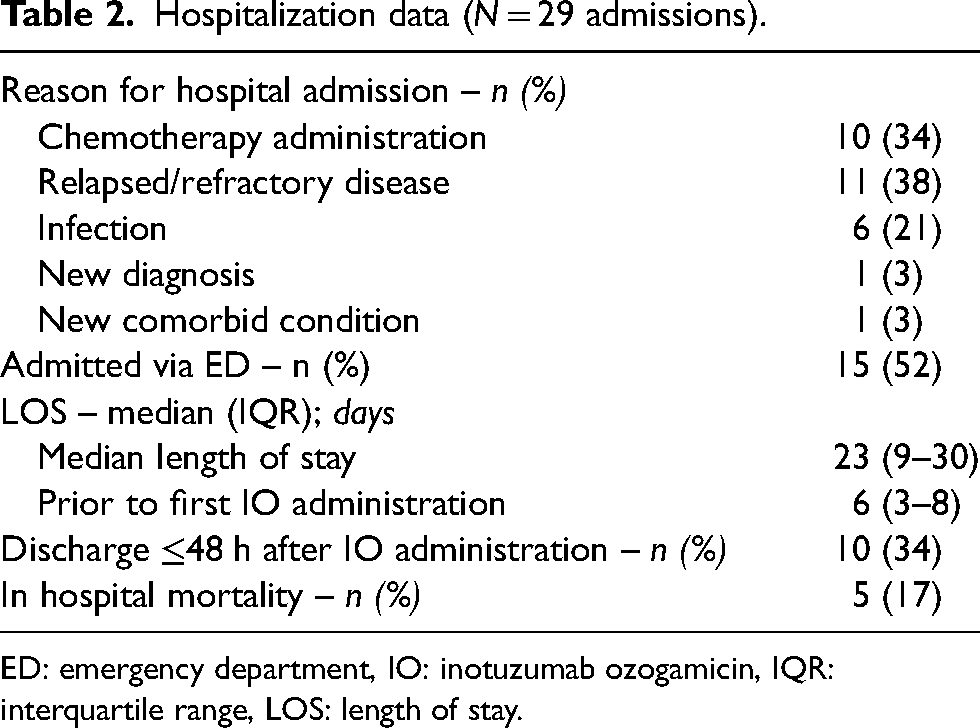
ED: emergency department, IO: inotuzumab ozogamicin, IQR: interquartile range, LOS: length of stay.
All uses of IO were approved by at least two BMT attending physicians. Justifications documented by prescribers for inpatient IO use per admission (N = 29) were ‘high disease burden/tumor lysis syndrome risk’ (30%), ‘use of an additional regimen requiring hospitalization’ (27%), ‘acute illness’ (21%), ‘transportation/housing/social issues’ (13%) and ‘relapsed disease’ (10%) (Table 3). Twenty-two patients received their first dose of IO during a hospital admission (76%), most initial doses were given within a week of admission (62%), and therapy was continued in the outpatient setting for 15 of 24 patients (63%). Of the four patients who were discharged and did not continue IO therapy in the outpatient setting, one was due to lack of response, one was readmitted for future cycles and then achieved remission, one had completed therapy, and one had a change in diagnosis. During nine admissions (31%) IO was administered as part of a combination chemotherapy regimen; seven of these were some iteration of hyperCVAD/mini-hyperCVD ± rituximab. Inpatient use of IO increased since 2017, with 78% of all inpatient-administered doses occurring in 2021 and the first half of 2022. The approximate acquisition cost for the 55 inpatient doses of IO was more than $2,000,000; the equivalent of 37.9 vials of IO was wasted at an approximate cost of $975,000 based on REDBOOK data.
Inotuzumab ozogamicin administration data (N = 29 admissions).
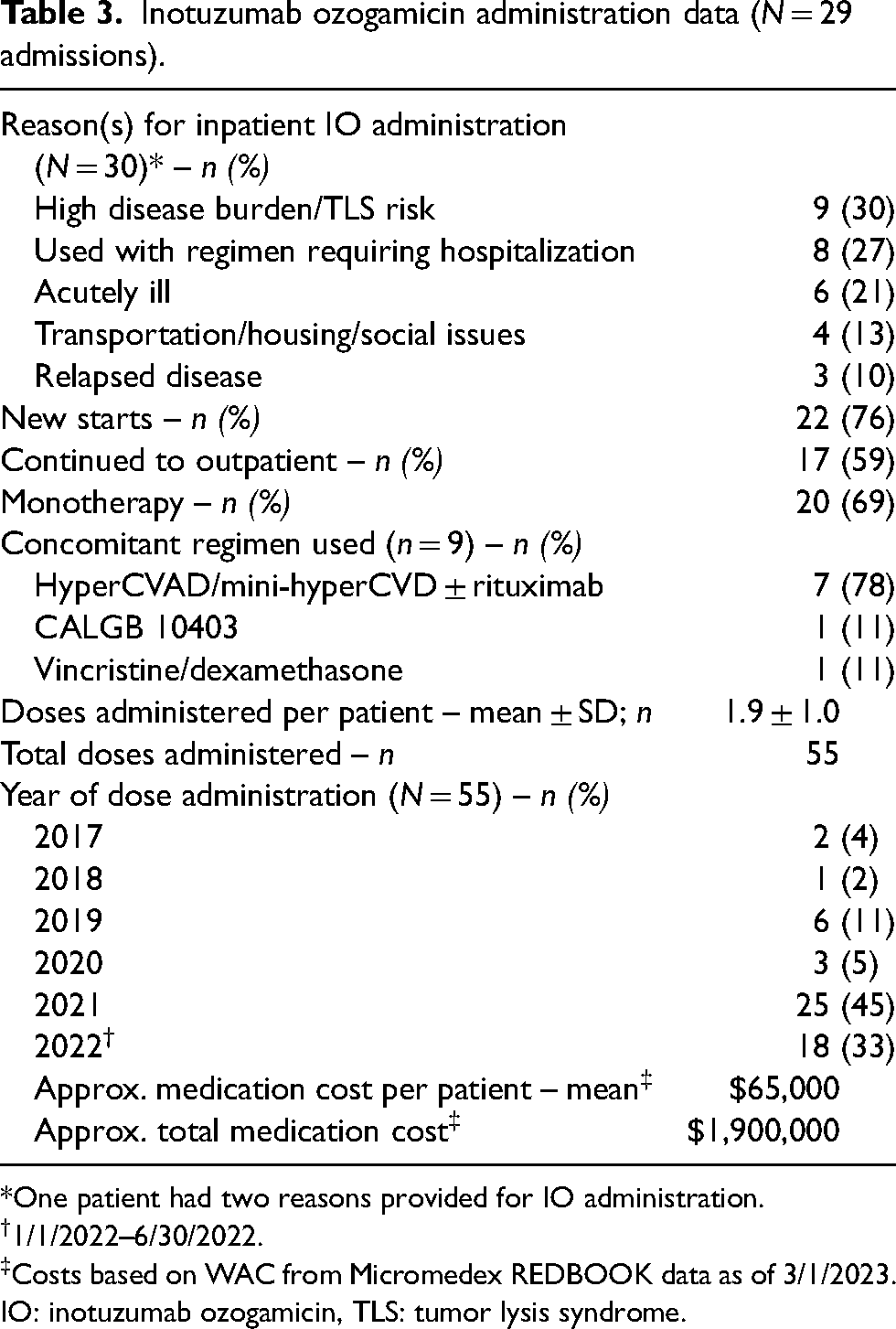
*One patient had two reasons provided for IO administration.
1/1/2022–6/30/2022.
‡Costs based on WAC from Micromedex REDBOOK data as of 3/1/2023.
IO: inotuzumab ozogamicin, TLS: tumor lysis syndrome.
Discussion
Overall, many patients were admitted for reasons that are not justified by the requirements for IO treatment. A third of patients were discharged within 48 h of dose administration, and more than a tenth received inpatient treatment due to lack of transportation or housing. Our study found similar results to Marks et al., which showed that while the overall hospitalization rate was lower than standard of care, 45.7% of subjects enrolled in the IO arm (N = 164) of the INO-VATE trial were admitted to the hospital for ‘scheduled treatment related to ALL.’ The authors acknowledged that this could have been affected by the fact that subjects were on protocol at the time. 4 Several opportunities for optimization of the inpatient use of IO were identified through this review. A third of admissions were for IO administered as part of a combination regimen; most of these were a variation of hyperCVAD or mini-hyperCVD ± rituximab. The paper cited by providers for the use of this combination is a single-arm phase 2 study published in 2018 investigating the use of mini-hyperCVD with IO ± rituximab. 5 In this paper, a post-hoc analysis comparing IO + mini-hyperCVD ± rituximab (n = 59) to historical data from similar patients receiving IO alone (n = 84) found improvement in objective response rate (ORR) [75% vs. 63%, respectively (P = .02)] and median overall survival (OS) [9.3 months vs. 5.6 months, respectively (P = .02)]. However, it is unclear from what population the historical controls were selected and how they were selected. A second single-arm phase 2 study from the same group investigating the use of IO + mini-hyperCVD ± blinatumomab ± rituximab was published in 2021. 6 In this paper, a post hoc analysis was also conducted comparing IO + mini-hyperCVD ± blinatumomab ± rituximab (n = 96) to historical data from similar patients receiving IO monotherapy (n = 84) that found improvement in median OS [13 months vs. 6 months, respectively (P < 0.001)]. 6 This study was also limited by its comparison to historical controls and unclear source of these historical controls. Based on these studies, the National Comprehensive Cancer Network (NCCN) guideline lists IO + mini-hyperCVD ± blinatumomab as an “other recommended regimen” for R/R Ph-negative B-ALL, with IO monotherapy as a preferred category 1 recommendation. 7 While the data may be lacking for the use of IO with these combination regimens, the administration of IO could have at the very least been completed prior to admission for the regimen or after discharge. Also of note, HyperCVAD-A can be safely administered in the outpatient setting negating the need for admission at all. Two patients received IO for indications that do not align with its FDA approval; CD22-negative disease, for which there is a lack of data, and treatment-naïve ALL, which is not recommended by the NCCN guidelines. 7 Had guidelines been in place to prevent the use of IO outside of FDA approved indications, 11 doses would not have been administered.
Four admissions (13.8%) were planned for the inpatient setting due to social, housing, or transportation issues, with an associated IO acquisition cost of approximately $380,000. This acquisition cost must be considered in addition to the indirect resources used during the hospitalizations. Currently, at our institution, there is a significant lack of coordinated systemic social support in place for patients requiring housing and transportation for chemotherapy administration in the infusion centers. With institutional patient and caregiver support in the form of rideshare vouchers and low or no-cost hotel rooms, these four admissions could have been avoided entirely for much less than the cost of IO alone. Additionally, outpatient administration in these instances would have allowed for acquisition at discounted 340B prices and for improved payor reimbursement as well as an expected increase in patient satisfaction. Of note, when the entire contents of a vial are not used to compound a dose in the inpatient setting, the remaining amount is discarded and cannot be billed for; however, waste may be billed to payors when used at infusion centers. A significant number of patients were discharged within 48 h of a dose of IO; in theory, the patients were medically stable for discharge and the doses could have been deferred to the outpatient setting. Had this occurred, 5 inpatient doses could have been avoided. Two patients received their last dose of IO within one week of death, indicating that administration of cancer directed therapy may not have been appropriate at that time.
Based on the results of this evaluation, appropriate inpatient use guidelines for IO were developed (Table 4). They are as follows: (1) restrict use to CD22-positive disease, (2) restrict use to R/R B-ALL pending FDA approval for newly diagnosed B-ALL, (3) prescribers should defer all IO administration to outpatient whenever possible, (4) restrict administration beyond the first dose to the outpatient setting unless inpatient care is deemed medically necessary, and (5) prohibit the use of mini-hyper-CVD or hyperCVAD-based regimens in combination (pending availability of high-quality data). CD22 disease positivity is defined per the phase 3 INO-VATE ALL trial by Kantarjian et al. as ≥20% blasts CD22-positive, with surface CD22 expression measured by fluorescence-activated cell sorting (FACS); immunohistochemistry analysis is allowed in patients with (1) dry tap or (2) if BM aspirate was inadequate and/or with insufficient circulating blasts for FACS. 2
Guidelines for inpatient use of inotuzumab.

Had the above restrictions been in place, the institution could have saved approximately $850,000 in the 5-year period that was studied. The recommendations were approved by the institution's Oncology P&T subcommittee, where IO was originally presented for formulary evaluation, and the guidelines were implemented across the health system. While our institution lacks a formal, multidisciplinary chemotherapy stewardship program, these guidelines and others have initiated discussions about the development of such a service. The findings regarding admissions related to lack of transportation and housing were also referred to the Oncology Transitions of Care Committee to improve social support for patients and caregivers.
There were several limitations to our evaluation including the retrospective nature and lack of consistency in documentation of prescriber rationale for use. Additionally, the small sample size may limit the generalizability of the results.
Conclusion
The inpatient use of IO represents a significant cost burden to the health system and precludes outpatient reimbursement for drug cost and administration. Appropriate use guidelines for IO are essential for the inpatient setting. A portion of the inpatient use of IO at UCSD Health could have potentially been deferred to outpatient administration.
Footnotes
Abbreviations
Author contributions
KNW and JGM designed the study; KNW collected the data; KNW and JGM performed the analysis and interpreted the results; KNW and JGM drafted the manuscript. All authors reviewed and approved the final version of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
