Abstract
Background
Inotuzumab ozogamicin (InO), a CD22-directed antibody–drug conjugate, is effective in relapsed or refractory B-cell acute lymphoblastic leukemia (R/R B-ALL), but hepatotoxicity remains a recognized adverse effect, most commonly sinusoidal obstruction syndrome (SOS). We previously described a distinct hepatotoxic pattern characterized by recent InO exposure, elevated liver function tests (LFTs), thrombocytopenia, and abnormal hepatic imaging, with biopsy demonstrating hepatic sinusoidal congestion (HSC) without venular occlusion. We termed this entity Calicheamicin Syndrome. This study expands upon that work through a retrospective cohort analysis.
Methods
Adult patients with R/R B-ALL treated with InO between 2018 and 2024 were retrospectively reviewed. Hepatotoxicity was defined as AST, ALT, ALP, or total bilirubin ≥2× the upper limit of normal within 30 days of InO administration. Patients were stratified by liver biopsy status, laboratory trends, clinical features, and imaging findings.
Results
Among 21 patients treated with InO, all developed LFT abnormalities, with peak values occurring a median of 16 days after the final dose. Six patients (28.6%) underwent liver biopsy, all demonstrating HSC without features of SOS. Biopsied patients experienced significantly deeper thrombocytopenia (median platelet nadir 26 × 109/l vs. 80 × 109/l; p = 0.03), while ALP elevations were similar between groups. Median time to hepatotoxicity improvement after InO discontinuation was 18 days, and no patients progressed to SOS.
Conclusion
Calicheamicin Syndrome represents a reproducible and reversible hepatotoxic entity distinct from SOS, characterized by InO exposure, transaminitis, thrombocytopenia, abnormal hepatic imaging, and HSC on pathology. Early recognition may facilitate monitoring and prevent progression to severe hepatic injury.
Keywords
Introduction
B-cell acute lymphoblastic leukemia (B-ALL) is an aggressive hematologic malignancy characterized by the proliferation of immature B-cell precursors in the bone marrow. Standard frontline therapy typically involves multi-agent chemotherapy regimens incorporating corticosteroids, vincristine, anthracyclines, and asparaginase, often followed by consolidation with allogeneic hematopoietic stem cell transplantation (Allo HSCT) in high-risk or relapsed patients.1,2 Despite these intensive therapies, relapse remains a major risk among adults. The therapeutic landscape has evolved considerably with the introduction of targeted immunotherapies, including monoclonal antibodies, bispecific T-cell engagers, and antibody–drug conjugates (ADCs). Among these, inotuzumab ozogamicin (InO), a CD22-directed ADC, has demonstrated significant efficacy in relapsed or refractory B-ALL (R/R B-ALL), offering a potential bridge to transplant and improved remission rates compared to standard chemotherapy.1–3
InO consists of a humanized monoclonal antibody linked to calicheamicin, a potent DNA-cleaving cytotoxic agent that induces double-stranded DNA breaks after reductive activation.4,5 The INO-VATE trial demonstrated significantly improved complete remission (CR) and minimal residual disease (MRD) clearance rates with InO compared to standard chemotherapy regimens, leading to its regulatory approval in 2017.6–9 Despite these therapeutic benefits, hepatotoxicity, including sinusoidal obstruction syndrome (SOS)/veno-occlusive disease (VOD), remains a serious and dose-limiting adverse effect, especially in patients undergoing Allo HSCT.6–8,10
InO hepatotoxicity and “calicheamicin syndrome”
SOS is a vascular liver injury characterized by sinusoidal endothelial cell damage, centrilobular necrosis, and venular obstruction that can culminate in hepatic failure.9,11–17 However, clinical experience with InO has revealed a broader hepatic toxicity spectrum, including cases that present with liver enzyme elevation and imaging abnormalities in the absence of classic SOS findings. McDonald et al. described histologic patterns of endothelial injury with calicheamicin-based ADCs, suggesting a direct toxic effect on hepatic sinusoids. 18
In our previously published case series, we reported six patients who developed hepatic sinusoidal congestion (HSC) following InO therapy. 19 Hepatic sinusoidal congestion describes congestion in the sinusoidal regions of the liver, likely from impaired venous outflow due to sinusoidal narrowing, portal vein insufficiency, other obstructions from neoplasms, or other hepatic injury or inflammation, but clinical symptoms do not manifest as severely as SOS.20–23 In our case series liver biopsies consistently demonstrated sinusoidal dilation and congestion without venular occlusion or centrilobular necrosis. These findings prompted us to propose the term “Calicheamicin Syndrome,” referring to a distinct, non-SOS hepatotoxicity driven by microvascular injury from the calicheamicin payload. The syndrome consists of four components including the use of InO, elevated liver function tests (LFTs), thrombocytopenia, and abnormal liver imaging, with symptoms all resolving with discontinuation of InO and supportive care including use of ursodiol.19,24,25 The syndrome could be identified clinically but supported diagnostically with a liver biopsy confirming HSC.
We conducted a retrospective cohort study of all InO-treated patients with B-ALL at Loma Linda University Cancer Center (LLUCC) between 2018 and 2024. Our objectives were to quantify the incidence of hepatotoxicity and biopsy-confirmed HSC, to characterize the biochemical and temporal patterns of InO-related liver injury, and to refine the clinical and mechanistic framework of Calicheamicin Syndrome. This expanded analysis provides further evidence that InO can induce a spectrum of hepatic injury distinct from SOS and highlights the importance of early recognition and monitoring of this emerging toxicity.
Methods
Study design and setting
We conducted a single-center retrospective cohort study of adult patients with r/r B-ALL who received InO at LLUCC between January 2018 and December 2024. The study was performed in accordance with institutional ethical standards. Institutional review board (IRB) approval was obtained, and patient data were de-identified prior to analysis.
Ethics statement
This study was approved by the LLU Health IRB (IRB# 5240644) and was determined to pose minimal risk to participants. The IRB granted a waiver of informed consent in accordance with 45 CFR 46.116 and a waiver of HIPAA authorization in accordance with 45 CFR 164.512, given the retrospective nature of the study.
Patient selection
Eligible patients were ≥18 years old, diagnosed with CD22-positive B-ALL, and received at least one cycle of InO. Patients with prior hepatic dysfunction due to viral hepatitis (hepatitis A, B, or C), alcoholic liver disease, or known hepatic metastases were excluded to minimize confounding.
Patients were included if they met criteria for InO-associated hepatotoxicity defined as
elevation in aspartate aminotransferase (AST), alanine aminotransferase (ALT), alkaline phosphatase (ALP), or total bilirubin (Tbili) ≥ 2 times the upper limit of normal (ULN) during or within 30 days after InO administration with or without clinical symptoms (right upper quadrant [RUQ] pain, ascites, or encephalopathy). Associated hepatotoxic medications such as azole antifungals, asparaginase, and methotrexate were reviewed to minimize other drug-induced causes of hepatotoxicity.
Data collection
Clinical and laboratory data were obtained from the institutional electronic health record. Parameters extracted included demographics, age, sex, ethnicity, body mass index (BMI), and performance status (ECOG). Treatment history includes prior chemotherapy regimens, number of InO cycles, cumulative dose (mg/m2), and stem cell transplant status. Laboratory trends included AST, ALT, ALP, Tbili, and platelet counts which were recorded at baseline (pre-InO), at the peak of hepatotoxicity, and after recovery or downtrend following InO discontinuation. Imaging data included abdominal ultrasound and CT imaging which were reviewed for hepatomegaly, steatosis, or sinusoidal abnormalities. Histopathology evaluated liver biopsy results to assess for HSC or SOS. SOS was defined per European Society for Blood and Marrow Transplantation (EBMT) 2016 diagnostic criteria. Hepatotoxicity was graded according to CTCAE version 5.0.
Definition of hepatic sinusoidal congestion and calicheamicin syndrome
HSC was defined as sinusoidal dilation or congestion within hepatic lobules on biopsy in the absence of venular occlusion, centrilobular necrosis, or histologic criteria for SOS.
Calicheamicin Syndrome was clinically defined as the presence of documented InO exposure, elevated LFTs ≥2 times ULN, thrombocytopenia temporally associated with hepatotoxicity, and abnormal liver imaging. Liver biopsy was obtained when LFT abnormalities persisted after InO discontinuation or when clinical differentiation between sinusoidal congestion and potential early SOS could not be confidently made based on clinical, laboratory, and imaging evaluation alone.
Outcome measures
The primary endpoint was the incidence of biopsy-confirmed HSC among patients treated with InO. Secondary endpoints included temporal relationship between InO administration, hepatotoxicity onset, and resolution, magnitude, and pattern of LFT and platelet changes, and clinical outcomes including hospitalization and symptom resolution.
Statistical analysis
Continuous variables were summarized as medians with interquartile ranges (IQR), and categorical variables as frequencies and percentages. Patients with biopsy-confirmed HSC were compared to those without biopsy using the Mann–Whitney U test for continuous variables and Fisher's exact test for categorical variables. Correlation between peak LFT elevation and platelet nadir was assessed using Spearman's rank correlation coefficient (ρ). A two-sided p < 0.05 was considered statistically significant.
Results
Patient characteristics
Table 1 describes the patients’ characteristics. A total of 21 patients with R/R B-ALL, who received InO, were included in this analysis. The median age was 31 years (range 7–83), 52% were female, and 67% were Hispanic. All patients’ LFTs were within normal limits prior to InO therapy, with median AST 24.5 U/l, ALT 17 U/l, ALP 95 U/l, and Tbili 0.3 mg/dl The majority received InO as salvage therapy for R/R disease for a median of 2 cycles (range 1–3 cycles) and median dose of 4.4 mg/m2 for InO. Four patients (19%) achieved CR, while the remainder had R/R disease at the time of analysis. No patients had pre-existing SOS, active viral hepatitis, or were receiving concurrent hepatotoxic medications at baseline.
Patient characteristics.

Liver function changes during InO therapy
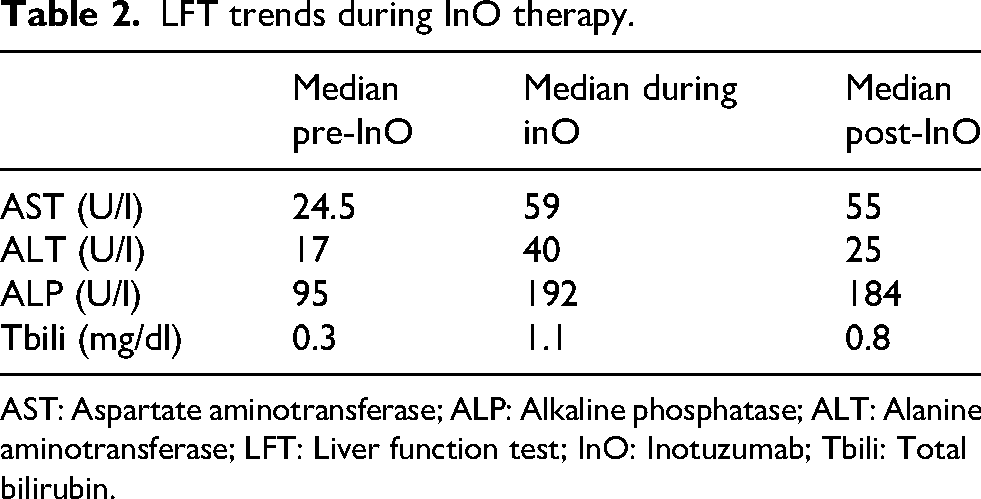
Table 2 and Figure 1 describe the LFT trends during InO therapy. Hepatotoxicity was a common finding in this cohort ranging from mild to moderate in severity. All 21 patients experienced some degree of LFT elevation during or immediately after InO therapy. Patients who received a liver biopsy received a median of 2 cycles and median dose of 7.5 mg/m2 for InO while patients who did not receive a liver biopsy received a median of 2 cycles and median dose of 1.6 mg/m2. Median AST rose from 24.5 U/l pre-treatment to 59 U/l during therapy, and median ALT increased from 17 U/l to 40 U/l, indicating about 2–3 times increase while on InO. ALP demonstrated the highest increase with a median of 95 U/l to 192 U/l during therapy. Median Tbili increased from a median baseline of 0.3 mg/dl to 1.1 mg/dl during therapy. LFT elevations generally peaked at a median of 16 days after the last InO dose. The majority of cases represented grade 1–2 transaminitis by CTCAE v5.0 criteria, and no patient developed fulminant hepatic failure or met EBMT diagnostic criteria for SOS. Clinical symptoms were mild; most patients were asymptomatic or reported RUQ abdominal discomfort. After cessation of InO, LFTs gradually declined, approaching baseline values within a median of 18 days. One patient experienced delayed normalization over approximately three months, but all patients eventually recovered with supportive care and careful monitoring.
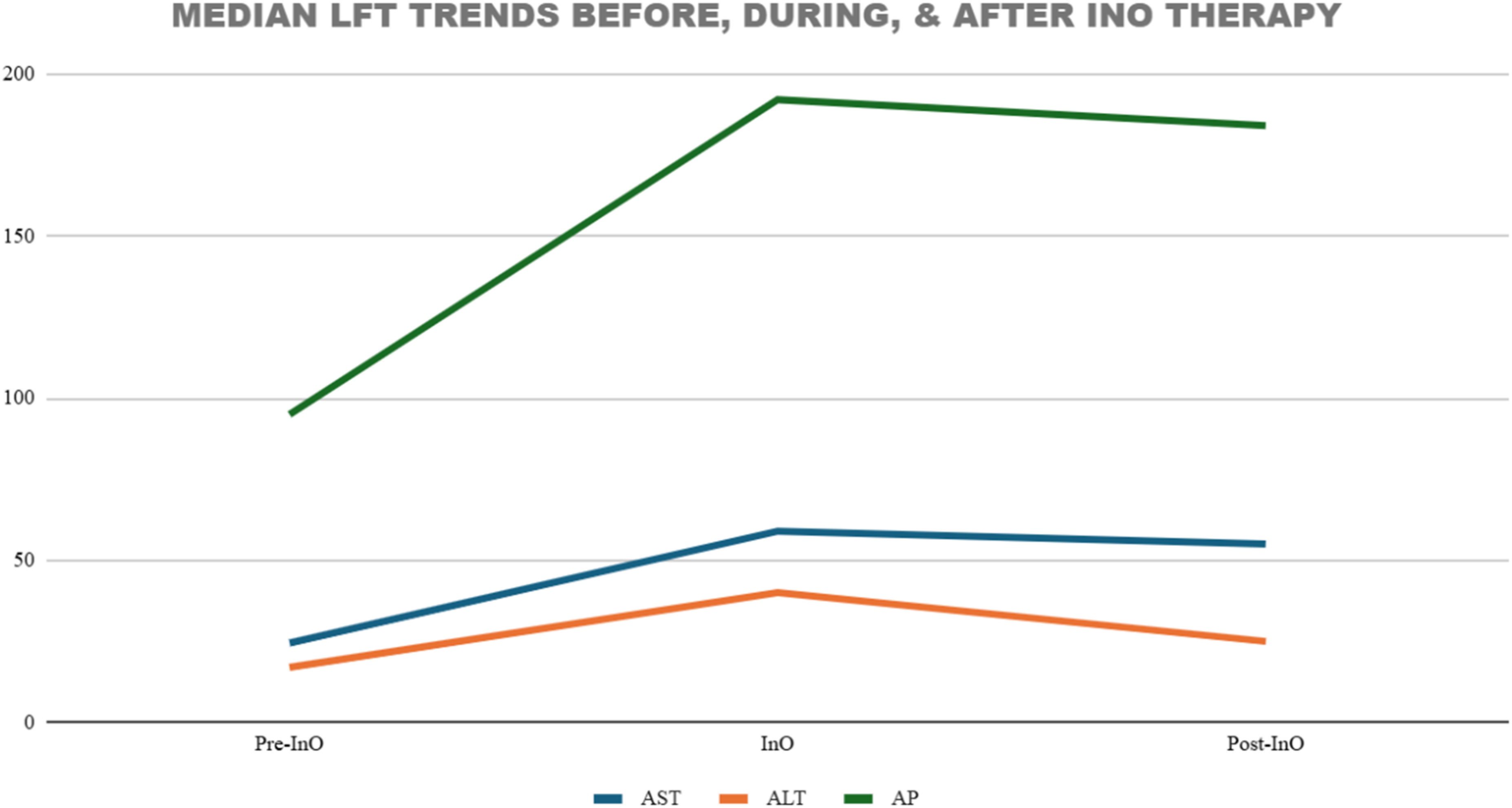
Graph of median LFT trends. AST: Aspartate aminotransferase; ALP: Alkaline phosphatase; ALT: Alanine aminotransferase; LFT: Liver function test; InO: Inotuzumab.
LFT trends during InO therapy.
AST: Aspartate aminotransferase; ALP: Alkaline phosphatase; ALT: Alanine aminotransferase; LFT: Liver function test; InO: Inotuzumab; Tbili: Total bilirubin.
Biopsy-confirmed hepatic sinusoidal congestion and “calicheamicin syndrome”
Six of 21 patients (28.6%) underwent liver biopsy due to persistent LFT elevation and clinical suspicion for deeper hepatotoxicity. All six biopsies demonstrated HSC without evidence of venular occlusion or classic SOS features as seen in Figure 2A-F. These biopsy-confirmed cases constitute the histopathologic core of the Calicheamicin Syndrome phenotype.
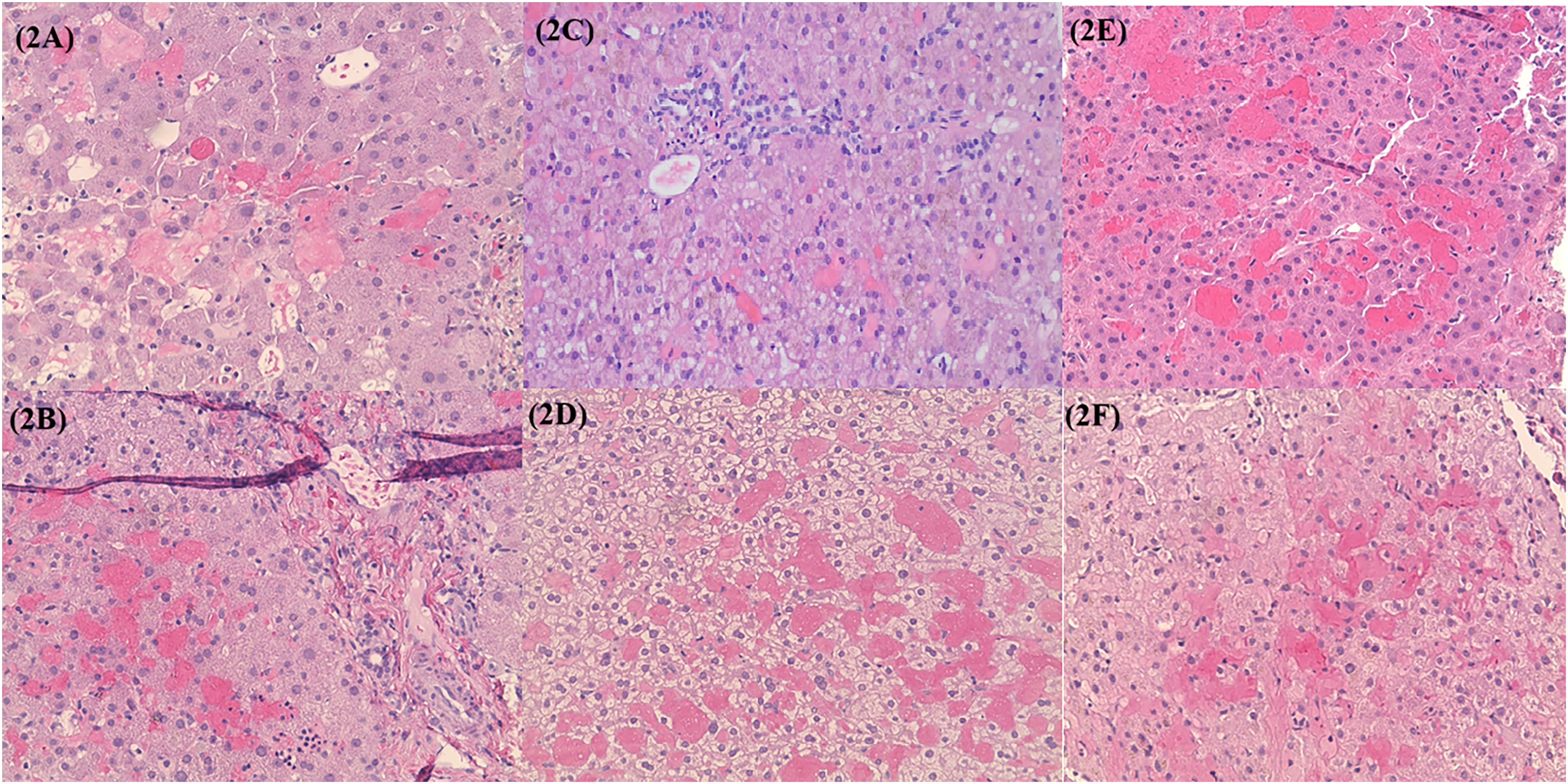
A–F. HSC of liver biopsies obtained from six patients. A. Liver biopsy demonstrating benign hepatic parenchyma showing sinusoidal congestion, mild cholestasis, and mild macrosteatosis. B. Liver biopsy demonstrating hepatic tissue with patchy mild sinusoidal congestion and dilation, mild portal and focal lobular inflammation, mild ductular reaction; patchy extramedullary hematopoiesis; focal minimal perisinusoidal fibrosis. C. Liver biopsies demonstrating mild macrovesicular steatosis, mild portal inflammation, sinusoidal dilation and congestion, and mild fibrous expansion of portal tracts. D. Liver biopsy demonstrating extensive sinusoidal congestion. E. Liver biopsy demonstrating benign liver parenchyma with marked sinusoidal congestion and focal minimal steatosis. F. Liver biopsy demonstrating moderate reactive hepatocyte changes including cellular swelling, occasional binucleation, mild nuclear atypia, mild lobular disarray; mild increase in portal fibrous tissues; moderate increase in stainable iron predominantly within hepatocytes and macrophages.
The median age in this subset was 31.5 years. Hepatotoxicity manifested at a median of 15 days after the final InO dose. LFT abnormalities were typically mild to moderate with AST, ALT, and ALP rising 2–4 times above ULN, and Tbili peaked between 0.6–2.7 mg/dl. The median time to LFT and overall hepatotoxicity improvement following drug discontinuation was approximately 17 days.
Thrombocytopenia was a consistent feature, occurring concurrently with LFT elevations. Platelet nadirs ranged at a median of 88 × 109/l and recovered in parallel with LFT normalization. Four of 6 biopsy-confirmed patients required hospitalization for monitoring or supportive management, with a median length of stay of 5.5 days. No patients progressed to clinical SOS, and all patients recovered with supportive care, shying away from hepatotoxic drugs, and using ursodiol 300 mg twice a day.
Radiographic findings on either abdominal ultrasound or CT abdomen/pelvis with contrast commonly included hepatomegaly, heterogeneous parenchymal enhancement, or mild ascites. Histopathologic examination in patients who underwent liver biopsy consistently revealed dilated and congested hepatic sinusoids with variable centrilobular involvement and mild perisinusoidal fibrosis or cholestasis. There was an absence of venular occlusion, fibrin deposition, or fibrotic obliteration, features that are typical of SOS.
Comparative findings between biopsy and non-biopsy patients
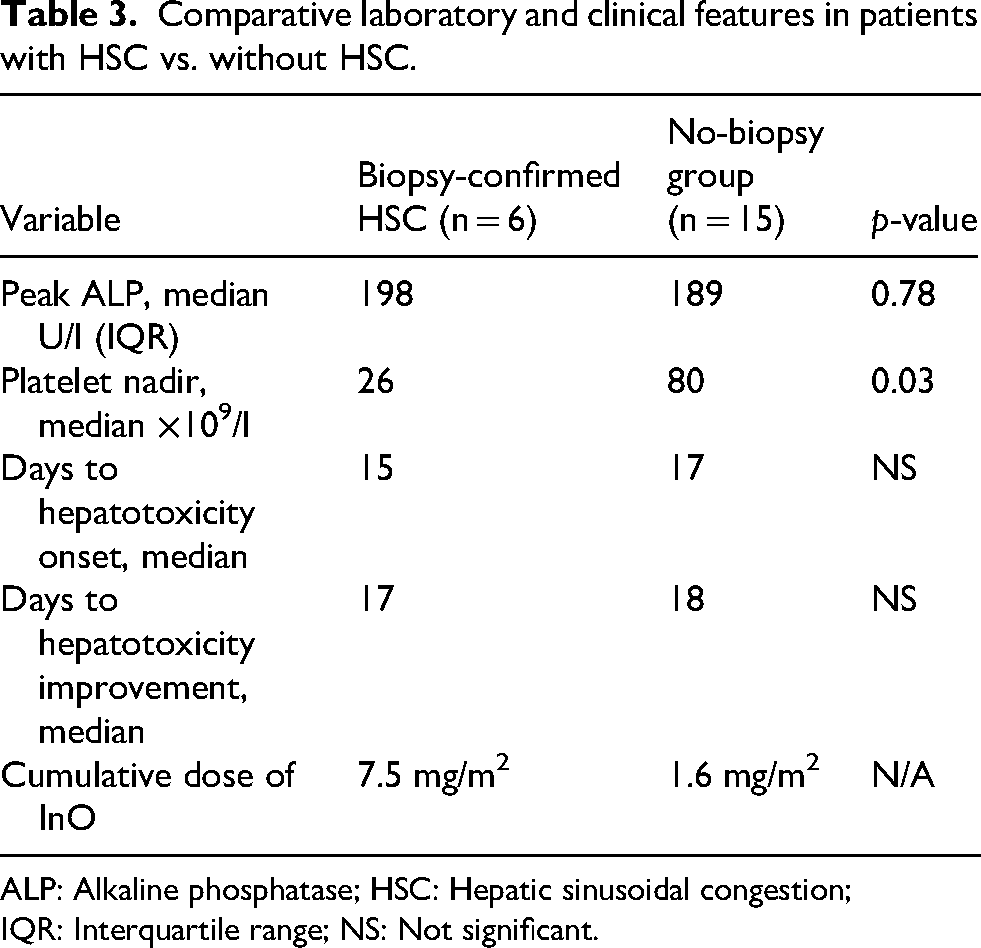
Table 3 describes laboratory and clinical features between patients with HSC and without HSC. Patients with biopsy-confirmed HSC experienced significantly deeper thrombocytopenia compared to the non-biopsy cohort with a median platelet nadir of 26 × 109/l vs. 80 × 109/l (p = 0.03). In contrast, peak ALP elevations were similar between groups with a median of 198 U/l vs. 189 U/l (p = 0.78). The timing of hepatotoxicity onset (median 15 vs. 17 days after the final InO dose) and time to LFT improvement (median 17 vs. 18 days) were also comparable.
Comparative laboratory and clinical features in patients with HSC vs. without HSC.
ALP: Alkaline phosphatase; HSC: Hepatic sinusoidal congestion; IQR: Interquartile range; NS: Not significant.
Comparative findings and clinical course
When comparing the six biopsy-confirmed patients with the remainder of the cohort, those with HSC received more cumulative InO exposure at a median of two cycles vs. one cycle and demonstrated more pronounced ALP elevation and deeper platelet nadirs. However, baseline demographics and comorbidities were otherwise similar between groups. None of the 15 non-biopsied patients developed clinical or radiologic SOS and their LFT elevations were transient, mild, and resolved without sequelae.
Across the entire cohort, discontinuation of InO and supportive management were universally associated with gradual recovery. No patients required defibrotide or liver-directed intervention, and no treatment-related deaths occurred.
Summary of key observations
These findings describe a self-limited pattern of hepatic injury temporally associated with InO administration and characterized by LFT elevation, thrombocytopenia, abnormal liver imaging, and the absence of venular occlusion or clinical SOS. These observations support the recognition of a distinct hepatotoxic entity, Calicheamicin Syndrome, reflecting endothelial injury from the calicheamicin payload that manifests as non-obstructive sinusoidal dysfunction rather than classical SOS.
Discussion
This study expands upon our previously published case series and provides the largest retrospective analysis to date examining hepatotoxicity associated InO outside the context of clinical SOS. 19 Through the review of 21 patients with R/R B-ALL treated at our institution, we identified a significant percentage of patients (28.6%) who received InO developed reproducible and reversible pattern of hepatotoxicity characterized by mild to moderate transaminitis, thrombocytopenia, abnormal liver imaging, and HSC in the absence of venular occlusion or clinical SOS. This collection of clinicopathologic data supports recognition of a distinct entity, Calicheamicin Syndrome, arising from the unique endothelial and microvascular effects of calicheamicin, the cytotoxic payload shared by InO and also gemtuzumab ozogamicin (GO) although the former was not studied in this analysis.4,5,18
Expanding the spectrum of InO-associated hepatotoxicity
Hepatotoxicity due to InO was initially characterized in the INO-VATE trial, which documented an increased incidence of SOS, particularly among patients proceeding to HSCT.6–8 However, the hepatic injury observed in our cohort represents a milder, non-obstructive form of sinusoidal injury distinguished by a lack of central venular occlusion and a milder course. Median onset of hepatotoxicity occurred approximately 16 days after the final InO dose, with improvement a median of 18 days following drug discontinuation and supportive management.
Histopathologic findings in all six biopsy-confirmed patients demonstrated sinusoidal dilation and congestion without the fibrotic venular obliteration typical of SOS. Importantly, none of these patients met EBMT 2016 criteria for SOS, emphasizing that this represents a parallel but mechanistically distinct process. 14 The differences in platelet nadirs between HSC and non-HSC patients in our study suggest that platelet sequestration and sinusoidal microvascular injury, rather than the magnitude of cholestatic enzyme elevation, distinguishes Calicheamicin Syndrome from transient, mild hepatotoxicity. Endothelial injury associated with InO can exist along a range where reversible sinusoidal congestion may precede overt SOS in susceptible individuals.16,17,20–23
Pathophysiologic considerations
Calicheamicin, a potent DNA-cleaving enediyne antibiotic, is responsible for the cytotoxic activity of InO and GO. It induces double-stranded DNA breaks after intracellular activation, leading to apoptosis of targeted CD22-positive B-cells.5–8 However, due to the liver's central role in ADC metabolism and clearance, off-target exposure to the calicheamicin payload in hepatic sinusoidal endothelial cells is plausible.4,18 Preclinical studies using nonbinding antibody–calicheamicin conjugates in cynomolgus monkeys demonstrated midzonal degeneration, endothelial loss, and sinusoidal microvascular injury even in the absence of antigen binding, implicating the calicheamicin component itself as the driver of hepatotoxicity. 4
We propose that the sinusoidal congestion observed in our cohort results from endothelial injury leading to sinusoidal dilatation, erythrocyte pooling, and platelet sequestration as seen in Figure 3. The temporal relationship between thrombocytopenia and LFT elevations supports this mechanism. Rather than marrow suppression, the thrombocytopenia likely reflects microvascular platelet trapping and destruction secondary to endothelial dysfunction and impaired hepatic thrombopoietin production.11,23 The reversibility of both LFTs and thrombocytopenia upon InO cessation further reinforces a transient, functional injury rather than permanent structural damage.
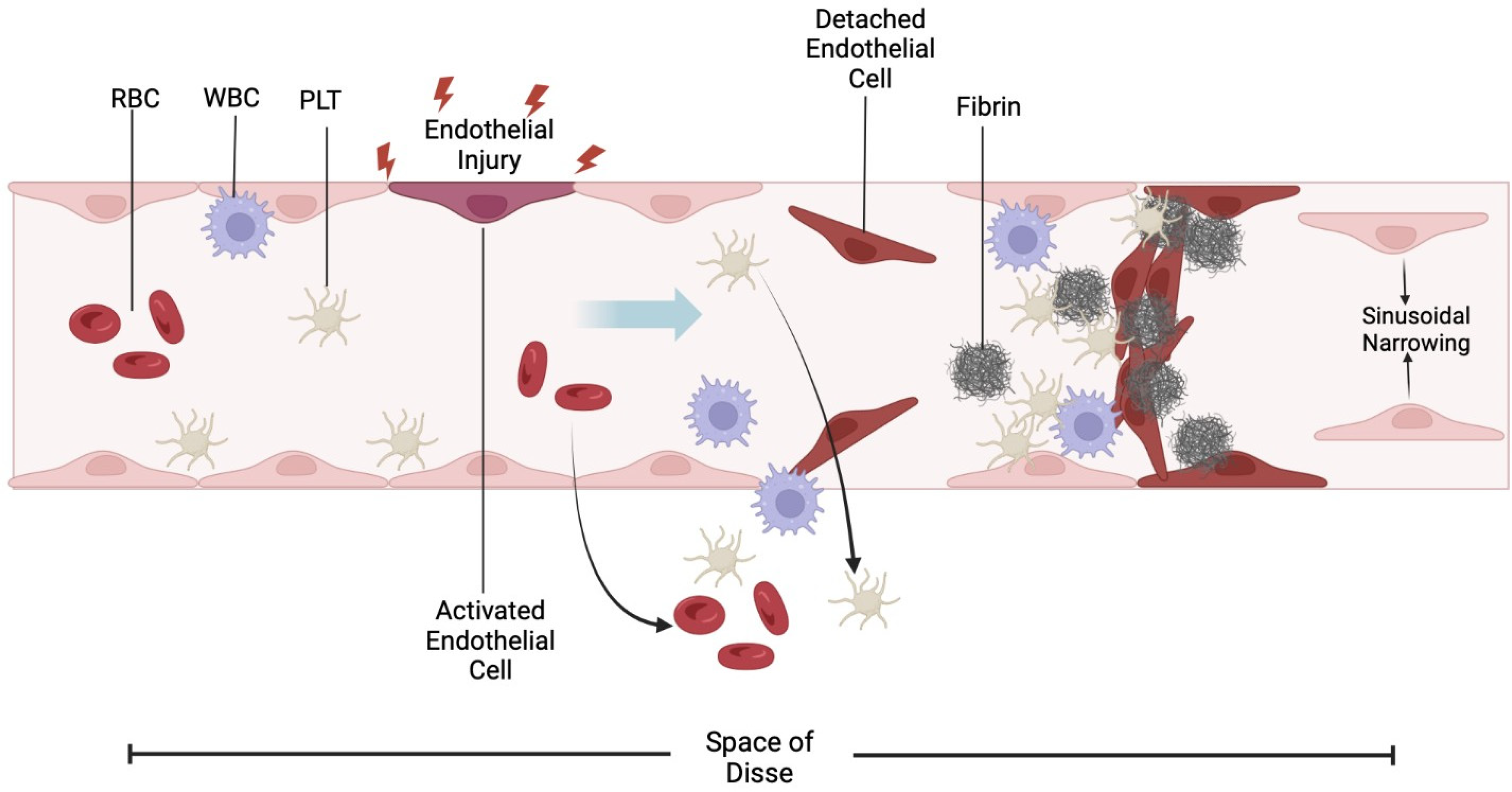
Diagram illustrating proposed hepatotoxicity via endothelial injury causing sinusoidal dilatation, erythrocyte pooling, and platelet sequestration leading to HSC. PLT: Platelet; RBC: Red Blood Cell; WBC: White Blood Cell.
Clinical implications
Awareness of Calicheamicin Syndrome is critical for clinicians managing patients on InO, especially for patients who may later undergo Allo HSCT. This study demonstrates that hepatotoxicity can occur even in Allo HSCT-naïve patients, underscoring that cumulative calicheamicin exposure, not prior transplantation, may be a key risk factor.9,12,13,15,16 Serial and close monitoring of AST, ALT, ALP, Tbili, and platelet counts is recommended during and after each InO cycle. A concurrent decline in platelet count and rise in LFTs should prompt early imaging and temporary InO cessation.
If abnormalities persist or clinical symptoms develop, liver biopsy can confirm the presence of HSC and support the diagnosis of Calicheamicin Syndrome.19,22,23 However, while liver biopsies can provide histopathologic confirmation of HSC, it is an invasive procedure with increased bleeding risk in thrombocytopenic patients, which is highly represented in this cohort. The clinical and laboratory findings along with recent InO exposure may be sufficient to establish a clinical diagnosis of Calicheamicin Syndrome. Liver biopsy results would not affect management and should be reserved in select cases where diagnosis remains uncertain or if there is concern for evolving SOS.
Supportive care through drug discontinuation, avoidance of concurrent hepatotoxic agents, and administering ursodiol remain the cornerstone of treatment for this patients’ cohort. No patients required defibrotide and all recovered with conservative measures. The recognition of Calicheamicin Syndrome provides an opportunity for early intervention before progression to SOS, preserving hepatic reserve and transplant eligibility.
Relationship to other calicheamicin-based ADCs
The phenomenon described here may potentially extend beyond InO. GO, a CD33-directed ADC conjugated to calicheamicin, has also been associated with hepatic injury, SOS, and fatal hepatotoxicity.3,5,17,18 Histopathologic findings from GO-related cases parallel those seen with InO, including sinusoidal dilation, congestion, and endothelial disruption. The shared payload and consistent histologic pattern suggest a class effect of calicheamicin-based ADCs. Recognition of this common mechanism may inform future ADC design such as linker modifications or alternative payloads to reduce hepatotoxicity risk while maintaining antitumor potency.
Limitations
This study is limited by its retrospective design, single-center nature, and relatively small sample size. Although biopsy confirmation strengthens diagnostic accuracy, histologic evaluation was available for only six patients, and not all patients underwent standardized imaging or serial laboratory assessments. Additionally, pursuing a liver biopsy in the setting of thrombocytopenia may pose a serious risk to patients suffering from Calicheamicin Syndrome, leading clinicians to rely on clinical judgment to detect this syndrome. The absence of prospective data and control cohorts precludes definitive causal inference. Nonetheless, the reproducibility of the clinical and histologic pattern across independent cases, combined with consistent temporal association to InO exposure, lends weight to the validity of this emerging entity.
Future directions
Further research is warranted to delineate the molecular and clinical determinants of Calicheamicin Syndrome. Prospective studies integrating biomarkers of endothelial injury could clarify the overlap between HSC and SOS.11,14,20 Given the higher rates of Calicheamicin Syndrome in patients who received higher median doses of InO, experimental models using hepatic sinusoidal endothelial cells exposed to calicheamicin conjugates may help define dose thresholds for endothelial toxicity.4,18 Additionally, pharmacogenomic or demographic factors such as sex, ethnicity, or body composition should be explored, as all six biopsy-confirmed cases in our cohort occurred in female and predominantly Hispanic patients. Identifying risk profiles could improve patient selection and monitoring strategies in future ADC trials.
Conclusion
This study identifies Calicheamicin Syndrome as a distinct and reversible hepatotoxicity associated with its quadriad components: History of InO use, elevated LFTs, thrombocytopenia, and abnormal liver imaging. The reproducible clinicopathologic pattern across our cohort supports a calicheamicin-mediated endothelial injury mechanism. Recognition of this entity broadens the understanding of ADC–related liver injury and underscores the importance of early detection and supportive management. As calicheamicin-based ADCs continue to expand in oncology, future prospective and mechanistic studies are warranted to validate this syndrome, refine monitoring strategies, and inform safer ADC design.
Practicing oncologists, hepatologists, pathologists, and pharmacists should be aware of InO toxicities and establish strategies for hepatic function monitoring, prevention, and management to provide quality care to patients undergoing treatment with InO. As the use of this therapeutic modality continues, further studies should explore demographic, clinicopathologic factors, and molecular pathology that may predict the development of Calicheamicin Syndrome.
Footnotes
Acknowledgements
The authors thank the patients whose clinical data contributed to this study and the clinical and pathology teams involved in their care.
Author contributions
Conceptualization – DT, MA
Methodology – DT, MA, DP
Software – DT, DP
Validation – PS, KL, MA
Formal Analysis – DT
Investigation – DT, KL, JC, NK, KB
Writing, Original Draft – DT
Writing, Review and Editing – DT, KL, JC, NK, PS, KB, MA
Visualization – DT, JC, PS, DP
Supervision - MA
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data sharing statement
The data supporting the findings of this study are available from the corresponding author upon reasonable request, subject to institutional review board approval and patient confidentiality requirements.
