Abstract
Objective
Myositis, an inflammatory disease affecting muscles, is a rare and potentially fatal immune-related adverse event associated with immune checkpoint inhibitors. There are limited data on its clinical features and management.
Case presentation
Atezolizumab, in combination with etoposide and carboplatin, was initiated in the patient diagnosed with metastatic small-cell lung cancer. After four cycles, maintenance atezolizumab was initiated. At the third visit of the maintenance therapy, the patient reported weakness, edema, and tightness in the muscles that had progressed over the course of a week. Mild solid-food dysphagia was also observed. Neutrophilic leukocytosis with elevated creatine phosphokinase (9234 U/L), erythrocyte sedimentation rate (111 mm/h), and transaminase levels were observed. A diagnosis of myositis was considered based on clinical findings. Atezolizumab was omitted and an oral 0.5 mg/kg/day dose of methylprednisolone was administered. The myositis resolved within 10 days. During the treatment of myositis, the patient underwent prophylactic cranial irradiation. The steroid dose was tapered off within 35 days and then atezolizumab was restarted.
Conclusion
The literature contains only a few case reports about atezolizumab-induced myositis, highlighting the challenges in defining its clinical features and management. Prompt diagnosis and treatment are crucial to prevent severe complications, such as myocarditis or respiratory muscle paralysis.
Introduction
Atezolizumab is an Fc-engineered humanized immunoglobulin G1 monoclonal antibody that enhances the immune response against cancer cells by binding and inhibiting programmed death-ligand 1. It has been approved by the Food and Drug Administration for the treatment of lung cancer, breast cancer, urothelial carcinoma, hepatocellular carcinoma, and melanoma. 1 Immune checkpoint inhibitors (ICIs) like atezolizumab have revolutionized oncology by facilitating cytotoxic T cells to target tumors, thereby providing crucial therapeutic options for various malignancies.1,2 Generally, ICIs have fewer side effects than cytotoxic chemotherapies. 3 Nevertheless, the broad immunostimulatory effects of ICIs can potentially induce immune-related adverse events (irAEs). 4
The irAEs most commonly occur in the skin, and gastrointestinal and endocrine systems. However, uncommon irAEs such as myositis have also been reported. Myositis is an inflammatory disorder that affects muscles, and leads to significant morbidity due to muscle weakness, pain, and damage. Although the incidence of ICI-associated myositis is < 1%, it can be potentially fatal, thus requiring high clinical alertness for prompt diagnosis and effective treatment.5,6
Some cases of atezolizumab-induced myositis have been reported.4,7–11 Thus, limited data are available regarding its properties and treatment. In this context, we present a case of an extensive-stage small-cell lung cancer patient developing myositis after atezolizumab administration and review the data in literature related to this topic. The aim of this report was to contribute to the understanding of clinicians, increase awareness, and help improve future clinical care.
Case presentation
In February 2021, a 61-year-old Turkish male patient with a 40-pack-year history of smoking and coronary artery disease was referred to our hospital (in Kocaeli/Turkey) because of a 5 cm mass in the middle lobe of the lung and bilateral multiple mediastinal lymph nodes on chest computed tomography. Adrenal metastases were also detected using positron emission tomography. The only drug he used was clopidogrel 75 mg/day. Small-cell lung cancer was diagnosed with a tru-cut biopsy from the lung, and etoposide (100 mg/m2), carboplatin (AUC-5), and atezolizumab (1200 mg) treatment for every 3 weeks was initiated intravenously. While thyroid function tests were within the normal range before starting the treatment, TSH of 18 (normal range: 0.27–4.2) mIU/L, free T4 of 0.3 (normal range: 0.85–1.6) ng/dL, and free T3 of 2.42 (normal range: 2.0–4.4) pg/mL were detected in the second cycle visit. Thyroid peroxidase antibodies (TPO Ab) and thyroglobulin antibodies (Tg Ab) were negative. Treatment with levothyroxine 100 µg/day treatment was started, and thyroid function measurements returned to normal within 2 months. After four cycles of chemotherapy and atezolizumab, a complete radiologic response was observed in the mediastinal lymph nodes and adrenal metastases, and partial regression was observed in the primary mass. Treatment was continued with maintenance atezolizumab (1200 mg every 21 days).
When the patient came for the third visit of the maintenance atezolizumab, he had stated that he had weakness and edema. In addition, a feeling of muscle tightness in the extremities was present. Muscle weakness and edema started in the lower extremities, worsened each day, and spread to the entire body within the last week. The patient also had mild solid-food dysphagia for the last 2 days. No ocular, respiratory, or cardiac symptoms were observed. There was no rash, Raynaud's phenomenon, or fever. The physical examination was unremarkable, except for edema in the extremities. The erythrocyte sedimentation rate (111 mm/h, normal value < 16 mm/h), serum creatinine (1.7 mg/dL, basal level 1.1 mg/dL, normal range: 0.7–1.18), and creatine phosphokinase (CPK) (9234 U/L, 46 × ULN) levels were elevated. The aspartate-aminotransferase (AST) was 215 U/L (6 × ULN), and the alanine-aminotransferase (ALT) level was 110 U/L (3 × ULN). Neutrophilic leukocytosis was seen in the total blood count (white blood cells count: 22.13 × 103/µL, neutrophils counts: 20.99 × 103/µL). The C-reactive protein was 0.9 mg/dL (normal value < 1 mg/dL). Serum electrolytes, albumin, troponine, brain natriuretic peptide, cortisol levels, and thyroid function test results were within the normal range. Serologic markers for hepatitis B and C, rheumatoid factor, anti-nuclear antibody (ANA), anti-smooth muscle antibody (ASMA), liver kidney microsome type 1 (anti-LKM-1) antibody, cyclic citrullinated peptide, proteinase-3, and myeloperoxidase antibodies were negative. No hematuria, myoglobinuria, or proteinuria was observed. Abdominal ultrasound findings were normal. There was no evidence of rhabdomyolysis or myocarditis on imaging or laboratory tests.
There was grade 2 suspected myositis and grade 4 CPK elevation. The patient refused muscle biopsy or electromyography and refused hospitalization. He was referred to nephrologists, rheumatologists, and a nutritionist. The patient's diet was arranged by the nutritionist for dysphagia. Rheumatologists recommended discontinuing atezolizumab and starting corticosteroid therapy with the diagnosis of myositis. In accordance with the recommendation of the American Society of Clinical Oncology (ASCO), 12 a 0.5 mg/kg/day (40 mg) oral dose of methylprednisolone was initiated. In Turkey, oral forms of prednisolone are available as 1 or 4 mg and oral forms of methylprednisolone are available as 4, 8, or 16 mg. Therefore, methylprednisolone was preferred for the ease of use of the patient. During the methylprednisolone administration, pantoprazole 40 mg/day for gastric prophylaxis, calcium carbonate 1000 mg/day, and vitamin D 880 IU/day for bone prophylaxis were also given orally. The patient was advised to drink plenty of fluids. After 10 days, the patient's CPK (1441 U/L), and creatinine (1.2 mg/dL) levels regressed. Besides, all the patient's symptoms improved. Therefore, the methylprednisolone dose was reduced to 32 mg, and continued with weekly tapering. The dose reduction scheme and the change in laboratory values are summarized in Table 1. During the methylprednisolone therapy, prophylactic cranial irradiation (PCI) was administered to the patient. One month after initiation of methylprednisolone; dysphagia, weakness, and tightness in the muscles completely disappeared. Atezolizumab was restarted after PCI was completed, and methylprednisolone was discontinued. According to these data; the Naranjo algorithm score 13 for myositis was 5 suggesting a probable association between nivolumab and myositis (see Table 2). The patient received the 12th cycle of atezolizumab treatment in June 2022. Since he moved to another city after this date, his follow-ups in our hospital were terminated.
Changes in laboratory values and steroid dose after the diagnosis of myositis (6 September 2021).
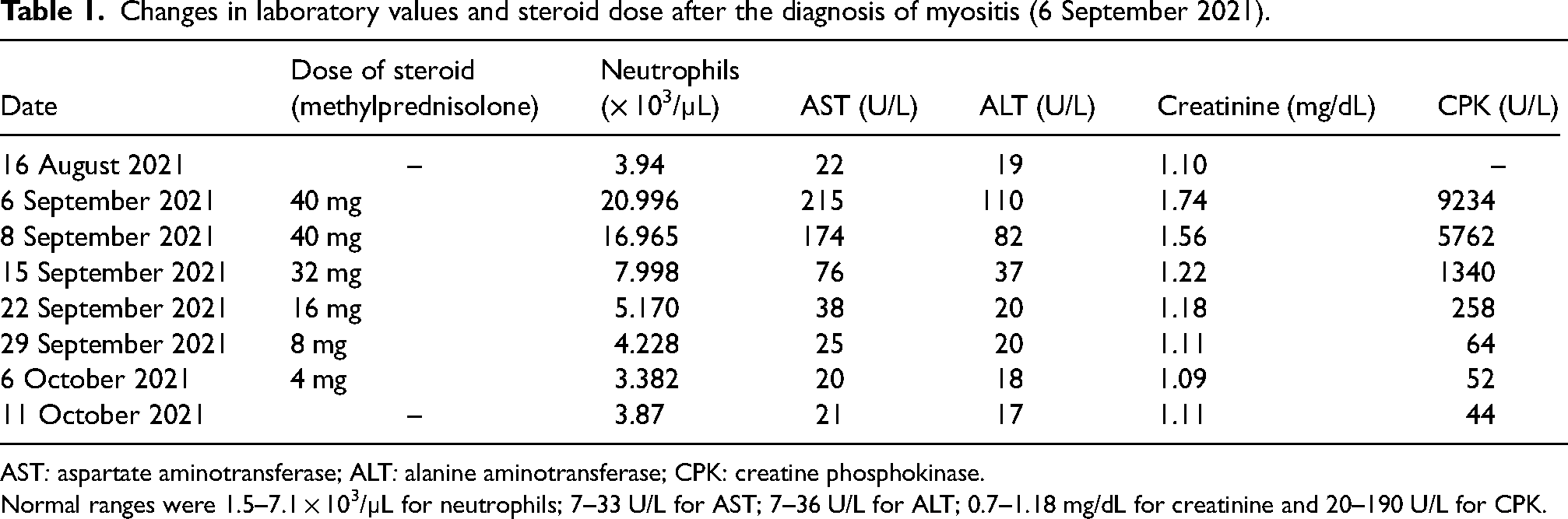
AST
Normal ranges were 1.5–7.1 × 103/µL for neutrophils; 7–33 U/L for AST; 7–36 U/L for ALT; 0.7–1.18 mg/dL for creatinine and 20–190 U/L for CPK.
Naranjo algorithm a for adverse drug reaction assessment.
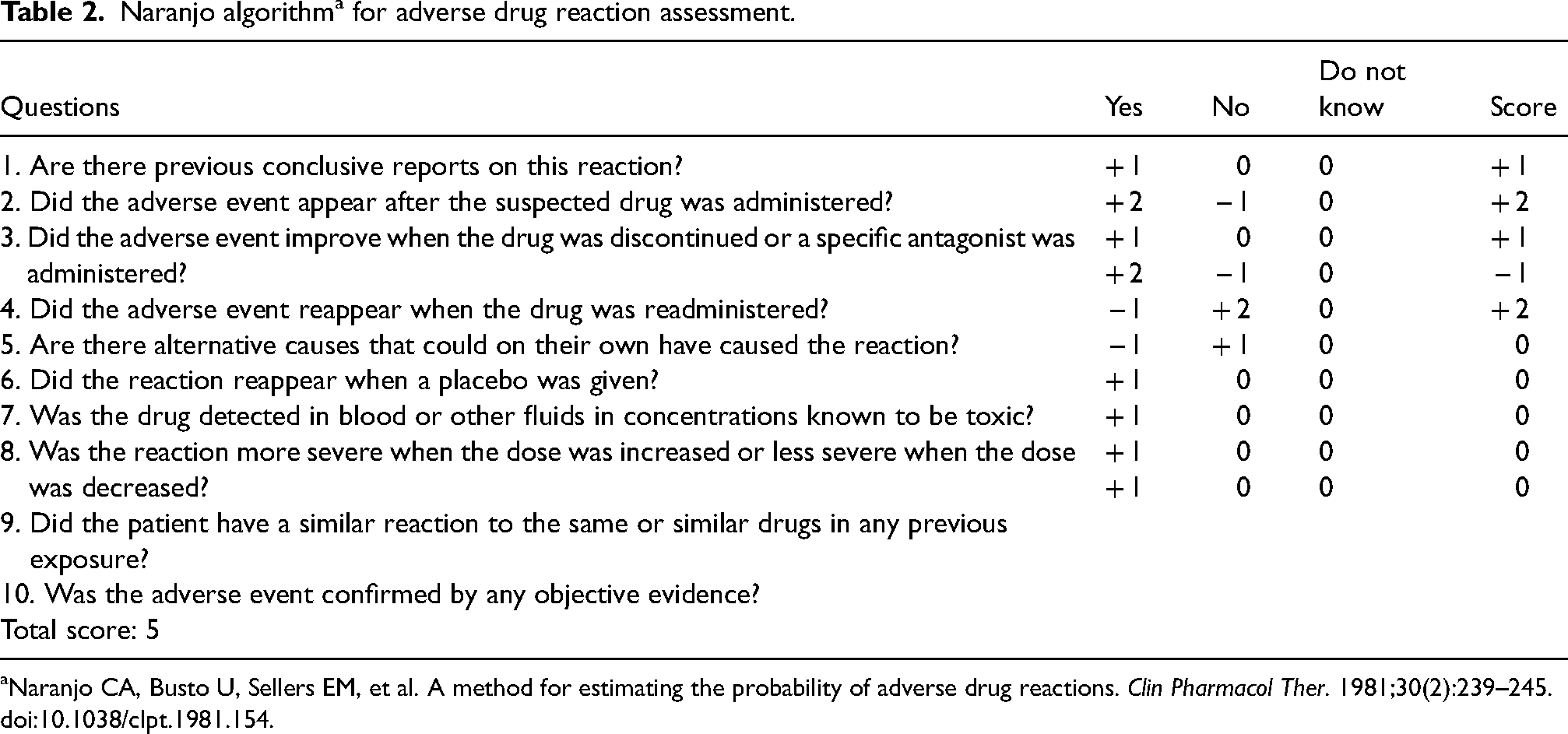
Naranjo CA, Busto U, Sellers EM, et al. A method for estimating the probability of adverse drug reactions.
Discussion
Immune checkpoint inhibitors have emerged as highly promising therapeutic strategies for the treatment of different types of cancers. This innovative approach leverages the power and effectiveness of the body's immune system to specifically target and eliminate cancer cells. 1 However, activation of the immune system can also lead to some irAEs. ICI-associated myositis is a rare irAE, with an overall incidence of 0.38%. 5 The case of atezolizumab-associated myositis is an even less frequent phenomenon. Its rarity creates difficulties in defining clinical features, diagnosis, and treatment. 4 In the current case, myositis was suspected in a patient with complaints of muscle weakness and mild dysphagia. High serum CPK levels indicate muscle damage. Muscle biopsy and electromyography could not be performed due to patient rejection. However, there was rapid improvement with an oral dose of 0.5 mg/kg/day methylprednisolone treatment, which strongly supported the diagnosis of myositis.
There have been a few case reports of atezolizumab-related myositis. One case involved a 74-year-old patient with metastatic urothelial carcinoma who developed myositis and myocarditis after receiving atezolizumab, leading to confusion with ischemic heart disease. 7 This patient subsequently developed severe hypercapnic respiratory failure and died. The other patient was a 67-year-old man with hepatocellular carcinoma and cirrhosis receiving atezolizumab + bevacizumab. 10 In the other two cases, atezolizumab caused overlapping of myositis and myasthenia gravis.4,8 One of these patients died because he did not benefit from treatment of irAE. Finally, the fifth case was an 80-year-old patient with metastatic urothelial carcinoma, who developed polymyositis after atezolizumab. 9 In addition, in a recent Korean study in which ICI-related severe neuromuscular myositis was recruited, four atezolizumab-induced Guillian-Barre syndrome variants were presented, but none of them were accompanied by myositis. All five pembrolizumab-related myocyte cases reported in this study had thymoma, concurrent myasthenia gravis, and hepatitis. One patient died because of organ failure, the others were treated with corticosteroid and IVIG (± plasmapheresis). 12 In all reported atezolizumab-related myositis cases, proximal muscle weakness was the predominant symptom, with varying degrees of myalgia and elevated CPK levels. However, it was remarkable that ptosis and dysphonia/dysarthria and/or dysphagia were reported in all cases due to weakness in the facial, throat, tongue, or esophageal muscles. The onset of myositis varied from several weeks to several months after initiating the atezolizumab therapy. In all cases, medium or high-dose steroids were used. In addition, plasma exchange or intravenous immunoglobulin was administered in some cases. Despite treatment, two patients died due to severe myositis. These cases underscore the need for high suspicion of myositis in patients who show proximal muscle weakness, ptosis, dysphagia, dysarthria, or other musculoskeletal symptoms while undergoing atezolizumab.
Management of irAEs is based on the assessment of severity using the Common Terminology Criteria for Adverse Events grading system. 14 Accordingly, weakness accompanied by mild pain is associated with grade 1 myositis, moderate pain and weakness limiting instrumental activities of daily living with grade 2 myositis, severe weakness and pain limiting self-care activities of daily living with grade 3 myositis, and life-threatening consequences with grade 4 myositis. According to the recommendation of ASCO, corticosteroids (prednisolone or methylprednisolone) are the primary treatment for mild to moderate irAEs (such as grades 1 and 2), with varying dosages of low (< 0.5 mg/kg/day), moderate (0.5–1 mg/kg/day), or high (1–2 mg/kg/day) depending on the severity. 12 Once irAEs resolve, a gradual weekly tapering of corticosteroid therapy is usually recommended. If irAEs are severe (grades 3 and 4) or do not improve with corticosteroids, other immunosuppressants such as mycophenolate mofetil, tacrolimus, tocilizumab, intravenously immunoglobulin, or plasma exchange may be administered additionally. 15 In general, for grade 1 events, ICI therapy can often be continued while managing the irAEs. However, for grade 2–4 events, immunotherapy is typically paused and may be resumed once irAEs have resolved, although permanent discontinuation may be required in some cases.15,16 In the current case, there was grade 2 myositis and grade 4 CPK elevation. ICI was omitted, oral moderate-dose (0.5 mg/kg/day) of methylprednisolone was started immediately and a relatively rapid response to methylprednisolone was obtained. After discontinuation of methylprednisolone treatment with tapering within 35 days, ICI was resumed.
According to the literature, the mean time of onset of myositis after first ICI administration was 5.6 ± 6.1 weeks and it was most reported in those patients receiving pembrolizumab and nivolumab. 5 In our case, myositis was post-atezolizumab and had a late onset (∼ 21 weeks after the start of treatment). However, some cases in the literature have developed myositis, even 34 weeks after the onset of ICI. 5
In the present case, myositis was preceded by thyroiditis. Similar cases of co-occurrence of ICI-related thyroiditis and myositis have been reported.4,5 This phenomenon did not lead to a change in the treatment plan. However, it has been reported that corticosteroid therapy may be inadequate in cases of myositis with myocarditis, respiratory muscle paralysis, myasthenic crisis, or rhabdomyolysis.4,5,10,12,17,18 In a meta-analysis about ICI-associated myositis, myocarditis was reported in 40.9% of cases.
5
Comparing the group of individuals who developed myocarditis to those who did not, several significant distinctions were observed in terms of clinical characteristics. The occurrence of myocarditis was significantly affected by the ICI regimen (
Notably, our patient underwent PCI during myositis treatment. The decision to proceed with PCI in the setting of active irAE requires careful consideration and multidisciplinary collaboration. In this case, ICI was interrupted because of the toxicity, and PCI was administered in that period.
Conclusion
The rarity of atezolizumab-induced myositis poses challenges for its diagnosis and management. To date, there is limited evidence on the specific management strategies for this irAE, and guidelines primarily rely on management approaches for myositis induced by other ICIs. In our case, moderate-dose oral methylprednisolone was effective in treating myositis. PCI was administered to the patient while steroid treatment was ongoing, and atezolizumab was resumed after discontinuing the steroids. This case is presented to remind medical oncologists to be aware of the side effects of ICIs and to demonstrate the importance of a multidisciplinary approach by sharing our experience with the treatment of myositis, a rare but potentially fatal side effect.
Footnotes
Authors’ contributions
Conception and design, acquisition of data, or analysis and interpretation of data: ES, drafting the article: ES, revising the article critically for important intellectual content: All authors, final approval of the version to be published: All authors: This article has been read and approved by all the authors. All authors met the authorship requirements stated previously. Each author declares that she believes the article represents honest work.
Consent to participate
Informed consent was obtained from the patient.
Availability of data and material
It can be obtained by asking the authors.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
