Abstract
Introduction
This study aimed to determine the stability of cetuximab: (1) under “in-use” conditions after dilution to 1 mg/mL in 0.9% sodium chloride in polyolefin bags and (2) as an undiluted solution (5 mg/mL) repackaged in polypropylene bags or kept in the vial after opening.
Methods
Ready-to-use 500 mg/100 mL vials of cetuximab solution were diluted to 1 mg/mL in 100 mL bags of 0.9% sodium chloride or repackaged as a 5 mg/mL solution into empty 100 mL bags. Bags and vials were stored at 4°C for 90 days and 25°C for 3 days. A syringe sample of 7 mL was taken from each bag for the initial determinations. The sampled bags were weighed to determine their initial weight and placed under the planned storage conditions. The physicochemical stability of cetuximab was estimated using validated methods.
Results
No changes in turbidity, no protein loss, and no changes in cetuximab tertiary structure were observed after 30 days of storage or when subjected to a temperature excursion of 3 days at 25°C and when stored at 4°C for up to 90 days, regardless of the concentrations and batches. The colligative parameters did not change under any of the tested conditions. No evidence of microbial growth was found in bags after 90 days of storage at 4°C.
Conclusion
These results support the extended in-use shelf-life of cetuximab vials and bags, which can be cost-effective for healthcare providers.
Introduction
Cetuximab is an epidermal growth factor receptor antagonist indicated for the treatment of squamous cell carcinoma of the head and neck and KRAS wild-type, epidermal growth factor receptor—expressing metastatic colorectal cancer. 1 It is available in 2 mg/mL (100 mg/50 mL and 200 mg/100 mL in the United States 1 ) and 5 mg/mL (100 mg/20 mL and 500 mg/100 mL in Europe 2 ) single-dose vials and administered as an intravenous (IV) infusion.1,2 Both weekly and biweekly IV infusion regimens are approved by the US Food and Drug Administration, 1 whereas the current approval by the European Commission is for the weekly dosage schedule only. 2
Currently, unopened cetuximab vials (2 and 5 mg/mL) are recommended to be stored in a refrigerator at 2°C to 8°C and, once in use, to be stored no longer than 24 h, unless opened in controlled and validated aseptic conditions.1,2 As the vials do not contain any antimicrobial preservative or bacteriostatic agent, from a microbiological point of view, the product should be used immediately after opening. 2 Extending in-use shelf-life of the prepared administration bag could allow for more flexibility of cetuximab administration and improve operations.
The primary objectives of this study were to determine the stability of cetuximab: (1) under “in-use” conditions after dilution to 1 mg/mL in 0.9% sodium chloride (NaCl) in polyolefin bags and (2) as an undiluted solution (5 mg/mL) repackaged in polypropylene bags or kept in the vial after opening.
Methods
Study design
The study was performed in the Pharmacy Department Control Laboratory of the Henri Mondor University Hospital Group from 26 May until 31 August 2020.
The study was conducted according to the consensus of the European Society of Oncology Pharmacy, Quality Guidelines of the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use and the European Pharmacopoeia (10th edition). 3
The equipment included: turbidimetry Cary 50 probe (Varian, Inc); 1 cm quartz microcuvettes (Hellma); ultraviolet (UV) Shimadzu UV-1800 UV/visible spectrophotometer (Shimadzu France); Perkin-Elmer LS45 fluorimeter; dynamic light scattering (DLS) system (Malvern Nano ZetaSizer); ion chromatography (IC) system comprising Thermo Scientific Ultimate 3000 high-performance liquid chromatography (HPLC), Sigma centrifuge 1–14 K, NPG-3400 RS pump, WPS-3000, TVRS injector, TCC-3000SD for four columns and VWD-3400 UV detector; size exclusion chromatography (SEC) system comprising Thermo Scientific Ultimate 3000 HPLC, ISO-3100 SD pump, WPS-3000 SL injector, TCC-3000 SD for four columns and PDA 3000 UV detector; Consort P901 pH meter (Bioblock Scientific); micro-osmometer; Fisher Scientific type 6 dissolver; Anton PAAR portable DMA 35 electronic densitometer; Eppendorf pipettes; and Sartorius analytical balance. The instruments were validated regularly in accordance with the relevant monographs (2.2.3, 2.2.5, 2.2.21, 2.2.29, 2.2.30, 2.2.35, 2.2.1, 2.5.33 and 2.6.1) of the European Pharmacopoeia (10th edition) 3 and used during their period of validity. The pipettes were validated by gravimetry. The controlled-temperature chambers were monitored continuously by the Sirius temperature-recording system (Jules Richard Instruments). During the test periods, the temperature remained within the accepted standards.
Preparation of bags and samples
Ready-to-use 500 mg/100 mL vials of cetuximab solution were diluted under validated sterile conditions (vertical laminar flow hood) to the concentration of 1 mg/mL in 100 mL bags of NaCl (n = 3) or repackaged as a 5 mg/mL solution into empty 100 mL bags (n = 3) or kept in the original vial (n = 3). Bags and vials were kept in a refrigerator (2°C; 8°C) at 4°C for 90 days and in a climatic chamber at 25°C (with 65% humidity) for 72 h (Figure 1). A syringe sample of 7 mL was taken from each bag for the initial determinations, referred to as D0 (reference). The sampled bags were weighed to determine their initial weight (needed for the calculation of possible water losses during storage) (Table 1) and placed under the planned storage conditions.
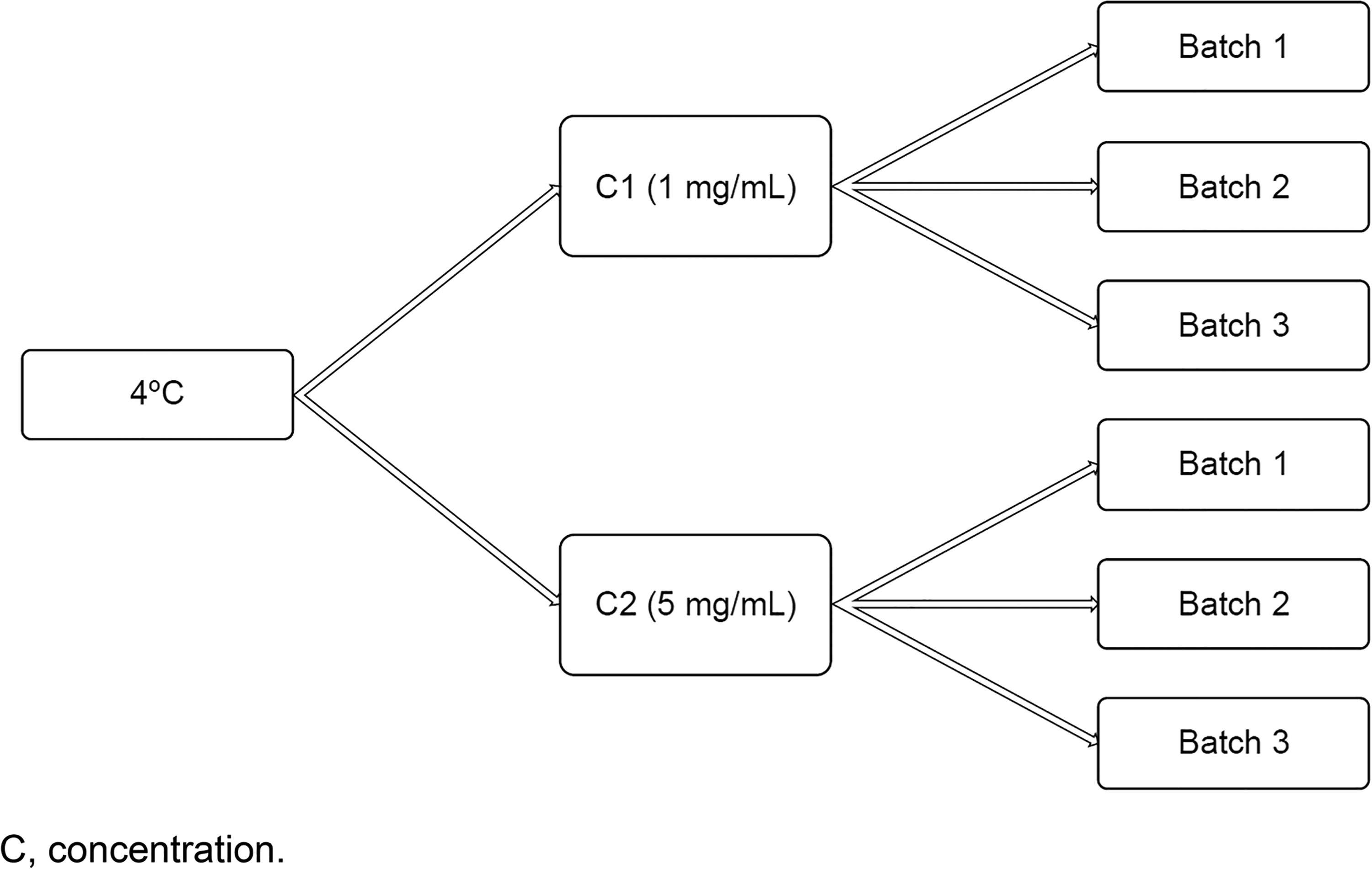
Organization of cetuximab samples.
Weight (g) of the prepared bags.

C: concentration; SD: standard deviation.
Assessments
The physicochemical stability of cetuximab was estimated using previously published methods that have been validated as stability indicators for antibodies4–13 and include: estimation of deamidation of asparagine residues and changes of charge by gradient IC; estimation of cleavage of disulfide bridges (fragmentation) by SEC; estimation of thermal stability curve (measurement of aggregation and determination of melting point) by DLS; turbidity assessments at 350 nm and aggregation index calculation (abs. ratio: λ350 /[ λ279 − λ350]); measurement of the optical density (OD) at 279 nm for calculation of the protein concentration; determination of tertiary structure integrity and tryptophan-type aromatic residues modification; derivative UV spectrophotometry and fluorescence spectroscopy; estimation of high molecular weight aggregates (dimers and low-order polymers) by SEC; estimation of submicron aggregates by DLS; sterility tests (bacteria, fungi, and yeasts) performed on samples at D90 in conformity with monograph 2.6.1 of the European Pharmacopoeia (10th edition) 3 ; and study of colligative parameters of density (U-tube method), pH, and osmolality.
The analyses were performed in triplicate unless otherwise instructed. The samples were collected under sterile conditions under a vertical laminar flow hood. The bags stored at 4°C were sampled at D0, D30, and D90. The bags and vials that had undergone a thermal excursion of 3 days at 25°C were sampled at D0, D3, and D30. All planned physicochemical analyses were conducted at each of these time points. Sterility test was performed only on bags held at 4°C and only at D90.
Spectrometric and turbidity measurements
For the measurement of turbidity, the analyses were performed on undiluted samples. The use of undiluted samples to measure turbidity is preferable, as it requires less manipulation and, therefore, minimizes the risk of contamination of the material or the solvent used. Samples diluted to 0.5 mg/mL in 0.9% NaCI were used for recording the scan between 200 and 600 nm, and samples diluted to 1 mg/mL were used for determining the protein concentration. The blank was measured in a cuvette containing 0.9% NaCI. The ODs at 279 and 350 nm were determined. The second derivative spectrum was analyzed between 250 and 320 nm. For fluorescence measurements, dilutions of 0.1 mg/mL in 0.9% NaCl were prepared.
Turbidimetry was used to estimate the quantity of visible protein aggregates by measuring protein density at 350 nm. At this wavelength, the intrinsic chromophores of the protein do not get absorbed. Only the aggregates in suspension diffract the incident light, thus causing pseudo-absorbance. The aggregation index was determined according to the following formula: λ350 /(λ279 − λ350) × 100 (where λ corresponds to the absorbance at the respective wavelengths of 350 and 279 nm). In agreement with Katayama et al., 14 an aggregation index <10 was evidence of the absence of soluble aggregates in the sample. In parallel, the protein content was estimated by measuring the absorbance at 279 nm in solutions diluted to 0.5 mg/mL. A decrease in this absorbance (>5%) would be manifested by a protein loss, especially due to adsorption during storage.
Second derivative UV spectroscopy was used to evaluate the impact of dilution and storage on the aromatic amino acids of the molecule. The position of the second derivative absorbance peak is sensitive to the polarity of the microenvironment of these amino acids and, thus, makes it possible to obtain an overall view of the tertiary structure of the molecule. The spectrum was analyzed from 250 to 320 nm. The operating conditions were the same as for the OD ratio. Five major peaks were monitored: 252-Phe, 258-Phe, 275-Tyr, 284-Thy/Trp, and 292-Trp.
Fluorescence was used to specifically examine the native fluorescence of tryptophan and study the possible changes in tertiary structure affecting the protein chains having high contents of this amino acid. Samples diluted to 0.1 mg/mL with NaCl were excited at 295 nm in a quartz cell maintained thermostatically at 25°C. The emission spectrum was recorded between 310 and 410 nm.
Ion chromatography
The samples were injected in pure form or diluted to 1 mg/mL. An excipient blank was injected to verify the absence of interference. The samples were centrifuged for 5 min at 10,000 rpm. The chromatograms obtained were compared with those on D0. The distribution of each variant was expressed as a percentage of the total area under the curve. Variations <3% were considered acceptable.
SEC chromatography
Samples diluted in 0.9% NaCI (1 mg/mL in 2 mL Eppendorf tubes) were centrifuged for 5 min at 10,000 rpm. The chromatograms obtained were compared with those on D0. The distributions of the main peak (monomer) and the associated peaks (dimers, oligomers, and high molecular weight peaks) were monitored. A monomer percentage >99% was considered acceptable. In addition, the occurrence of additional peaks (oligomers or fragments) was noted objectively.
Dynamic light scattering
DLS is a sensitive method for evaluating the size of population containing molecules, oligomers and aggregates from 0.3 nm to 10 μm. It measures the Brownian motion of the particles and relates to their size.
The samples in a quartz microcuvette (200 μL of each undiluted sample) were analyzed by a Nano S nanosizer particle counter (Malvern). The size and distribution of the hydrodynamic diameter as well as the possible occurrence of small aggregates (100–500 nm) were evaluated by integrating the intensity of the measured scattered light. A size variation of <3 nm over the course of time was considered acceptable. The occurrence of a peak of a population other than cetuximab (>10%) was considered unacceptable.
During a temperature rise, in the case of a dilution/concentration effect or simply during prolonged storage, the secondary, tertiary, and quaternary structures of a protein may change and lead to unfolding of the protein and ultimately to aggregation due to increased exposure of hydrophobic surfaces. These events can be observed by DLS because they cause large changes in protein size in the course of their denaturation. By raising the temperature progressively (1°C/min with 2 min of equilibration between each plateau) from 65°C to 80°C and measuring the diameter at each temperature, it is possible to plot the thermal aggregation curve. Comparison of the curves obtained for each duration and storage condition makes it possible to estimate overall but modest destabilization of the three-dimensional structure by energy accumulation. This is not detectable by the other methods, which do not reveal evidence of an already denatured state.
Colligative parameters
Density was measured by the oscillating tube method at 25°C. A variation of >0.01 g/mL in density was considered unacceptable.
An electronic pH meter with glass-combined micro electrode was used, and calibration was carried out prior to each series of measurements. The pH was measured at 25°C (temperature correction). A variation of 1 pH unit was considered unacceptable.
Osmolality was measured in a volume of 100 µL by the cryoscopic depression method, calibrated on a standard at 300 and 900 mOsmol/kg. A variation of 10 mOsmol/kg was considered unacceptable.
Sterility test
The sterility test was carried out only on D90. The type 2 test was used, as it is applicable to substances, preparations and products that must be sterile according to monograph 2.6.1 of the European Pharmacopoeia (10th edition). 3 The sterility test was carried out on three bags of each batch (the samples of the bags were pooled for each batch, then filtered by means of a Symbio Steritest pump [Merck Millipore]) and on the vial residues of the three batches. Filtration was carried out in two liquid media recommended by the European Pharmacopoeia: trypticase soy broth and thioglycollate broth. The media were then incubated at 25°C and 37°C for 14 days. The presence of a microbial bloom manifested by opalescence of the media was examined.
Results
Turbidity
The aggregation index remained within the predefined limits (<10) for all sampling times and both concentrations. Compared with D0, no change in turbidity at 350 nm after 30 days of storage was found, regardless of concentrations and batches. The same results were observed for samples subjected to a temperature excursion of 3 days at 25°C. These results demonstrate the absence of formation of light-diffracting aggregates during storage, both over 90 days at 4°C and after a temperature excursion of 3 days at 25°C.
Absorbance at 279 nm
On D0, the mean protein concentrations were 1.079 ± 0.008 mg/mL for the bags and 1.086 ± 0.005 mg/mL for the vials, compared with 1.028 ± 0.032 mg/mL for the bags and 0.999 ± 0.0064 mg/mL for the vials after 90 days of storage. These findings showed that protein concentrations in both bags and vials remained between 90% and 110% of the theoretical concentration (1 mg/mL). No protein loss during storage, especially adsorption, was observed, regardless of the evaluated concentration and storage temperature (Table 2).
Absorbances at 279 nm as a function of time for bags (with two concentrations) and vials on days 0, 15, 30, and 90 at 4°C.
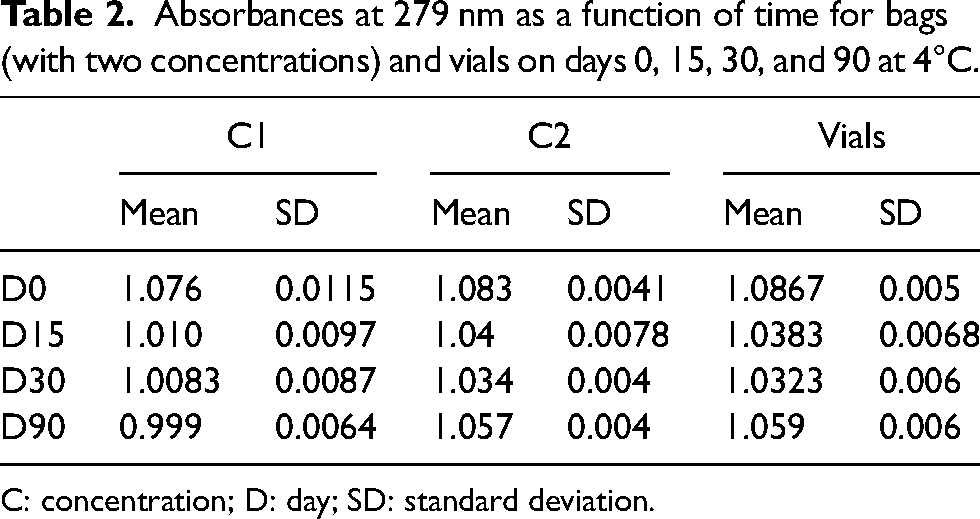
C: concentration; D: day; SD: standard deviation.
Derivative UV spectrometry and fluorescence: Tertiary structure
Derivative UV spectrometry in the aromatic acidic regions (250–300 nm) and fluorescence was used to estimate the alterations of the tertiary structure of the antibody during storage. As the position of the second derivative absorbance peak is sensitive to the polarity of the microenvironment of these amino acids, it is possible to obtain an overall view of the tertiary structure of the molecule. The five major peaks monitored were 252-Phe, 258-Phe, 275-Tyr, 284-Thy/Trp, and 292-Trp. The absence of change in the 250–270 nm region makes it possible to rule out perturbations of the phenylalanine environment by exposure to water during unfolding of the folded structure. Cetuximab stored at 4°C for up to 90 days retains its tertiary structure and remains identical to the initial substance (the slight shifts observed are <1 nm). The tertiary structure of cetuximab in bags and vials stored for 3 days at 25°C before being returned to 4°C remained unchanged for 30 days (Figure 2).
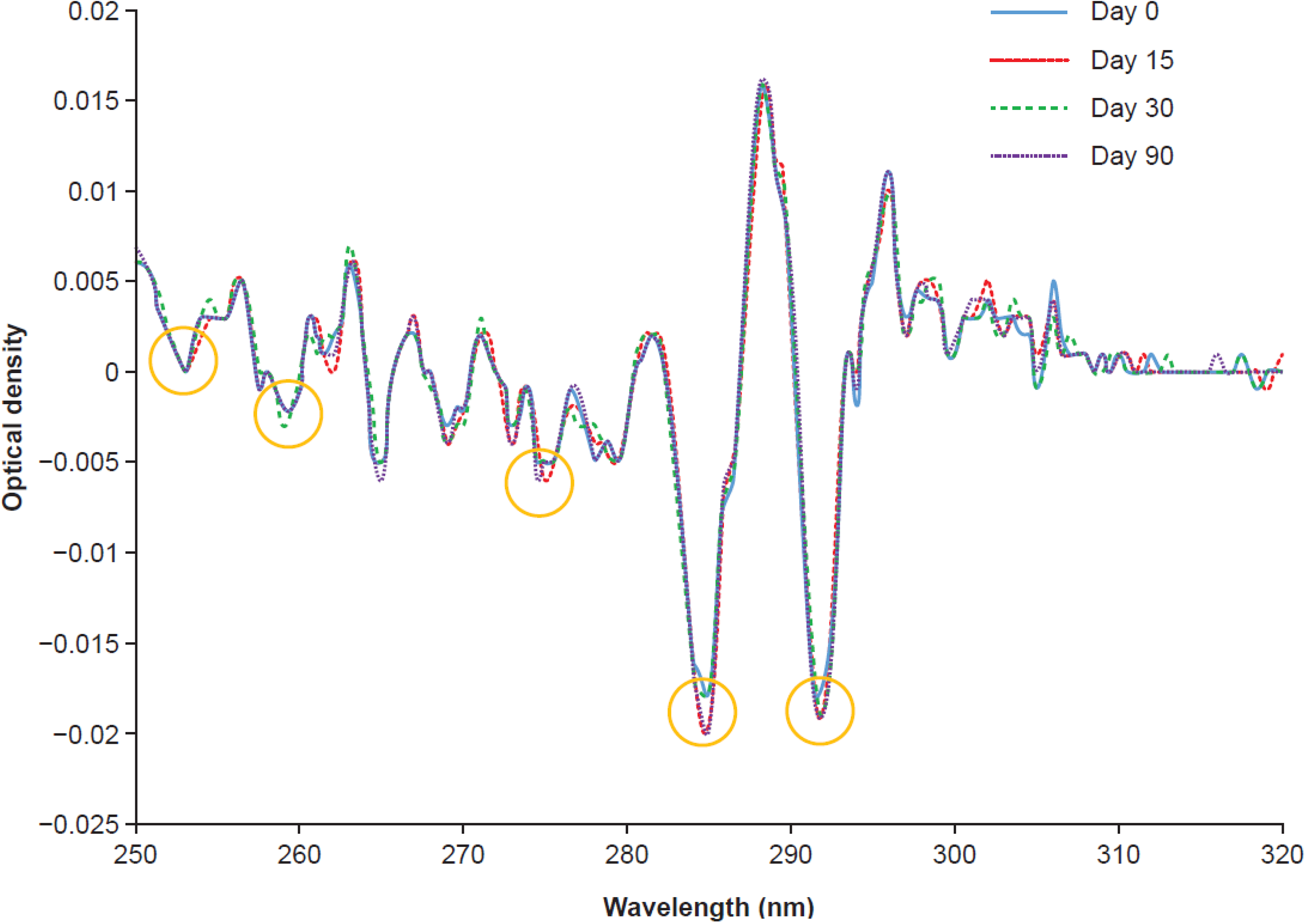
Second derivative ultraviolet spectrum of cetuximab diluted to 1 mg/mL or transferred to an empty bag as a 5 mg/mL solution and stored at 4°C.
Fluorescence
The fluorescence emission spectra did not change as a function of the storage duration (absence of shift >1 nm), confirming that the environment of the tryptophan residues was not modified (Figure 3). The same results were found for all bags and vials. Similar results were observed after storage of the vials for 30 days at 4°C.
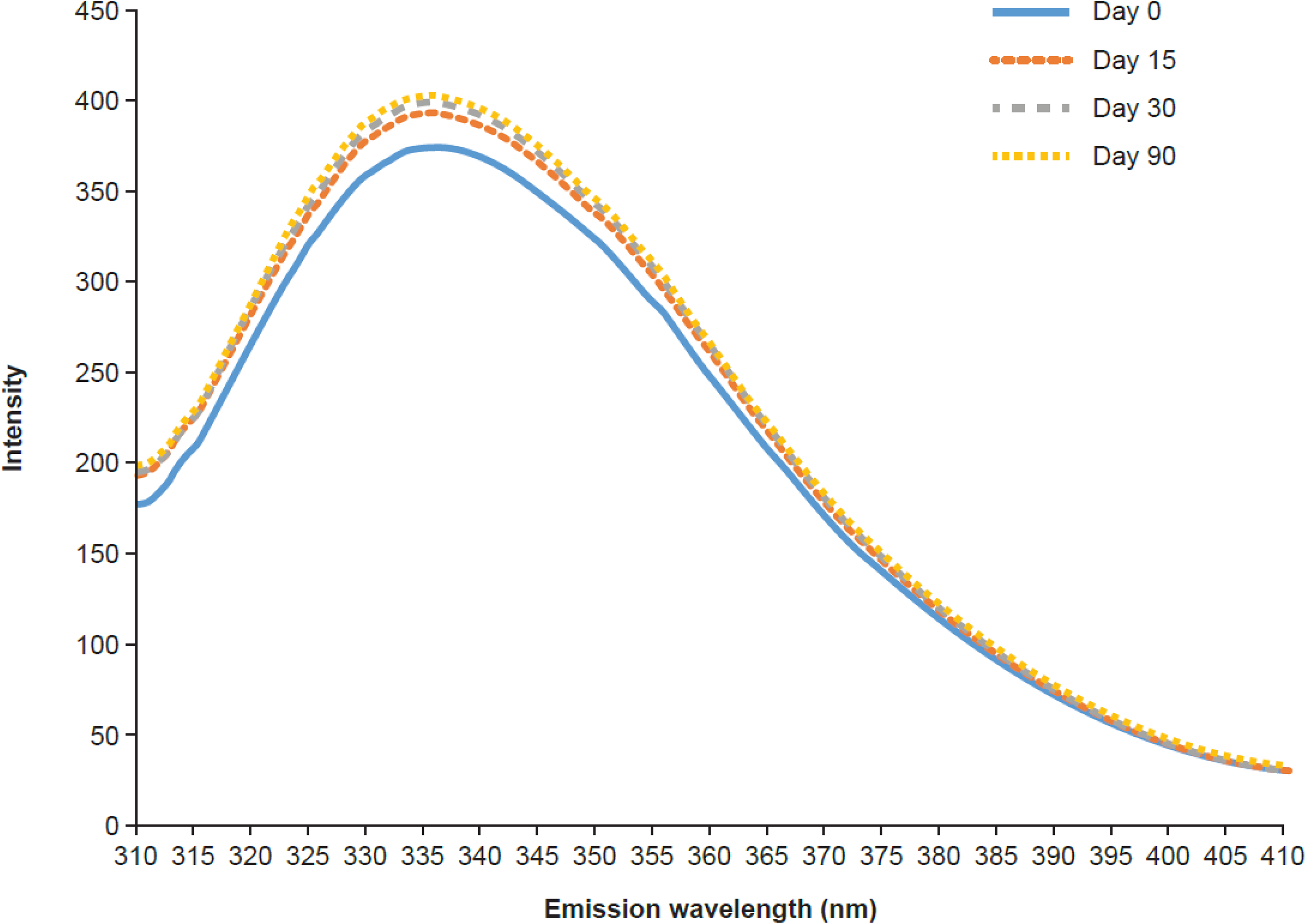
Fluorescence emission spectrum of diluted cetuximab stored for 30 days at 4°C.
Ion chromatography
Eight cetuximab isoforms/variants were found, consisting of four acidic variants situated before the main peak, one main variant and three basic variants. No new peak was observed, either in the acidic or basic part of the chromatograms that could indicate a change in variant distribution (Figure 4).
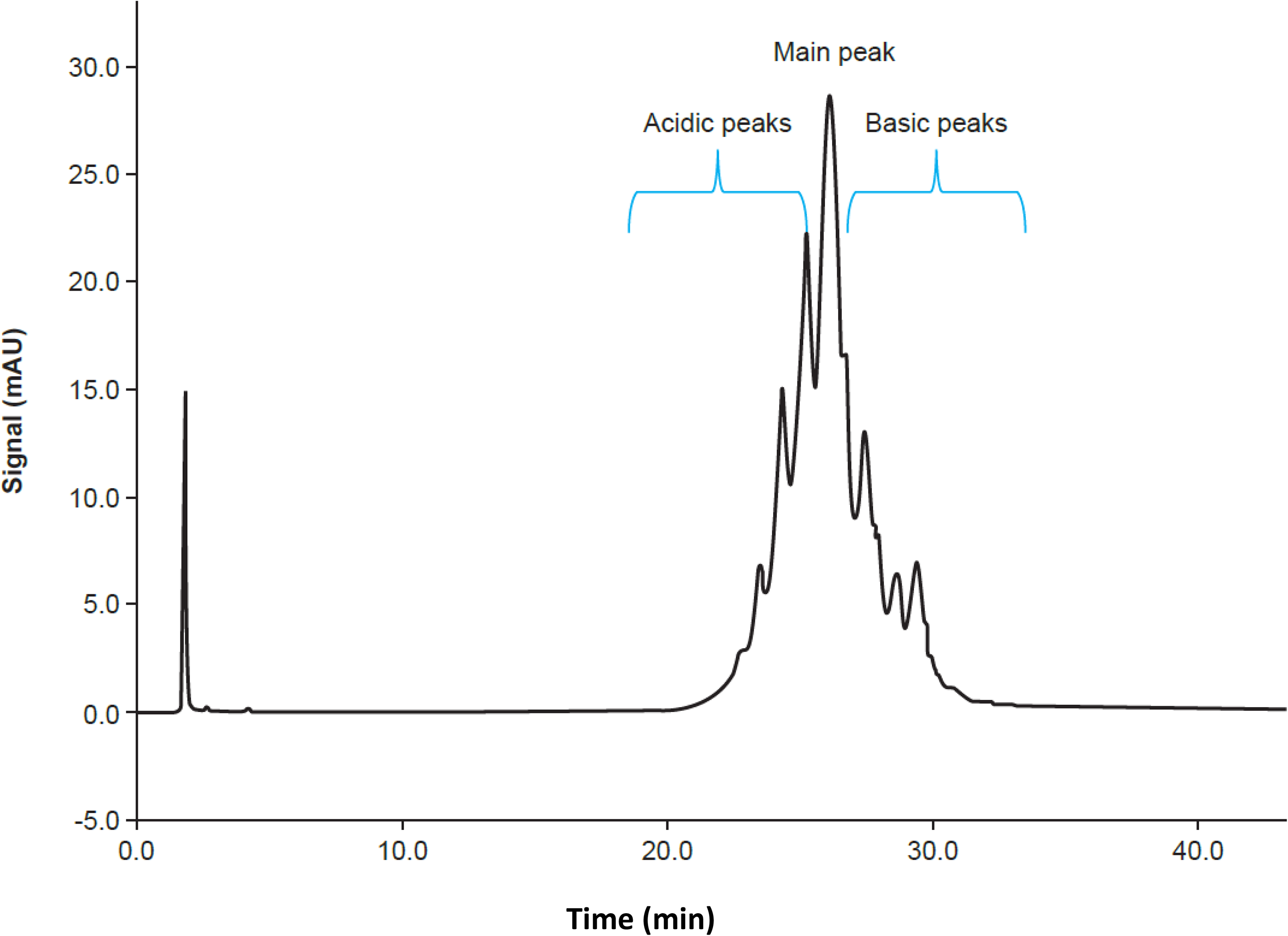
Ion chromatography profiles of cetuximab with eight identified peaks indicated (main peak = no. 5).
No change in the percentages of isoforms was observed after 30 days of storage at 4°C. A slight decrease in the main peak to the benefit of the acidic peak was observed on D90, but it remained <3% and had no impact on stability (Table 1).
A thermal excursion of 3 days at 25°C did not affect the stability of cetuximab, and similar results were observed after storage of the vials for 30 days at 4°C. These findings show that storing pure and diluted cetuximab for 90 days at 4°C does not increase its degradation by hydrolysis and isomerization.
Molecular exclusion chromatography
SEC allowed visualization of cetuximab monomers and possible oligomers and high molecular weight polymers, a sign of potential aggregation. For the vials, the monomer represented 99.84 ± 0.0166% of the total at D0 and 99.83 ± 0.011% of the total at D30 (Table 3), and no high molecular weight polymers were found. No additional peak was observed, regardless of the storage temperature and duration.
Percentages of the SEC peaks for bags (with two concentrations) and vials on days 0, 30, and 90.
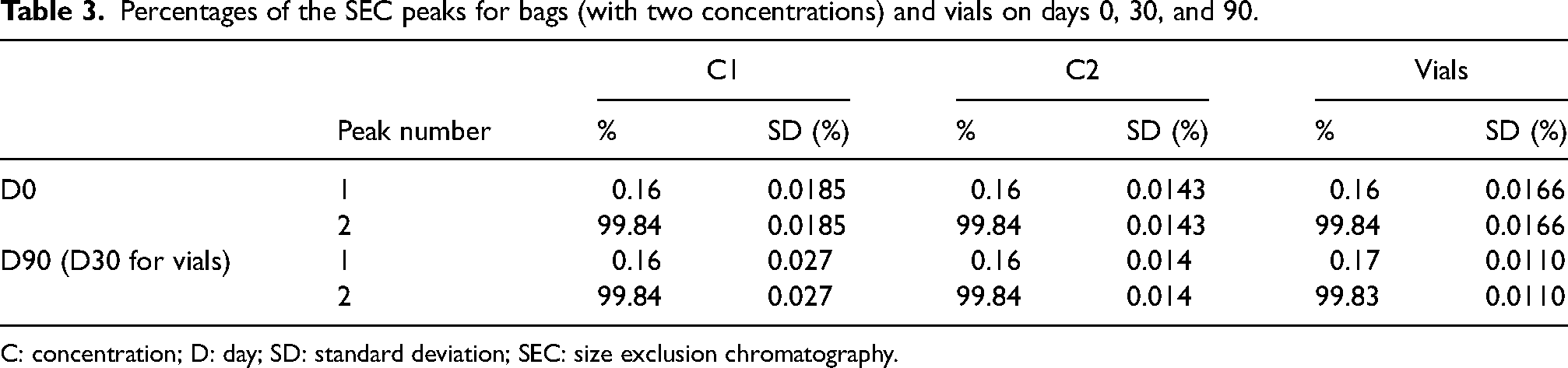
C: concentration; D: day; SD: standard deviation; SEC: size exclusion chromatography.
These findings, for both bags and vials, demonstrate the absence of oligomer formation and molecular modifications leading to fragmentations. Similar results were observed after storage of the vials for 30 days at 4°C and after storage of bags and vials for 72 h at 25°C.
DLS—hydrodynamic parameters
The mean hydrodynamic diameters on D0 are presented in Table 4.
Relative percentage of cetuximab isoforms in ion chromatography after storage at 4°C.
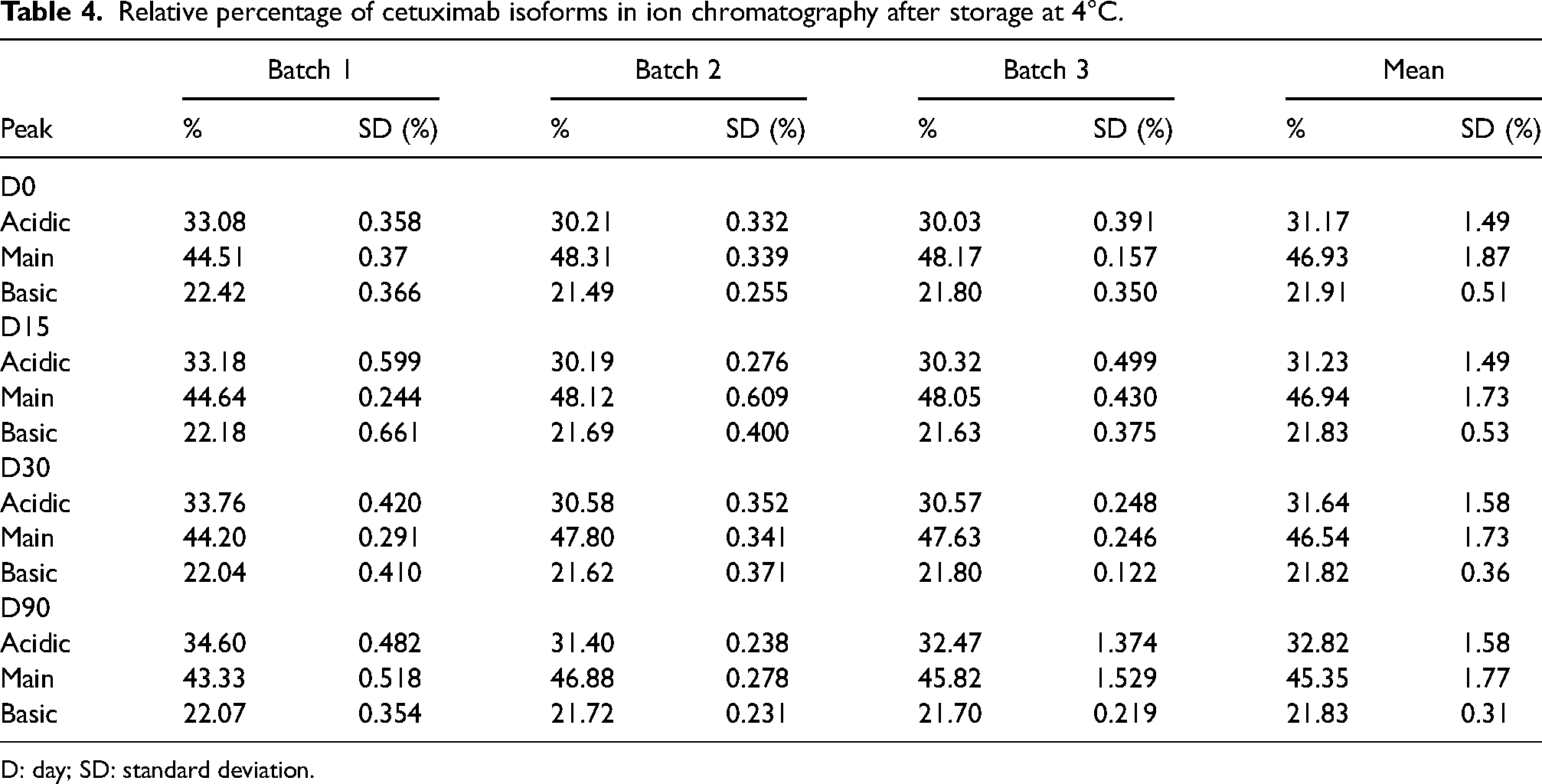
D: day; SD: standard deviation.
The very low polydispersity index values suggest that there is no dimerization or oligomerization, in agreement with the SEC results. Likewise, neither the hydrodynamic diameters nor the polydispersity index values evolved as a function of storage time, as shown by D90 results. No additional submicronic (50–1000 nm) population was observed. Furthermore, thermodynamic stability was not affected by concentration, temperature, or storage duration.
Colligative parameters (density, pH, and osmolality)
After 90 days of storage (30 days for vials), the density had not changed under any of the tested conditions. The same finding was obtained after a thermal excursion at 25°C (Delta <0.001). No difference in pH was found between the concentrations in the bags at D0 and D90. The initial pH of the vials was similar to that of the bags (5.47 ± 0.01) and remained stable during 30 days of storage (5.49 ± 0.048). The same observations were reported for the vials and bags subjected to a thermal excursion of 3 days at 25°C. Slight changes in pH (<0.5 pH units) did not affect the stability of cetuximab. The variations in osmolality remained <10 mOsmol/kg and did not impact cetuximab stability. The same observations were reported for the vials and bags subjected to a thermal excursion of 3 days at 25°C.
Sterility test
No evidence of microbial growth was found in bags after 90 days of storage at 4°C.
Discussion
We used several stability-indicating orthogonal and complementary methods to investigate the stability of cetuximab in vials after opening, after dilution with NaCl to a final concentration of 1 mg/mL in polyolefin bags and repackaged as a 5 mg/mL solution in polypropylene bags at 4°C. Overall, for these two concentrations and standard storage conditions (90 days at 4°C) or after a thermal excursion of 3 days at 25°C, no change was found in the physical or chemical stability of the antibodies in any of the three tested batches. Compared with freshly prepared bags (D0), no sign of physical instability was observed after 90 days of storage at 4°C. The chromatographic profiles by gel exclusion did not reveal formation of oligomers or breakdown of the molecular structure.
Similar to the findings of Farjami et al., 5 eight cetuximab isoforms/variants were found. IC did not reveal any change in distribution of ionic variants, signifying the absence of modification of the initial structure, especially deamidation. Derivative UV and fluorescence spectral analysis did not reveal any changes in the tertiary structure of the antibody during storage at 4°C for 90 days or after 3 days of thermal excursion at 25°C. The thermal denaturation curves were identical, suggesting the absence of thermodynamic destabilization of the entire three-dimensional structure. Identical results were observed for the vials after opening. Although cetuximab has been shown to be sensitive to mechanical stresses such as stirring, 15 no sign of aggregation was observed in this study. This finding demonstrates that typical handling procedures, such as dilution, bag filling, or withdrawing by syringes, do not alter the extended stability of cetuximab.
Current label restriction to single use is based on potential risk of microbiological contamination. Ensuring that strict precautions are taken, by opening the vial in a sterile atmosphere and maintaining sterile conditions throughout the period of vial use, is critical and allows for its multiple use. It should be noted that vials may be kept at 4°C for up to 30 days after opening if the assessed sterile conditions have been maintained and data have been recorded throughout the storage period of the opened vials. The bags were prepared from opened vials in a sterile insulator and stored in a Rapid Transfer Port that maintains sterility when transferring vials from the refrigerator to the isolator. The risk of contamination following the opening of the vials was assessed and the results of the risk analysis supported the extended in-use shelf-life of the opened vials.
A high number of samples was used for the sterility test to ensure that there is no contamination detected in any of the samples. The results showed that taking successive samples from the initially sterile samples did not lead to the introduction of contaminants that could have led to artifacts in the analytical determinations carried out. The risk of microbiological contamination should be assessed by each individual aseptic unit according to the risk factors identified in the production of sterile pharmaceutical preparations. These risk factors include quality of the environment, nature of the process, aseptic technique used, nature of the product, duration of administration, and incorrect composition. For our cetuximab preparation, these factors define whether a preparation is high or low risk. 16
Conclusions
The results of these analyses support the extended in-use shelf-life of cetuximab vials and bags. Cetuximab may be stored in vials (500 mg) for 30 days after opening and for 30 days after diluting in 0.9% NaCl to a concentration of 1 mg/mL or transferring into an empty bag. Cetuximab may also be stored for 90 days at 4°C without any noteworthy physical or chemical changes. Furthermore, the thermal cycling study conducted with storage of samples (bags and vials) for 3 days at 25°C affirms that this brief temperature excursion does not jeopardize the integrity of cetuximab. Cetuximab stored in this manner has sufficient stability to accommodate storage problems, such as interruption of the cold chain for 48 to 72 h.
In our stability studies, we control the concentrations used in practice by keeping them between a low and a high concentration. Dilution may have an impact on stability or, conversely, a highly concentrated form may adversely affect sample preservation. If no changes are observed at the two concentration points tested, the risk of seeing an effect on the concentrations between these two points is very low. In our study, we used the 5 mg/mL single-dose vials; however, our technique allows us to extrapolate the results to all the concentrations in this range (between 1 and 5 mg/mL), including the 2 mg/mL concentration of vials.
International studies investigating physicochemical stability of reconstituted IV solutions and ready-to-administer infusions concluded that their extended storage could facilitate pharmacy-based centralized preparation and result in cost savings.17–19 This is the first study investigating the physicochemical stability of cetuximab in opened vials and infusion bags when stored for longer periods than currently recommended. Our findings have implications for clinical practice, as an extended shelf-life for cetuximab can be cost-effective for healthcare providers, improve the efficiency of day-to-day operations, increase flexibility and convenience for clinical staff and reduce wastage.
Footnotes
Acknowledgements
The authors wish to thank Merck for technical support. Support for third-party writing assistance, provided by Aleksandra Erac-Zganec, of Nucleus Global, was funded by Merck Santé S.A.S., Lyon, France, an affiliate of Merck KGaA (CrossRef Funder ID: 10.13039/100004755). Opinions, recommendations, and views expressed in this publication are of the authors and should not be interpreted to represent the views or recommendation of Merck KGaA, Darmstadt, Germany. Authors recommend the practitioners to consult the product's current approved market specific prescribing information.
Author Contributions
VV contributed to conceptualization, methodology, formal analysis, writing-original draft preparation, and funding acquisition. GLG was involved in methodology, formal analysis, writing review, and editing. AA was involved in revision and approbation of the text. All authors were involved in writing-original draft preparation, writing review and editing, and final approval.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This manuscript was funded by Merck Santé S.A.S., Lyon, France, an affiliate of Merck KGaA.
